Abstract
Although the need for better medications for the treatment of psychiatric symptoms in people with Parkinson's disease (PWP) is not disputed, the approach and targets for these medications needs further attention. Psychiatric symptoms occur at higher prevalence in PWP—many start in the prodromal phase of the disease—and have complex associations and interactions with the motor symptoms and their treatments, begging the question of whether they may be mechanistically connected. In this manuscript, we review the current evidence for pharmacologic treatments of psychiatric symptoms in PWP and explore the potential next steps needed to develop new medications for psychiatric symptoms in PD.
Plain Language Summary
People living with Parkinson's disease (PWP) frequently experience psychiatric symptoms. The most common psychiatric symptoms in PWP are anxiety, apathy, depression, impulse control disorders, and psychosis. Psychiatric symptoms worsen quality of life and cause distress in PWP. Current treatments for psychiatric symptoms in PWP are not consistently effective, may have greater risks in PD, and some have not been specifically studied in PWP. This manuscript reviews the existing medication treatments for psychiatric symptoms in PWP identifying limitations and then suggests how to develop more effective treatments.
Introduction
Psychiatric symptoms such as anxiety, depression, impulse control disorder (ICD), psychosis, and apathy are highly prevalent among people with Parkinson's disease (PWP), yet they manifest in ways that may be distinct from similar symptoms in individuals without Parkinson's disease (PD). These differences, although well-recognized, remain underexplored and poorly understood, partly due to the complex neuropathological substrate of PD, including progressive neurodegeneration, Lewy body spread, dysautonomia, and dopaminergic depletion. The diagnosis and treatment of psychiatric symptoms in PD are further complicated by the influence of dopaminergic replacement therapies, which vary in delivery from continuous to pulsatile patterns and can themselves exacerbate or mask neuropsychiatric symptoms. Exploring the specific mechanisms underlying psychiatric symptoms in PD has led to attempts at their treatment targeting the hypothesized neurotransmitter systems or brain networks, but in some conditions like anxiety and depression, traditional antidepressants may not have the same efficacy or the disease heterogeneity in PD may require an approach that is tailored to mechanism at the patient level. In other conditions like apathy, ICD, or psychosis, treatment options with robust evidence are still lacking and more foundational research is needed to uncover novel mechanisms that can be targeted therapeutically. In this manuscript, we aimed to review the current level of evidence for treatment of these syndromes in PD, then to discuss the required next steps in order to develop new therapies targeting these symptoms (Table 1).
Current evidence for medications used to treat psychiatric symptoms in Parkinson's disease and suggested future directions for new treatments.

RCT: randomized controlled trial; SSRI: selective serotonin reuptake inhibitor.
Anxiety
The prevalence of anxiety in PD is three times higher than in non-PD populations of the same age. 1 Anxiety occurs in at least one-third of all people with PD (PWP) and is one of the main determinants of quality of life.2,3 International foundations focused on improving care for people with PD, such as the Parkinson Foundation, Parkinson's UK, and World Parkinson Congress, have consistently ranked anxiety as one of the top three unmet needs in the management of PD.4,5 Anxiety exacerbates motor symptoms, increases caregiver burden, and often increases social isolation because patients fear having a panic attack in public or have a social phobia. 6
In addition to occurring at increased prevalence in PWP many Diagnotic and Statistical Manual 5 (DSM 5) anxiety disorders have different epidemiology with later age of onset for anxiety disorders than is typical in non-PD populations and age of onset of PD is younger for PWP with anxiety compared to those without anxiety.1,2,5 Atypical anxiety syndromes which might be unique to PD are also highly prevalent and among the most difficult to diagnose and treat. 7 Anxiety associated with fluctuations in dopamine medication levels, or on-off state anxiety fluctuations is reported in up to 34% of PWP. A study of anxiety associated with fluctuations found that about one third of PWP experience a 3-fold increase in anxiety during off-periods. 8 Interestingly, in this sample of 200 PWP fluctuation associated anxiety occurred more in men while most other anxiety disorders are higher in women. Atypical anxiety may also occur in association with autonomic dysfunction or certain types of cognitive impairment. 7 A fear of falling is estimated to occur in over half of PWP often resulting in withdrawal from or avoidance of usual interests and social activities. 7
