Abstract
Background:
Psychotic symptoms are highly frequent in Parkinson’s disease (PD) patients and are associated with poor prognosis. They include hallucinations, delusions, and minor psychotic phenomena, including sense of presence, passage hallucinations, and illusions.
Objective:
To evaluate the frequency of psychosis in PD patients.
Methods:
A systematic review and meta-analysis of clinical trials, prospective and retrospective cohort studies, case-control studies, and cross-sectional studies reporting the frequency of psychosis, hallucinations, and delusions in PD.
Results:
Electronic database search wielded 3536 articles, an additional 91 were identified through citation chaining. Of these, 163 were fully inspected, 57 removed, and 106 included as relevant for neuropsychiatric events frequency, with 32 meeting our inclusion criteria (psychosis and/or specific psychotic phenomena). The pooled frequency of psychosis was 20.7% (95% CI 14.5 to 28.6; I2 = 94%, 15 studies; combined n = 2919). None of the pre-defined meta-regressions or subgroup analyses were statistically significant or helped explain the statistical heterogeneity. The pooled frequency of any form of hallucination was 21.6% (95% CI 14.7 to 30.6; I2 = 95%; 18 studies; combined n = 3161). Duration of PD at baseline and mean baseline Hoehn & Yahr stage helped explain the statistical heterogeneity in the meta-analysis of hallucinations.
Conclusion:
Based on the available evidence, around a fifth of PD patients experience psychosis or hallucinations. The risk of developing hallucinations is likely moderated by the disease duration, Hoehn & Yahr stage, and the cognitive status.
Keywords
INTRODUCTION
Parkinson’s disease (PD) is a synucleinopathy that affects millions of people worldwide and leads to progressive disability [1]. PD has been mainly defined and diagnosed on the basis of the motor impairment it causes, but it has become increasingly recognized that non-motor symptoms are a prominent feature of PD [2, 3].
Psychotic symptoms are frequent in PD patients [4] and are associated with poor prognosis such as greater physical disability, cognitive and affective dysfunction, caregiver distress, nursing home placement, and mortality [5–7].
The term psychosis has had several different definitions among the last decades. The clinical profile of psychosis in PD is different from other psychotic disorders, such as schizophrenia or mood disorders with psychotic features. Therefore, criteria applied to other psychiatric illnesses (like DSM) have limited utility for describing the diversity of psychotic phenomena in PD.
Psychotic symptoms in PD include hallucinations and delusions, which, simplistically, define psychosis. But there are also minor psychotic phenomena, which include sense of presence, passage hallucinations, and illusions.
There were no standardized diagnostic criteria for psychosis associated with PD until 2007 when the National Institutes of Health (NIH) National Institute of Neurological Disorders and Stroke (NINDS)/National Institute of Mental Health (NIMH) proposed diagnostic criteria for PD-psychosis, including minor symptoms, to better define and standardize the unique features of PD-psychosis [8]. This workgroup also presented and clarified the definition of psychotic symptoms. Delusions as false, fixed, idiosyncratic beliefs that are maintained despite evidence to the contrary and hallucinations as abnormal perceptions without a physical stimulus that can involve any sensory modality and may be simple or complex in form. However, they have also included presence hallucinations and illusions, which are not a part of the description of other psychotic disorders. Presence hallucinations consist of the experience that someone is present when nobody is there. These are not strictly hallucinations in that the sensory modality is not clear but are often classified as hallucinations. Illusions are misperceptions of real stimuli that are often visual in nature [8].
Estimates of the frequency of PD-psychosis vary widely among studies, with frequencies ranging from 20% up to 70% [9, 10]. There are few systematic reviews on epidemiology of psychosis in PD, most focusing on specific psychotic symptoms like delusions [11, 12] or visual and auditory hallucinations and minor psychotic phenomena but lacking focus on other psychotic symptoms [13].
Updated investigation on PD-psychosis epidemiology and characterization is mandatory to promote adequate assessment and management of psychosis and to prevent negative impact on PD prognosis.
The aim of the current study was to evaluate the frequency of psychosis and different psychotic phenomena (delusions, visual/olfactory/auditory/tactile hallucinations, illusions, sense of presence, minor psychotic phenomena) in PD patients.
METHODS
We conducted a systematic review evaluating the frequency of psychosis and different psychotic phenomena in PD patients.
