Abstract
The legalization of cannabis in many countries has allowed many Parkinson’s disease (PD) patients to turn to cannabis as a treatment. As such there is a growing interest from the PD community to be properly guided by evidence regarding potential treatment benefits of cannabis. This systematic review and meta-analysis aims to compile the best available evidence to help guide patients and their family, clinicians and researchers make informed decisions. A systematic search of the literature was conducted in June 2021. Five randomized controlled studies and eighteen non-randomized studies investigated cannabis treatment in PD patients. No compelling evidence was found to recommend the use of cannabis in PD patients. However, a potential benefit was identified with respect to alleviation of PD related tremor, anxiety, pain, improvement of sleep quality and quality of life. Given the relative paucity of well-designed randomized studies, there is an identified need for further investigation, particularly in these areas.
INTRODUCTION
Parkinson disease (PD) is the second most common neurodegenerative disorder with a prevalence of 1% in populations over 60 years of age [1, 2]. It is a multisystem illness impacting a wide range of motor and non-motor functions.
While the underlying pathophysiology of this illness is yet to be fully defined, some histopathological features are well described, most notably, degeneration of the dopamine producing neurons in the substantia nigra pars compacta [3]. Approximately 5–10% of PD cases demonstrate Mendelian inheritance. At least four genes have been shown to predispose to PD and these may also interact with environmental risk factors [4–6].
Human and laboratory studies have also demonstrated that oxidative stress and the inflammatory cascade have important roles in the pathogenesis of PD [7, 8]. The endocannabinoid system has been shown to have a neuroprotective function and in particular, antioxidant and anti-inflammatory actions [9]. There are more than 100 cannabinoids that have been identified from the cannabis plant. The two most researched cannabinoids are cannabidiol (CBD) [10] and tetrahydrocannabinol (THC) [11, 12]. CBD has gained interest as a PD treatment due to its anticonvulsant, anxiolytic, and analgesic effects. Similarly, THC has desirable analgesic, muscle relaxant, and antiemetic properties but also has problematic psychotropic effects [11, 12].
There has been growing interest in cannabis as a potential therapeutic agent for PD through counteracting oxidative cell damage and neuro-inflammation [13]. Additionally, it has been hypothesized that the antioxidant properties of cannabis could slow PD progression and improve PD symptoms [7, 8]. Tremor is one of the common motor symptoms in PD. There are studies demonstrating cannabinoid receptor activation ameliorating tremor in animal models [14, 15]. Cannabinoid 1 (CB1) receptor activation inhibits glutamate release and regulates subthalamonigral neuronal overactivity which in return reduces tremor [16, 17]. Dyskinesia resulting from levodopa and dopaminergic therapy is another common motor symptom in people with mid to late-stage PD [18]. Preclinical studies have shown that activation of CB1 receptors reduced dyskinesia in PD animal models [19, 20]. Cannabinoids have been shown to block dopamine transporters consequently reducing dopamine reuptake and dyskinesia [21]. There are also PD patient reports [22] and large survey results [37, 52] to suggest that cannabinoids can alleviate both motor and non-motor symptoms of PD.
The aim of this review was to interrogate the published and unpublished literature for evidence of treatment effects of cannabis in PD. We have focused on the potential effects on PD severity and progression, as well as effects on motor and non-motor symptoms.
METHODS
Search strategy
Searches for published and unpublished studies of cannabis treatment in people with PD were conducted on 14 June 2021. A three-step approach was utilized in searching for studies. First, an initial search of MEDLINE and EMBASE was undertaken using the following MeSH® terms: Parkinson disease, cannabis, marijuana, medical marijuana to develop a search strategy (Supplementary Tables 1 and 2). Second, the same search strategy was applied to CINAHL, PsycINFO, Web of science, Scopus, Proquest Dissertations, and CENTRAL databases using all identified keywords and index terms from the initial search. Public trial registers such as ClinicalTrials.gov and World Health Organization International Clinical Trials Registry Platform (ICTRP) were also searched for ongoing and unpublished studies. Lastly, bibliographies of all identified relevant articles were hand searched for additional studies.
Inclusion and exclusion criteria
Randomized controlled trials and non-randomized studies such as open label studies, before and after, case reports, chart reviews, surveys that evaluated therapeutic effects of cannabis or cannabis-based treatment in patients with PD were considered. Review papers were excluded but were assessed for additional relevant articles. Cannabis or cannabis-based treatment included any agent considered a cannabinoid whether used alone or combined with other cannabinoids or other agents, whether synthetic or a direct cannabis extract. Two commonly used cannabinoids are tetrahydrocannabinol (THC) and cannabidiol (CBD) [10]. The reported impact of any dose or treatment duration upon any motor and/or non-motor symptom of PD was included. The protocol for this review was registered in PROSPERO under ID: CRD42019124256.
Study outcomes of particular interest for this review were the Unified Parkinson’s Disease Rating Scale (UPDRS) [23] and Movement Disorder Society UPDRS (MDS-UPDRS) which measure the severity and progression of PD [24]. The UPDRS was developed in 1987 and has four parts: I) mentation, behavior, and mood, II) activities of daily living, III) motor examination, and IV) complications of therapy. Parts I, II, and IV require a PD medical specialist to evaluate the patient for each question according to a scale provided. Part III requires the specialist to perform a series of prescribed physical examinations and record the results according to the scales provided. Higher UPDRS scores relate to higher PD severity. The UPDRS was used in many of the reviewed studies to measure possible therapeutic effects of cannabis in PD. A reduction of between 4.1 and 4.5 on the total UPDRS is generally considered a minimal clinically important improvement [25]. The MDS-UPDRS is a revision of UPDRS completed in 2007. It has four parts: I) non-motor experiences of daily living, II) motor experiences of daily living, III) motor examination, and IV) motor complications [26]. A reduction of at least 7.1 on the total MDS-UPDRS score is considered a minimal clinically important improvement [27]. Other study outcomes included in this review were any questionnaires, scales, or assessments of PD motor and non-motor symptoms such as tremor, levodopa-induced dyskinesia (LID), anxiety, pain and quality of sleep. Lastly, quality of life and safety data were also collected.
