Abstract
Background:
Lack of participation of black and minority ethnic communities (BAME) in registered clinical trials is a concern as data emerging from these studies are used to licence new drugs or other interventions, even though findings made in such selected study populations have limited external validity in the aforesaid ethnic groups.
Objective:
We used Parkinson’s disease (PD), the fastest rising neurodegenerative disorder in the world, as an exemplar condition to test our hypothesis that participants from BAME communities are underrepresented in clinical trials.
Methods:
A systematic search of clinical trials registered on a Clinicaltrials.gov database which queried for PD with racial distribution data from 2017 to 2021.
Results:
Out of 266 trials considered, 54 trials were published in peer reviewed journals. Among these, only 23 (42.65%) publications reported data regarding the racial distribution of the participants. Out of these, five studies involved mixed racial participation and two trials included black subjects.
Conclusion:
We found that inclusion of under-represented BAME groups in recently published clinical trials is low, at only 21.57%, and is not even considered in most studies. Out of the reviewed trials, only 5 (21.75%) studies reported detailed demographic categories with black minorities enrolment. This constitutes a severe under-representation when compared to the proportion of Black or African American in the UK population (3%). Results of this study identified the need for better reporting of racial composition in clinical trials. We strongly recommend that future studies should consider ethnicity and other issues around diversity when designing and implementing the clinical trials, not only in the PD field but also beyond.
INTRODUCTION
Parkinson’s disease (PD) is a heterogeneous and complex multi-system disorder with a massive societal impact. PD has emerged as the fastest growing neurological disorder in disability-adjusted life years, deaths, and prevalence in the world with a global prevalence of PD that doubled between 1990 and 2016 and is projected to double again over 12 million by 2040 [1, 2]. In spite of reports to the contrary it is now widely recognised that PD affects patients across all age ranges, geographical locations, and racial and ethnic backgrounds worldwide [1].
The epidemiology of PD with a focus on diversity among non-white ethnic groups has been scarce and poorly studied. This notion could explain why PD appears to be relatively more frequent in Western populations [3, 4]. Little is understood about diversity in therapeutic and clinical trials in PD and related disorders, including issues around diagnosis and treatment that are important in relation to personalised medicine delivery and its enablers such as pharmacogenetics, body mass index, personality, cultural differences, otherwise known as the circle of personalised medicine [5, 6]. Several studies have reported ethnicity to be a predictor of mortality in PD [7]. Black patients tend to have a slightly higher risk of death than white patients and this finding has been replicated across several studies [8–11]. One study showed that women are consistently underrepresented in clinical trials in the PD field, and this is worrisome, because men and women with PD may have a different clinical presentation and exhibit a different response to treatment [12]. Certainly, studies outside the PD field have shown that trials of new interventions that were performed in predominantly men failed to identify adverse effects that only subsequently came to light in real life when larger numbers of women received the treatment [13].
One related issue is that, ideally, clinical trials involving PD patients should have a focus on BAME enrolment, particularly if trials are conducted in countries or localities with a multi-ethnic population. Inclusion of currently under-represented groups in clinical trials is imperative to determine possible differences in disease outcomes, response to interventions, and therapeutic benefits among different ethnic groups [14]. Enthusiasm for engagement of research is also echoed by BAME patients when surveyed. However, the participation of minority ethnic, specially the BAME community, in research remains a major issue. For instance, in cities like London where in many areas, BAME population may form over 13.3% of the overall population while in some areas such as Camberwell it may exceed 50% [15]. Another clinical trial literature review also revealed that only 17% of published US studies reported information about the enrolment of under-represented minorities in which the proportion of minority participants was much lower than in the general US population [16]. Approximately 6% of study participants were non-white in the reported studies while non-whites only represent 20% of the general US population age 60 and over [14, 16]. Overall, poor representation in research will lead to an incomplete picture of the true diversity of PD [17]. In this report, we present ethnicity-related data from PD studies registered on the publicly accessible database clinicaltrials.gov over a period of 5 years (between 2017 and 2021), aiming to explore and analyse the racial distribution of people taking part in the clinical trials.
