Abstract
Background:
Aquatic therapy is one therapy option for people living with Parkinson’s disease (PD). However, the optimal prescription, dosage, and delivery remain unclear.
Objective:
i) To generate consensus statements, ii) to establish evidence-based clinical practice aquatic therapy guidelines for PD.
Methods:
Seventy-three international experts were invited to participate in a 3-step modified Delphi study. Gaps in the aquatic therapy evidence, patient preferences, and stakeholder engagement were considered when developing the initial list of 43-statements identified by the research development group. Practice experts rated each statement on an 11-point Likert scale. Consensus for inclusion was set at a priori of ≥70% of respondents scoring an item ≥7. Two rounds of Delphi questionnaires were completed online, and the expert comments were analyzed using content analysis. An online consensus meeting with an expert subgroup (n = 10) then advised on the guideline’s acceptability and debated items until consensus for inclusion was reached.
Results:
Fifty experts participated in the Delphi round one (83% response rate) and 45 in round two (90% response rate), representing 15 countries. In round one, 35 statements met the criteria for consensus. Content analysis informed the revised statements in round two, where 12 of the remaining 16 statements met consensus. The final agreed aquatic therapy guidelines include key information about dosage, content, safety, contraindications, and the optimal aquatic therapy delivery throughout the disease course.
Conclusion:
Stakeholders, including international practice experts, informed a rigorous evidence-based approach to integrate the best available evidence, patient preferences, and practice expertise to inform these guidelines.
INTRODUCTION
Aquatic therapy is rapidly gaining global popularity for people living with Parkinson’s disease (PD) [1–6]. Aquatic therapy is a term used to describe the use of water as an exercise or rehabilitation medium to optimize physical function and participation for people presenting with varying needs and therapeutic goals [7–9]. Based on the hydrodynamic principles of water immersion, including hydrostatic pressure, buoyancy, viscosity, and thermodynamics, aquatic therapy is often used in the management of PD-specific motor and non-motor impairments [10, 11].
When targeting balance-specific impairments associated with PD such as postural instability, the unique hydrostatic pressure and buoyancy effects of water enhance sensory and proprioceptive input to increase muscle activation [12], while the therapist’s application of turbulence can further challenge postural control [12, 13]. In addition, engaging in aquatic therapy may yield added therapy benefits over land-based rehabilitation, such as the reduced risk of falls-related injuries and improved brain health [2]. There is emerging evidence to suggest that aquatic immersion enhances cerebral blood flow [2, 15], with a recent study demonstrating significant increases in cerebral blood flow following aquatic treadmill training compared to over-land treadmill training in healthy adults [16]. In addition, participation in community-based aquatic therapy appears to be well accepted by people with PD [6, 10]. It is associated with high levels of adherence [7, 17], which is important considering the challenges associated with engaging people with PD in exercise long-term.
There is growing evidence that aquatic therapy is feasible and safe for people with mild to moderate PD [7, 18]. Recent meta-analyses showed that aquatic therapy can be as effective as land-based exercise for improving mobility on the Timed Up and Go (MD –1.5 s, 95% CI –2.68 TO –0.32) [19], balance on the Berg Balance Scale (MD 3.1, 95% CI 1.2 to 5.0) [20], quality of life on the Parkinson’s Disease Quality of Life Scale-39 (MD –5.5, 95% CI –11 to –0.7) [20], reducing disability on the Unified Parkinson’s Disease Rating Scale motor (MD -4.6, 95% CI –7.5 TO 1.7) [11], and fear of falling measured using the Falls Efficacy Scale-I (SMD 0.72, 95% CI 0.19 TO 1.26) [1]. Heterogeneity exists across previous studies, with the results yet to be confirmed by large multi-centered trials [2, 19]. Variations in protocols and aquatic approaches, a lack of uniform intensity-level measures, and limited patient follow-up make it challenging for healthcare practitioners to bridge research-to-practice gaps and meet the requirements of evidence-based practice [2, 21]. While research showed positive cost-benefits for pain and physical function in people with lower-limb osteoarthritis who participated in a group-based aquatic program [22]; no trials have included an economic analysis to compare the cost-benefits of aquatic therapy in comparison with other land-based therapies for people with PD.
Since the research evidence underpinning the application of aquatic therapy in the management of PD is underdeveloped, no consensus exists for key components such as the optimal content, dosage, and delivery of aquatic therapy for people living with PD [19]. While the European Physiotherapy Guideline for PD [23] provides high-quality physiotherapy guidance, there are currently no internationally endorsed aquatic therapy guidelines for practitioners overseeing the delivery of aquatic therapy for people with PD. Clinical guidelines can help to facilitate evidence-based practice while also enabling practitioners to use their professional judgment based on the individual client’s needs and preferences [24–26]. The development of PD-orientated practice guidelines informed by an evidence-based practice framework [27] and complemented by consensus statements can provide healthcare professionals with practical recommendations for safe and effective aquatic therapy delivery [23, 28]. Furthermore, research has shown that using consensus guidelines to provide evidence-based best practice care can improve the overall quality of healthcare [29].
