Abstract
Background:
Parkinson’s disease (PD) is a debilitating neurodegenerative disease with both motor and non-motor manifestations. Available treatment reduces symptoms and is critical for improving quality of life. Treatment options include drugs, device-aided therapies, and non-pharmacological therapies. Complementary and alternative therapies (CATs) are also used in some countries.
Objective:
To examine the availability of PD treatment by country, and differences by national income as defined by the World Bank (high income countries (HICs), upper middle income countries (UMICs), lower middle income countries (LMICs) and low income countries (LICs)).
Methods:
This study was conducted by surveying International Parkinson and Movement Disorders Society members about availability of PD treatment. LMICs and LICs (LMICs/LICs) were analysed together.
Results:
There were 352 valid responses from 76 countries (41.5% from HICs, 30.4% from UMICs, and 28.1% from LMICs/LICs). Levodopa was widely available across all income groups (99%). Availability of other PD drugs decreased with national income. Availability of device-aided therapies decreased with national income (100% availability in HICs, 92.5% among UMICs, and 57.6% among LMICs/LICs). A similar trend was observed for CATs (37.0% availability in HICs, 31.8% in UMICs, and 19.2% in LMIC/LICs). Physiotherapy was the most available non-pharmacological therapy (> 90% respondents). Occupational therapy and SALT were less available in LMIC/LICs (49.5% and 55.6% respectively) compared to HICs (80.1% and 84.9% respectively).
Conclusion:
Our survey highlights significant discrepancies in availability of PD treatments between countries and income groups. This is concerning given the symptomatic benefit patients gain from treatment. Improving equitable access to PD treatment should be prioritised.
INTRODUCTION
Parkinson’s disease (PD) is a neurodegenerative disease presenting with motor symptoms, including rigidity, bradykinesia and shuffling gait, and non-motor symptoms, such as autonomic dysfunction, olfactory loss, sleep disorders, and cognitive impairment [1]. Conventional treatments for PD are targeted mainly at treating motor symptoms. These were defined arbitrarily based on group consensus and include levodopa combined with a decarboxylase inhibitor, dopamine agonists, monoamine oxidase B inhibitors (MAO-B inhibitors), and COMT inhibitors [2]. Efficacious, generic forms of most of these drugs exist. With disease progression, motor symptoms often become difficult to control. Device-aided therapies such as deep brain stimulation (DBS), levodopa/carbidopa intestinal gel (LCIG), and subcutaneously administered apomorphine are used to help manage motor fluctuations associated with long-term levodopa use [2, 3].
Various non-pharmacological therapies can be used to support the management of PD, including physiotherapy, speech and language therapy (SALT), and occupational therapy [2]. Complementary and alternative therapies (CATs) for PD, such as cannabinoids and acupuncture, are acknowledged as potential treatments, but high quality evidence to support clinical efficacy is currently scarce [2]. Mucuna pruriens is sometimes used as a naturally-occurring alternative to levodopa [4].
The global burden of PD appears to have more than doubled over the last 3 decades from 2.5 million individuals in 1990 to 6.1 million individuals in 2016 [5]. The WHO publishes a list of Essential Medicines which includes levodopa as the best-established and most efficacious treatment for PD [6]. Several studies have reported beneficial effects of levodopa on motor symptom relief, quality of life, and life expectancy [7, 8]. It is concerning that levodopa availability was reported in only 34% and 58% of primary care and hospital settings respectively in the WHO Atlas [9]. Availability generally decreased with income level of a country [9]. There are no published studies that examine global access to PD treatments. Access to medication is affected by many factors including the quantity of medication imported or manufactured, financial aspects of purchasing, distribution and supply chains within countries, prioritisation against other treatable comorbidity, case ascertainment, public awareness and health education, stigma, and use of local healers [10]. This pilot study sought to investigate access to PD treatment, focusing on the availability of conventional drugs, device-aided therapies, non-pharmacological therapies, and CATs at the country-level.
MATERIALS AND METHODS
We undertook a pilot survey of health professionals involved in the care of patients with PD. The survey was sent by email to all members on the International Parkinson and Movement Disorders Society (MDS) mailing list on 4 June 2020 and again on 23 June 2020. Responses up to and including the 8 July 2020 are represented here. The survey was written in English and had 14 questions. It was reviewed in draft and modified by the MDS Epidemiology Study Group (ESG) and then endorsed by the MDS. The questionnaire can be accessed in the Supplementary Material.
Study design
The pilot study had two main objectives. Firstly, to investigate the availability of PD treatment in different countries and secondly, by world region. PD treatment was subdivided into medication, including levodopa, MAO-B inhibitors, dopamine agonists, COMT inhibitors and amantadine; CATs, such as Mucuna pruriens; non-pharmacological treatment, including physiotherapy, occupational therapy and speech and language therapy; and device-aided therapies, including DBS, apomorphine and LCIG. Access to these treatments was analysed in terms of availability, financial accessibility, and potential physical barriers to access.
Data analysis
Responses to the survey questions were tabulated and graphically illustrated using Microsoft Excel. Narrative comments submitted by respondents were reviewed and categorised. Survey responses were first organised according to the geographical regions used by the United Nations Statistics Division (UNSD). These are based on continental regions further subdivided into sub-regions and intermediary regions, to obtain greater homogeneity in sizes of population, demographic circumstances, and accuracy of demographic statistics. For this report, countries in Eastern Africa, Middle Africa, Southern Africa, and Western Africa were classified as Sub-Saharan Africa.
Responses were then organized according to the country income classifications for the World Bank’s 2020 fiscal year [11]. This classification divides countries’ economies into four income groups: high (HICs), upper middle (UMICs), lower middle (LMICs), and low income (LICs). This considers a country’s Gross National Income (GNI) per capita, which can be affected by economic growth, inflation, exchange rates and population. Classification thresholds were adjusted for inflation annually using the Special Drawing Rights (SDR) deflator [11]. In our analysis, LMICs and LICs were considered together due to the low number of responses from these income groups. Responses that did not provide a country or job title were excluded. Responses that had “patient” or “retired” as the job title were also excluded. This led to eight responses being excluded.
