Abstract
Background:
Drug therapy is important for controlling symptoms in Parkinson’s disease (PD). However, it often results in complex medication regimens and could easily lead to drug related problems (DRP), suboptimal adherence and reduced treatment efficacy. A structured medication review (SMR) could address these issues and optimize therapy, although little is known about clinical effects in PD patients.
Objective:
To analyze whether an SMR improves quality of life (QoL) in PD.
Methods:
In this multicenter randomized controlled trial, half of the 202 PD patients with polypharmacy received a community pharmacist-led SMR. The control group received usual care. Assessments at baseline, and after three and six months comprised six validated questionnaires. Primary outcome was PD specific QoL [(PDQ-39; range 0 (best QoL) – 100 (worst QoL)]. Secondary outcomes were disability score, non-motor symptoms, general health status, and personal care giver’s QoL. Furthermore, DRPs, proposed interventions, and implemented modifications in medication schedules were analyzed.
Results:
No improvement in QoL was seen six months after an SMR, with a non-significant treatment effect difference of 2.09 (–0.63;4.80) in favor of the control group. No differences were found in secondary outcomes. In total, 260 potential DRPs were identified (2.6 (±1.8) per patient), of which 62% led to drug therapy optimization.
Conclusion:
In the current setting, a community pharmacist-led SMR did not improve QoL in PD patients, nor improved other pre-specified outcomes.
Keywords
INTRODUCTION
Nowadays, pharmacotherapy is still the keystone of treatment in Parkinson’s disease (PD), aiming to reduce both motor and non-motor symptoms. Although medication is initially effective, long-term treatment effects will be impaired, whereby patients experience fluctuations in drug response and side effects [1]. Besides, in more advanced disease stages, dosing frequency intensifies and additional medication is often needed [2]. The medication regimen complexity increases further due to comorbidities, and maintaining disease control might become more and more challenging [3]. Although medication adherence is crucial for achieving treatment success, non-adherence is common in PD: 13–67% of PD patients take less than 80% of their prescribed medication as intended [4–8]. This strikingly contributes to decreased quality of life (QoL), drug related problems (DRPs), failure of treatment, and increased health care costs [8–11]. Reasons are multifaceted, with complicated drug dosing regimens, polypharmacy, and lack of knowledge of the disease, its treatment, and proper drug use among them [6, 11]. Hence, PD demands a systematic approach for monitoring pharmacological care and the safety of medicines, as well as prevention of DRPs.
An intervention increasingly performed, and potentially effective for both improving adherence and optimizing treatment, is a medication review. This is defined as ‘a structured, critical examination of a patient’s medicines aiming to reach an agreement with the patient about therapy, optimizing the impact of medication, minimizing the number of DRPs and reducing waste’, subdivided in different levels of extensiveness, from prescription reviews to the more patient-centered clinical medication reviews [12]. Previous research on the effect of medication reviews—often focused on a specific disease or patient population (e.g., diabetes, hypertension, patients with polypharmacy, elderly, etc.)—demonstrated positive drug-related outcomes as improved adherence, less DRPs, and diminished use of potentially inappropriate medication [13–17]. However, clear evidence of a positive effect of a medication review on more clinical outcomes like QoL is lacking, since varying results have been reported to this regard [16–19]. This might be attributed to the different settings of care, different ways of performing medication reviews, heterogenous patient selection, and variable study designs.
In the Netherlands, community pharmacists perform medication reviews as part of standard care using a structured approach (structured medication review; SMR) [20], consistent with the clinical medication review approach according to Clyne et al. [12]. However, to define which patient (group) might benefit the most has shown to be difficult, and selection criteria have been changing over time [20–22]. Verdoorn et al. [23] recently found an improved QoL in a large population of elderly people receiving a medication review, where the patient-centered approach was crucial. Taking patient preferences into account may consequently lead to individualized care and more shared decision making.
In a small pilot study, positive effects on QoL of a medication review performed in an outpatient PD clinic were found [24]. The execution of SMRs in PD might tackle complex drug regimens and improve knowledge. Furthermore, by gaining more effect of drug treatment, PD related symptoms and thereby QoL might improve [25]. However, although of substantial importance from a patient’s point of view, little is known about the effect of an SMR in PD on QoL performed by community pharmacists in a primary care setting.
This study evaluated whether an SMR performed by community pharmacists led to improved QoL in patients with PD. Furthermore, its effect on the performance of activities in daily life, the experience of non-motor symptoms, and QoL in personal care givers was investigated.