The aims of this study were twofold. First, to establish consensus for statements about the optimal delivery, dosage, frequency, intensity, and content of aquatic therapy. Secondly, to support the clinical decision-making of healthcare practitioners by developing evidence-based aquatic therapy practice guidelines for people living with PD. These guidelines were developed based on current research evidence, patient values and perspectives, and international practice expertise resulting from the Delphi process. The overarching research question was:
What are the key considerations and components of an aquatic therapy program to inform the content of an aquatic therapy guideline for Parkinson’s disease?
METHODS
Similar to previous research, we employed a modified Delphi method to obtain expert opinion and gain international consensus to inform the practice-led component of the aquatic therapy guidelines (Fig. 1) [29–31]. The Delphi technique adhered to specifications in “Conducting and Reporting of Delphi Studies” (CREDES) [28] and previous publications [32]. This was to ensure a high measure of scientific rigor, credibility, and accuracy of the research outcomes [33]. The Delphi technique was chosen as it does not require face-to-face contact, thus reducing selection bias by facilitating international experts to participate [34]. The ease of access to the online Delphi questionnaires, and anonymity between experts, enables practice experts with divergent backgrounds to share their clinical knowledge and expertise without judgment [34, 35]. The Delphi technique is widely used as a process for making the best use of available information [33], particularly when the existing research evidence is conflicting or inconclusive, which was the case in this study [19, 35]. The Reporting Items for practice Guidelines in HealThcare (RIGHT) checklist was utilized to ensure further the correct development and reporting of the guidelines [36].
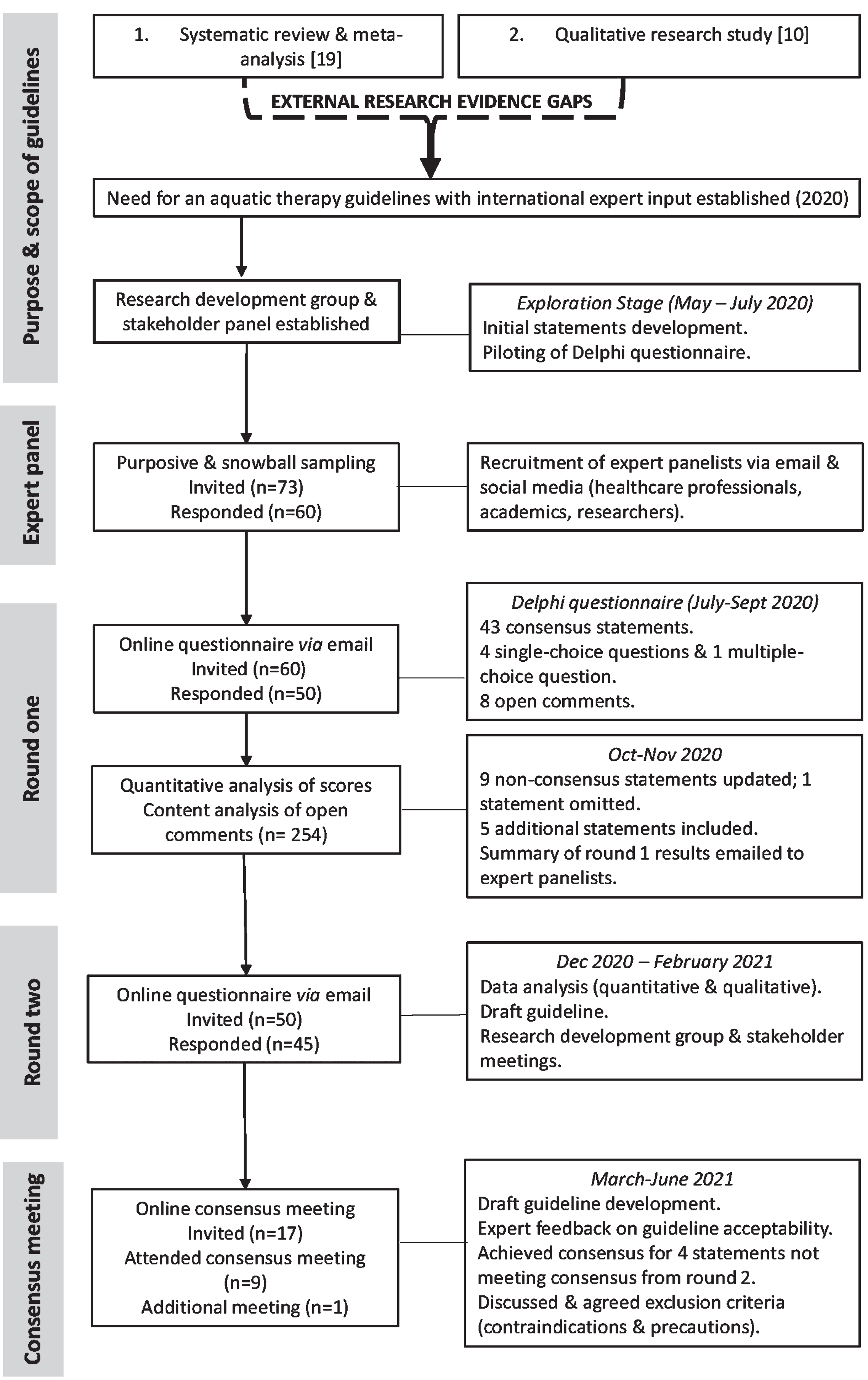
Guideline development process.