In the analysis of question 4 (Can you estimate how many patients with PD you see in a year?), question 5 (Which of the following PD medications are available in your country?), question 7 (How are medications funded?), question 9 (What proportion of your patients do you estimate use naturally occurring alternatives?), question 10 (What non-pharmacological therapies are available in your country for PD?), and question 11 (What advanced therapies are available in your country for PD?), the prevalence of availability was calculated based on the proportion of positive responses to the total number of responses from each country. Questions regarding availability of PD treatment were targeted at a national level. Responses to “other” options were analysed by two authors (JLY Cheong and ZHK Goh) using content analysis. Any responses that overlapped with available options were counted towards a response for that option. Question 8 (Which naturally occurring alternatives to medication are available in your country for PD (e.g., Mucuna pruriens)?) responses were analysed by two authors (JLY Cheong and ZHK Goh) also using content analysis. These responses were categorised into 4 main categories: Mucuna pruriens, cannabinoids, Ayurveda, and others. Question 6 (Estimate the proportion of your patients on the following medications) was analysed for frequency of use of conventional PD treatment. Non-responses to Question 6 were excluded from the analysis. Question 6 was targeted at the level of an individual’s clinical practice.
Analysis
Responses from HICs, UMICs and LMIC/LICs (3 groups) for different PD treatments were compared.
Responses were analysed for variability in reported availability by country. Countries with 5 or more responses were included in the analysis and categorised into HICs, UMICs, and LMIC/LICs. The reported availability of conventional PD medications, non-pharmacological treatment, and device-aided therapies were compared against other reported availabilities from countries within each income group to determine the coefficient of variation of each income group for each PD treatment. Microsoft Excel and GraphPad Prism Version 9.1.1 were used for the analyses.
RESULTS
Participants/demographics
The survey had a total of 360 participants (∼3.6% from a mailing list of ∼10,000). 352 valid responses were used (after 8 exclusions). Of these participants, 324 (92%) were medical doctors. The largest number of respondents were recorded from India 33/352 (9.3%), Brazil 26/352 (7.4%), and the USA 22/352 (6.3%) (Supplementary Material). There were 146 (41.5%) responses from HICs, 107 (30.4%) from UMICs, and 99 (28.1%) from LMICs/LICs (90 from LMICs and 9 from LICs).
The number of PD patients seen each year by health professionals decreased with national income. The proportion of respondents that saw > 200 patients with PD decreased from 57.9% in HICs to 30.7% in UMICs and 11.4% in LMICs/LICs (Table 1).
Table summarising availability of PD treatment by income group of countries
*Percentage of respondents who saw > 200 PD patients per year. **Percentage calculated as the proportion of positive responses compared to the total number of responses from each income group. ***Responses to the question regarding availability of CATs was qualitative, the accuracy of responses gathered may be different from that of responses to questions regarding the availability of other PD treatments, which were quantitative. DBS = deep brain stimulation. LCID = levodopa carbidopa intestinal gel.
Availability of conventional PD medications