Search and inclusion criteria
The literature search was performed by one of the authors (GD); the screening and decisions on inclusion were performed independently by three of the authors (IC, LP, and CS); data extraction and quality control were performed independently by two of the authors (IC and CS) and disagreements on screening or data extraction were resolved by discussion with two of the authors (GD and IC). This systematic review followed the reporting principles of PRISMA. The protocol was registered in PROSPERO as Neuropsychiatric events in Parkinson's Disease: a systematic review and meta-analysis [CDR42018118863].
The search strategy developed for all databases combined the terms Parkinson* OR Parkinsonism with psychosis, hallucinations, illusions, delusions, sense of presence, minor psychotic phenomena and were adapted from previous Cochrane systematic reviews of interventions for this condition, and The Cochrane Highly Sensitive Search Strategy for identifying RCTs in MEDLINE: sensitivity- and precision-maximizing version (2008 revision) was used. The search strategy was restricted to human participants only. All terms were searched as free-text and controlled vocabulary terms (i.e., Medical Subjects Headings (MeSH)).
The bibliographic databases MEDLINE, EMBASE, and the Cochrane Central Register of Controlled Trials (CENTRAL), and the clinical trials registries the WHO International Clinical Trials Registry Platform, and ClinicalTrials.gov, were searched for relevant citations for inclusion. Additionally, the reference lists from the identified articles were cross-checked for further potentially eligible studies.
Studies were included in this review if they 1) were clinical trials, prospective and retrospective cohort studies, case-control studies, or cross-sectional studies; 2) peer-reviewed papers written in English, Spanish, or French; 3) reported the frequency of psychosis, hallucinations, delusions, illusions, sense of presence, and/or minor psychotic phenomena; 4) diagnosis was made by clinical, structured, or semi-structured interview or through the administration of validated scales administered by experienced clinicians and not only by self-report questionnaire; and 5) included 25 or more participants.
The last search was performed in December 2020.
Data extraction and outcomes
Three reviewers independently screened the titles and abstracts yielded by the searches against the inclusion criteria and independently read the full text reports and determined if they met the inclusion criteria. Disagreements were resolved through discussion, with a fourth reviewer serving as the final arbitrator, and the authors sought additional information from the study authors, if needed, to resolve questions regarding eligibility.
Three reviewers then extracted data from the individual studies identified for inclusion onto a pre-piloted Microsoft Excel spreadsheet. This information included: authors; journal; year of publication; study design; setting; population demographics; sample size; intervention and control conditions; primary outcome; secondary outcomes; methodology for diagnosis (clinical or/and (semi)structured interview).
Data analysis
The analysis was performed in R version 4.0.3 using the meta package [14]. All outcomes were binary. Therefore, we calculated the pooled proportion of events was calculated with 95% confidence intervals, using a Sidik-Jonkman random effect models [15]. Statistical heterogeneity was reported using I2 [16]. To assess the robustness of the results and to identify potential methodological biases and subpopulations in which outcomes differed, we also conducted a series of a priori defined meta-regressions (percentage of male participants, mean disease duration, mean age onset and, mean disease stage (H&Y) and subgroup analyses (cognitive status, the methodology used for psychosis diagnosis and the study design).
To investigate the possibility of publication bias we created a funnel plot and conducted Egger’s test [17] for the primary outcome.
RESULTS
The electronic database search wielded 3536 citations, with an additional 91 were identified through citation chaining. Of these, 163 were fully inspected, 57 removed and 106 included as relevant for neuropsychiatric events prevalence. Of those, 32 met the inclusion criteria for our analysis (Fig. 1).
Search results flowchart.
Overall, 32 studies were included after full-text analysis (overview of included studies is provided in the Supplementary Material), all of which were published between 1996 and 2018. Fifteen studies with a total of 2919 patients were included for the global frequency of psychosis calculation, 18 studies for hallucinations frequency calculation, 8 studies for delusions frequency calculation, 4 studies for illusions calculation, 5 studies for sense of presence calculation, and 4 studies for minor psychotic phenomena frequency calculations. Studies that used vague diagnostic criteria, published as an abstract or poster or conference paper only, with incomplete data, duplicated publications and methodologically weak were excluded.