Ethics
This research received ethical approval from the University’s Faculty Research Ethics Committee (Ref: 2020_02_17_EHS). All stakeholders and experts named in the acknowledgments provided their written consent.
Research development group
The project was led by an experienced research team (the six listed authors) with extensive clinical practice and research proficiency in PD. They represented several disciplines (physiotherapy, neuroscience, implementation science, and medicine) from Ireland, Australia, and Italy. Four stakeholders representing people living with PD, residing in Ireland, and who had previous aquatic therapy experience (n = 2) formed our PD patient and public involvement panel (here forward referred to as the Parkinson’s panel). As part of the Delphi process, we asked the recruited experts to score aspects specific to clinical practice arising from gaps in the research evidence. As this was considered beyond the scope of lay participants, the Parkinson’s panel engaged in online consultation meetings during the entire research process. The panel also provided feedback to develop and refine the final guidelines.
Recruitment of international experts
In the context of this study, the term expert was defined as “healthcare professional, researcher, or academic with expertise in the treatment, service provision, research or analysis of aquatic therapy for people with Parkinson’s disease.” The inclusion and exclusion criteria were determined in line with this definition. Similar to previous research [31], snowballing and non-random purposive sampling was used to recruit a sample of more than 25 participants. This allowed for an anticipated dropout rate of 5%, in line with earlier research that recommends a minimum of 20 participants as an optimal sample size [37]. From May to July 2020, experts were given study information by email and social media platforms (Fig. 1). We also advertised by email thro-ugh national and international special interest groups (such as the Aquatic Therapy Association of Chartered Physiotherapists, Australian Physiotherapy Parkinson’s Community of Practice, Irish Society of Chartered Physiotherapists, International Aquatic Therapy Faculty, International Organization of Aquatic Physiotherapists). Prospective participants contacted the lead researcher (LC) and were assessed for eligibility. We intended to achieve a balance of international experts (male and female) from different geographical locations. In this regard, additional efforts were made to recruit experts from the USA and Canada to participate in the consensus meeting process and review the final guidelines. Endeavors to contact researchers and clinicians through appropriate organizations via email and social media platforms were unsuccessful.
Guideline scope
The population targeted by the guidelines
These guidelines are appropriate for adults with mild to advanced levels of disability (Hoehn & Yahr stages 1–4) resulting from a diagnosis of PD, including idiopathic PD, progressive supranuclear palsy, and young onset PD. The guidelines may also be appropriate for some people presenting with co-morbidities. However, it is advised that these individuals be screened by a qualified healthcare professional before commencing aquatic therapy.
Potential users of the guidelines
These guidelines were principally developed for physiotherapists treating people with PD in an aquatic therapy setting. Other potential users could include suitably qualified health care professionals (e.g., occupational therapists), exercise scientists, medical doctors, physical medicine, and rehabilitation physicians, along with people living with PD, their families, and carers.