Currently, there are no adequately powered RCTs with anxiety as the primary outcome to inform evidence based practice in PD. 5 RCTs for depression in PD show that several classes of antidepressant are efficacious for the treatment of depression, however, anxiety as a secondary outcome was not improved.9,10 A small trial of buspirone for anxiety in PWP found poor tolerability and a high rate adverse events affecting motor function. 11 As a result of the lack of RCTs for anxiety in PWP, a typical approach to treating anxiety is to follow the treatment algorithm used for the geriatric population which relies on antidepressants augmented by benzodiazepines. Benzodiazepines are often used in PWP and may be favored by providers due to faster onset of benefit compared to the 4–8 weeks required for antidepressants and for the potential to mask anxiety associated with akathisia or autonomic symptoms. Unfortunately, efficacy for the application of benzodiazepines in PD is unknown, but the increased risk of adverse events from benzodiazepine use in PWP is well documented such that PWP should follow the guidelines used for geriatric populations. 12 In addition to the lack of RCTs to guide evidence-based treatment of anxiety in PWP, the treatment approach is complicated by the fact that some manifestations of anxiety are specific to PD or are triggered by conditions that are uniquely associated with the treatment of PD. One such condition is on-off dopamine fluctuation associated anxiety described above which has panic-like episodes of anxiety occurring with oscillations in dopamine levels however there are no proven treatments for this type of anxiety. 8
Apathy
Apathy is a disabling non-motor symptom in PWP 13 and may impact protective behaviors like participation in physical exercise, potentially increasing the risk of death by nearly 3-fold. 14 Thus, the need for new therapeutics is warranted. Apathy is often treated as depression even though nearly half of PWP with apathy do not have concurrent depression. The complicated neuropsychiatric landscape of what is currently considered “Parkinson's disease” lends itself to multiple potential mechanisms that align with theoretical constructs of apathy. For instance, apathy in PwP has often been attributed to either executive and attentional dysfunction (“cognitive apathy”) or dopaminergically-responsive apathy associated with greater mesolimbic dopaminergic denervation (“motivational apathy”), 15 which aligns with a classic distillation of motivated behavior into affective/emotional content attribution, behavioral activation, and cognitive components. 16 As such, attempts to treat apathy in PWP have largely been based on “cognitive” or “motivational” mechanistic constructs by repurposing treatments used to treat cognitive or mood syndromes. Herein, we first summarize the existing state of evidence for pharmacotherapies for apathy by classes that target specific neurotransmitter systems or cognitive behavioral processes.
Dopaminergic approach
The evolving view on the core neurobiological underpinnings of apathy in PWP has somewhat limited the development of therapies targeting specific neurotransmitter systems or receptors. For instance, while cross sectional studies have shown associations between apathy and both lower17,18 or higher19–21 levodopa equivalent daily dose (LEDD), a recent longitudinal study in people with no baseline apathy showed that only lower cognition, more depression, and increasing motor severity predicted in apathy onset in PWP, notably excluding LEDD as a predictive factor of subsequent apathy. 13 On the other hand, striatal presynaptic dopaminergic terminal imaging was lower in apathetic individuals with early stage PD, 22 and in PWP treated with subthalamic nucleus deep brain stimulation (STN-DBS), acute stimulation of associative or limbic subregions of the STN can drive goal-oriented behavior 23 but the net result of the concurrent LEDD reduction following successful STN-DBS reduces impulse control disorder prevalence 24 and may increase apathy. 25
Given this apparent relationship between striatal dopaminergic tone and disorders of motivation, dopamine agonism has been explored as a treatment for apathy with some studies suggesting it may be more effective than brain stimulation, exercise-based treatments, or supplements. 26 With the various methods of enhancing dopaminergic tone in mind, a cross-sectional comparison of neuropsychiatric symptoms between people with PD taking pramipexole, ropinirole, or levodopa found that those on pramipexole had lower Neuro Psychiatric Inventory apathy subscores than those on levodopa (1.01 ± 1.7 vs. 1.87 ± 2.93; p = 0.02) and a nearly significant difference between pramipexole and ropinirole favoring the former (1.01 ± 1.7 vs. 1.54 ± 2.3; p = 0.06). 26 Of prospective trials using dopamine agonists (DA) to treat apathy in PD, some showed a benefit (rotigotine 16 mg daily, 27 piribedil 28 ) whereas others have shown trends toward improvement but failed to reach statistical significance,29,30 often in the setting of large placebo effects on apathy. Rasagaline, a type B monoamine oxidase inhibitor that primarily impacts oxidation of hydroxylated amines and dopamine rather than noradrenaline and 5-HT, did not impact apathy as a secondary outcome measure in a randomized study targeting depression in people with PD. 31 A secondary analysis of the ADAGIO trial, which targeted motor symptoms of PD with rasagiline, showed a non-statistically significant trend toward less apathy with rasagiline 32 but participants were also taking an antidepressant. In sum, correcting a dopaminergic deficit is typically seen as the standard approach in treating apathy but there may be conflicting evidence supporting this approach and it may be complicated by motor- and non-motor side effects of D2/D3 agonism.