No language, publication date, or status restrictions were imposed. Non-English articles were translated using google translate. If articles were found to report the same or similar data and from the same study, it was ensured that only one copy of the data was included. The article that had more data or a fuller description of the same data was nominated as the included article (e.g., if a journal article and conference abstract from the same study were ascertained, the former was included in this review). Two authors (BU and JC) independently screened and evaluated all articles. Any disagreements were resolved through a discussion with another author (MAO).
Data extraction and quality assessment
Quantitative data from included studies were extracted using a data extraction and assessment tool that was adapted from The Cochrane Public Health Group [28]. The extracted data included specific details on the interventions, populations, study methods and outcomes of significance to the review question and specific objectives.
We assessed the quality of all randomized studies using the criteria outlined in the Cochrane’s risk of bias (RoB) tool for conducting a systematic review [29]. Biases assessed for each study were: selection bias (random sequence generation and allocation concealment), performance bias (blinding of participants and personnel), detection bias (blinding of outcome assessment), attrition bias (incomplete outcome data), reporting bias (selective outcome reporting), and other (other sources of bias).
Statistical analysis
Where available, for randomized studies, treatment effects were measured as differences (treatment-control) in mean total UPDRS scores and meta-analyzed as weighted mean differences (WMD) utilizing a range of random effects models. The MDS-UPDRS was used in one RCT and is reported separately as it was determined that UPDRs and MDS-UPDRS scores could not be meaningfully combined.
Treatment effects reported in cross-over and parallel randomized studies were combined using the methods described in Curtain et al. 2002 [30] and Elbourne et al. 2002 [31]. Stata 15 (College Station, TX, US) packages metan, metaan and mvmeta were used to perform random effects meta-analyses. Data that could not be meta-analyzed due to heterogeneity in outcome measures and study designs have been presented in descriptive terms.
RESULTS
Search results and study selection
Electronic and hand searches returned 2,555 journal articles. After screening the titles and abstracts, 33 articles were found to be eligible for full-text screening. There were 26 randomized and non-randomized studies that met all inclusion and exclusion criteria.
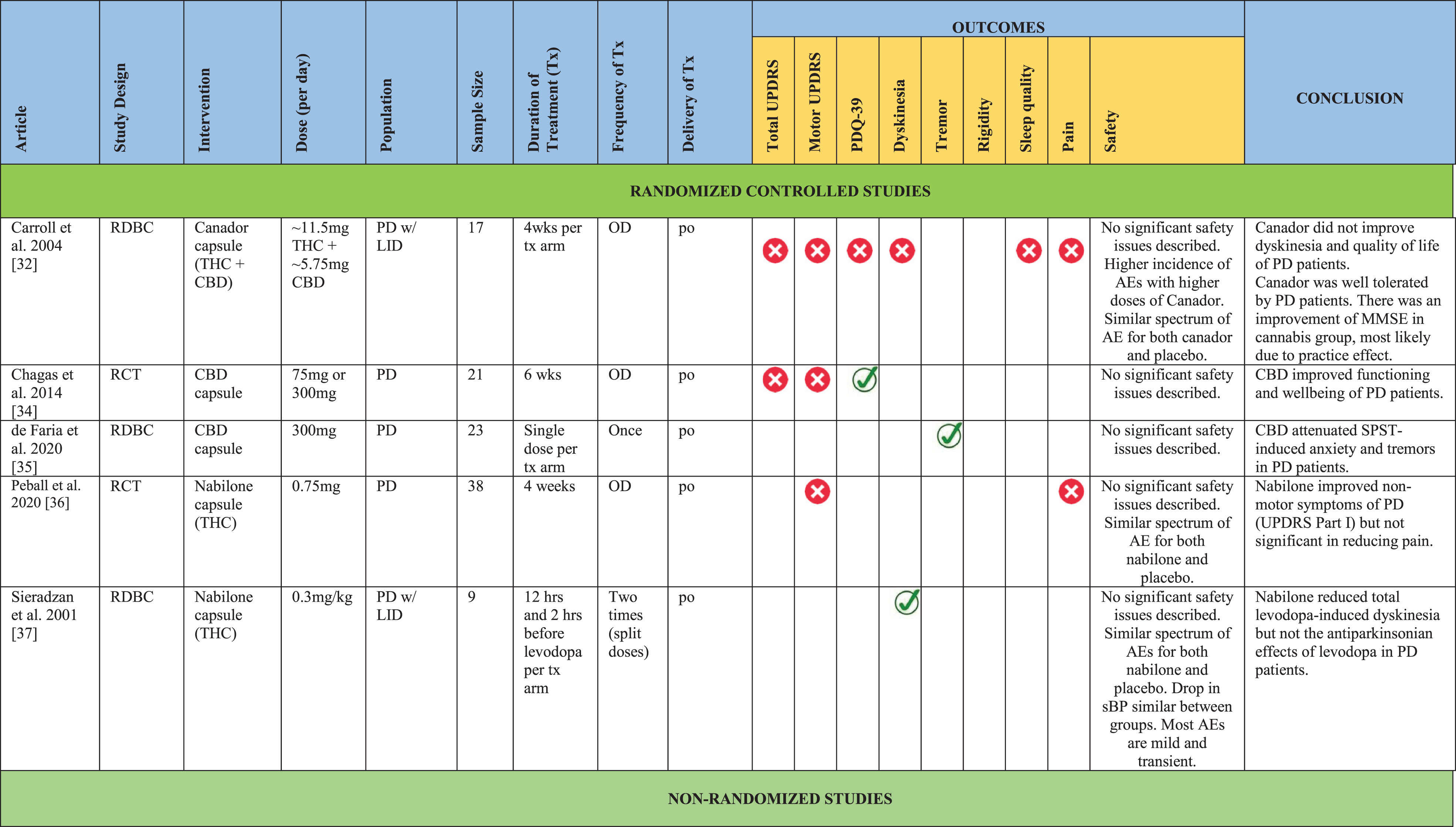
Six included articles were randomized studies [32–37] and twenty were non-randomized studies (NRS) and case reports [34, 38–56]. Two were published as both journal articles and conference abstracts [47, 48] and four journal articles presented essentially similar (or the same) results [33, 54]. Duplicates were identified during data extraction and only the most complete dataset included. Therefore, a total of 23 unique studies (5 randomized and 18 non-randomized studies) were included in this review. Figure 1 presents the PRISMA Flow diagram of the search, screening and selection process of studies. The reasons for excluding each article during the full-text review are also presented.