Evidence-based approach
Using the Delphi method, we employed a rigorous evidence-based practice (EBP) approach to integrate the best available evidence, patient values, clinical and research expertise to inform the guidelines [24, 38]. For the EBP pillar, ‘best external evidence,’ a recent systematic review and meta-analysis [19] of 14 randomized controlled trials (RCTs), exploring the optimal aquatic therapy dosage by applying the frequency, intensity, duration, type (FITT framework), was used to identify research gaps and inform the consensus statements. The systematic review, which was conducted by some of the authors (LC, MM, WO’C, AC) explored the effects of aquatic therapy on gait, balance, motor disability, functional mobility, falls, health and wellbeing outcomes in people with idiopathic PD [19]. The quality of the RCTs was evaluated using the PEDro rating scale [39]. Secondly, the EBP pillar, ‘Patient values and perspectives,’ was incorporated through the Parkinson’s panel, along with a recent qualitative study [10]. This qualitative analysis [10] was also conducted by some of the authors (LC, MM, WO’C, AC) and explored the preferences and opinions of people with PD about factors such as access to, participation in, and long-term adherence to aquatic therapy. The systematic review [19] and qualitative research findings [10] were synthesized, with gaps in the research identified and used to inform the content of the original statement list. In consultation with the Parkinson’s panel, the research team converted the list of statements into the Delphi questionnaire format under the headings: aquatic therapy delivery, location and pool environment, safety and supports, tailored aquatic program, dosage (frequency, intensity, duration), and elements of aquatic therapy. For the final EBP pillars, we sought to incorporate the ‘clinical expertise’ of the internationally recruited experts within the ‘context’ of the modified Delphi process, to inform and agree on the final aquatic therapy practice guidelines.
The Delphi process
The process of consensus-building and exploring the optimal aquatic therapy features for people with PD consisted of two rounds of online Delphi questionnaires (Fig. 1) using Qualtrics v2020 software (Provo, UT, USA) and an international con-sensus meeting. Only participants who completed round one were invited to complete round two. Follow-up email reminders were sent to non-responders 14-days after the original email invitation. Piloting of the round one questionnaire was performed with six qualified healthcare professionals independent of the study, with their feedback used to refine the format and readability of certain statements and questions. No statements were added or deleted following the piloting phase. In Delphi round one, an opening question asked participants to consider if aquatic therapy guidelines are needed (yes; no; maybe). Participants were instructed to score each of the 43 statements on an 11-point Likert scale (0 = unimportant, do not include in the guidelines; 10 = very important, must be included in the guidelines) and invited to add comments explaining their clinical reasoning. Participants were informed that a score of seven or above signified agreement [31, 40]. In addition, four single-choice questions and one multiple-choice question were included to explore opinions around dosage and types of aquatic therapy approaches adopted within clinical practice.
In Delphi round two, those statements not meeting the criteria for consensus, including the single and multiple-choice questions, were refined based on the participant’s collective scores and their qualitative feedback. Any changes made to the statements for round two were done via online discussion by the research development group and the Parkinson’s panel. An anonymized summary of the participants’ responses and their round one scores were then disseminated along with the round two questionnaire.
Data analysis
Quantitative data analysis for all Delphi questionnaire rounds were analyzed by the first author in consultation with a senior biostatistician, using SPSSv26 (IBM, NY, USA). Consensus was defined as an a priori median score of ≥7, rated by 70% or more of the experts’ [40]. The median and interquartile ranges were applied as they are considered more robust to outlier influences [34]. A combination of descriptive statistics and frequency distributions was conducted to analyze all quantitative data.
The first author, who was experienced in qualitative analysis, collated all open comments for analysis [31, 41]. A conventional content analysis approach was used to analyze all qualitative data, ensuring that the knowledge generated was grounded in the data, and captured the participant’s knowledge, expertise, and clinical reasoning process [41, 42]. Following round one, the anonymized qualitative data were coded separately by two reviewers (the first author and an experienced post-doctoral researcher) to establish and agree on an initial coding scheme [41]. The first author completed the rigorous process of grouping and categorizing the data from both Delphi rounds [43].
Consensus meeting
A consensus meeting was convened online via Zoom (Zoom Video Communications Inc., 2021) to review a draft version of the guidelines. A subgroup of panelists (n = 17) who completed both Delphi rounds were invited to partake in the consensus meeting. Potential panelists were asked based on their level of aquatic therapy expertise (≥6 years) and experience treating people with PD (Table 1). A draft of the guidelines presented as a two-page infographic, a preamble, and a list of all statements was shared with the expert subgroup two days before the meeting for them to review. During this meeting, experts were requested to discuss and comment on the guideline’s acceptability and establish if they represented current best practices. Four polls were completed amongst the experts for the four statements not meeting consensus from the Delphi round two questionnaire (Table 2). A checklist for screening those with PD before partaking in aquatic therapy, including risk factors and precautions, was reviewed and debated by the subgroup. The first author chaired the meeting with other research team members in attendance (MM, AC) and lasted approximately one hour. Due to different time zones, an additional meeting occurred with an expert from the USA (n = 1). This meeting followed a similar format to the consensus meeting, with a summary of key discussion points from the first meeting provided where further context was required.
Gender, professional expertise, academic profile, and demographic breakdown of the internationally recruited expert group
Delphi round one and Delphi round two statements meeting international expert consensus, those not meeting consensus, and those omitted with weighting and scores
PD, Parkinson’s disease; Md, Median; IQR, Interquartile range; % Score, Percentage of experts scoring the statement≥7 (Median score).