Cholinergic approach
The association between cognitive decline in PWP and cholinergic denervation 33 of the cortex that parallels nucleus basalis of Meynert (or cholinergic nucleus 4, Ch4) atrophy has led to hope that enhancing cholinergic tone might allow improvement in the “cognitive component” of apathy as it does with cognitive dysfunction. 34 The recent finding that Ch4 grey matter density was associated with apathy in PWP when adjusting for age, LED, and disease severity 35 may support this approach. One randomized, controlled trial tested the efficacy of rivastigmine in depression- and dementia-free PWP and showed an adjusted effect size of −0.9 (p = 0.031) favoring rivastigmine for reduction of the Lille Apathy Rating Scale (LARS). 36 Apathy worsened in some rivastigmine-treated participants in the open-label, long term phase of the study, but this was attributed to conversion to dementia in 50% of those receiving rivastigmine for 12 months. Notably, though there was concern about prior reports of worsening rest tremor with cholinesterase inhibition, it did not occur in this study. Following the hypothesis targeting the “cognitive component” of apathy, the intellectual curiosity and action-initiation LARS subscores were more responsive to rivastigmine than the emotion/affective subscores. However, in a randomized, crossover clinical trial of rivastigmine for mild cognitive impairment, there was no effect by treatment group of rivastigmine exposure on the Apathy Scale after 24 weeks of treatment, 37 where one would hypothesize that a more cognitively impaired group would stand to find greater benefit from cholinergic therapy. Overall, evidence from at least one well-controlled trial supports boosting cholinergic tone to treat apathy in PWP, but given the high prevalence of GI and autonomic issues in these patients and the non-specific receptor targeting of current cholinergic therapy, tolerability may limit the application of this approach.
Antidepressant approaches
While it may often be confused with or treated as depression due to the vegetative symptom overlap, apathy is distinct from depression. Selective serotonin reuptake inhibitors (SSRIs) or selective serotonin and noradrenergic reuptake inhibitors (SNRIs) are often attempted with apathy but have little supporting evidence. One open-label study randomized PWP to either an SNRI or SSRI (no placebo arm) and showed no statistically significant change from baseline in apathy. 38 In fact, a new observational study recently reported that SSRI, but not SNRI, use was associated with higher levels of apathy. 39 Interestingly, an open-label study of vortioxetine, a SERT inhibitor that also affects multiple HT receptors and alters other neurotransmitter levels, showed improvement in apathy after 12 weeks. 40
Other pharmacological approaches in apathy
Motivated by the adrenergic deficits found in PD, one group tried atomoxetine, a selective norepinephrine reuptake inhibitor indicated for use in attention deficit hyperactivity disorder, but only found a non-statistically significant trend toward improvement in apathy as a secondary outcome measure (difference in Apathy Scale change between groups at 8 weeks = −1.80 ± 1.22, p = 0.14). 41 Interestingly, a wash-out study of amantadine, which is typically repurposed to treat levodopa-induced dyskinesias, suggested worsening apathy after amantadine withdrawal, 42 which might indicate clinical effectiveness. A study on memantine, which works by blocking the flow of current through NMDA receptors, did not show improvement in apathy in PWP. 43 In Alzheimer's disease (AD), the noradrenergic and dopaminergic stimulant methylphenidate was found to improve apathy in a small clinical trial, and this response was predicted by acute cognitive enhancement with a dextroamphetamine challenge, 44 but stimulants have not been systematically studied for apathy in PWP.
Depression
Perhaps no other psychiatric symptom is so pervasively associated with PD than depression. Depression early in life increases the risk of PD by 2-fold, depression during the course of PD lowers quality of life, increases disability, and elevates the risk of suicide.45–47 An interdependent relationship between depression and PD is suggested by the finding that longer duration of PD or poorly treated motor symptoms predicts the failure of depression to remit with treatment. 48 Similarly antidepressant treatment may impact the course of PD treatment such that in early untreated PD (after controlling for depression) subjects taking tricyclic antidepressants had later initiation of dopaminergic therapy compared to those not taking antidepressants. 49 A challenge to addressing depression in PD is the frequent cooccurrence of apathy. It is possible that apathy is a within person mediator of depressive symptoms and that assessing treatment response in depression trials could be confounded by the presence of cooccurring apathy.50,51 In addition to these challenges, the evidence base for treatment of depression in PD with medications informed by clinical trials is modest and without clear distinction of comparative efficacy between antidepressant types.
