Abstract
Background:
Aquatic therapy offers an alternative physiotherapy approach to managing the motor and non-motor symptoms associated with Parkinson’s disease (PD).
Objective:
This review examined exercise prescription for aquatic therapy in PD and evaluated if aquatic therapy is as effective as land-based physiotherapy for improving movement, disability and wellbeing in people living with PD.
Methods:
A systematic search of eight databases was conducted to identify suitable randomized controlled trials from inception until August 2019. Aquatic therapy prescription data and outcomes of interest included gait, balance, motor disability, mobility, falls, mood, cognitive function and health related quality of life data was extracted and synthesised. A meta-analysis was performed where appropriate.
Results:
Fourteen studies involving 472 participants (Hoehn & Yahr scale I–IV) met the inclusion criteria. Eight were of modest quality, scoring 70–80% on the PEDro scale. Seven studies were included in the meta-analysis. Exercise prescription was highly variable and often insufficiently dosed. Similar gains were shown for aquatic therapy and land exercises for balance, motor disability or quality of life. A statistically significant difference was found for mobility as measured using the TUG (–1.5 s, 95 % CI –2.68 to –0.32; p = 0.01, I2 = 13%), in favor of aquatic therapy.
Conclusion:
Aquatic therapy had positive outcomes for gait, balance and mobility that were comparable to land-based physiotherapy in the early stages of PD. The optimal dosage, content and duration of aquatic interventions for PD could not be confirmed in this meta-analysis. Many trials appeared to be under-dosed and therapy duration was low, ranging from 3–11 weeks.
INTRODUCTION
Physiotherapy for people with Parkinson’s disease (PD) aims to improve movement, physical capacity, quality of life, disability and falls [1–3]. The content of a physiotherapy program varies according to individual needs [1, 4–6] and can include interventions such as progressive resistance strength training [7–9], cueing [10, 11], falls prevention education [7, 13], attention strategies [7, 12], therapeutic dancing [14, 15], cycling [16], upper limb training [17], Tai Chi [18], walking [19], and boxing [20]. Despite movement disorders and falls being key features of PD, the optimal elements of physiotherapy for this chronic neurodegenerative condition are not completely clear. There have been several large randomized trials of physiotherapy for people with PD [7, 22], and the results have been mixed. For example, Seymour et al. [23], Canning et al. [9], and Morris et al. [7] found some beneficial effects for resistance strength training, movement strategy training, and falls prevention education in people with idiopathic PD. Exercise training [24], gait training using virtual reality [25], and motor-cognitive training interventions [26] were also found to improve gait, balance and falls. Nieuwboer et al. [10] examined different exercise approaches outside of the clinic and showed that cue training in the home and community could have positive effects on gait, freezing and balance.
Aquatic therapy is one approach for managing the motor and non-motor symptoms in early PD [27]. Preliminary reports have shown favorable results for movement slowness, dystonia, balance, pain, quality of life and physical function [28–32]. Immersion in water affords people with PD an alternative environment in which to exercise [27]. Water’s physical properties of buoyancy, hydrostatic pressure and drag forces provide sensory feedback that may help to regulate motor output [33]. The physiological benefits of water immersion are well documented in healthy individuals [33–35]. More recently, cytokine modulation was observed to facilitate immune responses in people living with PD, with short-term and long-term benefits observed following aquatic therapy [36].
Preliminary literature reviews of mainly non-randomized controlled trials [37–39] reported that aquatic therapy may improve postural stability, motor function and gait in the early stages of PD. These reviews did not always report key elements such as the water temperature, water depth, accessibility of therapy, patient involvement in the treatment protocol design, location of pre-post assessments (in water or over-land), follow-up retention of aquatic therapy benefits, or the costs of aquatic therapy. Some recent trials have been reported, warranting a meta-analysis focusing exclusively on investigations using a randomized, experimental design. The primary aim of this review was to examine the therapeutic dosage and potential benefits of aquatic therapy for people living with Parkinsonism. The review sought to answer the following questions: What is the optimal frequency, intensity, duration, and type of aquatic exercise for people living with PD? Is aquatic therapy as effective as land-based physiotherapy/rehabilitation for improving gait, balance, motor disability, functional mobility, falls, health and wellbeing in people with idiopathic PD?