METHODS
We performed a systematic search of clinical trials registered on a publicly accessible database Clinicaltrials.gov which queried for PD. To maximise the results, the only search terms we used were “Parkinson disease” and “parkinson”. We limited the search to years 2017 through 2021 and only completed and published trials. We excluded unpublished trials or published abstracts only or where no racial inclusion data were reported.
The following inclusion criteria were utilised: 1) Humans, 2) English language, 3) Randomized Controlled Trial, observational study, and 4) a study completion date between January 1, 2017 and July 1, 2021. A total of 266 registered clinical trials were available in the ClinicalTrials.gov registry. Race or ethnicity data collected on both screened and enrolled individuals were obtained from published trials.
Literature search was independently done. The review of the literature search was done by three members of our team so that bias can be avoided, and scrutiny is thorough. The third team member was the final adjudicator to resolve potential disagreements on the inference from the data and this process was followed for screening, data extraction and critical appraisal. The studies had not specified a prior requisite for BAME data to be considered at entry point and as such these were retrospective analysis.
RESULTS
A total of 266 studies were conducted and published between 2017 and 2021 were registered on the clinicaltrials.gov database. Data from 54 were published in peer reviewed journals. Among these, only 23 publications reported data regarding the racial or ethnic distribution of the participants (Fig. 1). Of these, one publication was