Availability of conventional PD medications generally decreased across national income groups (Table 1). Of the drugs listed in the survey, levodopa was the most widely available. Responses across income groups reported 99% availability of levodopa. This was not the same for other PD medications. The availability of dopamine agonists decreased from 99.3% in HICs to 96.3% in UMICs and 80.8% among LMICs/LICs. Similarly, the availability of MAO-B inhibitors decreased from 99.3% in HICs to 88.8% in UMICs and 66.7% in LMICs/LICs. COMT inhibitors were reported to be available in 97.3% of responses in HICs, 83.2% of those from UMICs and 52.5% of those from LMICs/LICs. Availability of amantadine decreased from 96.6% in HICs to 86.0% in UMICs and 67.7% in LMICs/LICs (Fig. 1).
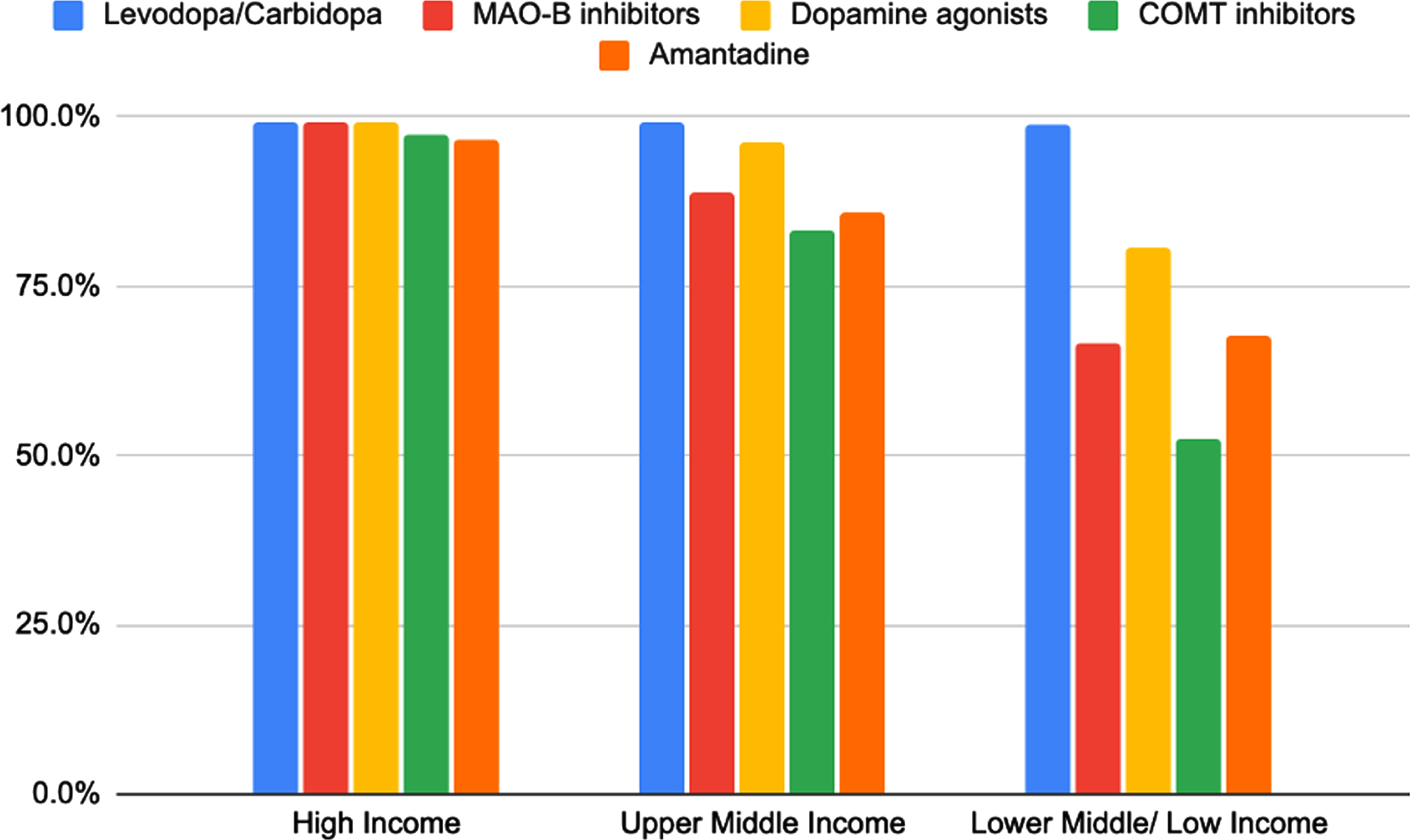
Figure of availability of conventional PD medications by income group of countries. Levodopa: HICs N = 145/146 (99.3%), UMICs N = 106/107 (99.1%), LMICs/LICs N = 98/99 (99.0%). MAO-B inhibitors: HICs N = 145/146 (99.3%), UMICs N = 95/107 (88.8%), LMICs/LICs N = 66/99 (66.7%). Dopamine agonists: HICs N = 145/146 (99.3%), UMICs N = 103/107 (96.3%), LMICs/LICs N = 80/99 (66.7%). COMT inhibitors: HICs N = 142/146 (97.3%), UMICs N = 89/107 (83.2%), LMICs/LICs N = 52/99 (52.5%). Amantadine: HICs N = 141/146 (96.6%), UMICs N = 92/107 (86.0%), LMICs/LICs N = 67/99 (67.7%).
Frequency of use of conventional PD medications were also found to decrease with income group (Table 2). 80.0% of responses from HICs indicated that > 75% of their patients were on levodopa compared to 69.8% among UMICs and 68.4% among LMICs/LICs. 9.0% of responses from HICs indicated that > 75% of their patients were on dopamine agonists compared to 16.5% among UMICs and 6.3% among LMICs/LICs. 6.9% of responses from HICs indicated that > 75% of their patients were on MAO-B inhibitors, compared to 4.2% among UMICs and 4.55% among LMICs/LICs. 1.4% of responses from HICs indicated that > 75% of their patients were on COMT inhibitors compared to 2.3% among UMICs and 1.9% among LMICs/LICs.
Table summarising frequency of use of conventional PD treatments according to income group
Percentages were calculated based on the proportion of positive responses for each option over the total number of responses.
The variability in the reported availability of conventional PD medications was highest among LMIC/LICs and lowest among HICs (Table 3). The coefficient of variation (CoV) for levodopa was 0.0% among HICs, 0.0% among UMICs, and 5.2% among LMIC/LICs. Similarly for MAO-B inhibitors, the CoV was 0.0% for HICs, 36.9% for UMICs, and 48.4% for LMIC/LICs. The CoV for dopamine agonists was 2.6% among HICs, 5.5% among UMICs, and 14.3% among LMIC/LICs. For COMT inhibitors, the CoV was 2.2% among HICs, 12.9% among UMICs, and 64.4% among LMIC/LICs. The CoV for amantadine was found to be 4.5% among HICs, 37.2% among UMICs, and 54.2% among LMIC/LICs. This gives an indication on possible discrepancies in availability of PD medications within countries.
Table showing coefficient of variation for each PD treatment according to income group
Countries with ≥5 responses were included. Reported availability was calculated for each country as the proportion of positive responses to total number of responses in each country. Coefficient of variation was calculated via GraphPad Prism Version 9.1.1.
Availability of CATs for PD
Availability of CATs for PD was also found to decrease with countries’ income. 37.0% of responses in HICs reported availability of CATs for PD, compared to 31.8% in UMICs and 19.2% in LMICs/LICs. Mucuna pruriens was the most frequently referenced CAT for PD across all income groups (Fig. 2). At least 85% of responses from each income group reported that fewer than 25% of PD patients use naturally occurring alternatives. Cannabinoids were most common among North, Central and South America, while Ayurveda are herbal therapies used in Southern Asia. Mucuna pruriens was more commonly used in North and South America, and Western and Southern Europe. Other CATs reported were gingko in South-east Asia, Australia and New Zealand, and Central America and caffeine in South-east Asia and East Asia.