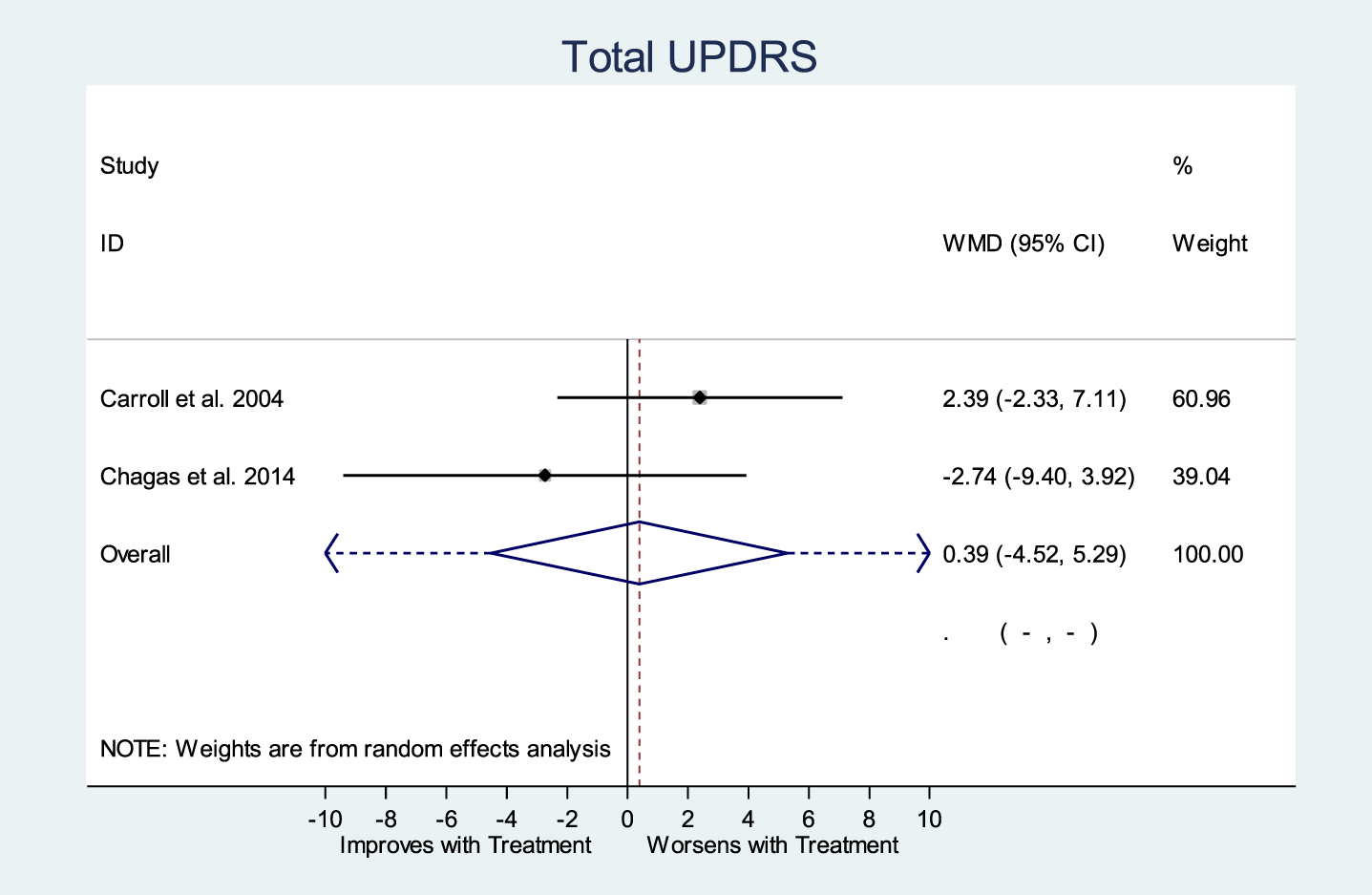
Meta-analysis of Total UPDRS Score (p = 0.877).
Population characteristics
All studies (randomized and non-randomized) investigated the effects of cannabis in people diagnosed with PD.
Various cannabis formulations were investigated in the five randomized studies. Two studies [32, 37] investigated the effect of cannabis treatment on LID in PD patients.
On the eighteen non-randomized studies (NRS), both prescribed and unprescribed cannabis treatments were investigated [39–41, 55]. Some studies investigated cannabis treatment in PD patients in relation to specific PD related disorders; namely, rapid eye movement [57] sleep behavior disorder [58], severe tremor [42], intractable tremor [46], and psychosis [56]. One study [45] explored dose escalation and safety of cannabidiol [10].
Cannabis intervention
The cannabis formulations used in the randomized studies were cannabis extract (∼11.5 mg THC and 5.75 mg CBD) [32], pure CBD capsules [34, 35] and synthetic THC [36, 37].
Of the NRS, four investigated pure CBD with specified doses [45, 58] whilst the remainder assessed unspecified cannabinoid formulations and doses [38, 59].
Study outcome measures
Only three of the five randomized studies measured UPDRS or MDS-UPDRS as an outcome [32, 36], whilst of the eighteen NRS, six studies reported some information of cannabis effects in terms of UPDRS or MDS-UPDRS [38, 56]. All NRS assessed outcomes related to PD symptoms such as LID, tremor, pain, improvement in sleep and quality of life. Some studies also provided safety data [38, 56] which were included in this review. Detailed study characteristics are presented in Table 1.
Characteristics of included studies
Reported study quality
The overall quality of the five randomized studies was considered high due to low risk of bias. The Cochrane’s risk of bias assessment revealed 88.57% and 11.43% measures of low and unclear risk of bias respectively for all randomized studies (Supplementary Figure 2). Reviewers’ evaluation for risk of bias is available on Supplementary Tables 3 and 4.
