Abstract
Background:
Postural instability is a major disabling factor in patients with advanced Parkinson’s disease (PD) and often resistant to treatment. Previous studies indicated that imbalance in PD may be reduced by low-intensity noisy galvanic vestibular stimulation (nGVS).
Objective:
To investigate the potential mode of action of this therapeutic effect. In particular, we examined whether nGVS-induced reductions of body sway in PD are compatible with stochastic resonance (SR), a mechanism by which weak sensory noise stimulation can paradoxically enhance sensory information transfer.
Methods:
Effects of nGVS of varying intensities (0–0.7 mA) on body sway were examined in 15 patients with PD standing with eye closed on a posturographic force plate. We assumed a bell-shaped response curve with maximal reductions of sway at intermediate nGVS intensities to be indicative of SR. An established SR-curve model was fitted on individual patient outcomes and three experienced human raters had to judge whether responses to nGVS were consistent with the exhibition of SR.
Results:
nGVS-induced reductions of body sway compatible with SR were found in 10 patients (67%) with optimal improvements of 23±13%. In 7 patients (47%), nGVS-induced sway reductions exceeded the minimally important clinical difference (optimal improvement: 30±10%), indicative of strong SR. This beneficial effect was more likely in patients with advanced PD (R = 0.45; p = 0.045).
Conclusions:
At least half of the assessed patients showed robust improvements in postural balance compatible with SR when treated with low-intensity nGVS. In particular, patients with more advanced disease stages and imbalance may benefit from the non-invasive and well-tolerated treatment with nGVS.
INTRODUCTION
Parkinson’s disease (PD) is one of the most prevalent neurodegenerative diseases and is characterized by a loss of dopaminergic neurons in the substantia nigra associated with intracellular aggregation of misfolded α-synuclein in form of Lewy bodies that extend through many brain regions along the course of the disease [1]. With more advanced disease stages gait and balance impairments as well as a spectrum of non-motor symptoms associated with PD (e.g., autonomic dysfunction, sleep disorders, mood disturbance, cognitive decline) become more prevalent and dominant for activities of daily living and quality of life. In particular, postural instability in advanced PD is a major cause of falling [2] that may only be alleviated by physical training, but remains resistant to pharmacological therapy or is even worsened by dyskinesia induced by dopaminergic treatment [3–5] or deep brain stimulation [6]. Accordingly, there is need for alternative principles and strategies for symptomatic treatment of postural instability in advanced PD.
In recent years, several non-invasive brain stimulation techniques have been employed to specifically target motor symptoms in PD that do not adequately respond to available pharmacological or invasive brain stimulation treatment options. Among these, galvanic vestibular stimulation (GVS)—a non-invasive technique to modulate vestibular afferent activity by weak electric currents [7]—has gained special interest due to the well documented close anatomic and functional connection between central vestibular networks and brain structures involved in PD pathology, especially the basal ganglia [8]. Specifically, a form of sub-threshold noisy GVS (nGVS; for a review see [9]) has been successfully administered to alleviate postural imbalance and other PD-associated motor and autonomic symptoms in a series of pilot studies on patients with PD [10–16].
Although the mode of action of nGVS is poorly understood so far, nGVS treatment effects in PD were previously repeatedly attributed to a mechanism known as stochastic resonance (SR) [8, 17]. SR is a phenomenon that can be observed in sensory systems, where the system’s response to weak, sub-threshold signals becomes enhanced in the presence of a weak amount of noise [18–20]. Typically, SR effects will critically depend on the noise amplitude insofar as signal transfer improves at intermediate noise intensities below threshold whereas too low or too high noise amplitudes will either not affect or disturb signal processing [20]. Accordingly, the characteristic signature for the exhibition of SR is a noise-induced modulation of the system’s output that follows a bell-shaped performance curve, which peaks at a specific intermediate level of noise that optimally facilitates signal transfer within the system.
Previous studies on nGVS effects in PD commonly limited the application of nGVS to one particular noise intensity and were thus not able to uncover the potential underlying treatment mechanism. To overcome this deficit, we studied nGVS effects on static postural stability in patients with PD across a broad range of stimulation intensities. By using different quantitative and qualitative criteria, we determined whether nGVS-induced modulations in balance of patients with PD are compatible with the presence of SR (i.e., a bell-shaped response curve) or follow other response dynamics. We further examined whether demographic or disease-related factors (e.g., duration of disease, severity of symptoms, etc.) may promote or prevent beneficial nGVS treatment outcomes in individual patients.