an observational study and 22 were randomized controlled clinical trials (RCT) related to PD. 30 studies were not included further because racial distribution details were not reported. The 23 studies included in the final analysis enrolled a total of 676 participants, of whom 363 (53.7%) were white and 46.3% were non-white. Out of the 23 studies, 11 (47.82%) included only white participants, and 7 (30.43%) only reported the ethnicity from their own countries of origin. Only five studies (21.74%) reported a mixed racial/ethnic composition (Fig. 2). An outline of the 23 included studies is presented in Table 1.
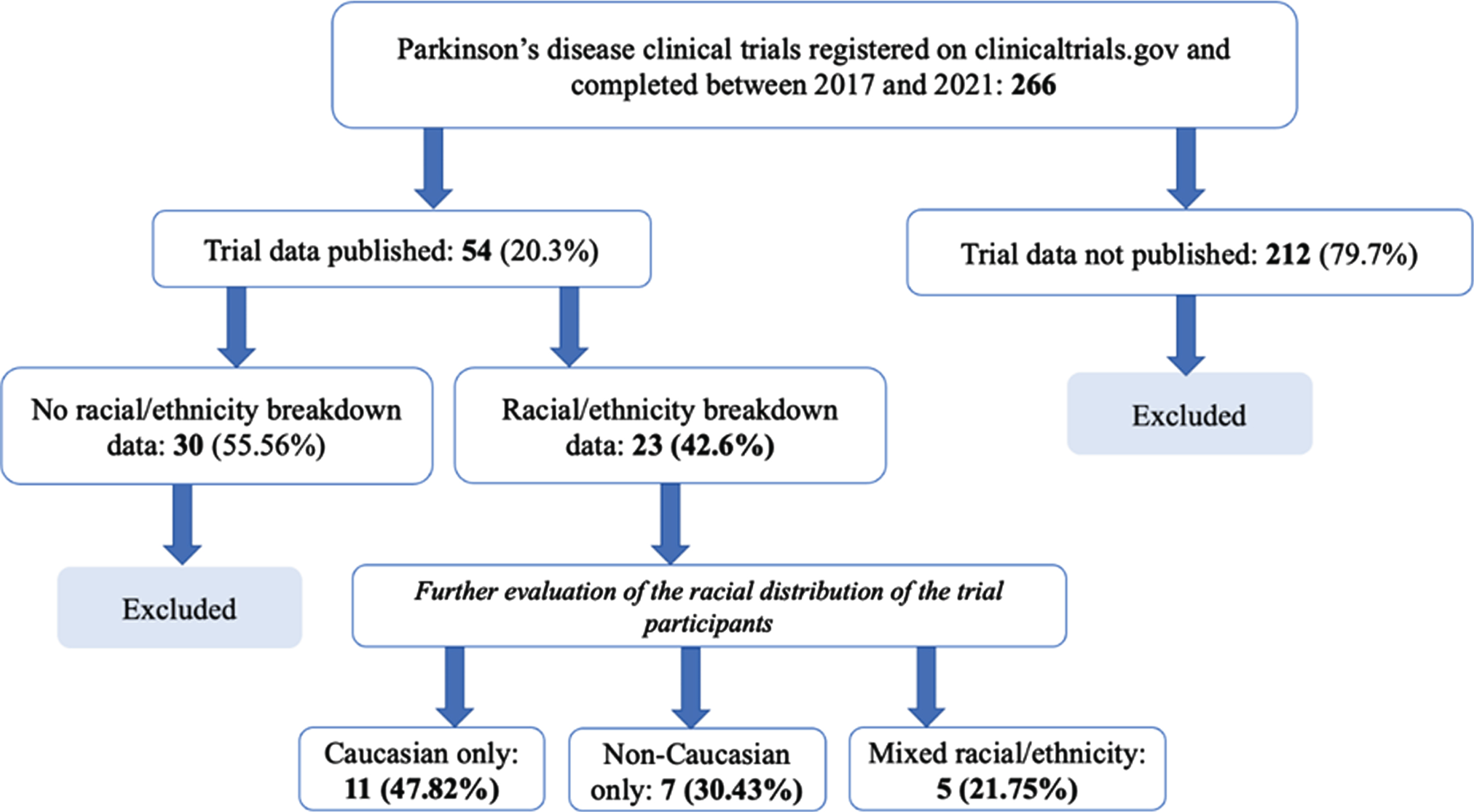
Flow chart to show racial inclusion related information (as allowed by the Office of National Statistic)- trial data published between 2017 and 2021.
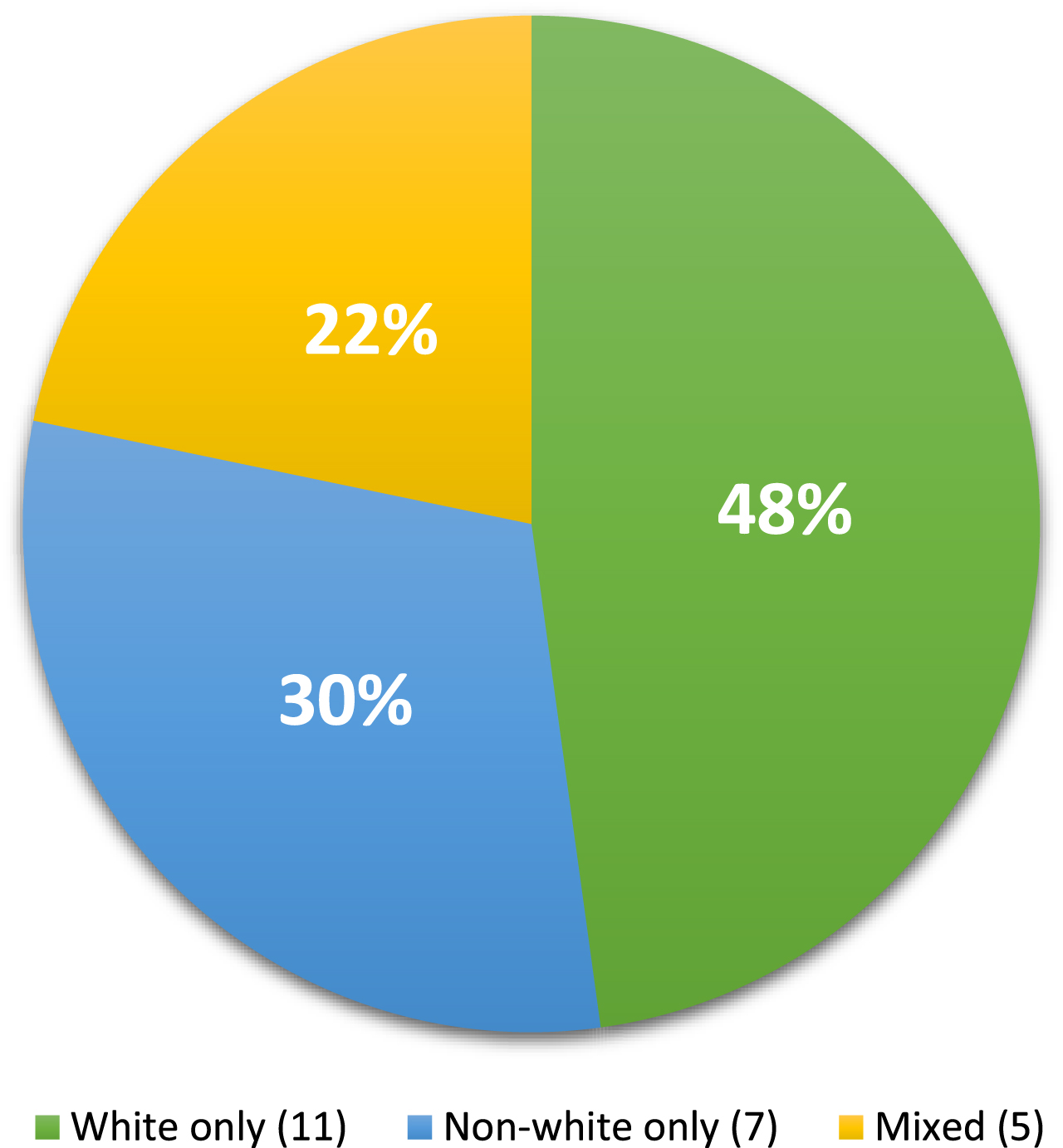
The pie chart shows the percentage of racial distribution in the 23 publications that reported data regarding the trial participation by different racial or ethnic population.
A summary of the ethnic breakdown reported in the published results of the 23 studies included in the final analysis
DISCUSSION
In this report, we analysed the ethnic distribution of research participants in clinical trials in the PD field over a period of 5 years (from 2017 to 2021), that were identified from a search of the clinicaltrials.gov database. Our results suggest that most clinical trials did not involve the minorities and mainly focused on Caucasians or races of the country of origin, even though the studies were performed in a country that had a multi-ethnic population character. Even studies performed in the urban areas of the UK and US often did not consider the need to include or consider multi-ethnic population base in recruited subjects, despite the marked diversity in these countries [41, 42]. Many other studies made no specific mention in of race or ethnicity in the study population, which raises concerns whether this diversity issue was properly addressed at all. This is in line with our findings that a total of 30 out of 54 published trials did not include racial or ethnicity participation. It is therefore of key importance to highlight the discrepancy between the pool available for recruitment and the final ethnic distribution of the study participants. In the current study as shown in the Table 1, countries like Turkey, Korea, Taiwan, Spain, HK, Slovenia, China, Malaysia, and Egypt have been represented to show isolated ethnic or racial populations (implied by default) using the clinical trials register that is publicly available. We recognize, however, that there are many more publications, from the same geographical territories, but not being reflected in the Clinicaltrials.gov and a detailed analysis in future will be undertaken.
The fact that ethnic minority subjects, especially black populations, are often not being included in PD studies, could have various implications. Current literature suggests that there may be differences in pharmacological response based on cultural differences and pharmacogenetics between races, indicating that a ‘one size fits all’ approach in relation to clinical trials data interpretation may not be sufficient [5, 44]. For example, a medication regime that is effective for Caucasians might not have a similar efficacy in another racial group or may not be even well-tolerated otherwise.