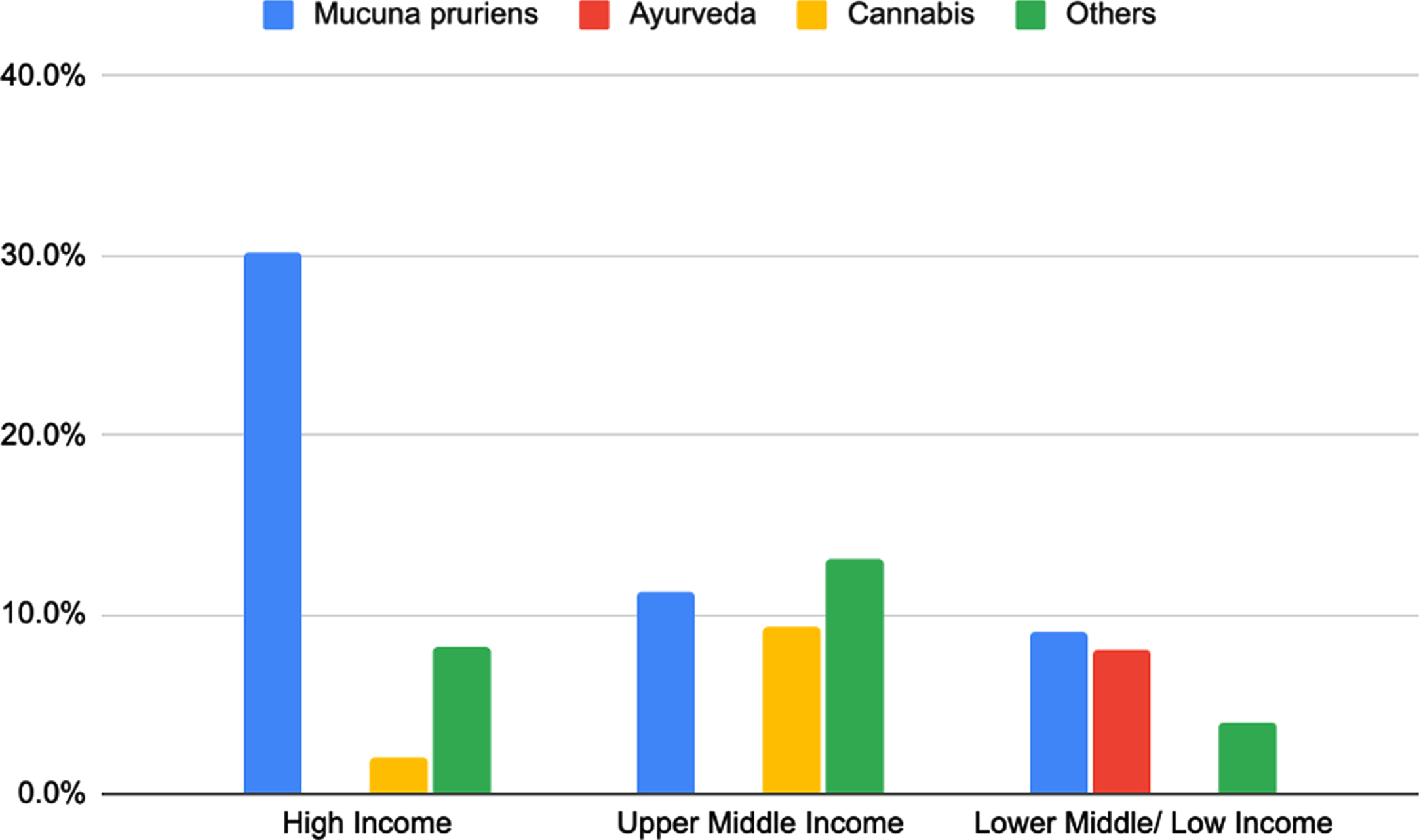
Figure of CATs for PD available according to income group of countries. Mucuna pruriens: HICs N = 44/146 (30.1%), UMICs N = 12/107 (11.2%), LMICs/LICs N = 9/99 (9.10%). Ayurveda: HICs N = 0/146 (0.00%), UMICs N = 0/107 (0.00%), LMICs/LICs N = 8/99 (8.10%). Cannabis: HICs N = 3/146 (2.10%), UMICs N = 10/107 (9.30%), LMICs/LICs N = 0/99 (0.00%). Others: HICs N = 12/146 (8.20%), UMICs N = 14/107 (13.1%), LMICs/LICs N = 4/99 (4.00%).
Availability of non-pharmacological PD treatment
Non-pharmacological treatments were reported to be widely available across all income groups, but there was no clear association between the availability of non-pharmacological therapies and national income. 99.1% of responses from UMICs indicate availability of non-pharmacological treatment for PD. This was followed by HICs at 98.6% and LMICs/LICs at 97.0%. Physiotherapy was the most available non-pharmacological therapy across all income groups (> 90%) and there was no clear difference in availability across income groups. Looking specifically at occupational therapy and SALT among LMICs/LICs, there was a significant decrease in proportion of availability of occupation therapy (49.5%) and SALT (55.6%) compared to HICs (80.1% and 84.9% respectively) and UMICs (78.5% and 87.9% respectively) (Fig. 3).
