Abstract
Background:
Fatigue is a frequent and troublesome symptom present from the early stages of Parkinson’s disease (PD).
Objective:
To examine the relationship between fatigue and the neuropsychiatric triad, which includes apathy, depression, and anxiety, in de novo PD.
Methods:
We performed a cross-sectional study including 197 patients with de novo PD and assessed fatigue using the Parkinson’s Disease Fatigue Scale (PDFS-16). We evaluated motor status using the Unified Parkinson’s Disease Rating Scale (UPDRS) part III score and evaluated neuropsychiatric status using the Ardouin Scale of Behavior in Parkinson’s Disease (ASBPD). We carried out univariate and multivariate analyses to model association between motor signs, non-motor signs, and fatigue risk.
Results:
Frequency of fatigue (28.9%) was of the same order of magnitude as that of apathy. PD patients with fatigue reported a lower quality of life than patients without fatigue (p < 0.0001). The ASBPD showed that patients with fatigue had higher scores for depressed mood (p < 0.0001), anxiety (p < 0.0001), and apathy (p < 0.0001). In the univariate analysis, fatigue score was positively correlated with apathy, depression, anxiety, and the neuropsychiatric triad as a whole, and to a lesser extent with female sex, hyperemotivity, and the UPDRS part III score. In the multivariate analysis, after adjusting for sex and motor status, the fatigue score remained significantly correlated with apathy (OR = 11.17 [4.33–28.78], p < 0.0001) and depression (OR = 4.28 [1.39–13.12], p = 0.01), but not with anxiety (OR = 0.94 [0.34–2.58], p = 0.9).
Conclusion:
We propose that the neuropsychiatric triad could be expanded to include fatigue.
INTRODUCTION
Fatigue is a common symptom at all stages of Parkinson’s disease (PD) [1]. However, its mechanisms are poorly understood and it is not clear whether it should be considered a motor symptom or a neuropsychiatric symptom belonging to the spectrum of the so-called neuropsychiatric triad, which comprises apathy, depression, and anxiety in PD [2–4]. Fatigue can be considered as having two dimensions: 1) perception of fatigue as reported by patients (subjective dimension), and 2) fatigability, which is defined as an objective change in motor and/or cognitive performance that can be measured and quantified (objective dimension) [5]. Recently, it has been suggested that fatigue in PD should be considered as separate from apathy and other neuropsychiatric signs such as anxiety and depression [6]. However, both fatigue and apathy can precede and accompany motor symptoms in PD, and some clinical and anatomical arguments support a close link between them [7–11]: 1) Lack of motivation, which is a seminal feature of apathy, is a significant predictor of fatigue and is strongly correlated with fatigue in PD [12, 13], 2) Improvement in fatigue and apathy has been reported in PD patients receiving dopaminergic treatment, particularly dopamine agonists with a high affinity for the mesolimbic D3 receptors, but also serotonergic receptor subtypes [3, 14–16], 3) Functional imaging studies of both apathy and fatigue have highlighted the dysfunction of the same brain areas: the ventral striatum, the cingulate cortex, and the supplementary motor area [4, 17–20].
The heterogeneity of some studies in terms of PD population, definition and measurement of fatigue, disease progression, and dopaminergic treatment may have blurred the putative link between fatigue and apathy, as well as between fatigue and anxiety and depression. Additionally, sleepiness is a common symptom in advanced PD and can be considered a confounding factor because many patients complaining of sleepiness use the term “fatigue” to describe this symptom. Similarly, PD patients also often report “fatigue” when talking about wearing off. For these reasons, we focused on patients with de novo PD as a relatively pure model to study whether fatigue might be a symptom related to apathy and, more broadly, to the neuropsychiatric triad of PD.
MATERIALS AND METHODS
Study design
The “Fatigue” de novo PD cross-sectional study was ancillary to the “Honeymoon” study, a French prospective and multicentric phase III trial conducted in patients with de novo PD [21]. The goal of the “Honeymoon” study was to describe neuropsychiatric signs in a de novo PD cohort and to test whether these non-motor signs were responsive to dopamine agonists. The precise methodology of the “Honeymoon” study has been described in detail elsewhere (ClinicalTrials.gov Identifier: NCT02786667) [21]. In the “Fatigue” de novo PD cross-sectional study, we considered only the baseline clinical evaluation and analyzed whether fatigue was a symptom related to apathy, depression, or anxiety, and finally to the neuropsychiatric triad as a whole.