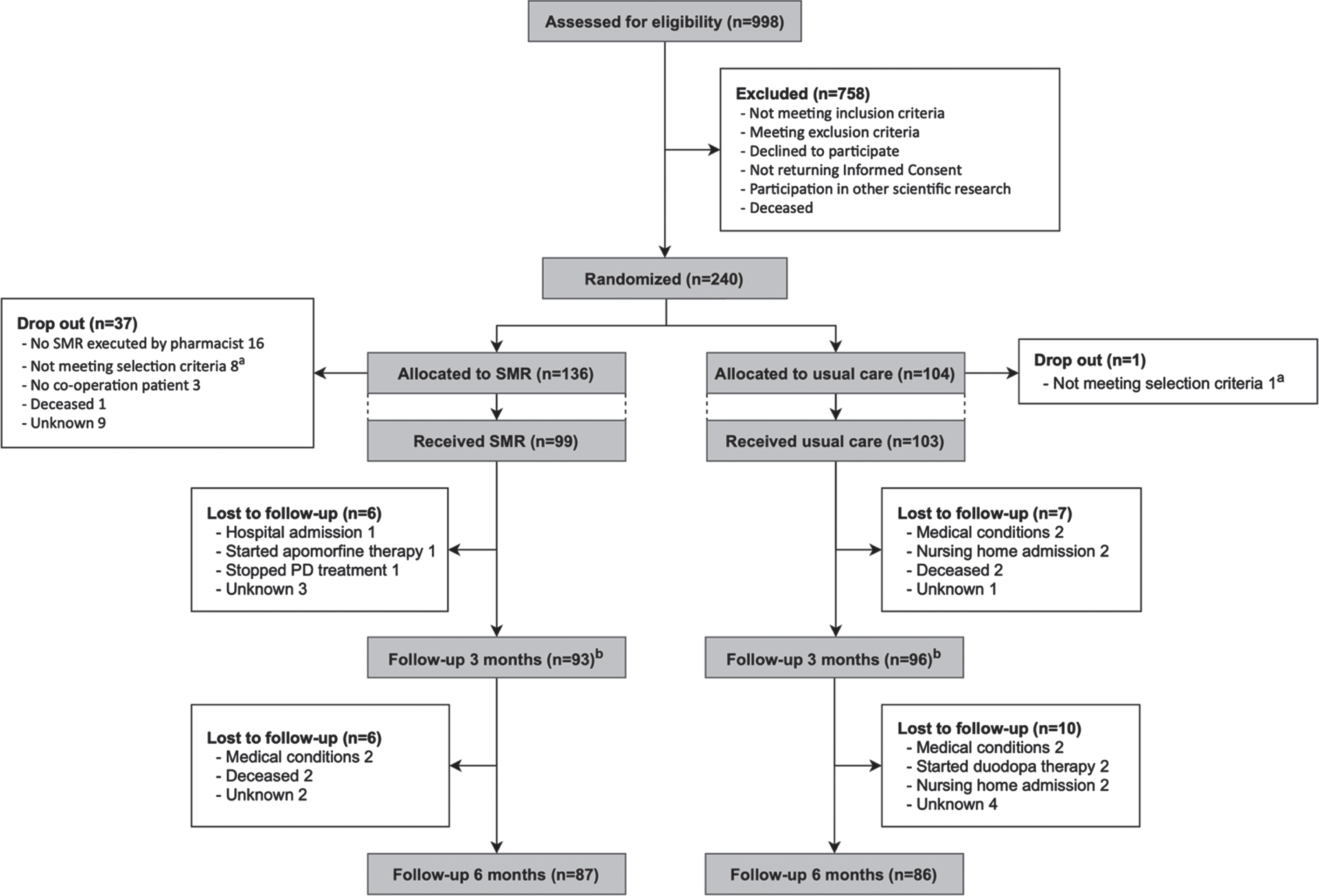
Flowchart. aThree patients started with advanced therapy, one received a recent SMR; two did not understand the questionnaires; one used only three medicines daily; one was diagnosed with atypical parkinsonism, one was admitted to a nursing home. bNo return of questionnaires: n = 9 (SMR group) and n = 5 (usual care group). SMR, structured medication review.
METHODS
Study design
This multicenter randomized controlled trial was conducted at three neurology outpatient clinics in the Netherlands in collaboration with community pharmacists and general practitioners (GPs), according to the trial protocol published previously [26]. The protocol was registered at trialregister.nl (NL4360). The study was performed in agreement with the principles of the Helsinki Declaration and in accordance with the Medical Research involving Human Subjects Act (WMO). The protocol was approved by the Medical Ethical Review Board Twente, the Netherlands.
Participants and randomization
Participants were eligible if they fulfilled the following criteria: 1) ≥18 years of age; 2) diagnosed with PD according to the UK-brain banking criteria [27]; 3) ≥4 different medicines daily; 4) ≥4 dosing moments daily; 5) expressing motor and non-motor symptoms; 6) living (semi-)independent in the region of the abovementioned outpatient clinics; 7) able to read and write the Dutch language. Key exclusion criteria were: 1) being unable to administer own medication, e.g., when requiring assistance from medical home care. Based on this criterion, participants receiving help from family or personal caregivers were not excluded; 2) having received a medication review within a year prior to the study; 3) having received advanced therapy (continuous apomorphine therapy, continuous levodopa gastrointestinal gel therapy or a Deep Brain Stimulator) within a year prior to the study, or scheduled to receive this within three months before start of the study.