METHODS
Identification and selection of studies
A systematic search of the literature was conducted for eight electronic databases (Embase, Cinahl complete, PubMed/Medline (Ovid), PsychINFO, Cochrane-CENTRAL, PEDro, Scopus, Web of Science) from inception to August 1, 2019. A comprehensive search of the titles, abstracts and keywords of the databases was performed using a predefined search terms (see full search strategy in Supplementary Material, Appendix I).
Reference lists in the articles identified, along with other reviews and key rehabilitation journals were manually searched to find additional trials, which were not identified through rigorous database searches. Titles and abstracts of potentially eligible trials were screened using a predefined criterion (Box 1) and Rayyan software, by the primary author. All articles were read by two independent review authors and key information was obtained using a predetermined data extraction form by the primary author [40]. All data was cross checked for agreement by the independent review authors (AC & MM). Methodological quality was independently assessed for six randomly selected studies and the remaining were crosschecked, with any disagreements resolved through discussion.
The preferred reporting items for systematic reviews and meta-analyses (PRISMA) guidelines were adhered to in this review. A review protocol was published on International Prospective Register of Systematic Reviews (PROSPERO ID CRD42018085996).
Inclusion criteria
Design Randomized controlled trial, published in English and peer reviewed Adults>18 years old Parkinsonism disorders (e.g., Idiopathic PD, Progressive Supranuclear Palsy, and Lewy Body Dementia) according to a confirmed diagnostic criterion Hoehn and Yahr Scale or Modified Hoehn and Yahr stages I–IV Include aquatic therapy conducted in a community-based swimming pool (public), a clinic or rehabilitation setting, or a hospital-based hydrotherapy pool (private and public) Primary outcomes: gait, balance, motor disability, functional mobility, mood, cognitive function and health related quality of life Secondary outcomes: economic assessment, participant’s input into study design, treatment compliance and participant’s safety Aquatic therapy versus land-based physiotherapy Aquatic therapy versus no therapy or usual care Aquatic therapy versus other aquatic therapy or water-based intervention Aquatic therapy as a co-intervention with land-based therapies versus land-based therapies
Participants
Intervention
Outcome
Comparisons
Assessments of characteristics of studies
Quality
The methodological quality of randomized controlled trials (RCTs) was assessed by two authors independently using the Downs and Black checklist [41] and the Physiotherapy Evidence Database (PEDro) Scale [42]. Both tools have reported reliability and validity, and have been used to appraise RCTs in previous systematic reviews [38, 44].
Participants
Trials including participants with Parkinsonism disorders with all levels of disease severity (Hoehn & Yahr I–IV) were included. Age, gender, disease severity, Levodopa equivalent dose (LED) and diagnostic criteria were recorded to compare participants across trials.
Intervention
For the purpose of this review, aquatic therapy was operationally defined as physiotherapy occurring in a pool environment, supervised by a qualified
healthcare professional. The experimental group could include varying aquatic therapy approaches (e.g., Ai Chi, Halliwick, Bad Ragaz Ring Method) [33] as long as it suitably targeted the outcomes of interest. Key information on the frequency, intensity, duration and type of aquatic therapy was extracted from each study.
Outcome measures
Primary outcomes of interest were gait measured using tools such as the Six-Minute Walk Test [45], freezing as measured by the Freezing of Gait Questionnaire [46], balance and postural instability assessed on the Berg Balance Scale [47], functional mobility measured using performance measures such as the Timed Up and Go [48, 49], and motor disability evaluated using motor impairment measures such as the Unified Parkinson’s Disease Rating Scale motor subsection III [50]. Health related quality of life (Parkinson’s Disease Questionnaire-39) [51], falls (Falls Efficacy Scale, Falls diary) [52, 53], depression (e.g., Yesavage test) [54], and cognitive function (SCales for the Outcome of PArkinson’s disease COGnition) [55] were also recorded. Patient or public involvement in any stage of the research study, treatment compliance and participant’ safety were also documented. The variables were further categorized based on a model of direct and indirect consequences of movement disorders [56], their effects on important motor functions and recommended goals of therapy, and intervention approaches.