Participants
All 199 patients included in the “Honeymoon” study were eligible for inclusion in the “Fatigue in de novo PD cross-sectional study”. For the “Honeymoon” study, PD patients who had been diagnosed for less than two years and had no significant cognitive impairment (defined as a score on the MATTIS Dementia rating scale > 130/144 [22] or on the Frontal assessment battery (FAB) [23] < 15/18) were included [21]. Patients receiving treatment with MAO-B inhibitors were included if their treatment had remained stable for over three months. Patients with PD undergoing treatment with levodopa or dopamine agonists were excluded.
Standard protocol approvals, registrations, and patient consent
The “Honeymoon” study was approved by the Ethics Committee of Grenoble, authorized by the National Agency for the Safety of Medicines and Health Products (AFSSAPS), and registered as NCT02786667. All patients included in the study gave their written informed consent in accordance with the ethical standards of the Committee on Human Experimentation of the institution and the Helsinki Declaration of 1975.
Clinical assessments
We assessed fatigue using the Parkinson’s Disease Fatigue Scale (PDFS-16) [24]. We used two validated scoring methods for this scale to assess fatigue as a quantitative and qualitative variable [24]. For the quantitative analysis, we used scoring method 1, which consists of scoring each item from 1 “strongly disagree” to 5 “strongly agree” according to a 5-point Likert scale. The total score for method 1 ranges from 16 to 80. For the qualitative analysis, we used scoring method 2, a binary scoring method that consists of scoring 1 for each item for which the patient selected “agree” or “strongly agree” and 0 for any other response. A score of≥8 indicates the presence of significant fatigue. We used the Ardouin Scale of Behavior in Parkinson’s Disease (ASBPD) to assess the whole behavioral spectrum that encompasses the so-called hypodopaminergic and hyperdopaminergic behaviors [25]. The ASBPD is a semi-structured clinical interview in which trained psychologists assess the severity of each hypodopaminergic and hyperdopaminergic item. It consists of 21 items, each of which are rated from 0 (no change in behavior) to 4 (severe change in behavior). We considered items rated≥2 as clinically relevant. In the present study, we focused on the hypodopaminergic items of the ASBPD: apathy, anxiety, depression, irritability, aggressiveness, and hyperemotivity.
The neuropsychological evaluation included assessment of global cognitive efficiency using the MATTIS Dementia Rating Scale (MDRS) [22] as well as assessment of executive function using the frontal assessment battery (FAB) [23].
We used the summary index of the Parkinson’s Disease Questionnaire for quality of life (PDQ-39) [26] to assess the functional impact of fatigue.
The Unified Parkinson’s Disease Rating Scale (UPDRS) parts I to III was used to assess motor and non-motor signs including fatigue and sleepiness.
We calculated UPDRS part III motor subscores and assessed PD motor subtypes as described previously [11, 27].
We also recorded use of psychotropic and chronic medications, including the number of patients using MAO-B inhibitors.
Statistical analysis
We compared the fatigued and non-fatigued groups using the chi-squared test for categorical variables and the Student’s t-test for quantitative variables. We modeled the neuropsychiatric triad, using the three ASBPD hypodopaminergic items: apathy, depression, and anxiety. We used univariate unconditional logistic regression models to model the association between fatigue, motor signs, and non-motor signs. We used multivariate unconditional logistic regression models to model association between fatigue and the neuropsychiatric triad. Variables with a p value < 0.2 in the univariate analysis were included in the multivariate analysis. We adjusted the odds ratios (ORs) and 95% confidence intervals (95% CI) for motor signs (UPDRS III). We performed all analyses using SAS software version 9.4 with a significance level of p≤0.05 (except for the comparison of the frequency of apathy, depression, and anxiety between groups, where the Bonferroni correction was used due to multiple comparisons).
RESULTS
Of the 199 patients enrolled in the “Honeymoon” Study, 197 had completed the PDFS-16 fatigue scale, and these were included in the present “Fatigue” study.