One study did not provide detail regarding the treatment allocation process [37] and for three studies, it was unclear if independent blinded assessors were utilized [34–36]. The detailed risk of bias and reviewers’ judgments across studies can be found in Supplementary Table 3.
The quality of non-randomized studies was considered as low due to a high for risk of bias. They were included in this review only in a narrative context and not used in meta-analysis.
Meta-analysis of total UPDRS score
Only two out of five randomized studies reported total UPDRS [32, 34]. Carroll et al. (2004) [32] provided their UPDRS raw data to us while for Chagas et al. (2014) [34], the UPDRS data were available in the article. Meta-analysis of these two studies using a random effects model is shown in Fig. 1. The overall estimate of the treatment effect was a marginal worsening of total UPDRS with a weighted mean difference of 0.39 (95% CI –4.52, 5.29; p = 0.877).
Of the eighteen non-randomized studies (NRS), only three presented data on total UPDRS or MDS-UPDRS. One open label study using 150–400 mg/day of pure oral CBD reported an improved total UPDRS score [56] while two other open-label studies of ∼1600 mg/day of pure oral CBD reported an improvement in total MDS-UPDRS scores [45, 46].
Meta-analysis of all UPDRS Parts were also undertaken. There was no evidence of an effect with regards to UPDRS Parts I, II, III, and IV (Fig. 2). Peball et al. (2020) [36] presented MDS-UPDRS Part I and reported significantly less deterioration in non-motor symptoms (MDS-UPDRS Part I) for THC treated patients compared to placebo [36], whilst MDS-UPDRS Parts II-IV did not show significant differences between groups.
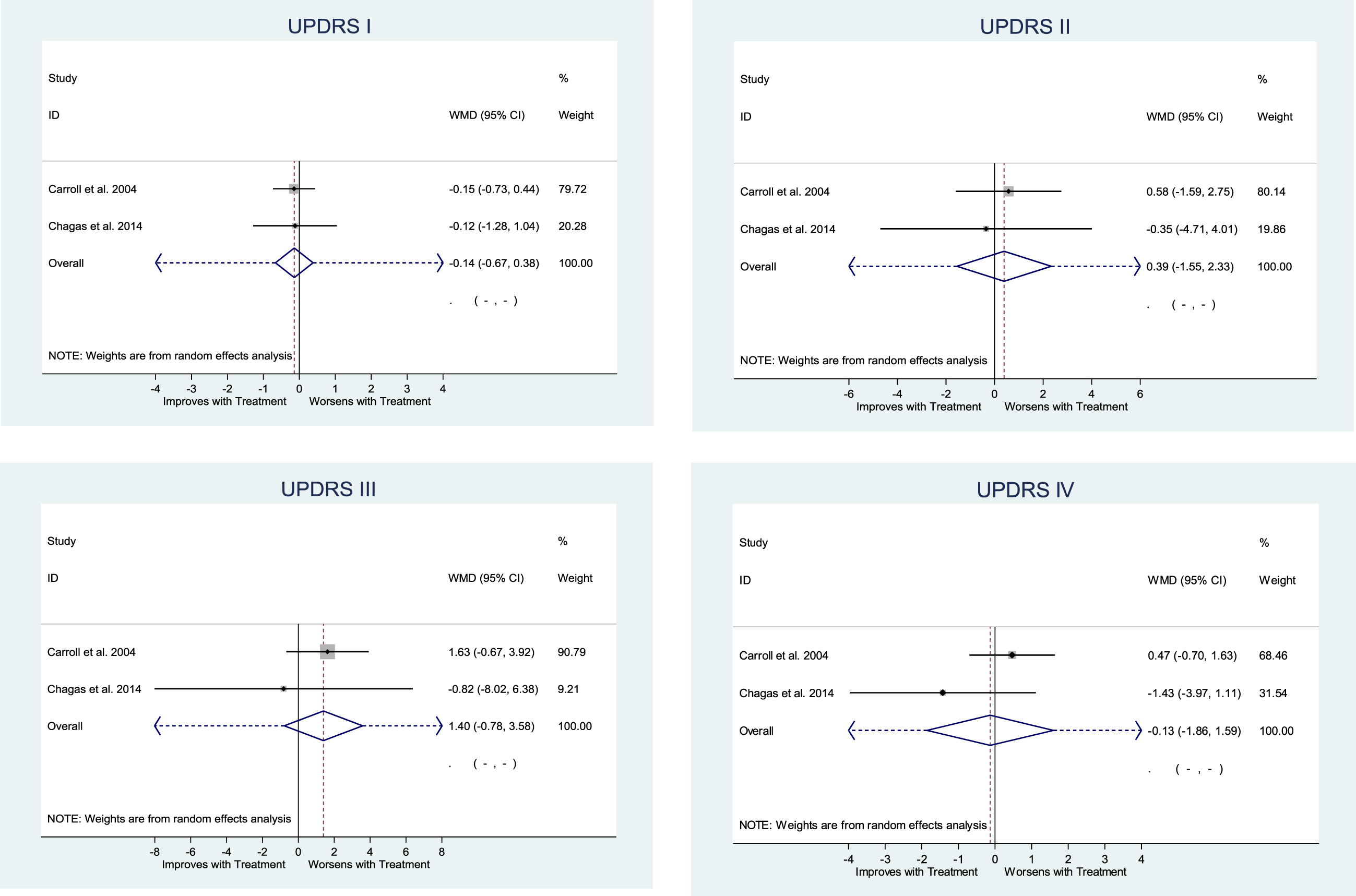
Meta-analysis of UPDRS Part I (p = 0.596), Part II (p = 0.692), Part III (p = 0.209), Part IV (p = 0.880).
The three randomized studies that presented UPDRS and MDS-UPDRS data [32, 36] differ in their study design (cross-over vs. parallel RCT), cannabis products investigated (THC alone, CBD alone, THC: CBD), the cannabis dose used (∼11.5 mg THC and 5.75 mg CBD, 75 or 300 mg CBD, or 0.75 mg THC), and study population (early-stage to mid-late stage PD).