MATERIALS AND METHODS
Participants
Fifteen patients with PD (age 61.1±9.4 years, 4 females) participated in the study and provided written informed consent prior to inclusion. Detailed patient characteristics are provided in Table 1. Each patient underwent a complete physical, neurological, and neuro-otological examination, as well as a standardized clinical scoring of disease stage and symptom severity (Hoehn & Yahr Scale, H&Y; Movement Disorders Society - Unified Parkinson’s Disease Rating Scale, MDS-UPDRS) by an expert neurologist (AZ). Non-motor symptoms such as depression or cognitive impairment were further assessed by the Beck Depression Inventory (BDI) and the Montreal Cognitive Assessment (MoCA). Patients had a mild to moderate disease severity with H&Y of 2.1±0.5 and MDS-UPDRS of 23.5±20.3. None of the patients showed additional signs of atypical Parkinsonism or manifest vestibular or auditory disorders. Patients were examined while taking their regular medications. L-dopa was the basic medication in all patients (mean daily dose: 590±248 mg). Fifteen age-matched healthy controls (age 57.7±4.7 years, 7 females; Supplementary Table 1) were included in the study to establish normative data. All participants gave written informed consent prior to study inclusion. The ethics committee of the medical faculty of the University of Munich approved the study protocol, which was conducted in accordance with the Declaration of Helsinki.
Clinical characteristics and global stimulation effects of patients
MDS-UPDRS, Movement Disorders Society - Unified Parkinson’s Disease Rating Scale; H&Y, Hoehn & Yahr Scale; SR, stochastic resonance; PD subtype: ART, akinetic-rigid type; ET, equivalent type; L-dopa: DA, dopamine agonist; COMT, COMT inhibitor; MAO, MAO-B inhibitor; SSRI, selective serotonin reuptake inhibitor; Chl, cholinesterase inhibitor; comorbidities: D, depression; CI, cognitive impairment; PN, peripheral neuropathy.
Galvanic vestibular stimulation
Vestibular noise stimulation (i.e., nGVS) was applied via a pair of 4.0 cm×6.0 cm Ag-AgCl electrodes attached bilaterally over the left and right mastoid process. The skin at the electrode sites was cleaned with abrasive gel and subsequently dried. Zero-mean Gaussian white noise stimulation with a frequency range of 0–30 Hz and varying peak amplitudes of 0–0.7 mA was delivered by a mobile constant current stimulator (neuroConn®, Illmenau, Germany).
Experimental procedures
Body sway was recorded for 30 s on a posturographic force plate (Kistler, 9261A, Kistler Group, Winterthur, Switzerland) at 40 Hz while patients were standing with their eyes closed (Fig. 1A). This procedure was performed eight times, while patients were stimulated with a different amplitude of nGVS (ranging from 0–0.7 mA, individually randomized order per patient) in each trial. Patients were blinded to the exact stimulation order. Between trials, patients were given at least 30 s breaks to recover and were allowed either to hold on a handrail or to sit down.