According to the PDFS-16, 57 of these patients (28.9%) had significant fatigue. Patients with fatigue did not differ significantly from patients without fatigue in terms of age, disease duration, or cognitive status (Table 1). Sleepiness as assessed by item eight of the UPDRS part I was very low in our cohort (mean score: 0.50, SD: 0.75) and did not differ between the two groups (p = 0.10).
Patient characteristics
†comparison between de novo PD with and without fatigue. Student’s t test for quantitative variables. Chi-squared test for qualitative variables. UPDRS, Unified Parkinson Disease Rating Scale; MDRS, Mattis Dementia Rating Scale; FAB, Frontal Assessment Battery; PDQ-39, Parkinson Disease Questionnaire 39 items.
Bivariate analysis showed that there was a higher frequency of female patients in the fatigue group compared to the non-fatigue group (p = 0.009). Additionally, the fatigue group was significantly more impaired regarding both motor and non-motor status (Table 1). Patients with de novo PD with fatigue had higher ASBPD scores than patients without fatigue for depressed mood (p < 0.0001), anxiety (p < 0.0001), and apathy (p < 0.0001). Although patients with fatigue were more frequently being treated with antidepressants, MAO-B inhibitors, and anxiolytics than patients without fatigue, the difference between the two groups was not statistically significant. PD patients with fatigue reported a lower quality of life than patients without fatigue, with an average PDQ-39 score twice as high as that of the patients without fatigue (p < 0.0001).
As a next step, we compared the frequency of apathy, anxiety, and depression in PD patients with and without fatigue (Fig. 1). Results were consistent and showed a significantly higher proportion of apathy, anxiety, depression, and the neuropsychiatric triad in the fatigue group compared to the non-fatigue group (p < 0.0001) (Fig. 1). We found that 22.8% of PD patients with fatigue had none of anxiety, apathy, or depression, compared to 77.9% of PD patients without fatigue. The inverse was also true; fatigue was significantly more frequently reported among patients with de novo PD with depression, apathy, and anxiety (p < 0.0001).
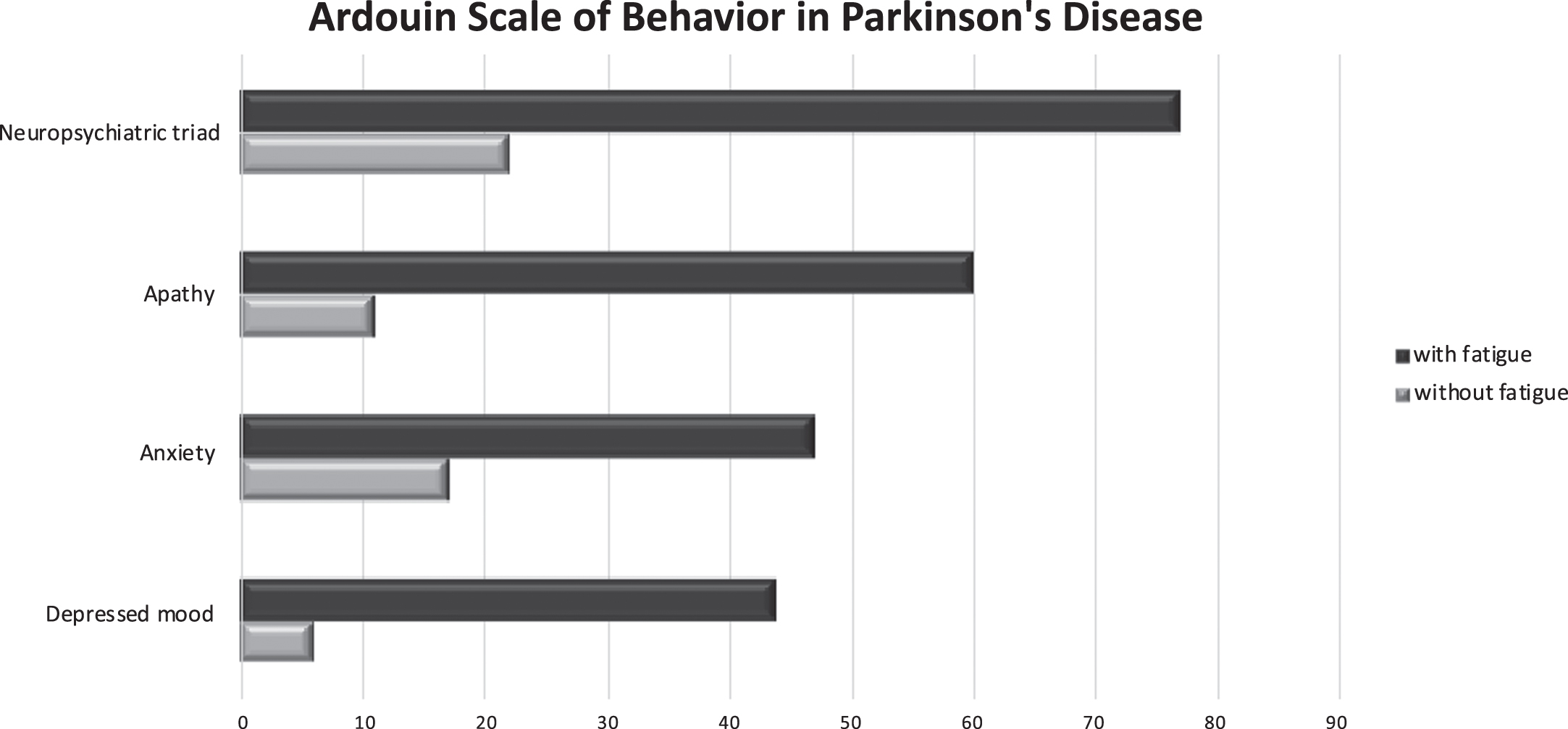
Frequency of apathy, anxiety, depressed mood, and the neuropsychiatric triad in fatigued and non-fatigued de novo PD patients according to Ardouin Scale of Behavior in Parkinson’s Disease (ASBPD). *Level of significance due to Bonferroni correction: p < 0.025.