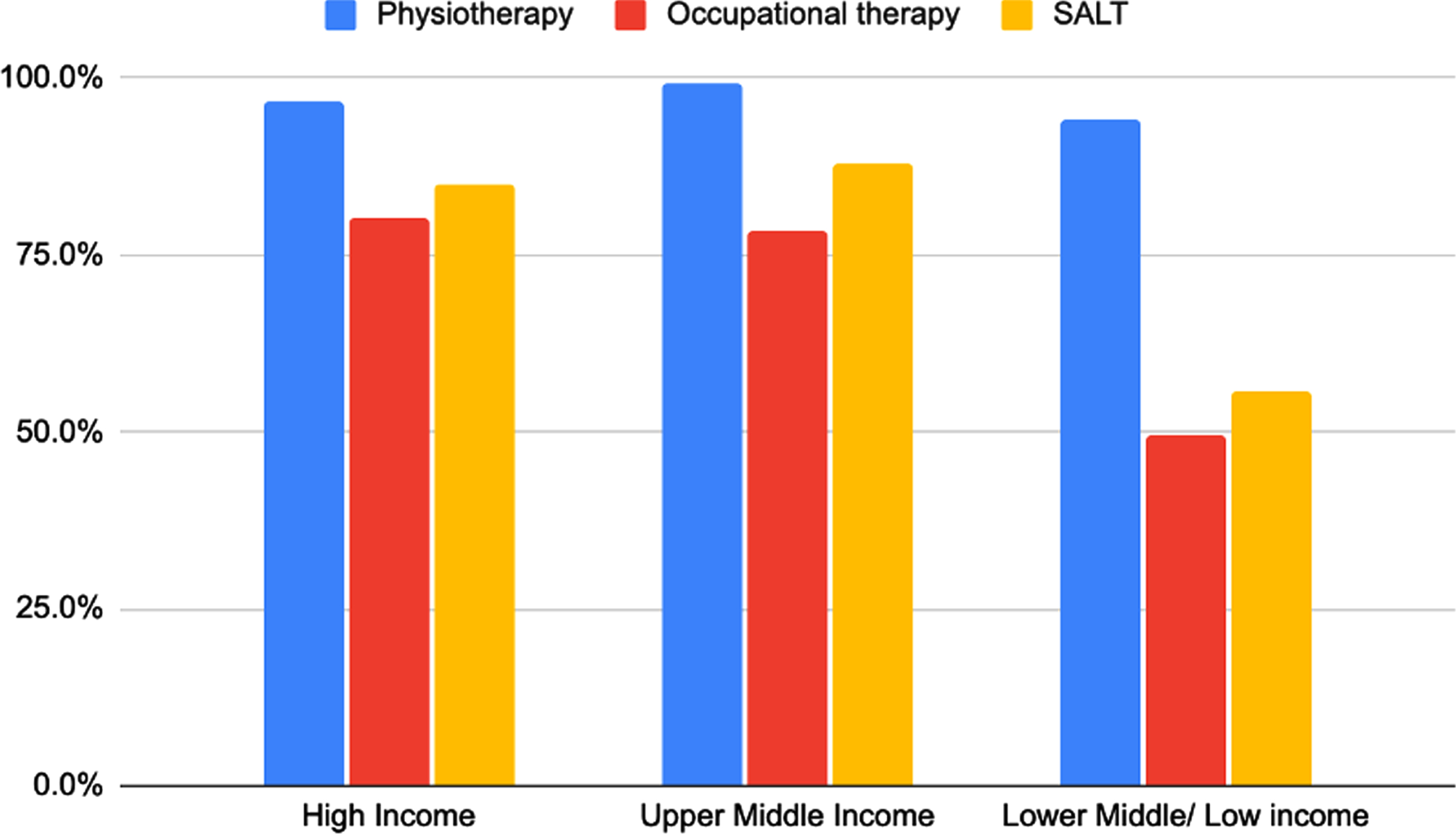
Figure of non-pharmacological therapies available according to income group of countries. Physiotherapy: HICS N = 141/146 (96.6%), UMICs N = 106/107 (99.1%), LMICs/LICs N = 93/ 99 (93.9%). Occupational therapy: HICs N = 117/146 (80.1%), UMICs N = 84/107 (78.5%), LMICs/LICs N = 49/99 (49.5%). SALT: HICs N = 124/146 (84.9%), UMICs N = 94/107 (87.9%), LMICs/LICs N = 55/99 (55.6%).
According to additional comments by respondents, nutritional therapy and psychological therapy were more common among Northern (6.1%), Central (15.4%), and South (7.3%) Americas, and Northern Europe (13.3%). Various exercise therapies such as yoga and tai chi were reported among respondents from South-eastern (2 responses) and Southern Asia (2 responses) and Australia and New Zealand (3 responses). Dance and art therapies were reported in Australia and New Zealand (3 responses) and South America (3 responses). Acupuncture was reportedly used in Eastern Asia (1 response). It was noted that among respondents from Africa, non-pharmacological therapies except physiotherapy were limited.
The variability in the reported availability of non-pharmacological treatment was generally highest among LMIC/LICs except for physiotherapy. For SALT, the CoV increased from 28.9% among HICs to 37.3% among UMICs and 66.7% among LMIC/LICs. The CoV for occupational therapy was 29.9% among HICs, 25.7% among UMICs, and 43.7% among LMIC/LICs. The CoV for physiotherapy was similar across income groups with 5.9% among HICs, 5.7% among UMICs, and 6.0% among LMIC/LICs (Table 3).
Availability of device-aided PD therapies
The availability of device-aided therapies decreased with national income. 100% of responses from HICs reported availability of device-aided therapies, compared to 92.5% among UMICs and 57.6% among
LMICs/LICs. The availability of DBS decreased from 94.5% in high income countries to 91.6% in UMICs and 52.5% in LMICs/LICs. Similarly, availability of apomorphine decreased from 76.0% among HICs to 39.3% among UMICs and 27.3% among LMICs/LICs. The availability of levodopa-carbidopa intestinal gel showed a drop from 95.2% in HICs to 21.5% in UMICs and 8.1% among LMICs/LICs (Fig. 4). Deep brain stimulation (DBS) was the most available device-aided therapy among UMICs and LMICs/LICs at 91.6% and 52.5% respectively. Levodopa-carbidopa intestinal gel was the most available device-aided therapy in HICs (95.2%), but DBS was similarly available (94.5%).