RESULTS
Forty-five experts (female n = 37; male n = 8) representing 15 countries completed both rounds of the Delphi questionnaire (Table 1). Most (n = 38, 84%) were qualified physiotherapists. Eight participants were involved in or had previously completed aquatic therapy research in the field of PD.
Round one
Fifty experts completed round one’s questionnaire (83%). The majority (88%, n = 44) of respondents agreed that aquatic therapy guidelines for people with PD were required, with 12% (n = 6) answering ‘maybe.’ Twelve of the forty-three statements included in round one did not meet consensus (see Supplementary Table 1). The research team excluded one statement after round one because it scored≤1, with only 14.6% of the panelists scoring it seven or more (Table 2). Data gathered from the single and multiple-choice questions are presented in chart format within the Supplementary Material (see Supplementary Figures 1–5). The six open comments generated a total of 253 comments, an average of five per expert. The inductive content analysis yielded five main categories and 17 generic categories (see Supplementary Table 2).
Round two
Forty-five experts completed round two (90% response rate). Consensus for inclusion was attained for 12 of the 16 Likert statements (consensus range 72.7–97.7%) (see Supplementary Table 3). Agreement was achieved for the four statements (Md≥7) without reaching consensus (53.3–66.7%) (Table 2). The open comments generated 121 comments, an average of 2.6 comments per expert. Content analysis of all available comments yielded no other categories.
Consensus meeting
Overall, there was consensus among the panel of experts (n = 10) from eight countries that the guidelines were acceptable and representative of clinical practice. Consensus was also achieved for the four statements not reaching agreement from round two (Table 2; see Supplementary Table 4) where ≥70% of participants scored the poll ‘yes’ to include the statements in the guideline recommendations. Prior to poll voting the statements were refined according to expert feedback and discussion (see Supplementary Tables 4 and 5), along with the expert’s recommendations for amendments to the guideline infographic (see Supplementary Guideline Infographic).
Evidence-based aquatic therapy guidelines: Expert recommendations
Aquatic therapy delivery: Expert consensus was gained for all seven statements about aspects of aquatic therapy delivery, such as how soon after diagnosis, people with PD are referred for aquatic therapy and how aquatic therapy services should be provided and adapted during the disease course (Table 2). The findings are presented in a flow diagram to provide practical signposting for healthcare practitioners when referring, screening, assessing, and delivering aquatic therapy programs for people with PD (Fig. 2; see Supplementary Guideline Infographic).
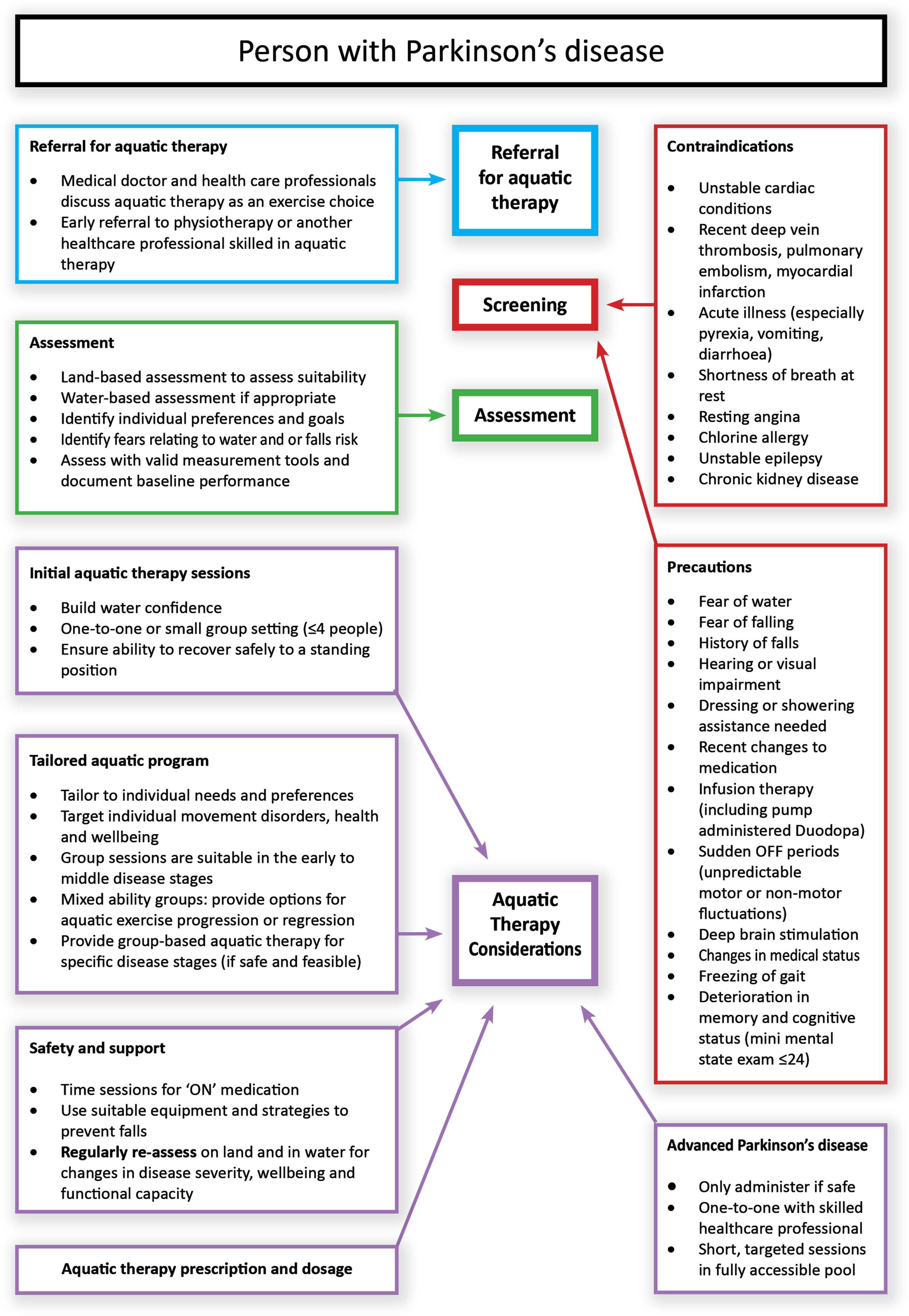
Flow diagram of processes and considerations for aquatic therapy delivery for people with Parkinson’s disease.
Location and pool environment: Experts agreed that access to aquatic therapy within the community setting is beneficial for people with PD. Most experts agreed that a fully accessible, heated pool is preferable for people with PD (Table 2) and is included in the flow diagram (Fig. 2). The content analysis provided further context about factors such as the disease stage and functional capacity, which need to be considered by clinicians when identifying pools suitable for individuals with PD (see Supplementary Table 2). Some experts advised that warm pool temperatures (≥33°C) need to be used with caution. They suggested that warmer water may be appropriate only for lower intensity activities or when treating people with advanced PD with reduced physical capacity.