No single study in this review investigated the long-term effects of cannabis including that on PD progression.
Effects of cannabis on tremor
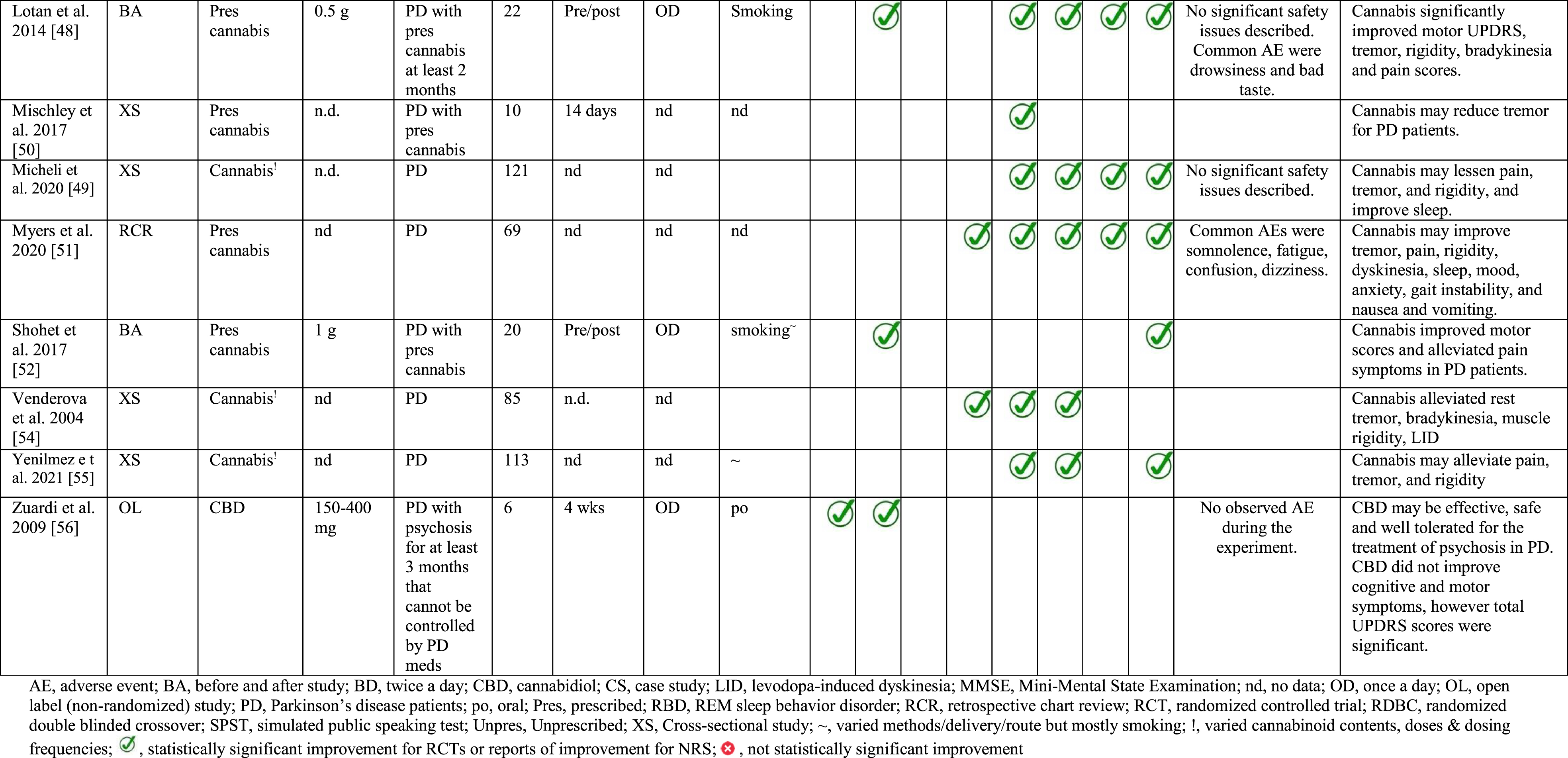
One randomized study demonstrated a decrease of tremor amplitude after administering a single CBD 300 mg dose compared to placebo (p = 0.022) [35].
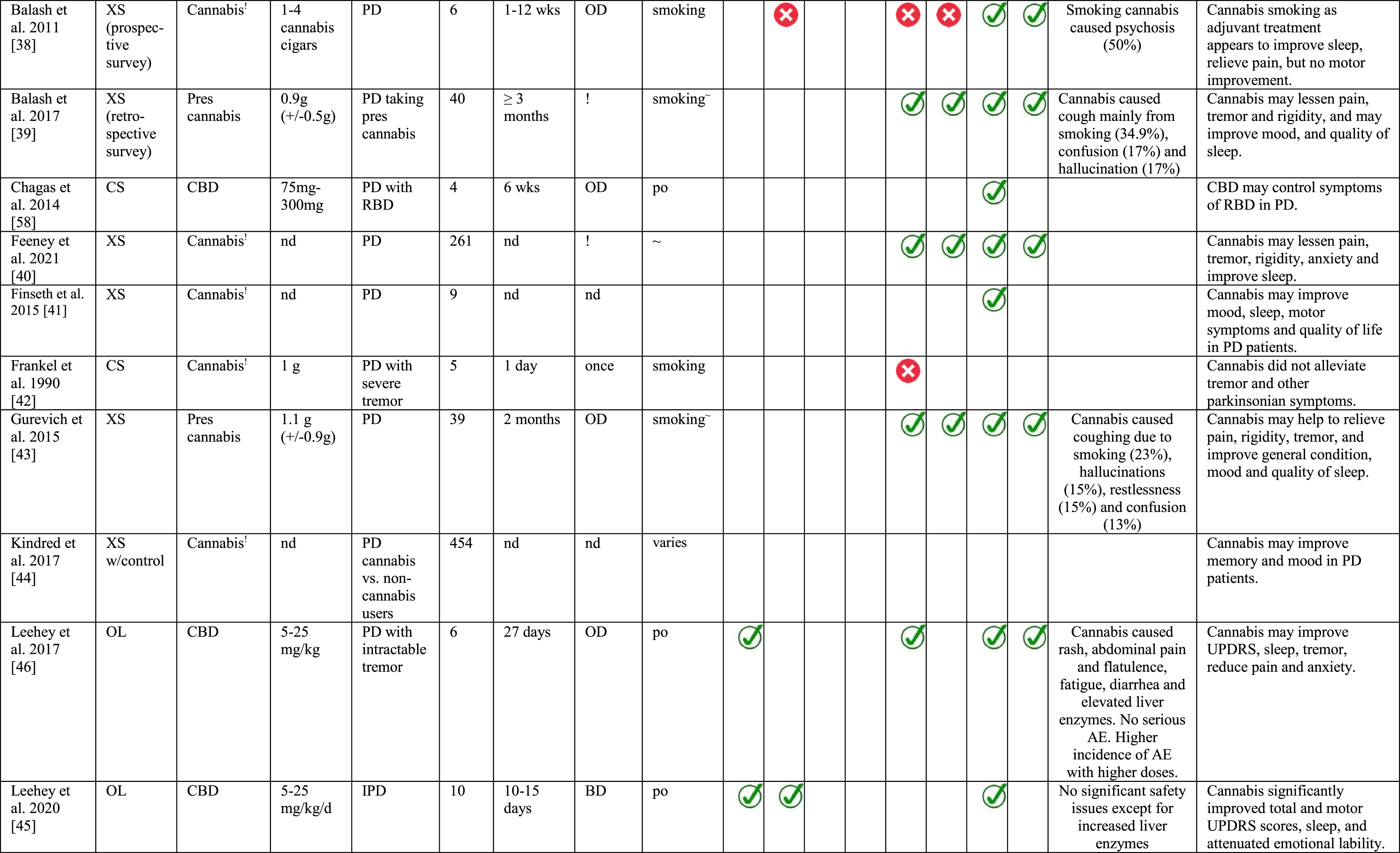
Eleven out of the eighteen NRS that measured tremor reported an improvement following cannabis treatment [38–40, 54].
Twenty-two PD patients in Israel reported reduction of their tremor after smoking medical cannabis measured by UPDRS Items 20 and 21 [48]. Also in Israel, another study similarly reported that smoking prescribed medical cannabis for at least 3 months showed improvements in tremor following telephone interviews [39]. Another study also showed that inhalation of cannabis by PD patients suffering tremor alleviated in their symptom as detected by a movement monitor wore by participants for 14 days during treatment [50].
An electronic survey in the US of 261 respondents with PD who were cannabis users in the past six months and taking various forms of cannabis be it THC, CBD or mixed taken orally found that 25% of these respondents reported considerable improvement in tremor, while 38% reported moderate improvement, 24% reported slight improvement and only 13% reported no improvement [40]. Also, from a telephone survey of 39 PD patients taking medicinal cannabis approved by the Ministry of Health in Israel, 59% reported decrease in tremor symptoms [43]. A survey sent via mail to PD patients in Prague revealed 85 out of 339 respondents (25.1%) were already taking cannabis for PD [54]. Of these cannabis users, 31% reported improvement of rest tremor and that a minimum of 3 months of cannabis treatment provided more constant and consistent benefits compared to short term treatment. Finally, a survey of cannabis users with PD in Germany revealed that 25% of respondents felt significant relief in tremor from taking cannabis mostly via oral route [55].
Effects of cannabis on levodopa induced dyskinesia