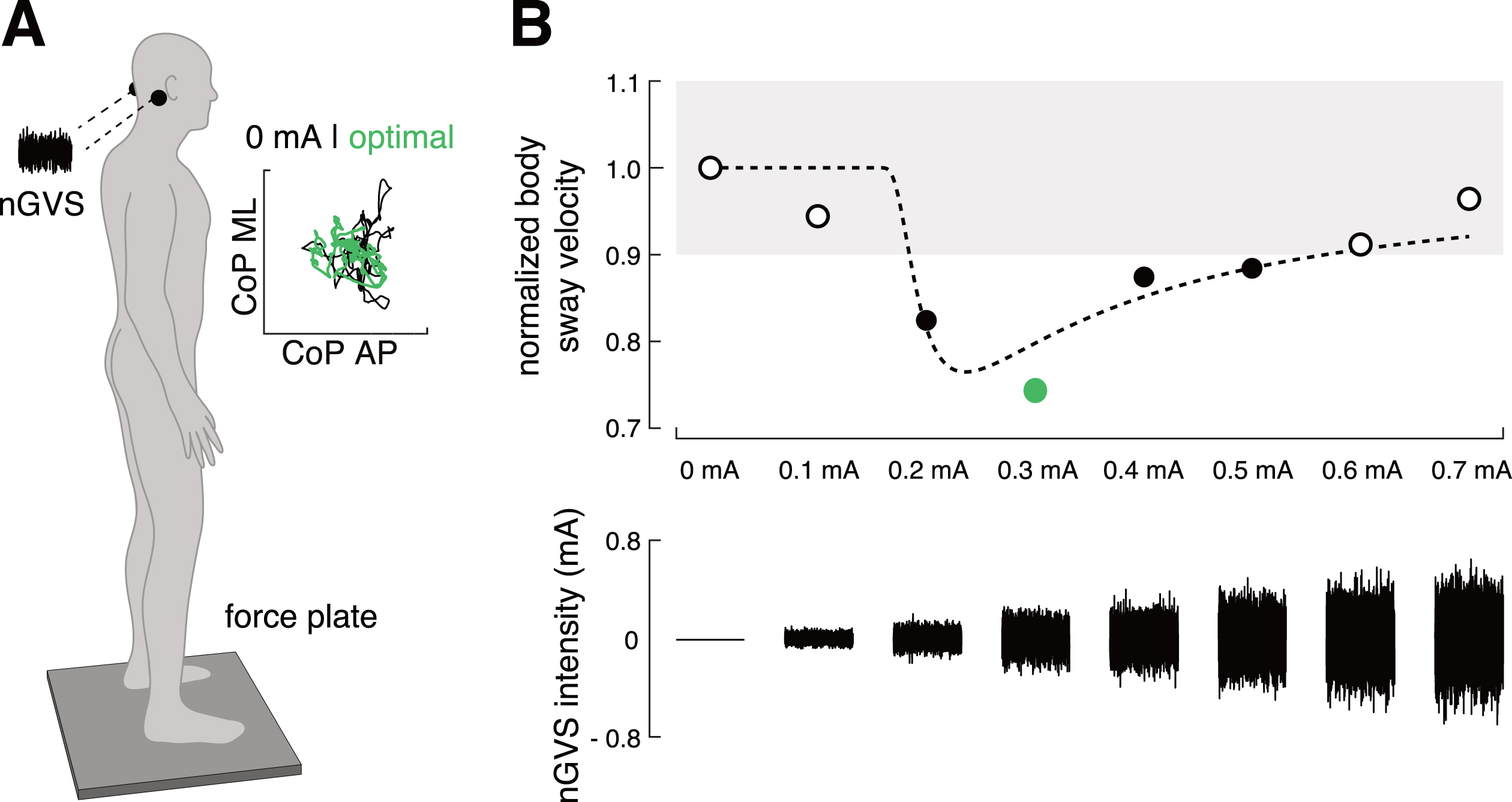
Experimental setup and procedures. A) Effects of noisy galvanic vestibular stimulation (nGVS) on static balance in patients were measured on a posturographic force plate. Velocity of body sway was calculated from the resultant center of pressure (CoP) trajectories (inset). B) Exemplary modulation of body sway (simulated data, upper panel) across the administered nGVS intensities (lower panel) that follows a bell-shaped performance curve indicative of the presence of stochastic resonance (model fit: dashed line). Filled black dots indicate body sway reductions greater than the minimal clinically important difference (grey area). The green filled dot indicates the optimal reduction of body sway at a particular nGVS level.
Data and statistical analysis
For each stance trial, mean sway velocity was calculated as the primary outcome measures based on the recorded radial center-of-pressure (CoP) trajectory using the formula SV = 1/T × ∑ i |ri+1 - r i |, [mm/s], where T is the total trial duration (i.e., 30 s) and r i is the radial CoP distance of the ith sample. For further analysis, sway velocity measures from the 8 stance trials were normalized to sway velocity obtained during 0 mA stimulation (i.e., sham condition).