Lastly, we analyzed the association between fatigue and the whole neuropsychiatric triad, but also between fatigue and each component of the neuropsychiatric triad separately (Table 2). In the univariate analysis, the fatigue score was significantly associated with each component of the neuropsychiatric triad and also with female sex, hyperemotivity, and, to a lesser extent, with the UPDRS part III score. When looking at the multivariate analysis after adjusting for sex and motor status, the fatigue score remained significantly correlated with the whole neuropsychiatric triad (OR = 11.55 [5.33–25.02] p < 0.0001), namely with apathy (OR = 11.17 [4.33–28.78], p < 0.0001) and depression (OR = 4.28 [1.39–13.12], p = 0.01). The correlation with anxiety no longer reached a significant level (OR = 0.94 [0.34–2.58], p = 0.9).
Univariate and multivariate analysis
*univariate analysis. **multivariate analysis.
DISCUSSION
The frequency of fatigue in our study (28.9%) was slightly lower than in published data for drug-naïve de novo PD cohorts, which report percentages from 36% to 55% [1, 29]. However, in most studies, fatigue was assessed using other fatigue scales, which have distinct measurement properties that could partly explain the observed differences [30]. It is interesting to note that a recent study using the Italian version of the PDFS-16 found a frequency of fatigue of 22% at baseline in a de novo PD cohort [11].
Bivariate analysis showed that fatigue was more frequent in female than in male patients (p = 0.009) (Table 1), which is in accordance with some literature but not all [1, 31]. Importantly, in line with previously published data, the average PDQ-39 score was twice as high in the fatigued group (p < 0.0001), positioning fatigue as a key determinant of quality of life in de novo PD [1, 8]. We subsequently demonstrated correlations between fatigue, apathy, depression, anxiety, and quality of life (p < 0.0001). In the univariate analysis, fatigue score was positively correlated with apathy, anxiety, and depression. When looking at the multivariate analysis after adjusting for sex and motor status, the fatigue score remained significantly correlated only with apathy and depression (Table 2). These results raise questions about the links between fatigue and the neuropsychiatric triad of PD.