The multi-center 12-week RCT Study of Antidepressants for Depression in PD (SAD-PD) demonstrated that paroxetine and venlafaxine were both more effective than placebo for treating depression in PD however the number needed to treat (NNT) for paroxetine was 13 and 24 for venlafaxine. 9 For context consider that in non-PD populations SSRIs (like paroxetine) have a NNT of 7 and that in general a “good” NNT is considered to be <10. 52 Further mudding the water on the efficacy of SSRIs in PD, an 8-week RCT of paroxetine CR, nortriptyline, and placebo found that while nortriptyline was superior to placebo for depression paroxetine CR was not. 10 Paroxetine CR is a controlled release form of the paroxetine used in the SAD-PD trial cited above. The difference in demonstrated efficacy between alternate forms of paraoxetine is concerning even though it is possible that 8-weeks was too short for the controlled release form of paroxetine (although regular paroxetine separated from placebo in SAD-PD by week 4) and the sample size of 52 was too small given that the effect size for paroxetine CR was 0.51.
New and atypical antidepressant approaches have been tried for depression in PWP however they have produced mixed or only preliminary results. A 12-week open label trial in n = 30 PWP of vortioxetine, a serotonin agonist and reuptake inhibitor (SARI), found signal for efficacy with a 52.7% reduction in depression symptoms from baseline to 12-weeks which suggests further study in an RCT is merited. 40 In addition to ‘traditional’ antidepressants, pramipexole, a dopamine agonist, was found to have antidepressant efficacy compared to placebo when used in PWP however the effect was small improving the Beck Depression Inventory by only 1.9 points. 53 Although it is tempting to consider antidepressant response to pramipexole in PWP as evidence that depression in this context is mechanistically linked to PD, there is evidence that pramipexole has mood elevating properties in non-PD populations as well. 54 Rasagiline, a monoamine oxidase inhibitor (MAOI), used to treat the motor symptoms of PD also failed to show efficacy for depression in PWP. 31 In summary, clinical trial evidence for the treatment of depression in PWP is limited for monotherapy approaches, there are no RCTs on comparative efficacy between antidepressant classes or for treatment resistant approaches such as combination therapy with two antidepressants or other augmentation strategies after monotherapy trials fail.
Impulse control disorders
Impulse control disorders (ICDs) in the context of PD refers specifically to excessive goal-directed behavior, such as excessive gambling, sexual behavior, eating, or spending, in the setting of dopamine replacement and is a subset of behaviors that cause disability or affect quality of life under the broader umbrella of “impulse control behaviors”, which includes related manifestations like punding (originally described in methamphetamine addicts) or dopamine dysregulation syndrome. Given that there is a correlation between the incidence of ICDs and exposure and dose of dopamine replacement, specifically dopamine agonists with higher affinity for D2 and D3 receptors, the mechanism is proposed to involve dopaminergic tone in at least two ways: (1) pulsatile inhibition of the “indirect” basal-ganglia-thalamocortical parallel loops that involve decision-making networks, 55 which seems more common in impulse control behaviors like punding; (2) Off-target effects of dopamine replacement on the D3 receptors in the mesolimbic system, especially since the ventral tegmental area is relatively intact in PD and still provides dopaminergic innervation to the ventral striatum. 56 The latter has supported the idea of what has been shown experimentally: increased positive feedback learning and impaired negative feedback learning 57 related to enhanced and more continuous dopaminergic tone throughout the reward/learning circuitry. Thus, treatment of ICDs through taper of dopamine replacement therapy seems straightforward, but a conundrum arises when this precipitates other disabling motor and non-motor symptoms of PD, raising the need for alternative treatments to control ICDs like how amantadine may be used for levodopa-induced dyskinesia when levodopa cannot be tapered.
Dopaminergic approach
In obsessive-compulsive disorder, which is considered more of an anxiety-related, avoidance behavior than an enhancement of goal-oriented behavior as in PWP manifesting ICDs, dopaminergic blockade can be therapeutic but that is not an option in PWP due to the effect on motor and autonomic function. However, longitudinal studies of PWP support the idea that ICDs can resolve with reduction of dopaminergic therapy with a specific focus on dopamine agonists, 58 given their stronger dose-effect relationship. 59 While dopaminergic therapy reduction is not always possible, changing the timing or receptor specificity of dopamine replacement may be helpful. Interestingly, switching of pulsatile, oral levodopa to continuous intestinal levodopa gel led to reduction in ICDs in PWP 60 regardless of DA use status. Similarly, long-acting DAs such as rotigotine and extended release pramipexole might be associated with a lower incidence of ICDs than short-acting DAs, 61 but this has not been tested in prospective trials. A reduction of LEDD and/or wean from DAs can often be achieved with bilateral STN DBS, facilitating a reduction in appetitive behavior in one randomized, controlled trial of DBS. 62 An observational study comparing the course of ICDs in PWP undergoing DBS surgery or not showed a reduction in ICD prevalence with DBS that was mediated by reduction in total LEDD. 63 New DAs or partial agonists with D1/D5 receptor specificity, 64 avoiding D3 agonism that might drive ICDs behaviors, are in development but no outcomes on ICDs are available in publication.