One randomized study shown that THC at a dose of 0.3 mg/kg alleviated dyskinesia (p = 0.05) [37] while another randomized study using ∼11.5 mg THC and ∼5.75 mg CBD total dose reported no significant difference between cannabis and placebo groups (p = 0.09) [32]. The former study used only one outcome measure, i.e., total dyskinesia score while the latter used a range of dyskinesia assessments. The sample sizes for both randomized studies were small (n = 26) and both used a crossover design in which THC psychoactive effects may have made it difficult to blind patients and to some extent, investigators and outcome assessors.
A survey of 85 PD patients who were cannabis users found 14.1% reported an improvement in dyskinesia after taking cannabis of unspecified cannabinoid content [54]. A retrospective chart review of PD patients in a single center found that 69 PD patients were already using medical cannabis for their condition and were reporting dyskinesia improvement after taking unspecified cannabis formulations [51].
Effects of cannabis on anxiety
Data from a randomized study indicated that a single CBD administration reduced anxiety in PD patients who underwent the simulated public speaking test (SPST) compared to control as evaluated by the visual analog mood scales anxiety factor (p = 0.021) [35]. As this study used only a single CBD administration and induced anxiety experimentally, its results are not easily generalizable. Also, in another RCT [36], participants from a THC treated group reported reduction of their anxiety levels compared to placebo as measured by MDS-UPDRS Item 1.4 (p = 0.044).
In an open label study of CBD, 3 out of 6 PD patients who were treated for 2 weeks reported less anxiety symptoms after taking CBD [46]. Also, in an electronic survey in the US accumulated 261 respondents with PD reporting improvement of anxiety after taking cannabis [40]. 35%, 43% and 15% respondents reported considerable, moderate and slight improvement in anxiety, respectively and only 6% did not.
Effect of cannabis on sleep quality
A randomized double-blind trial [36] of nabilone (THC) reported fewer sleep problems in the treated group compared to placebo (p = < 0.001). The placebo group in this RCT experienced difficulties getting a full night’s sleep (Item 1.7 total score: 2.30) compared to nabilone (THC) treated group who suffered some sleep problems but these did not lead to difficulties getting a full night of sleep (Item 1.7 total score: 1.00)
In an open label study, CBD treatment for 2 weeks in PD patients was reported to improve participants’ quality of sleep [45, 46]. Other NRS also reported cannabis of unspecified cannabinoid content to improve sleep quality [38–41, 49].