Fatigue associated with the neuropsychiatric triad
Some clinical and pathophysiological data in the literature corroborate the associations found in the present study. Firstly, a prediagnostic case-control study demonstrated that fatigue arose in patients with PD a few years before motor signs as a prodromal state in parallel with neuropsychiatric disturbances, particularly depression and anxiety [8]. Moreover, other studies in de novo PD have reported that fatigue is correlated with anxiety, depression, and apathy [9, 29]. In recent years, several structural and functional neuroimaging studies have highlighted similarities in the dysfunction of some brain areas and circuits involved in both apathy and fatigue in PD, particularly serotonergic denervation within the ventral striatum and anterior cingulate cortex [4, 19]. Furthermore, two recent neuroimaging studies using resting-state functional MRI in drug-naïve PD patients have highlighted an altered connectivity within the sensorimotor network and outlined the role of the supplemental motor area in PD fatigue, which is interesting since this area is also involved in apathy in PD [18, 33].
Fatigue in association with apathy and depression
Recently, computational modeling approaches to understanding motivated behaviors have given rise to the concept of effort-based decision-making, in which the cost-benefit evaluation underpins the willingness to engage and sustain goal-directed behaviors [33, 34]. According to the effort-based decision-making model, apathy may arise in the case of hypersensitivity to effort and/or blunted sensitivity to rewarding outcomes [33, 34]. This model is highly consistent with the theoretical model of fatigue, which postulates that an imbalance between motivational input and level of perceived exertion reduces the work output [35, 36]. Interestingly, negative affect, which characterizes depression, may also alter the cost/benefit evaluation and thereby reduce motivational input or increase perceived exertion. Decrease in emotional resonance (i.e., reward deficiency syndrome) and emotional distress, which are prominent features of apathy in de novo PD, may therefore partially account for the simultaneous occurrence of apathy, depression, and fatigue in de novo PD [32]. Intriguingly, correlation between fatigue and anxiety did not persist in the multivariate model when controlling for sex and motor status. This raises the possibility of a common denominator that links apathy, depression, and fatigue, but not anxiety and fatigue.
Fatigue and anxiety
Anxiety is characterized by exaggerated or prolonged feelings of nervousness or alertness. This behavioral sign frequently accompanies depression and apathy in PD [37]. Although it is underpinned by dysfunction within limbic cortico-basal circuits, anxiety also involves dysfunction within the fear circuit, particularly the amygdala [37]. Thus, to some extent, anxiety distinguishes itself from depression and apathy. Anxiety does not therefore appear to be a determining factor for fatigue and may instead be considered a comorbidity.
Anhedonia, the missing link?
There is a broad overlap between anhedonia, apathy, and depression [34, 39]. Although apathy is associated with anticipatory anhedonia, which participates in a diminished emotional resonance that leads to a failure to “energize” and trigger a specific action, depression might be associated with consummatory anhedonia, an inability to experience pleasure that is related to decreased hedonic perception of reward [34, 38–42]. Interestingly, anticipatory and consummatory anhedonia can each lead to an imbalance between motivational input and perceived exertion, resulting from perception of fatigue in accordance with the model of fatigue described above [35]. Conversely, fatigue could reflect a broader neuropsychiatric clinical spectrum that encompasses a set of amotivated behaviors, namely anhedonia, apathy, and depression. Thus, anhedonia could be the missing link connecting apathy, depression, and fatigue. In line with this, a previous study in patients with de novo PD showed that apathy was strongly correlated with fatigue and anhedonia, and that presence of fatigue was significantly associated with cognitive and behavioral dimensions of apathy [13].
Fatigue as an independent manifestation
Although our results support the hypothesis of a close link between fatigue and the neuropsychiatric triad, 22.8% of patients with de novo PD with fatigue in our cohort experienced neither apathy nor depression. Conversely, 22.1% of PD patients without fatigue experienced at least one of anxiety, apathy, and depression. Various hypotheses may be considered in order to explain the incomplete overlap between fatigue and the neuropsychiatric triad.
Firstly, fatigue can be considered multifactorial and related to numerous other non-motor signs including sleepiness, pain, dysautonomia, and other comorbidities [43–47].
More deeply, fatigue could be seen, at least in some patients, as an independent symptom related to another pathophysiological dysfunction. In particular, previous imaging studies have emphasized decreased connectivity within the salience network and increased connectivity within the default mode network in PD patients with fatigue [18, 48]. The salience network has a key role in the detection of behaviorally relevant stimuli including interoceptive information processing [49]. Interestingly, in a recent model of exercise-induced central fatigue, McMorris and coworkers highlighted the crucial interplay between interoception and motivation in perception of effort and fatigue [50]. One might speculate that disruption within the salience network alters the cognitive and motivational attributes of relevant stimuli, thereby contributing to apathy, depression, and biased perception of effort allocation and perceived exertion, resulting in fatigue. Fatigue could therefore reflect a broader pathophysiological and clinical spectrum ranging from interoceptive dysfunction to motivated behavior disorders, thus expanding the neuropsychiatric triad of PD.