Non-dopaminergic approaches
Few clinical trials have been performed specifically targeting ICDs in PD. Naltrexone, a competitive opioid receptor antagonist, was tested in a randomized, double-blind, placebo-controlled trial in 50 PWP with ICDs and did not meet its primary endpoint of the change in Clinical Global Impression-Change response, yet showed a statistically significant greater decrease in the Questionnaire for Impulsive-Compulsive Disorders in Parkinson's Disease-Rating Scale (QUIP-RS) over 8 weeks. 65 Given the association with excessive D2 activation and hypersensitization of the striatum discussed above, ICDs might be seen as a parallel to levodopa-induced dyskinesia. 66 Thus, amantadine, a mixed mechanism drug with NMDA receptor antagonism, was trialed in a small cross-over study specifically targeting pathologic gambling in PWP. Amantadine reduced gambling and the Yale-Brown Obsessive Compulsive Scale 67 in PWP with persistent gambling despite switch from DA to levodopa and gambling counseling. However, we don’t have direct evidence of its effect on other classic ICDs nor on the overall ICD severity. Further, amantadine is not tolerated in all PWP due to its potential to unmask psychosis and worsen autonomic and cognitive function.
It is known that high doses of clonidine can engage postsynaptic receptors and enhance prefrontal adrenergic cortical activity. Based on this theory and evidence of impulsivity reduction in heroin addicts with its use, clonidine, an α2-adrenergic receptor agonist, was used in a randomized, double-blind, controlled trial in PWP with ICDs. There was a non-significant trend toward a larger proportion of participants with a reduction in QUIP-RS of 3 points or more (42% vs. 35% in clonidine 75 mg BID vs. placebo). 68 While that primary endpoint was negative, the clonidine group experienced a larger QUIP-RS reduction at 8 weeks of treatment (11.0 vs. 3.6 points) and had a higher Parkinson's Disease Questionnaire-39, a PD-specific quality of life scale. Surprisingly, orthostatic hypotension and daytime sleepiness were no more common with clonidine than placebo. Another study looked at adrenergic tone enhancement with atomoxetine 40 mg daily in a double-bind, placebo-controlled, crossover trial and showed reduced reflection impulsivity and risk taking in gambling and improved stopping accuracy on the Stop Signal Task while enhancing sustained attention, though none of the participants had ICDs at baseline. 69 In summary, a few agents have shown promise in affecting behavioral outcomes associated with ICDs in PWP but only naltrexone showed a statistically significant improvement in a secondary outcome measure in an RCT with a small N and has yet to be reproduced.
Psychosis
Hallucinations and delusions, collectively called psychosis, occur in ∼60% of PWP at some point in the disease course. 70 Although psychosis is often associated with advanced disease, in some cases it occurs early and may be a sentinel symptom of worsening prognosis. In a cohort of incident PD followed for seven years, hallucinations were the first in a series of symptoms including recurrent falls, dementia, and nursing home placement that were associated with an increased relative risk of death of 2.7 when two or more occurred during follow up. 71 In addition, psychosis is associated with the highest costs, an increase in caregiver strain, more frequent and prolonged hospitalizations, and is the strongest predictor of nursing home placement in PD.71–74
An important question to be answered to inform the treatment of psychosis in PD is whether the mechanism is similar enough to that of other “primary” psychotic disorders to be relevant. At first pass the phenomenology of psychosis in PD appears to be different than that of schizophrenia in that visual hallucinations, rather than auditory, are the most common perceptual disturbance. 75 Llorca et al. found auditory hallucinations were experienced by 83% of patients with schizophrenia compared to 42% PWP, and visual hallucinations were reported by 88% of PWP while only 55% of schizophrenics had them. Another difference is that negative symptoms, apathy, avolition, blunted affect, impaired cognitive function, and social withdrawal which considered an integral part of schizophrenia, are not considered as part of PD psychosis syndrome and often occur in the absence of psychosis in PD. In schizophrenia it is hypothesized that negative symptoms are due to reduced dopamine in the mesocortical pathway, while the positive symptoms of hallucinations and delusions are thought to be due to excessive dopamine in the mesolimbic pathway. While a similar mechanism may be at work in PD psychosis, these “negative symptoms” in PD are attributed to the overall neurodegenerative process and potentially other neurotransmitters such as acetylcholine.17,22,33 The role of dopamine replacement in PD psychosis remains controversial, because levodopa infusion does not induce hallucinations, levodopa dose is not consistently associated with risk in prospective studies, and psychosis occurs often in unmedicated patients. 76 Therefore the role of dopamine medication reduction as a treatment for psychosis requires more study.