Safety and supports: Expert consensus was gained for 13 statements about the safety and support needs of people with PD, such as fear of water and or falling, dressing, and showering requirements. Agreement about group size, level of supervision, and equipment requirements to support safe transfers in the pool environment was also achieved (Table 2). There was consensus for providing support for accessing community-based aquatic classes and the role of skilled healthcare professionals in providing basic training to patient support workers and other identified community pool workers. Experts agreed that people in the advanced disease stages could benefit from aquatic therapy, with one-to-one therapy sessions provided by skilled healthcare professionals indicated (Table 2). The key recommendations are outlined in the flow diagram (Fig. 2).
Tailored aquatic program: Expert consensus was achieved for all four statements (Table 2). The majority of respondents agreed that the aquatic program ought to be tailored according to individual goals and preferences and level of impairment, with access to groups for specific disease stages optimal where available (Table 2). The main recommendations are illustrated in the flow diagram (Fig. 2). It is recommended that they be considered in line with the exercise prescription recommendations presented in Fig. 3, using the frequency, intensity, time, type (FITT) framework. (See Supplementary Guideline Infographic).

Key exercise prescription and dosage recommendations for aquatic therapy.
Dosage: Frequency of aquatic therapy: The expert panelists agreed that performing at least two sessions per week is optimal for people with PD engaging in community-based aquatic therapy (Fig. 3). Where people with PD only have access to one session per week, there was consensus that a home exercise program prescribed by their primary physiotherapist is recommended to be provided (Fig. 3). When people with PD undergo intensive rehabilitation within a rehabilitation or hospital facility, experts agreed that between two to five sessions of aquatic therapy per week could improve rehabilitation outcomes if feasible (see Supplementary Table 4).
Dosage: Intensity of aquatic therapy: In the absence of specific guidance around aquatic therapy exercise prescription, there was consensus that the American College of Sports Medicine (ACSM) physical exercise guidelines for people with chronic diseases [44] could offer guidance for clinicians when designing and progressing aquatic therapy programs. Expert feedback highlighted that the level of intensity achieved for each individual is dependent on their medical status and co-morbidities. During the active component of the aquatic class, moderate to high levels of intensity, determined using valid and reliable measurement tools, are recommended (Fig. 3). There was agreement among the experts that performing cardiovascular aquatic exercises at a reasonably vigorous-intensity level, or a predetermined heart rate is essential, particularly in the earlier disease stages. While the experts agreed that to support people to progress, the number of repetitions, sets, and rest periods are essential components of the aquatic therapy program, they also indicated that these could be challenging to monitor, report, and control for during group-based aquatic therapy in comparison to one-to-one aquatic interventions where there is likely to be a higher staff-to-patient ratio.