Written informed consent was obtained from all participants.
After baseline measurements, patients were randomly allocated to either the intervention group or control group in a 1:1 ratio with a blinded blocked randomization with block sizes of four and eight. Definite inclusion for patients in the control group was immediately post randomization, while patients in the intervention group needed to receive an SMR to be definitely included. Due to high drop out of patients in the intervention group before receiving an SMR, the randomization process was revised during the study course and performed in a 4:1 (intervention group: control group) ratio, in order to meet the calculated power in time.
Intervention
Community pharmacists performed an SMR as a one-time assessment at the beginning of the trial within the intervention group. The control group received usual care and did not receive a medication review during follow-up. All participating community pharmacists were offered an accredited training in advance focusing on PD, its pharmacological treatment, the study protocol, and a uniform approach in performing SMRs. This approach was based on the Dutch Systematic Tool to Reduce Inappropriate Prescribing (STRIP)-procedure, a Dutch standardized method for pharmacists for performing SMRs, which is part of the multidisciplinary guideline Polypharmacy in the Elderly [20]. This tool puts high emphasis on the involvement and contribution of the patient as an equal partner in evaluating problems regarding medication use or dosing, drug interactions, and adverse reactions. Subsequently, potential DRPs were identified for which interventions were proposed, taking into full account the patient’s views, beliefs, and preferences about their medication regimen.
DRPs were grouped by the researchers, based on the explanation of the pharmacist, according to classification of Strand et al, on which the STRIP-method is based [28]. Categories were: additional drug therapy required, unnecessary drug therapy, ineffective drug therapy, dosage too low, dosage too high, (potential) adverse drug event, clinical relevant contra-indication or interaction, and drug usage problems. Interventions were classified as drug interventions (starting, stopping, replacing, or changing the dosage of a drug) and other (e.g., giving information or advice according to drug usage).
The protocol allowed for consulting a GP, neurologist, and other specialists by the pharmacist, if needed. Any proposed adjustments in the drug regime were discussed with the patient and involved physicians. After completing the SMR, pharmacists documented all proposed and implemented interventions. In case of uncertainty regarding implementation, medication prescriptions during follow-up were checked for changes as well as documentation in electronic patient files. When inconclusive, the pharmacist was contacted for verification. Clear data collection and documentation was required to ensure continuity of care and to facilitate cooperation between health care professionals, which we addressed with a uniform recording file (see the Supplementary Material).
Outcomes
Information from participants regarding medication regime and dosing was verified by their pharmacists. Since definitions vary, polypharmacy was defined as using≥4 drugs daily [29]. Severity of PD was measured by Hoehn & Yahr stage (HY) [30]. Comorbidity scores were calculated using the Rx-Risk Comorbidity Index (range 0–46) [31].
At baseline and after three and six months, participants (and when applicable, their personal care givers) completed a set of six validated questionnaires. The primary outcome was the PD specific QoL at six months, measured by the Parkinson’s Disease Questionnaire-39 (PDQ-39) (range 0–100) [32]. Higher scores represent worse QoL. A difference of≥1.6 points in the PDQ-39 indicates a clinically relevant or minimally important difference (MID) [33]. Also, a sensitivity analysis was performed to measure the effects of an SMR in patients in whom at least one proposed intervention was implemented.
The secondary outcomes were 1) daily life activities and physical disability (AMC Linear Disability Scale (ALDS) (range 0–100) [34]; 2) non-motor symptoms (Non-Motor Symptoms Questionnaire (NMS-Quest) (range 0–30) [35]; 3) general QoL and health status (EuroQoL (EQ)-5D-5L) (range –0.446–1) and EuroQoL Visual Analogue Scale (EQ-VAS) (range 0–100) [36, 37]; 4) QoL of PD care givers (PDQ-Carer) (range 0–100) [38]. For the ALDS, EQ-5D-5L, and EQ-VAS higher scores imply better performance respectively better general QoL and health status. Higher NMS-Quest and PDQ-Carer scores imply the experience of more non-motor symptoms respectively worse QoL of care givers.
All SMR files were analyzed regarding potential DRPs and proposed and implemented interventions and reported descriptively. Additionally, exploratory analyses were performed to analyze whether a subgroup of patients could be identified that would benefit most from an SMR.
Statistical analysis
A difference of 1.6 points in the PDQ-39 score was assumed clinically relevant [33]. Based on own expectations, we assumed a between-group difference of six points in PDQ-39 score post intervention.
Based on previous investigation reporting a standard deviation of 15 [33], we performed a preliminary sample size calculation. To detect a clinically important difference with a type I error of 5% and a power of 80%, a total of 198 participants was required.