The so called “minor phenomena”, illusions, passage hallucinations, and sense of presence are highly prevalent in PD and are often considered “benign” although they are associated with a higher risk of fully formed hallucinations and more advanced psychotic symptoms. 77 In 5950 PWP the prevalence of attenuated psychotic symptoms was 43.1% and they were independently associated with incident psychosis (HR 3.0, 95% CI 2.4–3.9, p < 0.001). Despite conveying increased risk of psychosis minor phenomena are usually not treated because relative to the distress they cause the risk benefit of treating them is sometimes unfavorable. 78 Further, while there is evidence that early treatment of hallucinations and delusions with antipsychotics in schizophrenia may mitigate progression, and treatment of advanced psychosis in PD may decrease mortality and nursing home placement, there is little to no evidence supporting the role of early treatment when only minor phenomena are present in PD. 79
Historically, the biggest challenge to treating psychosis in PD has been that most antipsychotics block dopamine type 2 receptors which leads to a significant worsening of motor symptoms. In addition to worsening motor function, an increased risk of mortality is associated with the use of both typical and atypical antipsychotics in PWP. 80 The current best evidence medications for treating psychosis in PD are those that have little action at dopamine receptors, clozapine and quetiapine, or a newer medication, pimavanserin, that has no action at dopamine receptors. 81 Quetiapine is the most frequently used despite having equivocal evidence of efficacy.82–84 The efficacy of low-dose clozapine is supported by two RCTs, however clozapine is less often prescribed due to required blood monitoring and greater rate of adverse effects.85,86 Pimavanserin, a selective serotonin 2A receptor inverse agonist that does not block dopamine receptors, has FDA-approval for the treatment of psychosis in PWP based on evidence from a multi-center RCT, but may need 4 weeks to have benefit and is difficult to obtain outside of the USA. 87 Although only supported by secondary outcome data from RCTs focusing on demented PD populations and meta-analyses, cholinesterase inhibitors, e.g., rivastigmine, galantamine, donepezil, appear to decrease the frequency and severity of hallucinations suggesting that cholinergic mechanisms may also play a role in psychosis in PD. 88
Future directions: The need for more therapeutics
There is consensus among movement disorders specialists that there is a need for more treatments for neuropsychiatric symptoms in PWP, but the needed next steps vary by the state of existing therapies in the non-PD population and the specificity of the clinical syndrome to PWP (Figure 1). For instance, disorders that have evidence-based treatments in the non-PD population, like anxiety, should have effective treatments repurposed into PD populations for cases where dopaminergic tone optimization is not effective. In other conditions more specific to PD or neurodegenerative conditions, like apathy, we may need more foundational research to deconstruct the multiple mechanisms at play while developing novel agents that target said mechanisms. Herein, we discuss specific issues related to future development of pharmacotherapies for each of the clinical syndromes where existing evidence is summarized above:
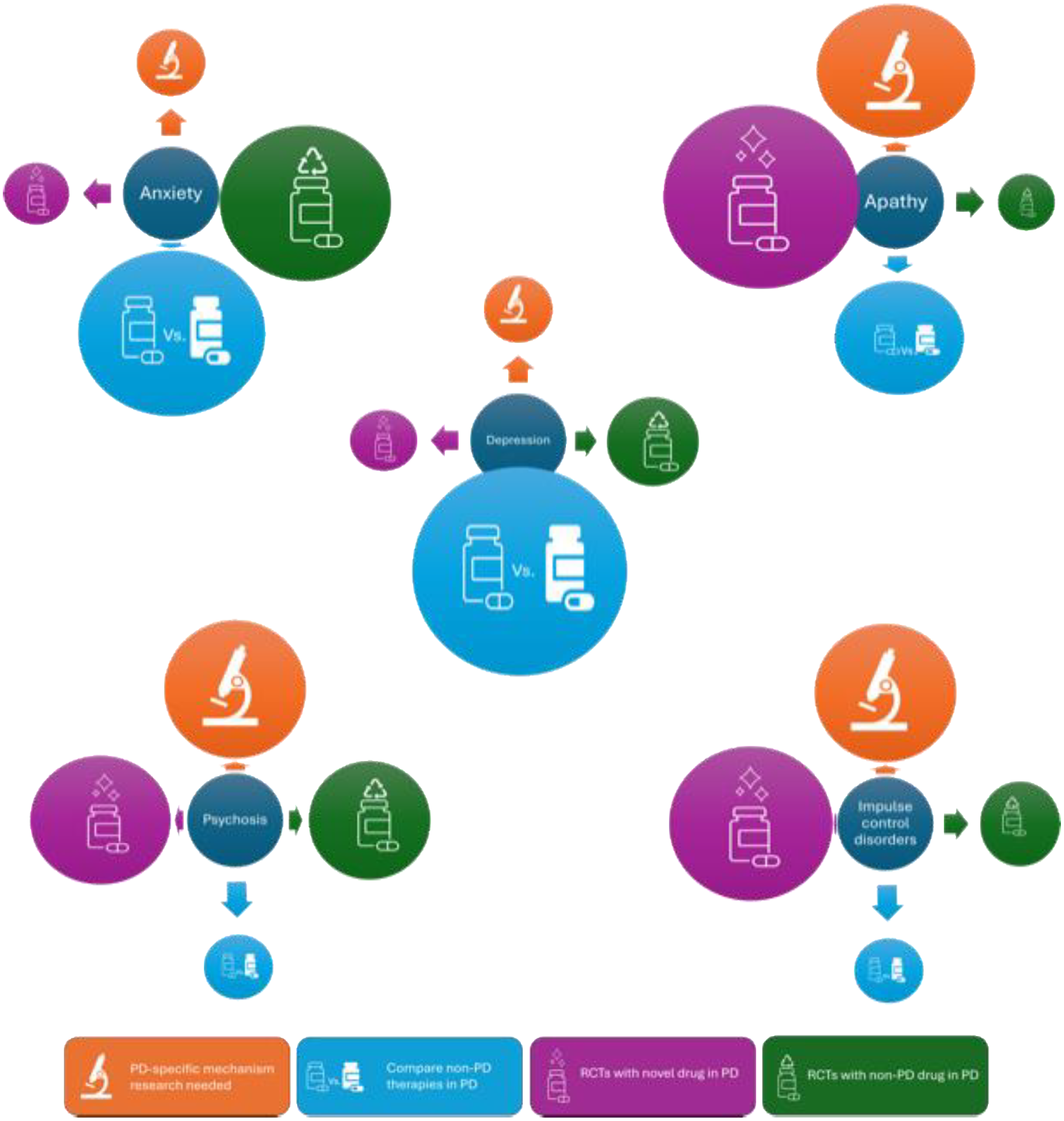
Approach for developing new medication treatments for psychiatric symptoms in Parkinson's. Size of circles represent proposed emphasis needed on the corresponding stage of research toward new therapies in each psychiatric syndrome in PD.
Future directions for anxiety