Dosage: Duration of aquatic therapy: The respondents agreed that sessions should last between 30 to 60 minutes to achieve maximal therapeutic effects. The expert’s feedback did highlight, however, that this may vary according to different aquatic activities (e.g., shorter 30-minute session for high-intensity aerobic exercises), individual functional capacity (e.g., shorter 30-minute session to limit fatigue), and the disease stages (e.g., shorter 30-minute for people with advanced PD in a one-to-one setting). Following round two of the Delphi questionnaires, the optimal duration of aquatic therapy was inconclusive (Table 2). Most experts who engaged in the consensus meeting process agreed that at least 12-weeks of aquatic therapy is recommended to achieve optimal outcomes (Fig. 3). In addition, there was consensus among most experts that continuous participation in community aquatic therapy was preferable to support long-term adherence and maintain therapy effects (Fig. 3). (See Supplementary Guideline Infographic).
Aquatic therapy elements: The expert panelists agreed that a multicomponent aquatic program targeting different movement disorders and non-motor symptoms should be included (Table 2). When designing and developing an aquatic therapy program, key elements for consideration have been included within the guidelines (Fig. 3).
DISCUSSION
This modified Delphi study aimed to gain consensus rigorously and systematically on a core list of statements to inform new aquatic therapy guidelines for people living with PD. As outlined in the guidelines, people with PD can benefit from access to information on exercise and its benefits at the time of diagnosis, with aquatic therapy one option for consideration [10]. Findings from our qualitative study [10] suggest that early access to aquatic therapy services, delivered by suitably qualified healthcare professionals with PD knowledge, may enhance patient outcomes and increase long-term adherence for some people with mild to moderate disease. These guidelines support the timely delivery of an individually tailored aquatic program for people with PD, an approach that is widely supported in the literature [45] and other available guidelines [23, 46].
As part of the initial screening process described, the list of contraindications and precautions agreed by the expert group can be used to assess suitability and optimize safety and independence during participation. This list reflects screening criteria previously outlined, for example, in the UK and Australian aquatic physiotherapy guidelines [47, 48]; however, specific criteria for PD need to be individually considered. For example, orthostatic hypotension [49], cardiac disease [50], freezing of gait [51], and sudden “off” periods causing motor fluctuations [52] need to be screened for by skilled healthcare professionals before beginning aquatic therapy. In addition, our guidelines recommend that fear of water and or falling associated with the pool environment and safety when dressing and showering, which were reported by a small portion of people with PD [10], are identified during assessment. These issues may be addressed by providing key supports to individuals, as well as introducing graded exercise activities to enhance water confidence during the initial aquatic therapy sessions, within a safe pool environment [6, 10].
Regarding dosage, the recommended duration of 30–60-minute aquatic therapy sessions aligns with the aquatic therapy research for people with PD [18–20]. Most experts agreed that to achieve optimal outcomes, at least 12-weeks of aquatic therapy is recommended. This recommendation can be considered in conjunction with the client’s needs, therapy goals, and service capacity. This recommendation is consistent with other research evidence, suggesting that prolonged strength training [53, 54], aerobic training [55–57], dance therapy [58, 59], and Tai Chi [60, 61] can produce long-term beneficial effects for people with PD engaged in physiotherapy and exercise. Notably, the findings from our meta-analysis [19] found short-term positive effects on balance, gait, and mobility in comparison to land-based exercise or physiotherapy, following between 3–11 weeks of aquatic interventions. Short-term benefits gained from aquatic therapy in people with PD could be considered better than none [23, 62]. Owing to the progressive nature of PD, the expert group recommended that practitioners encourage people to continuously engage in community-based aquatic therapy throughout the disease course as a means of maintaining their physical function and wellbeing. This is supported by the literature, which indicates that regular and consistent exercising in people with PD is associated with positive long-term effects on mobility, physical activity, and health-related quality of life [63, 64].