Risk of bias
Due to the different study designs included in our review, we assessed the risk of bias using two methods, namely the Newcastle-Ottawa Scale (NOS) in case control studies and cohort studies and the Quality Assessment Checklist for Prevalence Studies (adapted from Hoy et al.) for prevalence studies. Studies were considered of high quality on the NOS if these had more than 4 points. Full data are provided in the Supplementary Material.
Primary outcome: global frequency of psychosis
The pooled global frequency of psychosis was 20.7% (95% CI 14.5 to 28.6; I2 = 94%; 15 studies; combined n = 2919; Fig. 2). Regarding the meta-regressions conducted, we found that none of the regressors were statistically significant. Additionally, none of the regressors were able to explain the statistical heterogeneity in the analysis. Regarding the subgroup analyses conducted, we found that none of the subgroups were statistically significant (Table 1).
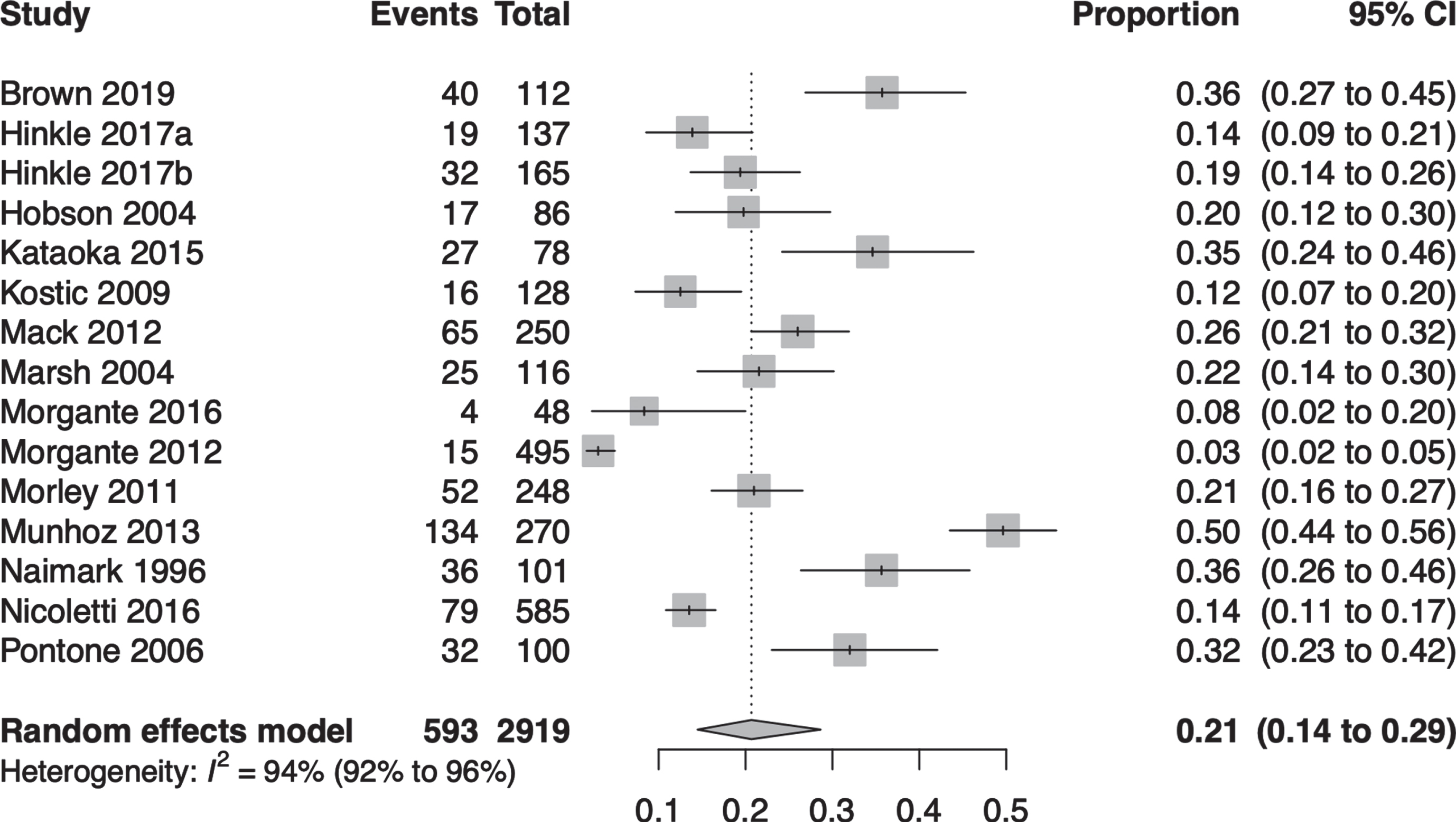
Pooled global frequency of psychosis.