To determine whether SR-like dynamics were present in the balance responses of patients to varying nGVS levels, we tested three increasingly rigorous criteria that built on one another in analogy to a previously proposed approach [21, 22]: 1) The first criterion tested whether body sway of patients improved for at least one particular nGVS level compared to sham condition (i.e., 0 mA nGVS). 2) The second criterion was based on a visual inspection of response dynamics of body sway across increasing nGVS level by three experienced human raters (i.e., MW, FS, and AZ). Each rater had to evaluate whether (in addition to the fulfillment of the first criterion) nGVS-amplitude-dependent changes of body sway in individual patients were further compatible with a bell-shaped response curve with improvements of performance at intermediate stimulation intensities that is indicative of the presence of SR. This evaluation was based on a plot of the normalized nGVS-dependent changes in body sway and a concomitant plot of a theoretical SR-curve that was fit on the data using a goodness-of-fit statistic [21, 22] (Fig. 1B). The criterion was met if at least two of the raters identified the presence of SR-like dynamics. 3) The third criterion additionally evaluated whether improvements at intermediate nGVS levels were greater than the minimal clinically important difference (MCID; defined as half the standard deviation for normative data [23]) for changes in body sway velocity. MCID for sway velocity was 2.3 mm/s calculated based on the posturographic recordings of the 15 age-matched healthy individuals standing with eyes closed for 30 s.
Based on the three criteria, patients were classified as showing solely optimal improvement and no SR (criterion 1), exhibiting weak SR (criterion 1 & 2) or showing strong SR (criterion 1 & 2 & 3). Potential correlations between SR classification and age, gender, disease duration or disease severity (H&Y, MDS-UPDRS) were analyzed using Spearman’s rank correlation. Results were considered significant at p < 0.05. Statistical analysis was performed using SPSS (Version 26.0, IBM Corp., USA).
RESULTS
Administration of nGVS at intensities ranging from 0–0.7 mA was well tolerated and did not cause disequilibrium in any of the examined patients. Two patients noticed nGVS treatment at higher intensities in the form of a light tickling sensation on the skin. The remaining patients did not perceive nGVS at any of the tested intensities.
Initially, we determined whether sway velocity was reduced by at least one particular nGVS intensity compared to sham condition (i.e., 0 mA nGVS). This criterion was met by every patient with an optimal improvement rate of on average 19% (range: 3 to 49%) at a mean nGVS intensity of 0.3 mA (range: 0.1 to 0.5 mA).
In the second step of analysis, an established SR-curve model was fitted on the individual patient outcomes (Fig. 2). Three experts were asked to independently rate for each patient by visual inspection of individual fitted curves whether nGVS-induced modulations of sway velocity followed a bell-shaped performance curve. Based on their judgements, SR-like balance responses to nGVS treatment were present in 10 patients (67%) with an optimal improvement rate of on average 23% (range: 3 to 49%) at a mean nGVS intensity of 0.3 mA (range 0.1 to 0.5 mA). Also, on the group level average responses of these patients closely followed the theoretic bell-shaped SR-curve fit with optimal improvement at intermediate noise intensities (Fig. 3). In the remaining 5 patients (33%), body sway velocity did not show any consistent changes but rather fluctuated randomly across the tested nGVS intensities. Four of these patients had a very mild impairment (MDS-UPDRS < 10, on-medication).