When designing an aquatic therapy program for people with PD, the experts agreed that including a range of different exercise elements, tailored, and progressed according to the individual needs, is recommended. Findings from the meta-analysis [19] indicate that there is low to moderate quality evidence from a small number of aquatic therapy trials (n = 14) to support the inclusion of functional mobility, balance, gait, and posture training within the aquatic therapy program for people with mild to moderate PD. Most of the included trials [19] incorporated a warm-up, cool-down phase, and a core exercise component targeting flexibility, aerobic fitness, strength and endurance, mobility [5, 13], balance [5, 66], posture [5], walking [7, 68], obstacle training [4, 7], and dual-task training [3]. For example, a trial by Volpe et al. [5] included specific water-based exercises, primarily performed in a standing position, designed to targeted postural deformities in people with PD, such as performing trunk extension and flexion to challenge the individual’s point of stability in the water. While the buoyancy and hydrostatic effects of water immersion can enable people with PD to perform such exercises in water, these exercises would be difficult to perform on land due to risk of falls and injury. In addition, the systematic review findings also indicate some preliminary evidence for using established aquatic therapy approaches such as Halliwick [4, 66] and Ai Chi [69]. Methods such as Bad Ragaz Ring method (BRRM) and Watsu require further investigation to confirm their effects in PD. These established aquatic methods incorporate core exercise components such as joint mobilization (BBRM, Ai Chi), stretching (Watsu), muscle strengthening (BRRM), muscle relaxation (BBRM, Watsu), pain modulation (BRRM), proprioception (Ai Chi, BRRM), trunk stabilization and rotation (Halliwick, BRRM, Ai Chi), and endurance (Halliwick) [70].
It is also important to consider, the findings from other exercise literature, which supports the inclusion of a range of challenging and complex exercises [71]. For example, including highly challenging balance training could lead to more robust improvements in balance [72]. Adding resistance exercises [53, 73], alongside cognitive training and dual-task training [3] could optimize the results for people with PD engaging in aquatic programs. In line with the growing body of research highlighting the importance of high-intensity aerobic exercise in slowing disease progression [55, 74], these guidelines recommend that aquatic exercises are performed at a vigorous intensity, particularly in the earlier disease stages. At present, no substantial evidence exists to support the efficacy and safety of aquatic exercise performed at a higher intensity [75], with few studies including valid intensity measures [19]. Future trials comparing the feasibility and safety of high-intensity aquatic programs, along with the optimal pool temperatures, and aquatic therapy programs delivered as an early intervention for people with PD are warranted. In addition, further research is needed to establish the optimal intensity measures for water-based exercise, including how to reliably measure cardiac function [2].
Strengths and limitations
A significant strength of these practice guidelines was the inclusion of different professions with extensive expertise and years of experience from over 15 international countries. The active engagement of patient stakeholders (Parkinson’s panel) during all stages of this study also ensured that the guidelines are relevant and feasible to implement in practice [40, 76]. While limited research evidence [19] may limit the strength of guideline recommendations, the high recruitment and Delphi completion rates by practice experts indicate the relevance, timeliness, and demand for aquatic therapy guidelines to assist in decision-making for all healthcare professionals [77].
Males were underrepresented in the consensus process. This may be explained by the number of female physiotherapists who formed a large portion of our expert panel and tend to work in neurology, geriatrics, and public healthcare [78]. Nevertheless, the sample was representative of a diverse range of research and practice experts. This study was conducted during the Covid-19 global pandemic. Due to the evolving landscape, information around changes to practice including, minimizing social contact, group size, pre-screening, additional hygiene, and pool cleaning measures, required for the safe delivery of aquatic therapy during the pandemic, was not included when developing the consensus statements and guideline recommendations [79]. It is advised that healthcare professionals implement any additional guidelines in line with national and local public health guidance, alongside individual organization’s policies and procedures.
CONCLUSION
At present, there is an unmet need for aquatic therapy guidelines in PD. In this regard, we present internationally agreed guidelines for prescribing and delivering aquatic therapy for adults with PD. These evidence-based practice guidelines are a valuable reference for healthcare professionals and could facilitate improved clinical outcomes for people living with PD.
Footnotes
ACKNOWLEDGMENTS
We thank all the expert panelists who completed the Delphi questionnaires. We acknowledge with thanks those who contributed their time and expertise to the consensus meeting process: Emily Dunlap (USA), Dr. Emine Eda Kurt (Turkey), Fleur Terrens (Australia), Josefa Domingos (Portugal), Judy Goroncy (Australia), Johan Lambeck (the Netherlands), Jacqueline Pattman (UK), Dr. Lucia Cugusi (Italy), Moira Smith (Australia), and Dr. Osnat Fliess Douer (Israel). We thank our Parkinson’s panel for their input and expertise. We also acknowledge the work of Dr. Simon Haines (La Trobe University, Australia) for his contribution to the qualitative content analysis and Parkinson’s Victoria.
The first author received funding in part for a PhD fee waiver for this research from the School of Allied Health, University of Limerick. All other authors received no financial support for the research, authorship, and/or publication of this article.
CONFLICT OF INTEREST
The authors declare that there is no conflict of interest.