Clinical experience and emerging evidence suggest that ethnicity may be a determinant of heterogeneity, with differences in epidemiology, clinical manifestations, and mortality. For example, variations in the catechol-O-methyltransferase enzyme (COMT), which influences the rate of levodopa metabolism, have been described, with a possibly increased prevalence of the variant that metabolises levodopa quicker among the native African people as compared to a white population [45]. Similarly, Asian patients also appear more likely to experience levodopa-induced dyskinesias due to a higher frequency of high COMT activity, hence lower doses of dopaminergic drugs are often recommended for this specific population [46]. Differences between ethnic groups are evident when considering comorbidities, such as a higher prevalence of diabetes among the Asian population, although further research is needed to establish whether this or other comorbidities influences the symptom expression of PD [47]. Another important issue relates to compliance to the prescribed intervention, which may well vary across different populations. And just as unexpected and sometimes serious adverse effects have been described in women (who were underrepresented in the original seeding trials), we might expect to see similar adverse effect in non-white PD populations who did not participate in the original studies which led to the registration of a particular intervention.
There are socio-cultural issues that also need to be addressed. Conclusions from studies that focussed entirely on white populations may not be acceptable or even appropriate in some cultures and as such recommendations in guidelines may be bespoke. For example, studies demonstrate that yoga and meditation enhance proprioception and balance in individuals diagnosed with PD and that such programmes are safe and effective for patients with PD [38]. However, these studies report no involvement of black, and very little Asian participants, so it is difficult to envisage how this physical intervention could be adopted in these ethnic groups as there are differences in cultural perceptions [38].
Personalised medicine for PD is not just a genetic approach to treatment but encompasses various aspects of treatment, particularly treatment disparities among different racial distributions. Clearly, increased knowledge on the role of ethnicity in PD and individualising the treatment of both motor and non-motor symptoms based on the susceptibility of specific subtypes of PD to side effects (subtype- specific treatment), genetic framework, personality, pharmacological, pharmacogenetics, lifestyle-related issues as well as socio-demographic, in particular racial distribution in term of therapeutic intervention which may be helpful for monitoring symptom expression and treatment response of PD, address inequalities in health care delivery worldwide and improve the delivery of personalised medicine [6, 48].
On the other hand, our study revealed that most of the studies were conducted in single-centres and this would highlight the possibility of requirement of pan-global collaborations for appropriate ethnic or geographical representations in PD research.
We totally agree that multi-sectorial strategy involving governments, pharmaceutical organizations, and other key stakeholders is also necessary to increase the minority ethnic enrolment in PD research. This includes the possibility for collaboration of these partnerships with global consortia studying the genetic etiology of PD [Genetic Epidemiology of Parkinson’s Disease (GEoPD; https://www.geopd.net), The International Parkinson Disease Genomics Consortium (IPDGC; https://www.pdgenetics.org)], and the initiation of Global Parkinson’s Genetic Program -GP2 by the Michael J Fox Foundation in promoting diversity and placing more emphasis on minority recruitment in PD research.[49]
The main strengths of this report include its originality and its importance in identifying apparent inattention to the under-enrolment of minority persons into PD clinical trials within the research community. There are some limitations to this study as we limited our search to publications of studies registered on clinicaltrials.gov only. We were therefore limited by the availability and accuracy of information in published papers. However, findings from this study clearly show a low frequency of reporting racial composition of PD trial populations and an overwhelming Caucasian predominance in nearly all PD sample populations where racial composition was reported.
Conclusion
PD affects all racial and ethnic groups globally; and the minority populations are often associated with a substantial disease burden. Expanding the access to clinical trials or other research studies and ensuring a representative enrolment are of utmost importance in PD research. There is an urgent need to recognise and address the ways to improve the “research effect” among our minority ethnic population and start collecting data regarding such initiatives where available, and to identify as well as overcome possible barriers in PD trial participation.
CONFLICT OF INTEREST
The authors have no conflict of interest to report for this article.