Lastly, we need to consider the link between fatigue and motor and cognitive cortico-basal ganglia circuits. Bivariate analysis showed that de novo PD patients with fatigue had higher UPDRS part III (p = 0.02) and bradykinesia (p = 0.014) motor scores than the patients without fatigue (Table 1). Thus, we cannot rule out a more severe disease progression or higher motor fatigability and comorbidities in the fatigued group [46]. However, motor status did not appear to be the main driver of fatigue in our cohort, as fatigue—when adjusted for UPDRS-III score—remained highly correlated with apathy and depression (Table 2). In the same way, cognitive status was not correlated with fatigue score (Table 1).
Limitations
Our study has several limitations. The “Honeymoon” study was not designed specifically to study fatigue and its interactions with the whole spectrum of motivated behaviors in de novo PD. Furthermore, we did not assess sleepiness, pain, or dysautonomia with dedicated scales, nor did we consider comorbidities or physical activity levels, which may all be confounding or causative factors for fatigue even in the early stages of PD [11, 51]. Larger prospective and longitudinal studies examining the entire spectrum of motivated behaviors and all confounding factors are needed in order to better characterize fatigue and its pathophysiology.
In conclusion, we found that apathy and depression were significantly correlated with fatigue regardless of motor and cognitive status and propose that fatigue could be included in the neuropsychiatric triad of PD. Complaints of fatigue in de novo PD therefore require careful consideration of the presence of apathy and/or depression in order to manage patients accordingly.
Footnotes
ACKNOWLEDGMENTS
We thank all participants for taking part in this study. We thank Jennifer Dobson for proofreading this manuscript.
This study was ancillary to the “Honeymoon” study, for which UCB provided financial support. The sponsor had no role in the study design, data analysis and interpretation, or in writing the manuscript.
CONFLICT OF INTEREST
Dr. Béreau reports reimbursement of travel expenses to scientific meetings from ELIVIE, Medtronic and Boston Scientific, honoraria from Merz Pharma and Allergan for lecturing, outside the submitted work
Dr. Castrioto reports research grants from France Parkinson Foundation and Medtronic, reimbursement of travel expenses to scientific meetings from ELIVIE, Medtronic and Boston Scientific, honoraria from Novartis for lecturing, outside the submitted work.
Dr. Fraix reports reimbursement of travel expenses from Merz, AbbVie, honoraria for scientific counselling from AbbVie.
Dr. Azulay reports honoraria for consultancies and advisory boards from Abbvie, Merz, Allergan, Medtronic, Ever Pharma.
Dr. Moro reports honorarium from Medtronic and Newronica for lecturing, and an educational grant from Boston.
Dr. Tranchant reports reimbursement of travel expenses from Merz, and honoraria from Allergan for lecturing.
Dr. Anheim reports travel grants and/or honoraria from Actelion Pharmaceuticals, Johnson and Johnson, Teva, LVL, Aguettant, Orkyn, AbbVie and Merz.
Dr. Thobois reports grants from France Parkinson, FRM, ANR, PHRC, UCB, Neurodis, personal fees from Aguettant, Boston Scientific, Novartis, non-financial support from Zambon, Abbvie, Elivie outside the submitted work.
Ms. Lhommée, Ms Klinger, Ms Bichon, Ms Schmitt, Dr. Durif, Ms Longato, Ms Philips, Mr. Pelissier, Dr. Broussolle report no relevant disclosure related to this study.
Dr. Krack reports a research grant from UCB for the present study, research grants from Swiss National Science Foundation, ROGER DE SPOELBERCH Foundation, Bertarelli Foundation, Annemarie Opprecht Foundation, Parkinson Schweiz, Boston Scientific, and Aleva, lecturing fees paid to employing institution (Inselspital) from Boston Scientific, as well as reimbursement of travelling expenses to scientific meeting by Zambon, outside the submitted work.