A modified intention-to-treat analysis was used, which essentially meant that to be included in the analyses, patients needed to complete baseline questionnaires and the intervention group patients needed to receive an SMR—whether or not any subsequently interventions were implemented. A mixed model repeated measurement analysis was performed for all continuous variables. Missing individual questions in the PDQ-39 questionnaires were imputed using Expectation Maximization, according to the PDQ-39 manual [39].
In the exploratory analyses, intervention group patients with a PDQ-39 difference score of ≤–1.6 points (clinically relevant improvement) versus > –1.6 points (no clinically relevant improvement, or deterioration) were compared on patient characteristics. The analyses were both performed with complete cases, and - as a sensitivity analysis - based on multiple imputations. Data were analyzed using SPSS version 24. p-values≤0.05 were considered statistically significant.
RESULTS
Between May 2014 and December 2018, 240 patients were randomized (Fig. 2). Drop out was high in the SMR group for different reasons. The execution of an SMR appeared challenging to initiate despite prior commitment of the community pharmacist. In other cases, the execution was delayed, with consequence that initially eligible patients in the meanwhile started other PD treatment (e.g., continuous levodopa gastro-intestinal gel therapy, Deep Brain Stimulator) and met exclusion criteria, whereby they could not continue study participation. A total of 202 PD patients ended up in the modified intention-to-treat analysis, together with 109 home care givers. Baseline characteristics and questionnaire data are summarized in Table 1. No differences were found between both study groups.
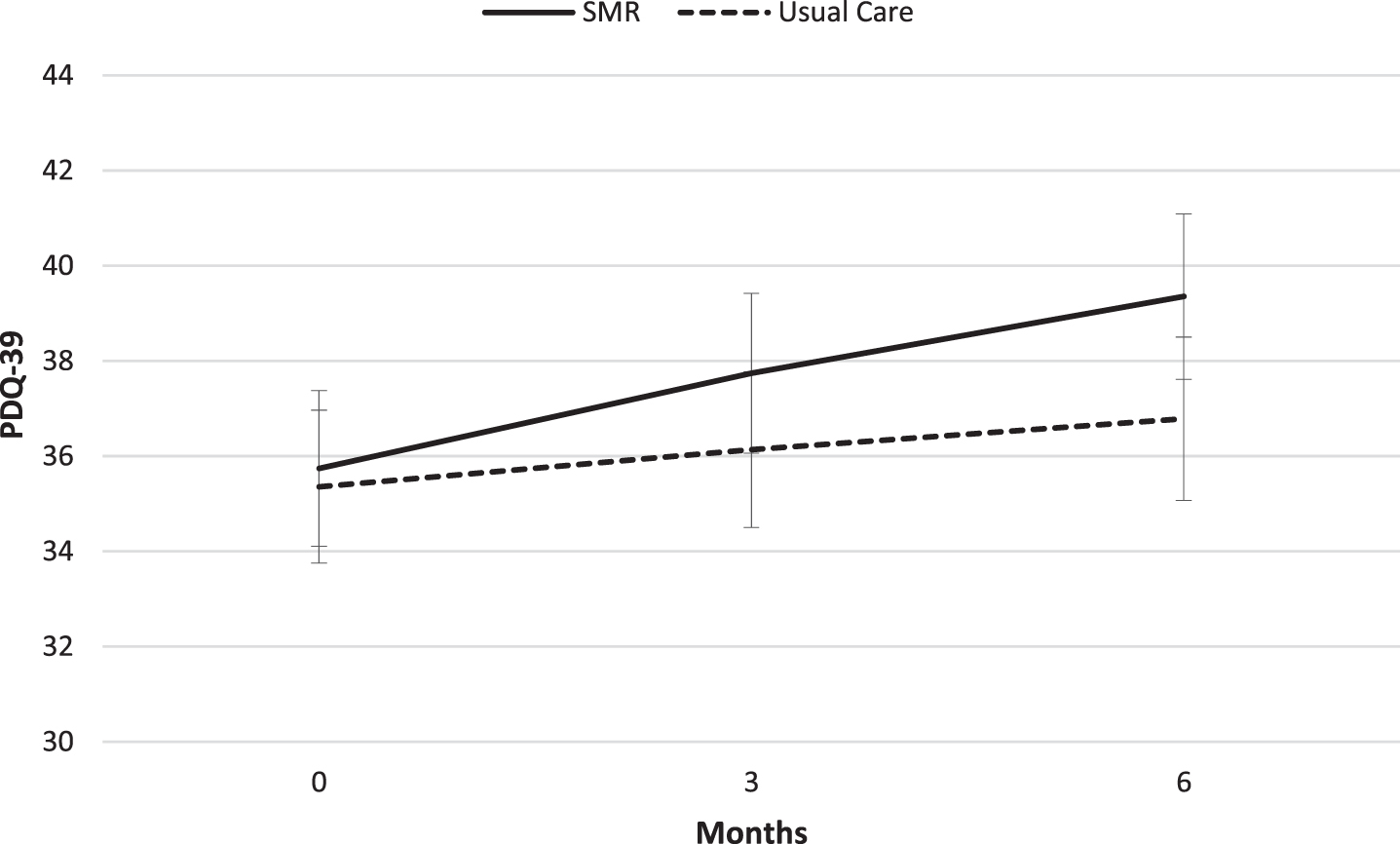
Mean PDQ-39 score during follow-up (range 0 (best QoL) – 100 (worst QoL). PDQ-39, Parkinson’s disease questionnaire-39; SMR, structured medication review.