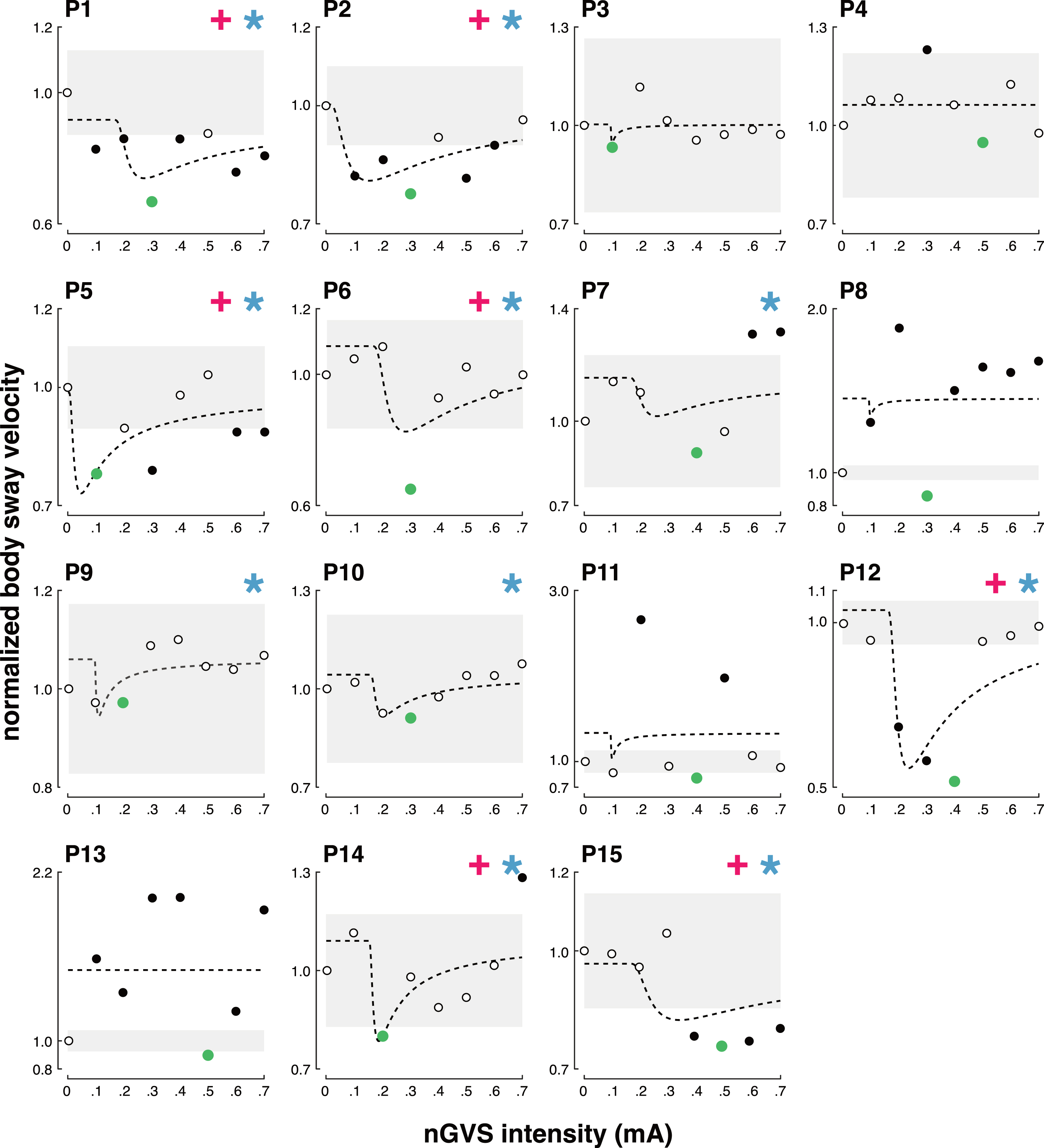
Individual effects of low-intensity vestibular noise stimulation on static balance. Normalized body sway responses to noisy galvanic vestibular stimulation (nGVS) are plotted against the administered nGVS levels for each individual patient. Dashed lines represent the stochastic resonance (SR) model fits. Black filled dots indicate body sway modulations greater than the minimally important clinical difference (grey area). Green filled dots indicate optimal reductions of body sway at particular nGVS levels. Blue asterisks denote those patients that exhibit SR-like responses according to three human judges (weak SR). Pink crosses denote those patients that additionally show clinically meaningful improvement of body sway (strong SR).