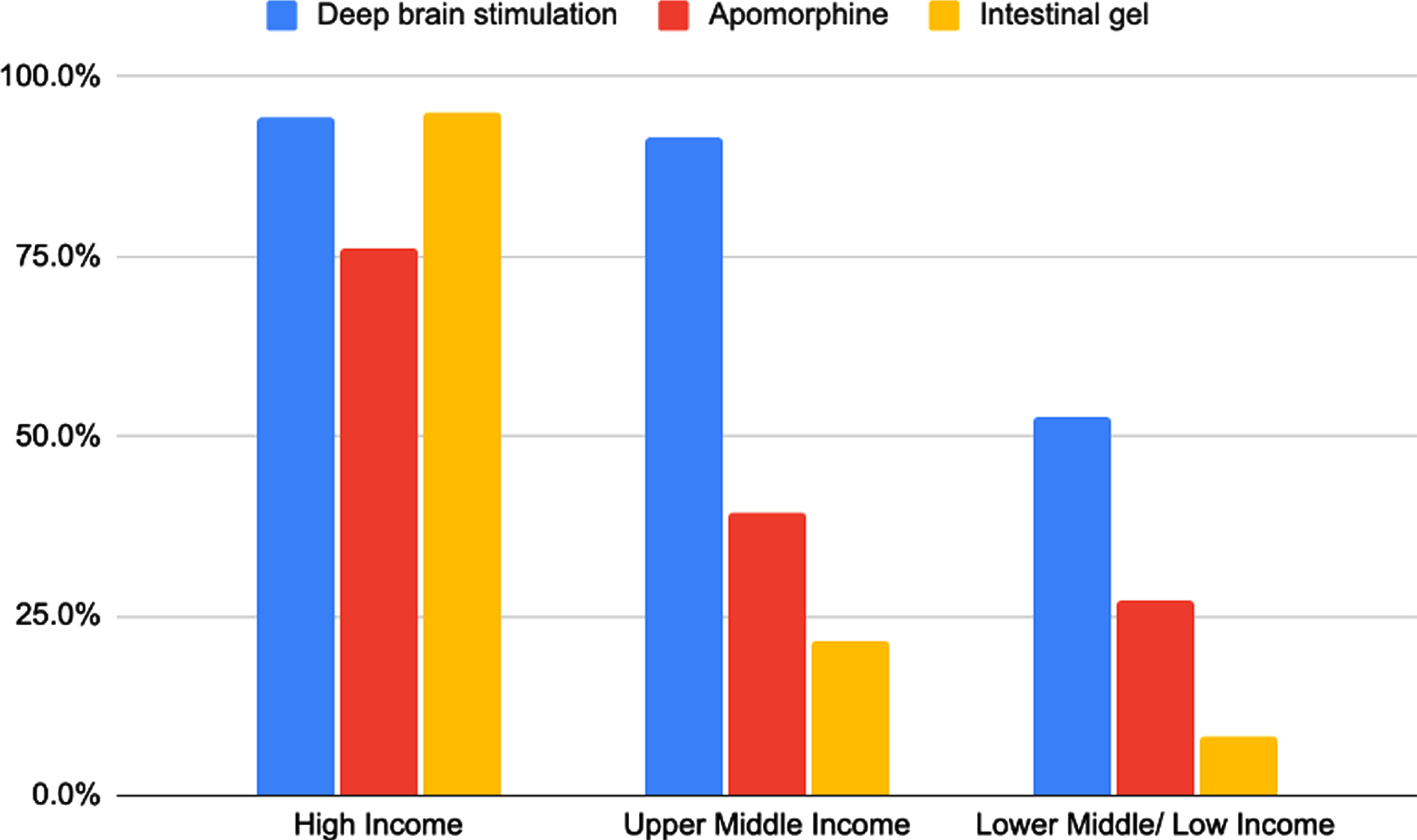
Figure of device-aided therapies available according to income group of countries. Deep brain stimulation: HICs N = 138/146 (94.5%), UMICs N = 98/107 (91.6%), LMICs/LICs N = 52/99 (52.5%). Apomorphine: HICs N = 111/146 (76%), UMICs N = 42/107 (39.3%), LMICs/LICs N = 27/99 (27.3%). Levodopa carbidopa intestinal gel: HICs N = 139/146 (95.2%), UMICs N = 23/107 (21.5%), LMICs/LICs N = 8/99 (8.1%).
The variability in the reported availability of device-aided therapies was highest among LMIC/LICs. For DBS, the CoV for HICs was 14.0% and 5.9% among UMICs, compared to 82.3% among LMIC/LICs. The CoV for apomorphine similarly was 40.8% among HICs, 94.4% among UMICs, and 133% among LMIC/LICs. The CoV for LCIG was 0.00% among HICs, 123% among UMICs, and 156% among LMIC/LICs (Table 3).
Funding of PD medications
The availability of fully government funded PD medication decreased drastically with income from 50.0% in HICs to 33.6% in UMICs and 3.0% in LMICs/LICs (Fig. 5). 79.8% of responses from LMICs/LICs reported that patients fully pay for their PD medications.