Baseline demographic and clinical characteristics
Data presented as mean±SD unless otherwise noted. aRepresenting quality of life of care givers. ALDS, AMC Linear Disability Scale; EQ-5D-5L, EuroQoL 5D-5L; EQ-VAS, EuroQoL Visual Analogue Scale; HY, Hoehn & Yahr stage, IQR, Interquartile range; PDQ-39, Parkinson’s disease questionnaire-39; PDQ-Carer, Parkinson’s disease questionnaire for PD care givers; NMS-Quest, Non-motor symptoms questionnaire; SMR, Structured medication review.
Primary outcome
Not all PDQ-39 domains were normally distributed, hence normally distributed difference scores were used in repeated measurement analysis and presented in Table 2. Baseline PDQ-39 scores were similar in both groups. There was no difference in change after six months in PDQ-39 overall score or in any PDQ-39 subdomain between both groups, except for ‘Emotional well-being’. Patients in the SMR group scored significantly worse in this subdomain compared to the usual care group, with a difference of 5.54 (p = 0.01 (1.28;9.81). The major of this difference occurred between 3 and 6 months, in which an increase in PDQ-39-score from 3.17 is seen in the SMR group, whereas this score in the usual care group decreases with 1.69 points. This is also seen in the ‘course-over-time difference’, representing differences in the course of PDQ-39 scores during follow-up between both groups. This course also significantly differed in subdomain ‘Emotional well-being’. In all other domains and in the overall PDQ-39-score; courses over time were equal.
PDQ-39 scores at baseline and mean difference scores after 3 and 6 months
Data presented as mean±SE unless otherwise noted. aRange from 0 (best QoL) – 100 (worst QoL). bΔI-C: a negative change is in favor of the SMR group. cp-value based on ‘group-by-time’-interaction. *Significant difference; p < 0.05. PDQ-39, Parkinson’s disease questionnaire-39; SMR, structured medication review.
In both groups, PDQ-39 scores had increased after six months. All but one (subdomain ‘Cognitions’) observed differences were in favor of the usual care group, however, not significant. Figure 2 visually shows the change of actual PDQ-39 overall scores in both groups.
In the sensitivity analysis, the SMR group comprised only patients in whom at least one proposed intervention was implemented (n = 71). Compared to the usual care group, there was no significant treatment difference (2.51 (–0.41;5.43), or course-over-time difference (p = 0.45) in PDQ-39 overall score. Also, subdomain analyses showed almost similar results.
Secondary outcomes
Questionnaires
Of secondary outcomes presented in Table 3, only ALDS score was not normally distributed, and data were therefore presented as difference scores. Although both the ALDS treatment difference and the NMS-Quest, EQ-5D-5L, and EQ-VAS treatment effects were in favor of the control group, none was significantly different. The same applies to the course-over-time differences.
Data presented as mean±SE unless otherwise noted. aBased on ‘group-by-time’-interaction. bChange in favor of the usual care group. ALDS, AMC Linear Disability Scale; EQ-5D-5L, EuroQoL 5D-5L; EQ-VAS, EuroQoL Visual Analogue Scale; IQR, Interquartile range; NMS-Quest, Non-motor symptoms questionnaire; SMR, structured medication review.
PDQ-Carer scores of the SMR group (n = 51) and the usual care group (n = 58) showed no significant differences in both treatment effect and course-over-time (Table 4).
Secondary outcomes: mean PDQ-Carer scores at baseline and follow-up
Data presented as mean±SE unless otherwise noted. PDQ-Carer range: 0 (best QoL) – 100 (worst QoL). aA negative change is in favor of the SMR group. bBased on ‘group-by-time’-interaction. PDQ-Carer, Parkinson’s disease questionnaire for PD care givers; SMR, structured medication review.
SMR process and DRPs
Community pharmacists of 82 pharmacies were involved in this study. The average time spent on the execution of an SMR was 101 minutes (SD 57) for community pharmacists, and 16 minutes (SD 15) for GPs. The latter was estimated by the involved community pharmacist.