Meta-regressions and subgroup analyses for frequency of psychosis
We found no evidence of publication bias (p value for linear test = 0.20). The funnel plot for this outcome can be found in the Supplementary Material.
Secondary outcomes: hallucinations, delusions, illusions, sense of presence, and minor psychotic phenomena
The full details about the secondary outcomes can be seen in Table 2.
Secondary outcomes
The pooled global frequency of any form of hallucination was 21.6% (95% CI 14.7 to 30.6; I2 = 95%; 18 studies; combined n = 3161; Fig. 3). Regarding the meta-regressions conducted, we found that none of the regressors were statistically significant. However, the mean baseline Hoehn & Yahr stage and the mean baseline duration of PD explained 13.53% and 17.43% of the model’s statistical heterogeneity, respectively. Subgroup analysis based on cognitive status was statistically significant (p value for interaction), while based on diagnosis method was not (Table 3).
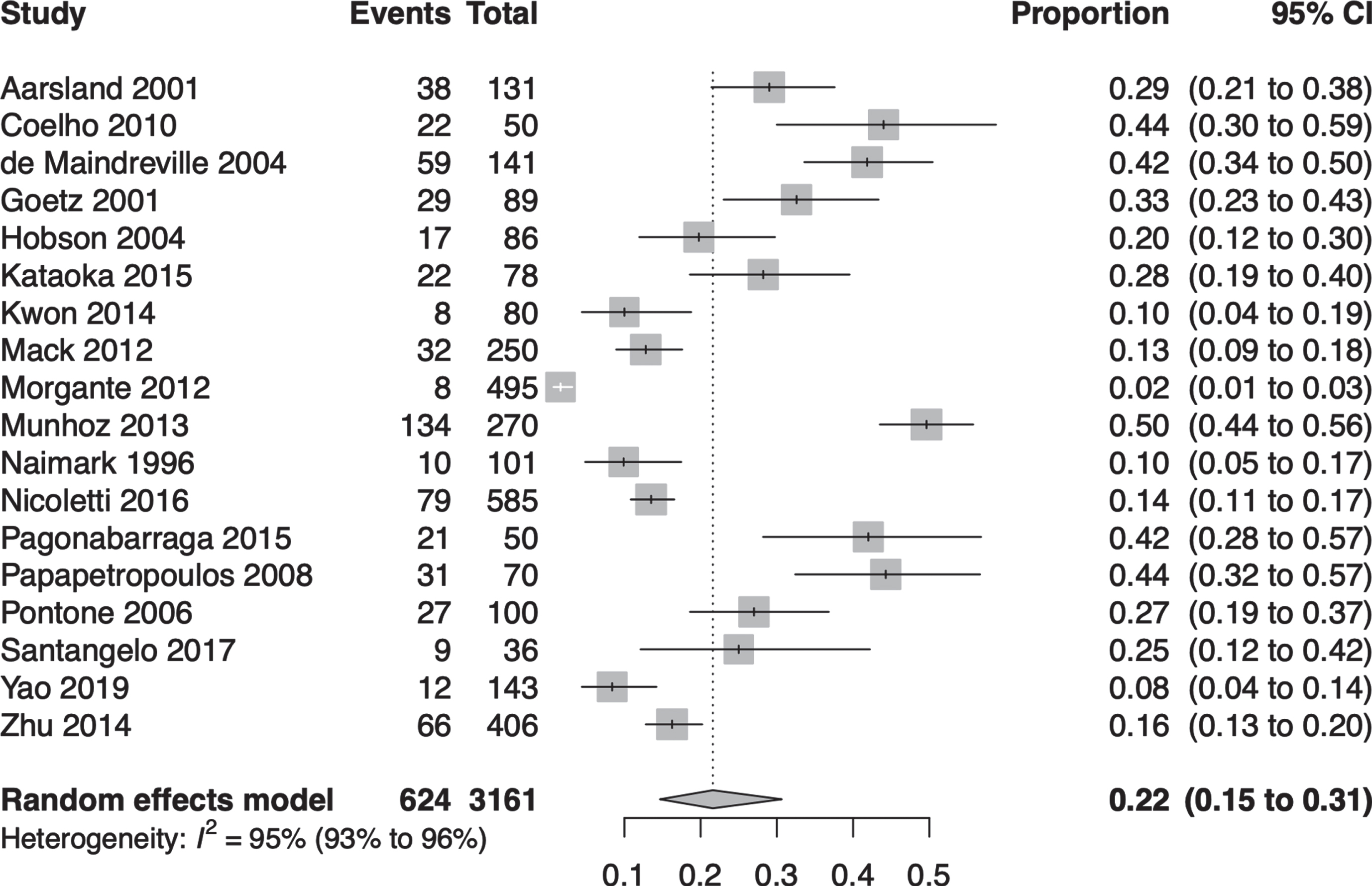
Pooled global frequency of hallucinations.