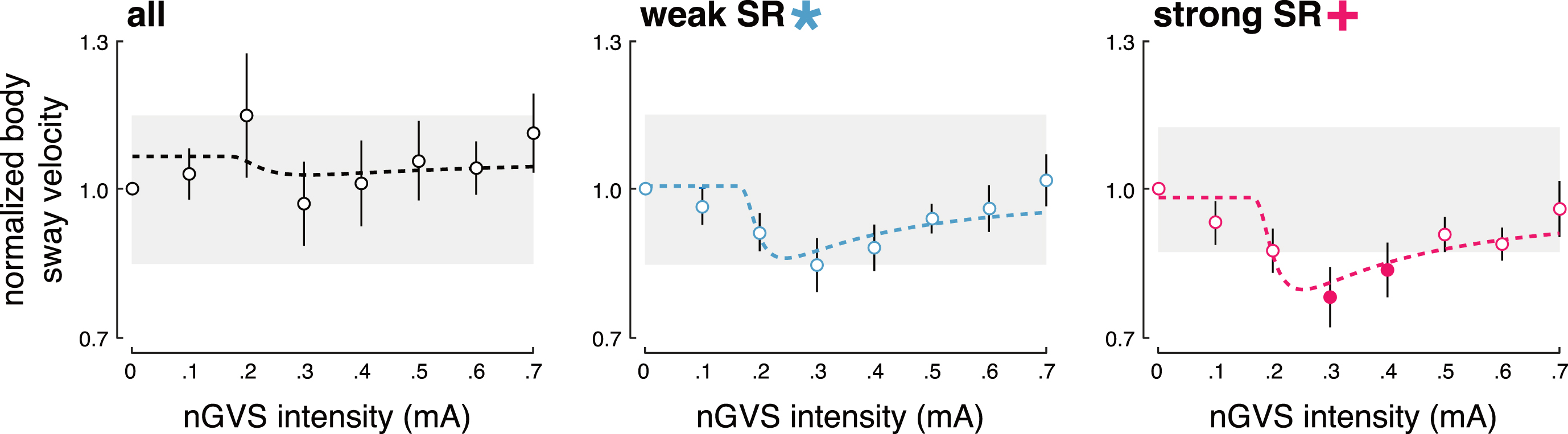
Group average effects of low-intensity vestibular noise stimulation on static balance. Group average normalized body sway responses (mean±SEM) to noisy galvanic vestibular stimulation (nGVS) are plotted for each of the administered nGVS levels for all patients (left panel), those patients exhibiting weak stochastic resonance (SR; middle panel), and those exhibiting strong SR (right panel). Filled dots indicate body sway modulations greater than the minimally important clinical difference (grey area).
Subsequently, we identified those patients that not only exhibited SR-like responses to nGVS treatment, but also showed clinically meaningful improvements in static balance (i.e., reductions of sway velocity greater than the MCID; Fig. 2). The criterion for the presence of strong SR was met by 7 patients (47%) with an optimal improvement rate of on average 30% (range: 21 to 49%) at a mean nGVS intensity of 0.3 mA (range: 0.1 to 0.5 mA). Exhibition of strong SR was also apparent on a group level with considerable improvements of static balance at intermediate noise intensities (Fig. 3).
In a final step, we explored whether any demographic or disease-related factors might facilitate or prevent the exhibition of weak or strong SR in response to nGVS treatment. Correlation analysis revealed an association between disease severity (MDS-UPDRS) and the presence of SR (R = 0.45; p = 0.045, not corrected for multiple comparisons) only. Patients with advanced forms of PD and imbalance were accordingly more likely to exhibit SR-like responses at clinically meaningful effect sizes when treated with nGVS.
DISCUSSION
Previous studies provide accumulating evidence that PD-associated postural imbalance and other motor and autonomic symptoms might be attenuated by a treatment with low-intensity vestibular noise stimulation (i.e., nGVS) [10–16]. These beneficial effects were consistently attributed to the exhibition of SR in vestibular sensorimotor pathways, without, however, providing sufficient evidence for the latter assumption. Since a better understanding of the mode of action of nGVS in PD is important for potential future therapeutic applications, we explicitly examined whether nGVS treatment influences postural instability in patients with PD in a way compatible with the presence of SR. We found that treatment responses in two thirds of the examined patients closely followed an SR-like bell-shaped performance curve with optimal balance improvements at intermediate noise intensities (i.e., between 0.1 and 0.5 mA). The average optimal nGVS intensity was found at 0.3 mA, which conforms with previous reports on healthy individuals [21] and equates to approximately 60% of the estimated detection threshold of vestibular afferent responses to GVS [24]. In half of the patients, nGVS treatment further yielded balance improvements at clinically meaningful effect sizes. Balance responses in the remaining third of patients did not show any systematic dependency on nGVS and hence likely reflect variation in the performance metric (i.e., body sway) rather than any therapeutic effect. The observed responder rate to treatment in patients is considerably higher than previously reported for young healthy individuals, where nGVS-induced balance responses compatible with SR were only rarely reported [22]. Taken together with the observed association between PD severity and responder rate, this suggests that particularly patients with advanced postural imbalance and motor symptoms might benefit from a treatment with nGVS. For these patients, treatment options to alleviate imbalance, prevent falls, and preserve mobility and self-care are currently limited to physical therapy, while other therapeutic approaches (such as L-dopa or deep brain stimulation) have limited effects or even aggravate postural instability by dyskinesia [3].