In the 99 executed SMRs, 260 potential DRPs were identified, resulting in 2.6 (±1.8) DPRs per patient. Most concerned ‘drug usage problems’ (36%), ‘additional drug therapy required’ (20%), and ‘unnecessary drug therapy’ (12%) (Supplementary Table 1).
Of the proposed interventions, 193 (74%) were drug interventions, 56 (22%) concerned information regarding optimizing drug use and effect, 6 (2%) monitoring and 5 (2%) other. Table 5 presents an overview of drug interventions classified by drug class. Nineteen percent was anti-Parkinson drug related.
Proposed drug interventions classified by drug class
ATC, Anatomical Therapeutic Chemical classification; NSAIDs, non-steroidal anti-inflammatory drugs.
In 161 cases (62%), a proposed intervention resulted in drug therapy optimization. Specifically regarding drug interventions, 102 (53%) of the 193 got implemented (Fig. 3).
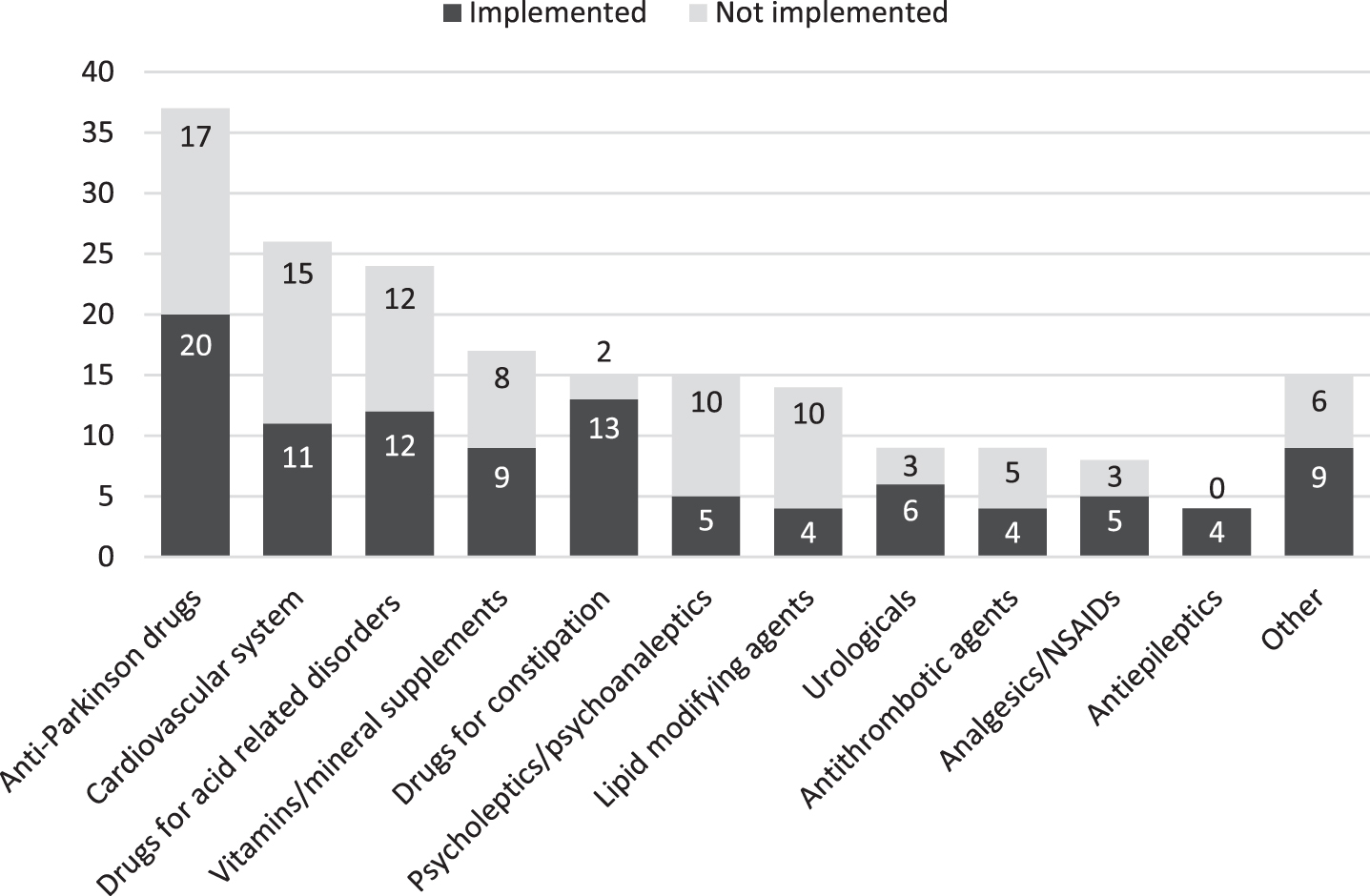
Implementation of proposed drug interventions classified by drug class. NSAIDs, non-steroidal anti-inflammatory drugs.