Meta-regressions and subgroup analyses for frequency of hallucinations
Regarding the pooled estimated frequency of the different forms of hallucinations was 16.5% for visual hallucinations, 7.1% for auditory hallucinations, 3.3% for olfactory hallucinations, and 2.6% for tactile hallucinations. The pooled estimated frequency of delusions was 6.1%. The pooled estimated frequency of illusions was 11.5%. The pooled estimated frequency of sense of presence was 30.2%. The pooled estimated frequency of minor psychotic phenomena was 23.6%.
DISCUSSION
This is the most complete systematic review on frequency of PD-psychosis, focusing frequency of psychosis as a whole and specific psychotic symptoms, and to the best of our knowledge the only available meta-analysis.
Although estimates of the frequency of PD-psychosis vary widely among studies [9], the global frequency of psychosis in this meta-analysis was 20.7%.
The variability in reported frequency of PD-psychosis may be attributed to use of different inclusion criteria in PD population, different criteria to define psychosis and inconsistencies in which psychotic phenomena were examined as well as the methodology used for psychosis diagnosis. Inclusion criteria in PD samples differ in PD duration, H&Y stage, age onset and cognitive status could possibly influence the psychosis frequency.
We conducted a meta-regression analysis to investigate the relationship between psychosis frequency and the study inclusion criteria namely age at baseline, sex, age of PD onset, duration disease, and disease stage (Hoehn & Yahr stage), and we found that none of the regressors were statistically significant (Table 2). The global frequency of psychosis was not evaluated in advanced stage patients and the only study that included advanced-stage patients only evaluated the frequency of visual hallucinations [18]. Our results contrast with previous studies that found the frequency of psychosis was associated with earlier age onset of PD and a longer PD duration [19]. Regarding the subgroup analyses conducted, the methodology used for psychosis diagnosis (using a clinical interview or/and (semi)structured interview versus just applying a rating scale) and cognitive status (demented versus non-demented) were not statistically significant (Table 2) which may be partially explained by the greatest difficulty in diagnosing psychosis in patients with dementia. The last aspect was somewhat surprising since prevalence studies consistently found hallucinations as well as delusions to be significantly more frequent in demented than non-demented PD patients [19–21] and identified cognitive impairment and dementia to be independent risk factors for hallucinations [19].
Likewise, the criteria used to define psychosis differ among studies and some inconsistencies in which psychotic phenomena were examined were found with some studies focusing only on hallucinations and not assessing delusions or minor psychotic phenomena. In fact, minor phenomena have been excluded from most studies on PD-psychosis. For these reasons, it is difficult to estimate the true prevalence of PD-psychosis. We must remember that there were no standardized diagnostic criteria for psychosis associated with PD until 2007 when the NIH NINDS/NIMH proposed diagnostic criteria for PD-psychosis, including minor symptoms, to better define and standardize the unique features of PD-psychosis [8]. Our meta-analysis included studies prior to the definition of those diagnostic criteria of psychosis which may influence the results.
The pooled global frequency of any form of hallucination was 21.6% (95% CI 14.7 to 30.6; I2 = 95%; 18 studies; combined n = 3161; Table 3). Regarding the meta-regressions conducted, we found that none of the regressors were statistically significant. However, the mean baseline Hoehn & Yahr stage and the mean baseline duration of PD explained 13.53% and 17.43% of the model’s statistical heterogeneity, respectively. Subgroup analysis based on cognitive status was statistically significant (p value for interaction), while based on diagnosis method was not (Table 4), which is in line with previous studies identifying dementia as an independent risk factor for hallucinations [19].