To address the need for evidence-based treatments for anxiety in PD a reasonable first step would be to conduct a RCT using the best-evidence medication for anxiety in the general population, e.g., SSRI. One advantage of this approach is that some portion of anxiety disorders occur before the onset of PD and may be “idiopathic anxiety disorders” unrelated to the neurodegenerative process and would be expected to respond to conventional therapies. Another advantage is that non-responders or partial responders from this RCT would provide information that could inform studies to determine novel mechanisms of anxiety in PD. For example, if failure to respond was associated with the presence of on-off dopamine fluctuations that might suggest a dopaminergic intervention was needed rather than an SSRI. While mechanistic studies of anxiety in PD should go forward regardless, a strategy that starts with a RCT of a medication known to be efficacious for anxiety in non-PD populations provides hope for at least a subset of patients to find relief from suffering and may provide information that informs future studies of novel mechanisms of anxiety in PD.
Future directions for apathy
Given the degree of disability caused by apathy in PWP and the lack of robustly effective therapies, there is substantial room for further therapeutic development in this space. One challenge of repurposing existing medications to address apathy in PWP is the dose-limiting side effects encountered with these agents. While there might be some evidence for boosting the overall cholinergic tone with cholinesterase inhibition, PWP can often be bothered by off-target effects such as hypersalivation, GI upset, and nausea and skin reactions to transdermal formulations are common. New pharmacotherapies targeting specific cholinergic receptor subtypes that play a role in apathy might avoid some of these off-target effects and could be pursued, similar to how a α4β2-nicotinic receptor partial agonist has been tried for deficits in attention underlying gait disorders. 89 The association between apathy and cognitive impairment may suggest a role for targeting cognitive enhancement, especially executive functions of decision-making and planning, as possible treatment approaches for “cognitive apathy”. For instance, boosting prefrontal cortical adrenergic tone with repurposed stimulants such as methylphenidate or other non-stimulant adrenergic augmentation in PD-associated apathy should be addressed in an RCT. Lastly, while there is already mounting evidence that traditional SSRIs are ineffective for apathy, newer antidepressants with mixed receptor properties, like vortioxetine, should be explored further in clinical trials on apathy.
Future directions for depression
For the treatment of depression modest RCT evidence supports the efficacy of several agents across most of the major classes of antidepressants, e.g., SSRI, SNRI, tricyclic, a dopamine agonist pramipexole, and suggests that SARIs may also be useful. A next step for identifying new treatment approaches for depression in PD would be to consider comparative efficacy trials and augmentation trials exploring options for non- and partial responders. These trials should be conducted using protocols that allow for the natural complexity of treating depression in PD such as co-occurring anxiety and apathy, so that their impact on treatment response can be studied. Approaches that examine the potential antidepressant benefit of new PD medications should also continue, as they have been, by including secondary outcome measures that assess mood and anxiety. Any medication that treats both motor and psychiatric symptoms will be especially valuable for PWP as it would reduce the risk of polypharmacy and the pill burden in cases where both conditions exist. Mechanistic studies of depression in PWP continue to be important as the connection between the two syndromes includes prodromal depression as a risk factor for PD and a reciprocal exacerbation of symptoms, e.g., depression worsens PD symptoms and PD symptoms confound antidepressant response suggesting the possibility that a shared biology exists.