Reasons for not implementing were: new insights after consultation with GP (n = 34) or medical specialist (n = 10), changes refused by the patient (n = 8), or unknown (n = 39). Furthermore, in 16 cases additional information or advice was given, not related to any DRP (e.g., ‘discuss the care giver’s burden of care with physician’).
Exploratory analyses
In the SMR group, 87 patients completed six months follow-up measurements. Based on PDQ-39 difference scores, 19 patients showed clinically relevant improvement (mean –6.3, SD 4.8), and 68 showed clinically unaffected results or deterioration (mean 6.6, SD 7.6). No differences were found between these groups in gender (p = 0.37), HY (p = 0.76), age (73.4 vs. 71.9 (–2.3;5.3), PD duration (p = 0.55), number of daily medicines (7.2 vs. 7.5 (–1.5;0.8), number of daily intake moments (p = 0.74), and comorbidity score (4.5 vs. 4.6 (–1.1;0.9). Analyses based on multiple imputations showed similar results.
DISCUSSION
In this study, a community pharmacist-led SMR had no effect on overall QoL in a general PD population in primary care. On subdomain ‘Emotional well-being’ even worse QoL was seen after an SMR. Also, no effect on disability score, non-motor symptoms, or QoL of care givers was seen. We will discuss various reasons that might provide an explanation for our results.
In about one-fifth of patients in the SMR group no DRPs were found. These patients might have no need for improvement and should in retrospect preferably be excluded from receiving an SMR with more tailored selection methods. Also, only 62% of all proposed medication modifications were eventually implemented. This is in line with previous research in primary care setting in non-PD conditions, with implementation rates varying between 48–71% [14, 40]. However, no clinical improvement is expected in the other 38%. Regarding drug modifications, in 44 of the 91 not-implemented modifications there was a rationale after consultation with a physician. For example, with extra medical (history) information, blood pressure monitoring or laboratory tests, indications for prescribing and dosages were still correct, or DRPs were accepted since stopping the drug would worsen a patient’s health status. Also, solutions for potential DRPs turned out to be previously tried without success. Occasionally, the patient refused modification.
Even if DRPs were solved, the sensitivity analysis showed no difference in QoL. The assumption of drug optimization leading to clinical improvement was not confirmed by our data. It is conceivable that no clinical improvement is seen when the implemented modification is only administrative (n = 7), for example when medication was already stopped, but the pharmacist had not been informed or was not yet aware. It is remarkable though, that despite the strong pharmacy-patient liaison in the Netherlands, and a strong commitment of pharmacists to surveillance of medication, apparently not all prescription changes—both in primary and secondary care—are known by the community pharmacist. This leaves room for improvement; when not all health care providers are well informed about the actual treatment status, it facilitates the occurrence of false assumptions regarding medication and might harm the medication safety. Also, when preventive drugs were started or discontinued, QoL would not directly be influenced, perhaps even negatively if after starting side effects occur. Furthermore, representing daily practice, there was delay in the implementation of some modifications, and a potentially desired effect might not be measurable or not have its maximum impact yet in the months of follow-up left. Our final explanation is that changes in medication schedule might only be small contributions in ameliorating the life of PD patients; an optimized medication schedule might not be a big enough contribution. It seemed not to tackle all DRPs or symptoms patients experience due to PD and its necessary treatment.
An interesting fact is that a notable number of patients randomized to the SMR group did not receive an SMR. This was mostly due to logistic constraints: SMRs are time-consuming and demand cooperation between multiple health care providers and the patient. Since some initially eligible patients failed the selection criteria while awaiting their SMR, for example due to nursing home admission, study participation became impossible. However, it did not influence our results in aiming to assess the actual effect of an SMR in a primary health care setting. With the modified intention-to-treat analysis approach, as acknowledged in the study protocol, the effectiveness of the SMR could be assessed [26]. This was irrespective of whether or not proposed interventions were eventually implemented.
Compared to other studies, Verdoorn et al. [23] found clinical improvement measured by EQ-VAS six months after a medication review by community pharmacists in community-dwelling persons, ≥70 years of age, with≥7 drugs. The medication reviews in our study were executed according to the same method; however, the patient selection differed. Although both included community-dwelling persons, we selected a patient group with a specialized and complex disease, of which treatment takes place mainly in secondary care and of which the population in a community pharmacy is only small. Stuijt et al. [24] also found a significant improvement of QoL six months after a clinical pharmacist-led medication review, specifically in PD patients. However, it concerned a pilot study with only 23 patients. Patients had on average 4.6 DRPs, and the implementation rate of proposed modifications was 86%. The main differences with our study were the fact that SMRs were executed in a PD outpatient clinic by a clinical pharmacist specialized in PD drug treatment, with close collaboration with other involved physicians. This pharmacist had direct access to hospital patient records and every SMR was discussed with the neurologist. Also, the study population contained generally advanced disease stages, whereas almost 70% in our study had HY scores of 1–2.5. The lower HY scores can be explained by the primary care setting, and the inclusion of PD patients still being able to live (semi-)independently.