Regarding the pooled estimated frequency of the different forms of hallucinations was 16.5% for visual hallucinations, 7.1% for auditory hallucinations, 3.3% for olfactory hallucinations, and 2.6% for tactile hallucinations. Our results are similar to other studies showing that visual hallucinations are characteristic, but auditory, olfactory, tactile hallucinations, and multisensory hallucinations have also been described [18, 26].
The pooled estimated frequency of delusions was 6.1% and illusions was 11.5%. The pooled estimated frequency of sense of presence was very high, with 30.2% of patients experiencing this symptom. Sense of presence was the most common type of psychotic symptom in our meta-analysis, although only five studies (971 patients) have evaluated it. Sense of presence is a complex symptom, often difficult to described by patients as it cannot be related to a specific sensory modality and should be asked systematically when evaluating psychotic symptoms. The frequency of minor psychotic phenomena was 23.6%. Given the high frequency of this symptom and its clinical relevance, as it may be associated with greater depressive symptoms and worse quality of life [26], minor psychotic phenomena should be asked routinely to patients and should be included in screening rating scales. Although minor psychotic phenomena include sense of presence, passage hallucinations and illusions, the included studies applied different criteria to define minor psychotic phenomena, so we had to evaluate these phenomena separately.
The methodology used for psychosis diagnosis, namely methods of ascertainment (the use of self-report questionnaires only versus the administration of different scales by a clinic or a clinic, or (semi) structured interview) could also theoretically influence the frequency of psychosis. We must have in mind that we only included studies in which the diagnosis was made by clinical, structured, or semi-structured interview or through the administration of validated scales by experienced clinicians and excluded studies that diagnosed psychosis only by self -report questionnaire. Maybe for this reason, when we compare the psychosis frequency calculation using different diagnostic methodologies, the difference in frequency was not statistically significant. It is possible that patients may not report psychotic symptoms spontaneously in self report questionnaires or even deny them in direct questions. The clinical psychiatric evaluation may be more sensitive to detect psychotic symptoms especially in patients with cognitive impairment and lack of insight. If we have included all studies, probably the aspect of methodology of ascertainment would be more evident.
We also did not find differences in the frequency of psychosis between different study designs, represented by the methodology used to conduct the study.
None of the included studies evaluated the global frequency of psychosis and only one study evaluated the frequency of visual hallucinations in advanced stage patients [18]. Further studies are needed to determine the frequency of psychosis at advanced/late stages as well as its characteristics.
This study has several limitations. First, few studies (small number of patients) were included to review frequency of specific psychotic symptoms (tactile and olfactory hallucinations, delusions, illusions, sense of presence, minor psychotic phenomena). Second, several studies reporting the frequency of psychosis in samples lower than 25 patients were excluded as well as those in which diagnose was made based solely in self- reported scales which can also be interpreted as a strength. Third, limitations of subgroup and sensitivity analyses are well established and the fact we do not have statistically significant results does not imply that the examined covariates are not of clinical importance. Fourth, we did not evaluate the possible influence of dopaminergic therapy in psychosis frequency which could be an interesting element to study. The main strength of this review is the inclusive approach, with focus not only on the frequency of psychosis but also on different psychotic symptoms, some of them not previously studied in a systematic way. Other strengths of this review were the assessment of articles risk of bias and bias publication.
Footnotes
ACKNOWLEDGMENTS
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
CONFLICT OF INTEREST
Joaquim J. Ferreira: no conflict of interest to report. No specific funding was received for this work. Received grants from GlaxoSmithKline, Grunenthal, Fundação MSD (Portugal), TEVA, MSD, Allergan, Novartis, Medtronic. He received consultancy fees from GlaxoSmithKline, Novartis, TEVA, Lundbeck, Solvay, BIAL, Merck-Serono, Merz, Ipsen, Biogen, Acadia, Allergan, Abbvie, Sunovion Pharmaceuticals, Zambon, Affiris. He also participated in advisory boards for Bial and expert testimony to Novartis.
The other authors have no conflict of interest to report.