How could nGVS-induced SR impact postural imbalance in PD? There is growing evidence that PD is linked to a dysfunction of the vestibular system, in particular deficits within vestibulospinal reflex pathways (for a review see [8]). nGVS-induced SR in vestibular signal transfer has been shown to not only sensitize vestibular perception per se [21, 26], but to also improve the responsivity of vestibulospinal reflexes in healthy individuals and patients with vestibular hypofunction [27, 28]. These ameliorating effects of nGVS on vestibular-related balance function were further linked to an increased static and dynamic balance performance of healthy individuals while standing and walking under vestibular-challenging circumstances [29–31] and in patients with chronic bilateral vestibular impairment [32]. Hence, nGVS could positively impact postural instability in PD by restoring the reduced vestibulospinal balance responses of affected patients [12].
In addition, nGVS has been suggested to induce central neurochemical changes in basal ganglia loops [12, 33]. PD is linked to a progressive degeneration of dopaminergic neurons (e.g., in the substantia nigra) that results in decreased on-site levels of GABA release. On the other hand, application of nGVS in a parkinsonian rat model has been shown to specifically promote GABA release in the substantia nigra, thus improving locomotor and balance performance [33]. Hence, nGVS could potentially help to transiently normalize the neurochemical balance in the motor circuitry of the basal ganglia. This mode of action would imply an inhibition of the substantia nigra pars reticulata (a structure that is overactive in patients with PD) which may be linked to amelioration of postural symptoms associated with PD [12, 33]. Thereby, nGVS may resemble the effect induced by L-Dopa treatment. Other sites of action in the basal ganglia such as the caudate nucleus or putamen may be considered based on c-FOS expression profiles following nGVS in rat models [34].
The findings of this study should be interpreted in light of certain preconditions. First, in accordance with previous studies [10, 16], we examined nGVS effects on balance while patients were continuing their regular dopaminergic medication. This might to some extent mask the stimulation effects [13]. On the other hand, postural imbalance in PD has been shown to be rather unresponsive to treatment with L-dopa especially in advanced disease stages [5]. Furthermore, in a realistic future treatment scenario, nGVS would presumably be applied complementarily rather than acting as a substitution for established pharmacological treatment (e.g., with L-dopa). Secondly, in addition to the rather small sample size of examined patients, our study cohort was mainly focused on patients with mild to moderate disease severity (H&Y: 2.1±0.5). Among these, the more affected patients had more benefits from nGVS treatment. Hence, follow-up studies are required to examine whether nGVS treatment of balance deficits is equally or even more effective in patients at more advanced stages of PD.
Footnotes
ACKNOWLEDGMENTS
The authors thank Katie Göttlinger for copy-editing the manuscript. The authors thank Lorenz Assländer for providing recourses for data analysis. The study was supported by the German Federal Ministry for Education and Science (BMBF, IFB 01EO1401).
CONFLICT OF INTEREST
MW received funding from the neuroConn GmbH (DC stimulator).
JL reports speaker fees from Bayer Vital, Biogen and Roche, consulting fees from Axon Neuroscience and Biogen, author fees from Thieme medical publishers and W. Kohlhammer GmbH medical publishers, non-financial support from Abbvie and compensation for duty as part-time CMO from MODAG, outside the submitted work.
AZ reports speaker fees and research support from Dr. Willmar Schwabe GmbH. He serves as an associate editor for Frontiers in Neurology –Section Neuro-otology and Section Translational Research in Rehabilitation.