Data analysis
The results from each trial were combined to calculate the overall effects of aquatic therapy compared to a control intervention using standard meta-analysis methods, when two or more trials included the same outcome measures [40]. All of the outcome variables were continuous variables, therefore the weighted mean difference method was applied by calculating the change in the mean and standard deviation from baseline to the first assessment post intervention [57]. The mean difference (MD) was calculated for both the aquatic therapy and control groups for each trial, and was combined using the random effects model of meta-analysis and inverse variance [58]. The random effects model of meta-analysis was adopted as it assumes that the true treatment effects in the individual trials may be different from each other [40]. A statistical heterogeneity of I2 ≥50% was considered substantial heterogeneity [59].
RESULTS
Flow of studies through the review
From a total electronic search yield of 258 possible studies, fourteen studies were deemed eligible for inclusion (Fig. 1).
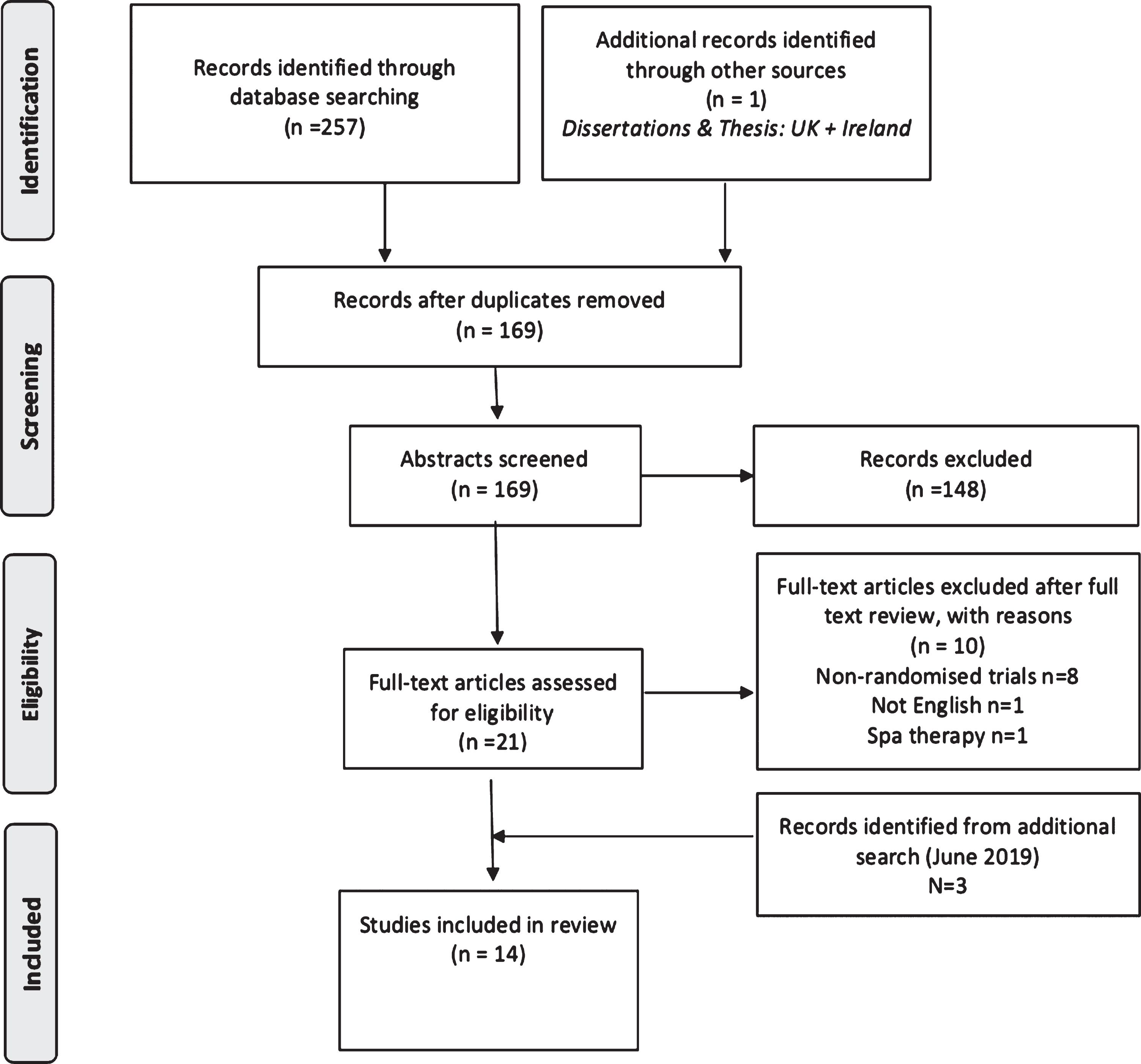
PRISMA flowchart.