A case series of 4 PD patients treated with oral CBD (3 patients 75 mg CBD and one patient 300 mg CBD) for 6 weeks had prompt, significant and persistent reduction of rapid eye movement [57] sleep behavior disorder (RBD) [58]. In fact, 3 out of 4 participants had elimination of RBD symptoms after CBD treatment such as yelling, talking, pushing kicking, or punching during sleep.
An electronic survey of PD patient in the US of 261 respondents reported substantial improvement in sleep disorders after taking cannabis. 46%, 31%, and 15% respondents reported considerable, moderate, and slight improvement in sleep disorders with only 9% who did not [40].
In a telephone survey of 39 PD patients taking medical cannabis in Israel, 59% reported improvement of sleep quality after taking cannabis [43] while in a German survey, 27% of PD cannabis users reported significant improvement of sleep quality [55]
An open label study of purified CBD taken for between 10 and 15 days reported an improvement of sleep quality in PD patients [45]. Twenty-two PD patients who received medical permission to smoke cannabis and were additionally treated with 0.5 g of cannabis (unspecified cannabinoids) showed 12 of these 22 PD patients reported significantly less sleep problems while 8 also had mild relief [48].
Effects of cannabis on pain
In an open label study of oral CBD, three of six PD patients reported significant reduction of pain levels [46]. NRS reporting the use of prescribed cannabis of unspecified cannabinoids also noted immediate pain reduction [48, 55].
In a telephonic survey in Israel of PD patients granted medicinal cannabis prescriptions, 64% reported improvement in pain [43]. Also, an electronic survey in the US of 261 respondents with PD reported substantial alleviation of pain after taking cannabis [40]. 35%, 36%, and 15% of respondents reported considerable, moderate, and slight reduction of pain with only 14% experienced no effect in reducing pain.
Moreover, twenty-two PD patients who inhaled 500 mg of medical cannabis (unspecified cannabinoids) reported a significant reduction of pain [48]. The same outcome was achieved when fourteen PD patients who smoked cannabis were assessed on their pain threshold using thermal quantitative sensory testing revealing a significant reduction of subjective perception of pain after inhaling cannabis [52]. In a survey of PD patients using cannabis in Germany, 50% of responders reported significant reduction of pain after taking cannabis [55].
Effects of cannabis on quality of life
Quality of life has been explored in a randomized study using the Parkinson’s Disease Questionnaire-39 (PDQ-39) scale [34]. Treatment with CBD, 300 mg/day for 6 weeks, reduced feelings of stigma associated with PD (p = 0.038) and improved overall activity of daily living (p = 0.022) positively affecting overall quality of life [34]. No effect was noted at a dose of 75 mg/day for 6 weeks. Carroll et al. (2004) [32] reported no improvement in QoL (∼11.5 mg THC+ ∼5.75 mg CBD/day). Participants who took higher CBD and THC doses experienced psychotic events.
Safety
There were no significant safety events or concerns reported in the five randomized trials [32, 34–37]. Mild severity adverse events [60] were detected across both cannabis treatment and control groups in all randomized studies. However, a higher incidence of AEs was associated with higher cannabis dosing especially products with THC [32]. For cannabis products with THC, psychological side effects were common such as drowsiness, forgetfulness, insomnia and nightdreams [32, 36]. These AEs resolved when the cannabis doses were reduced or when treatment was stopped.
Common cannabis products and routes of administration
Both inhalation and oral routes of cannabis administration have reportedly provided significant improvement in some PD related motor and non-motor symptoms. More numerous studies have investigated the safety of oral CBD products.
For example, an open label safety analysis of pure CBD taken orally in 10 PD patients demonstrated that high dose of CBD, ∼1600 mg/day (or 20 mg/kg/day), is associated with mild adverse reactions such as liver enzymes elevation, diarrhea, fatigue and somnolence [45] and resolved when the dose was reduced, or treatment was stopped. A double-blind trial of 21 PD patients using 75 mg CBD, 300 mg CBD and placebo taken orally demonstrated similar side effects in all groups [34]. An open label study of 6 PD patients suffering psychosis for 6 months were treated with 150–400 mg of CBD for 4 weeks demonstrating significant reduction of psychotic symptoms [56]. CBD also decreased UPDRS total scores signifying potential benefit in PD symptoms overall. Lastly, case series presentation of 4 PD patients suffering with RBD were treated with CBD (3 patients 75 mg/day and 1 patient 300 mg/day) demonstrated substantial reduction of their RBD symptoms [58].
However, there are also evidence supporting the use of pure THC, or mixed THC and CBD suggesting some benefits. Most of cannabis products (unspecified cannabinoids) used in various NRS were taken orally with less than half of respondents were inhaled cannabis.
An electronic survey of PD patients in the US accumulated 261 respondents who reported improvements in tremor, stiffness, pain and sleep disorders. 40% of these respondents were taking cannabis via inhalation, and at least 50% were taking cannabis orally either by drinking, eating, or spray. 32% of respondents were using THC (high or low), 36% CBD (pure, high or low), and 12% mixed cannabinoids [40].
A questionnaire web-based survey of 113 PD-related cannabis users in Germany took medicinal cannabis mostly via oral route (∼70%), and few took via inhalation (∼30%) [55]. 40% take CBD only, 10% THC only, 20% both THC and CBD, while the rest did not know the cannabinoid contents. Inhaling THC compared to oral CBD was reported to produce more significant efficacy and improvement of motor symptoms such as freezing (80% of inhalation THC versus 24% of oral CBD users). However, there were only 4 of 5 responders reported improvement of inhaled THC.
DISCUSSION
This review revealed no strong evidence suggesting clinical benefits of cannabis in treating patients with PD. Treatment duration in the two randomized studies included in meta-analysis was, however, short; 4 and 6 weeks [32, 34], respectively. Treatment of at least 3 months would seem a more appropriate period especially in trials investigating PD with motor symptoms uncontrolled by levodopa [61]. Also, some participants did not attain the target treatment dose due to cannabis related AEs particularly psychological side effects associated with THC [32]. The use of an optimization phase prior to a double-blind trial may assist in ensuring each participant attains the maximal beneficial dose without or with minimal undesired side effects. Finally, there were relatively few randomized studies, all of small sample size for which there was heterogeneity in the cannabis products used (THC alone, CBD alone, and combination), dose, study design, and participants’ stage of PD.