Future directions for ICDs
In order to develop targeted therapies for ICDs, the foundational understanding of the syndrome may need to be deconstructed and reinterpreted using existing frameworks for behaviors observed in other clinical syndromes. For instance, PWP experiencing an ICD may share some aspects of mania with increased goal-oriented behavior and impulsivity. The parallels between a bipolar affective disorder and the spectrum of motivational behavior disorders that can co-occur in PWP (ICDs and apathy often co-occur like mania and depression in bipolar illness) warrant consideration of whether ICDs should be treated with a “stabilizing” approach. For instance, observational studies from two separate cohorts have suggested an association between SSRI use and ICDs risk in PD90,91 and those with ICDs had larger mood changes between the “on” and “off” medication states, 91 which parallels the potential for SSRIs to precipitate mania when treating bipolar depression. Thus, one wonders if mood stabilization approaches with quetiapine, depakote, lamotrigine or new agents would be effective. This could be answered with small studies for proof of concept and safety. With regard to the effects of mesolimbic D3 receptor agonism, there is space for the development of D1 (apomorphine or similar?) or D1/D5-specific agonists that might address motor symptoms with less impact on motivational behavior and negative or positive reinforcement learning. Apathy measures should be included in any ongoing clinical trials of these agents for motor benefit and consideration should be given to specifically targeting apathetic PWP in early, proof-of-principle studies.
Future directions for psychosis
Dopamine's antagonistic relationship between motor improvement and psychosis worsening is the biggest challenge in developing new treatments for psychosis in PD. This does not necessarily mean that antipsychotic therapies that involve dopaminergic mechanisms should be abandoned. For example, antipsychotic drugs that have a greater ratio of cortical to striatal dopamine type 2 receptor binding are both more efficacious for psychosis and less likely to cause motor side effects in non-PD populations, e.g., clozapine cortical > striatal D2 binding, haloperidol cortical = striatal D2 binding. 92 Therefore, drug development that emphasizes a more favorable cortical to striatal binding ratio could be a strategy for dopamine-based antipsychotic medications in PD. Another direction would be to continue to explore molecules, like pimavanserin, that modulate serotonin. In fact, this mechanism may explain the action of clozapine, which binds to multiple serotonin receptors including 5-HT2A receptors. Finally, an approach that targets the cholinergic system seems appropriate given the secondary outcome data demonstrating psychosis improvement with acetylcholinesterase inhibitors in cognitively impaired PD. A timely example of how modulating the cholinergic system might help treat psychosis comes from the newly FDA approved antipsychotic for treating schizophrenia, xanomeline and trospium chloride. 93 Xanomeline is a M1 and M4 muscarinic receptor agonist that does not block D2 dopamine receptors. Xanomeline is being investigated for use in treating psychosis in people with Alzheimer's disease.
As PD is increasingly recognized as a neuropsychiatric disorder rather than a “movement disorder”, and symptomatic therapies for motor symptoms extend functional ability well into the advanced phases of disease when psychiatric conditions become even more common, better treatments are needed for psychiatric symptoms that are tolerable to people with autonomic dysfunction and polypharmacy. For some psychiatric disorders, like depression existing therapies for non-PD should be studied in larger, randomized, controlled trials. On the other hand, some conditions like anxiety, apathy, ICD, or psychosis, may have disease-specific mechanisms that need to be better understood or have PD-specific limitations on the use of existing therapies. Here, it is essential to better understand contributing mechanisms so that drugs from other indications can be repurposed or novel agents can be developed to target these syndromes.
Footnotes
Acknowledgements
We would like to acknowledge our patients as the primary motivation to conduct this review to guide the development of better and more specific treatements for psychiatric symptoms in PD.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was not directly funded but KAM and GMP are supported by NINDS/NIH (R21NS128391-01A1) and 1R01MH123552 respectively.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: KAM: Dr Mills has received funding from Parkinson's Foundation, Michael J. Fox Foundation, and NINDS/NIH (R21NS128391-01A1). He receives clinical trial research support from UCB Pharma. He has done consulting with Tilosia, LLC. GMP: Dr Pontone is funded by 1R01MH123552. GMP has done consulting with Acadia Pharmaceuticals Inc and GE Healthcare.