Characteristics of studies
Table 1 presents the characteristics of the studies included within the review. A total of 265 participants contributed to the aquatic therapy analysis, with sample sizes ranging from six [60] to 34 people [61] with PD. Both males and female participants were included in all studies with the exception of one that excluded females [62] due to cultural factors. Participants presented with mild to moderate disease symptoms (Hoehn & Yahr stage I–IV). Eight studies involved testing during the ‘ON’ medication phase [28, 63–67] and five studies in the ‘OFF’ phase, with medication withheld between eight [62] to 12 h before assessments [30, 69].
Characteristics of studies included in review
SD, standard deviation; IQR, interquartile range; AT, aquatic therapy; PEDro, Physiotherapy Evidence Database.
Methodological quality
The methodological quality of the studies varied from low [60] to high [28] according to the Physiotherapy Evidence Database Scale (PEDro) and Downs and Black checklist (Table 1). The median score for methodological quality using the PEDro scale was seven out of ten (IQR 7,7). Eight of the 14 included studies scored between seven and eight implying moderate quality [28–31, 62–65], with a high risk of bias recorded for blinding of therapists and subjects, and intention to treat analysis (Table 2).
Methodological quality using the Physiotherapy Evidence Database (PEDro) scale
Total PEDro score ranges from 0 to 10 with a higher score indictive of better quality; Y, yes (=1); N, no (=0).
The Downs and Black checklist was also used to further guide and assess the methodological quality of the papers included, with a median score of 23.4 (IQR 22,24). Only one trial [28] scored a maximum score of 27 indicative of a low risk of bias, with a priori sample size calculation and time period for recruitment of participants the foremost items recorded as high risk of bias (Table 3).
Methodological quality using the Downs and Black checklist
Note: Total Downs and Black score ranges from 0 to 27 with a higher score indictive of better quality; Y, yes (=1); N, no /unable to determine, UTD (=0).
Frequency
Table 4 summarizes the frequency, intensity, time and type (FITT) of aquatic interventions implemented across all included trials. The frequency of intervention is described as the number of aquatic therapy sessions completed per week [70]. The majority of studies provided either two sessions [29, 67–69] or five sessions per week [28, 63–65].
FITT framework with targeted outcome variables and treatment components for the aquatic therapy intervention (n = 14)
AT, aquatic therapy; RPE, rate of perceived exertion; HRR, heart rate reserve; ACSM, American College of Sports Medicine.
Intensity
The intensity of interventions were heterogeneous across the studies with eleven failing to clearly define or objectively measure the intensity levels of participants [28, 63–69]. Studies varied from using the Borg Scale [29], self-selecting pacing [61], or by increasing the number of repetitions [62] as set out by the American College of Sports Medicine (ACSM) guidelines [71] for older people with chronic conditions.
Time
The length of each session varied from 30 [64] to 60 min [28, 65–67] for the majority of interventions. The intervention period ranged from three [61] to 11 weeks [69]. The overall intervention time (Table 1) ranged from 360 [60] to 2,400 min [28, 65]. Six studies included a follow-up assessment period varying from 17-days [60] to six months [31, 64].