Some potential areas of benefit requiring further investigation were identified; reduction of tremor, alleviation of some non-motor symptoms particularly anxiety, pain, sleep quality, and improvement of QoL.
This review suggested a potential benefit of cannabis in controlling tremor, a common PD motor symptom. The one randomized study [35] to investigate this effect demonstrated that the administration of a single CBD 300 mg dose effectively reduced tremor amplitude compared to placebo. Other NRS [48, 54] supported this outcome where cannabis notably reduced tremor. However, the measured outcomes were subjective experience rather than quantitative measurements. These results suggest further research is warranted, especially the effect of CBD over a longer treatment period.
Another area that cannabis may have a benefit is on the alleviation of non-motor symptoms. One randomized study [36] demonstrated less deterioration of non-motor symptoms, measured by MDS-UPDRS Part I, for a THC (nabilone) treated group compared to placebo. A survey of PD patients in Germany reported benefits from cannabis in relieving non-motor symptoms such as pain, muscle cramps, depression, and sleep disorders [55]. Most of the 113 PD cannabis users in this survey reported taking cannabis orally while some via smoking, using either THC or CBD only, or combination cannabis products. A similar outcome was observed in a US survey [40]. Around 44% of respondents reported cannabis (THC, CBD, or mixed) effectiveness in treating anxiety, pain and sleep issues [40].
Possible beneficial effects in relation to anxiety, sleep, and pain reported from NRS and surveys should be investigated using more robust experimental designs. This is particularly warranted given results of cannabis efficacy in other medical conditions. In a double blind randomized trial [62] and a retrospective case series [63] of participants with social anxiety disorder and healthy volunteers, cannabis was reported to reduce anxiety levels in participants. Preclinical and clinical studies have also consistently shown cannabis to possess analgesic properties [64–66]. A systematic review of clinical studies demonstrated cannabis as an effective pain control in chronic, non-cancer, pain populations [67]. Lastly, a systematic review of clinical studies aiming to find patient reported reasons for taking medical cannabis revealed that cannabis was mainly sought to treat pain, depression, and anxiety [68].
Another promising area of benefit for PD patients is the effect of cannabis, especially, CBD in improving QoL. Many PD patients experience stigma because of their condition. Experienced stigma is an important key determinant of QoL in PD [69]. Shame arises from increased physical dependence, need for help, and deteriorated body image [70]. It is also important to note that difficulty in performing activities of daily living [70] is independently associated with a higher perception of stigma towards self. CBD may improve the overall ability to carry out activities of daily living which as a result may reduce negative perception of self [71]. This could explain why CBD showed significant outcomes in both carrying out ADLs and stigma experience domains of PDQ-39 [34]. This outcome has also been observed in treatment-resistant epilepsy treated with CBD [72]. It should be noted, however, that the meta-analysis in this review of UPDRS Part II [70] showed no treatment effect (Fig. 2). Again, while there is a considerable amount of observational evidence with respect to the QoL effects of cannabis treatment, until definitive RCTs are performed its benefits remain suggestive.
CONCLUSION
This review did not find strong evidence for the beneficial use of cannabis in PD patients. No clinically significant improvement in the overall symptoms of PD was detected from robust studies though NRS and surveys suggested potential benefits. Relatively few RCTs were identified. These had small sample sizes and were highly heterogeneous in the cannabinoids investigated, their methods of measurement, and study design. Only three studies used the UPDRS/MDS-UPDRS.
Conversely, there are some number of non-randomized studies, case series, and surveys identified in the cannabis literature. These, generally, reported favorable therapeutic effects, which when compared to the equivocal or absence of effect seen from the few RCTs would suggest bias. However, notable improvements were seen for THC and CBD in treating non-motor symptoms of PD particularly in reducing anxiety, pain, and improving quality of sleep. There are also potential benefits of CBD in improving QoL and attenuating tremor. There were no major safety adverse events described.
Although this review revealed possible areas of clinical significance, robust and sufficiently powered randomized controlled studies are lacking. Well-designed studies are needed to accurately estimate the treatment effects of specific cannabis formulations for each potential therapeutic application as well as to identify therapeutic doses.
Implications for clinical practice and future research
This review provides some evidence that cannabis may improve quality of life of PD patients and alleviate tremor and some non-motor symptoms but better designed RCTs are needed to confirm these observations. A more focused study objective may provide more meaningful and positive outcomes such as evaluating CBD/THC effects in tremor or pain. Additionally, a longer treatment period and supporting MDS-UPDRS scale with specific motor and non-motor scales would be beneficial for future studies.
Footnotes
ACKNOWLEDGMENTS
The authors thank some of the authors of studies selected for this review who also helped recover missing data. The authors also want to thank Gold Coast University Hospital librarians for their assistance in conducting the literature search.
CONFLICT OF INTEREST
Berzenn Urbi has been an associate investigator for clinical trials supported in kind by BOD Australia.
Simon Broadley has received honoraria for attendance at advisory board meetings and speaker’s fees from Biogen Idec, Sanofi-Genzyme, Roche, Novartis, Merck, Bayer-Schering and Teva. Dr. Broadley received support to attend International scientific meetings from Biogen Idec, Sanofi-Genzyme, Roche, Novartis, Merck and Bayer Schering. Dr. Broadley has been a principal investigator for clinical trials sponsored by Biogen Idec, Sanofi-Genzyme, ATARA Biotherapeutics, Novartis, Alexion and CBio.
Arman Sabet received honoraria for attendance at advisory board meetings and speakers fee from CSL, BOD Australia, Eisai, Seer medical, Teva and Novartis. Dr. Sabet received support to attend scientific meetings from Biogen Idec, Sanofi-Genzyme, Roche, Novartis, Merck, Allergan, Ipsen and CSL. Dr. Sabet has been a principal investigator for clinical trials supported in kind by BOD Australia.
Saman Heshmat has been an associate investigator for clinical trials supported in kind by BOD Australia.
Joel Corbett, Ian Hughes, Maame Owusu, and Sarah Thorning have no financial disclosures.