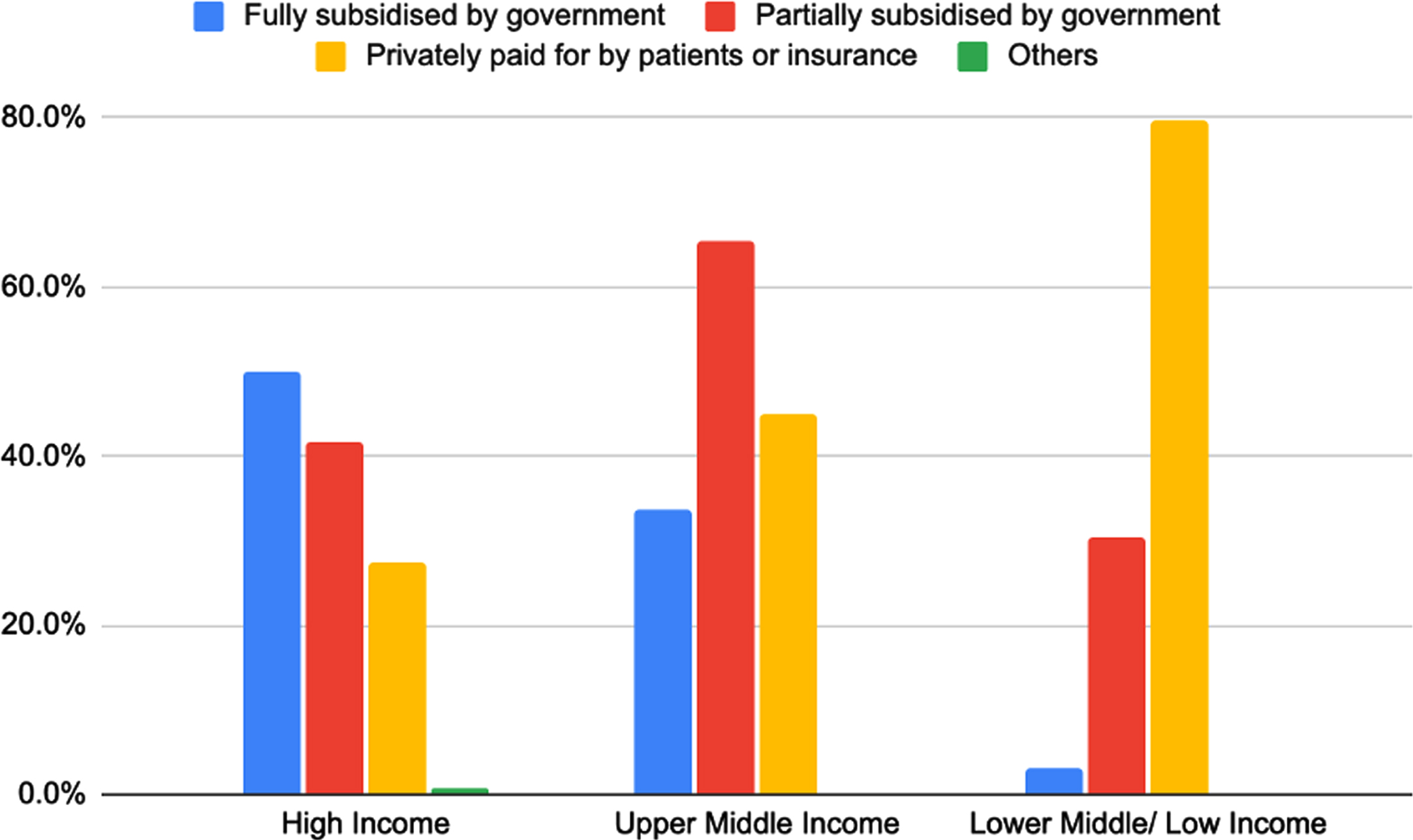
Figure of type of funding for PD treatment according to income group of countries. Fully subsidised by government: HICs N = 73/146 (50.0%), UMICs N = 36/107 (33.6%), LMICs/LICs N = 3/99 (3.0%). Partially subsidised by government: HICs N = 61/146 (41.8%), UMICs N = 70/107 (65.4%), LMICs/LICs N = 30/99 (30.3%). Privately paid for by patients/insurance: HICs N = 40/146 (27.4%), UMICs N = 48/107 (44.9%), LMICs/LICs N = 79/99 (79.8%).
DISCUSSION
This pilot study represents a first attempt to obtain a snapshot of global availability of treatment of PD and it highlights important gaps in availability of therapies. Given the established benefits PD treatments have on relieving symptoms and improving quality of life for PD patients, it is concerning that a significant proportion of patients, particularly in lower income countries, may not be receiving treatment. Furthermore, this is framed against an alleged doubling of the global burden of PD over the last 3 decades [5]. Whilst this observation requires further examination, it is striking that it is in LMICs/LICs where burden appears to be increasing the fastest and these are also the countries with the lowest availability of efficacious, generic treatment.
In this survey, the availability of conventional, CATs and device-aided PD therapy decreased from high to low income countries. According to the WHO Atlas (second edition), which provides data on the availability of levodopa in primary and secondary health settings, levodopa is only available in 12.5% of Africa compared to 79.1% in Europe [9]. While our survey results indicated a much higher percentage availability of PD drugs (99% availability of levodopa across national income groups), this is likely to reflect availability at the country-level, but not the distribution of treatments within a country. Our survey respondents may have tended to be from larger medical centres with greater availability of treatments and responses may not reflect availability in rural areas.
The frequency of use of levodopa was generally high across income groups, which may have been associated with levodopa being on the WHO list of Essential Medicines. However, among other conventional PD medications, it was interesting to note the patterns in frequency of use among countries of different income groups. Among MAO-B inhibitors and COMT inhibitors, the frequency of use was highest among HICs, while majority of responses from LMIC/LICs indicated that they were used by < 25% of their patients. This may reflect the lower availability of MAO-B inhibitors and COMT inhibitors due to their higher costs [12]. Interestingly the frequency of use of dopamine agonists appeared higher among UMICs and LMICs/LICs compared to HICs. A possible reason for this might have to do with the availability of ergot dopamine agonists as an essential drug for infertility in lower income countries [13, 14]. However, non-ergot dopamine agonists and COMT inhibitors are not widely available and generally remain a costly option for PD treatment in certain countries [13, 16]. Further investigation may be useful to confirm this trend and identify possible reasons for it. Amantadine appeared to have a lower frequency of use among HICs and UMICs compared to LMIC/LICs, despite being relatively more costly compared to other available conventional PD medications [14]. Its lower frequency of use in HICs may possibly be affected by its main use as an adjuvant therapy to other conventional PD treatments or in the management of dyskinesias.
With regards to non-pharmacological therapies for PD, our survey did not identify a clear trend between the availability of therapies and national income. Our findings were similar to that which has been observed from other studies. A study from Brazil reported that 43.5% of PD patients received physiotherapy, while 75.2% of patients did not engage in other therapies such as occupational and speech therapy [17]. In comparison, a study on the use of physiotherapy for PD patients in the Netherlands reported that 60% of PD patients received physiotherapy [18]. Potential determinants go beyond national wealth and the prevalence of health education, support for self-management and the structure of health systems are also important.