This raises the question whether the current setting of executing SMRs in PD could be optimized to obtain more effect. Especially since it turned out to be an intensive investment for the pharmacists (and probably also other involved physicians), both regarding time and logistics. Recently, the Dutch guidelines concerning medication reviews in primary care have been updated [22]. At the time of study start, guidelines for selecting patients for an SMR recommended patients being≥65 years using≥5 medicines, with either renal or cognitive impairment or risk of falling [20]. Strict cut-off points for age, number of medicines, and specific risk factors are now left out. Although patients≥75 years using≥10 medicines still seem a good target group, selection is recommended to be merely based on the degree of vulnerability, which is nevertheless tightly related with the previous criteria. Examples of signs of vulnerability are so called geriatric giants (immobility, postural of cognitive impairment, incontinence) or a recent hospital admission, especially when possibly due to DRPs (e.g., falling, obstipation, syncope, confusion). Furthermore, the focus of an SMR should be more on existing health related problems, possible side effects and personal treatment goals, since clinical significant effects after preventive adjustments in medication without health complaints beforehand are assumed to be only small. Lastly, more emphasis is put on involving the medical specialist to review medication when patients are mainly treated by them, and drug treatment is highly specialized or complicated.
Since the HY scores were relatively low, experienced health problems in these patients, or rate of vulnerability, might thus far be not too relevant. Besides, the minimal drug use for study participation was also lower compared to what current guidelines propose. This new insight was not known at time of study start. However, exploratory analyses did not show any specific patient criterion for better patient selection, although the power of the analyses is not sufficient for definitive conclusions, and an exploratory analysis assessing vulnerability is not feasible.
Based on our data and the recommendations in the updated guidelines, it might be interesting to explore whether PD patient selection based on rate of vulnerability (with presumably higher HY scores and medication consumption), pre-existing health related problems, or possible side effects will enhance the clinical effect of an SMR in future research. Besides, we support closer communication and cooperation between primary care and the medical specialist. Even though this was encouraged in the study design, it turned out that the neurologist was not always involved. Next to close collaboration, the added value of a more intensified PD training program for pharmacists could be analyzed in future studies [24].
Although we did not find any improvement in QoL, an SMR might result in an optimized medication schedule. When leading to less DRPs, side effects and hospitalizations, the intervention could still be effective. An additional cost-effectiveness analysis might reveal whether the intervention might perhaps save costs, as was also found in other research [18, 41].
Our study had several limitations. First, since the execution of an SMR takes time, we carried out follow-up measurements three and six months after the SMR date. Of all SMRs, 83% was executed within three months after baseline measurement. Nevertheless, although most SMRs were performed in a short time span, SMR patients often had more time between baseline and first follow-up measurement compared to the usual care patients. This might have led to a small underestimation of the results, since the SMR group had more time for PD to progress. Second, not all pharmacists participated in the PD training. However, all received information in writing, and were in close contact with the research team. We must also note that the offered PD training is not part of standard care and hence our outcomes might even be an overestimation. It might also positively affect the usual care of control group patients, and therefore reduce the effect size of the SMR in our study. However, we think this effect in the usual care group is only minor; although pharmacists with more knowledge of PD might abolish more DRPs, the collaborating pharmacists were well informed and agreed that control group patients should not receive a full SMR with actively interviewing the patient during study participation. Third, since patients have to get used to any drug modification, a follow-up of six months might be too short to measure outcome differences between both groups, also regarding the impact of preventive drug changes. Lastly, a blinded randomization was not feasible. This might have led to increased awareness of medication use or overestimated expectations of the SMR group patients beforehand, by which the intervention effect might be influenced.
In conclusion, structurally reviewing medication by trained community pharmacists in general PD patients in primary care did neither improve QoL, nor improve disability, non-motor symptoms, general health status or QoL of PD care givers. Time and logistic constraints were observed in the daily practice of executing SMRs, and better PD patient selection criteria could not be determined based on exploratory analyses. We assume that a selection based on clinical complaints due to potential DRPs, optimized (digital) communication, and a thorough involvement of health care providers in secondary care could improve the setting for SMR execution, and might make this intervention in PD more effective. Future investigations are therefore needed.
Footnotes
ACKNOWLEDGMENTS
The authors would like to thank all patients and involved health care providers, especially the cooperating pharmacists, for their effort and contribution.
This work was supported by the Royal Dutch Pharmacists Association (Koninklijke Nederlandse Maatschappij ter bevordering der Pharmacie, KNMP). This funding had no role in the analysis or interpretation of the results in this study.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