Unsurprisingly, device-aided therapies were less available in lower income regions. As with other aspects of PD management, even in countries where device-aided therapies are widely available, these are not always accessible to patients. Contraindications to therapy include social considerations, such as support at home to manage equipment [3] which are likely to be more scarcely available in low income countries. A study in Denmark found that patients living west of the ‘Great Belt’ were less likely to have started device-aided therapy than those on the east side. 10% of PD patients started device-aided therapy in the Capital region compared to 7% in Central Jutland from 2008 to 2016 [19]. This was thought to be due to differences in the availability of movement disorder specialists in each region, infrastructure, and access to public transportation, particularly for patients in rural regions [19]. Therefore, despite the seemingly greater availability of device-aided therapies in HICs, this may not always reflect greater availability for patients in specific regions.
In the present study, the use of CATs for PD decreased with decreasing national income. This may be due to the high cost of CATs for PD. A Korean study reported that the average monthly cost of CATs for PD was 102.30 USD compared to 72.80 USD for conventional PD medications [20]. A study in Singapore similarly reported that the cost of CATs for PD was 12.4 times higher than that of conventional PD medications [21]. In Sweden, 20% of PD patients reported spending 50 euros on CATs for PD [22]. In addition, while the overall use of CATs for PD reported in our results was low, this may not be an accurate reflection on the actual use of these therapies as patients may not have reported use to their physicians. For example, studies in Singapore and Argentina suggest that only 16% and 57% of PD patients respectively inform their physicians about their use of CATs [21, 23]. Reasons include the assumption that physicians do not need to know and fear of a negative response [24]. The use of CATs for PD may similarly have been underreported in our survey.
Variability in availability of PD treatment
Our survey results suggest that there is a greater variability in the reported availability of various PD treatments in LMIC/LICs, most notably among device-aided therapies. This may reflect discrepancies in availability within countries due to geographical barriers, where therapies may only be available in more urban parts of a country or financial challenges, where patients may not be able to afford the treatment. The potential reasons for the observed increased variability among LMIC/LICs and treatment gap will be discussed in more detail in the following sections.
Financial burden of PD treatment
Our survey suggested that the availability of fully government-funded PD medication decreased drastically with national income and that a large proportion of patients in LMICs/LICs pay in full for their PD medications. Various studies on the cost of PD medication in African countries have found that patients spend a significant proportion of their monthly income on PD medications at 33 USD in Tanzania, 10 USD in Ethiopia, and 48 USD in Kenya [25–27]. In addition, patients are also required to pay for consultations and transportation costs. In a region where almost 50% of the population live on less than 1.25 USD a day [28], patients may not be able to afford medications, even if they are available. In contrast, PD medications are effectively reimbursed in most European countries through their respective national healthcare systems, making treatment more available, but also widening the treatment gap between higher and lower income countries [29].
Limitations
Our study has several limitations. Firstly, we received a small number of responses from LMICs and especially LICs, which may have affected the accuracy of our findings from this group. As countries were not evenly represented, this may have skewed the results towards the situation in countries with more responses. This may be in part due to there being a lower proportion of specialist PD healthcare professionals or MDS members in these countries. The WHO Atlas reported that there were 0.03 neurologists per 100 000 population in Africa compared to 4.84 in Europe [9]. Moreover, because respondents were more likely to be those from larger medical centres in urban areas, survey responses are unlikely to capture availability at a local level or in rural settings.
Secondly, as our survey results are reported by healthcare professionals, there may be incongruence between prescription of PD treatment and patients’ use of treatment due to other factors affecting access, such as financial limitations and access to local pharmacies, particularly in rural areas, as highlighted above, leading to treatment gaps within countries.
Thirdly, our survey results were based on the knowledge base of the respondents rather than national formularies and may not have been entirely accurate. This may be particularly relevant to questions such as the CATs available in countries and may not reflect the true availability of such treatment options.
Fourthly, our survey took place at the height of the COVID pandemic in June and July 2020. Countries’ governments at the time had been channeling efforts towards the fight against the pandemic and many healthcare professionals were redirected to support these efforts. Healthcare professionals may therefore have been occupied and less likely to have been able to find the time to complete our survey. This may have contributed towards the lower response rate to our survey. A follow-up survey may therefore be important to increase response rates and provide a more accurate picture of availability of PD treatment in countries.
Further studies
This study provides a first snapshot of the global distribution of availability of different types of PD treatments. In view of the treatment gaps identified between and within countries, similar studies on a country or regional level would be beneficial in analysing availability of and distribution of PD treatment in greater detail within countries, to develop a clearer picture of the extent of problems with availability and identify barriers to access. Ultimately, these studies would be helpful in developing region-specific solutions to improving availability of PD medications for patients.
With regards to funding of PD medications, our survey question did not distinguish between patients who funded their medications out of their own pockets and those who funded them through private insurance. Future surveys on availability of PD medications could consider distinguishing between these forms of private funding to provide a more accurate depiction of the financial burden on patients.
PD is a complex neurodegenerative disease with both motor and non-motor complications. Management of non-motor complications such as depression, sleep disorders, and dementia have not been well explored in our present survey and further studies investigating the extent of these complications and availability of options to manage them would be beneficial.
CONCLUSION
In conclusion, this study, as an overview of global availability of PD medication, highlights that there are significant gaps in availability of various PD treatments in many countries. Of concern from our results is that this decrease in availability is often associated with a decrease in income of a country. This is compounded by the fact that most PD patients in LICs are required to fully fund their own treatment so that even where medications are available within a country, they may not be available to the people who need them. Given the fact that PD prevalence among LICs may have risen at a higher rate than that in HICs despite stagnation in availability of PD medications in those countries, this suggests that the problem is only going to increase in magnitude in the coming years. Improving availability of PD medications should be a priority, so that patients receive the treatment they require to improve their quality of life.