Type
All studies included a five to 10-min warn-up and cool-down period consisting of whole body stretching, cardiovascular or relaxation activities. The main neuromuscular training components consisting of Ai Chi movements [30, 70], dual-task aquatic exercises [67], specific gait training exercises [29, 61], tailored aquatic therapy programs [60, 62], and perturbation based balance exercises [28] were compared to land-based physiotherapy or usual care [29]. Zhu et al. [64] compared aquatic obstacle training to ‘traditional’ aquatic therapy consisting of balance training using the Halliwick method.
Environmental factors and treatment setting
Water temperatures differed from 30°C to 34°C across studies. Pool depths, reported in all but six studies [28, 67], varied from 0.6 to 1.5 meters. The level of immersion at which study participants exercised in water were waist depth [62] or the mamillary line [61, 65] in three trials. Four studies took place in a hydrotherapy pool in an outpatient rehabilitation setting [31, 64]. Four studies were in an inpatient rehabilitation setting, [28, 66] four in public swimming pools [30, 70] and one in a community hydrotherapy pool [29].
Adverse events, participants involvement, and economic evaluation
Eight investigations reported no adverse outcomes following participation in the aquatic interventions, although six studies did not specify whether or not adverse events were monitored or documented during the intervention period [60, 70]. No trials reported data for economic cost, and no studies reported patient or public involvement in any stage of the research study.
Effects of aquatic therapy compared to land-based physiotherapy
Of the 14 trials included in the review, seven studies were included in the meta-analysis [28, 70]. The effects of aquatic therapy versus conventional land-based physiotherapy for balance, motor disability, mobility and health related quality of life were included in the meta-analysis as they were the most common outcomes of interest. Carroll et al. [29] could not be included in the meta-analysis as they published data as median and interquartile range values and the data were skewed. Zhu et al. [64] compared aquatic obstacle course training to an aquatic therapy arm, which could not be included in meta-analysis comparing aquatic therapy to land-based physiotherapy. Volpe et al. [61] did not report data for the control groups as they compared healthy subjects matched for age, body mass index and gender to the experimental group of people with PD.
Gait
Five studies [29, 66] reported outcomes relating to gait. Freezing of gait was assessed in three trials [29, 66] using the self-evaluating Freezing of Gait Questionnaire (FOGQ). Zhu et al. [64] found significant improvements in both the aquatic obstacle therapy group and traditional aquatic therapy group (p < 0.001). Carroll et al. [29] and Clerici et al. [66] observed no differences in the FOGQ between the aquatic therapy group and usual care control group or intensive motor-cognitive group respectively. Volpe et al. [61] found significant improvements in spatiotemporal parameters (stride length, stride period, stance period, speed, cadence) in participants with PD who received underwater gait training, with findings from 3D gait analysis demonstrating a significant improvement in hip, knee, ankle and shoulder ranges of motion (p < 0.05). While two studies observed no significant improvements in gait variability [29, 60], or step amplitude, turn time and cadence [60] following four [60] and six [29] weeks of twice weekly aquatic therapy.
Balance
Six studies were included in the meta-analysis for the Berg Balance Scale (BBS) when comparing aquatic therapy and aquatic Ai Chi to land-based physical interventions [28, 65] (refer to Fig. 2, or for a more detailed forest plot, see the Supplementary Material: Appendix II, Supplementary Figure 2). Small non-significant effects were observed for balance on the BBS in favor of the aquatic therapy and Ai Chi groups (2.08 points, 95% CI –0.43 to 4.60; p = 0.10, I2 = 37%), while Palamara et al. [31] found long term gains in BBS scores were maintained in more people with PD who received aquatic therapy in addition to intensive land-based rehabilitation. Kurt et al. [63] found greater improvements in dynamic balance for the aquatic Ai Chi group when compared to land-based exercises, measured with the Biodex-3.1 (anteroposterior index p < 0.001, mediolateral index p < 0.001, overall balance index p < 0.001). Mean velocity of the center of pressure (COP) was better in the aquatic therapy group compared to land-based physiotherapy (p = 0.01; 95% CI –5.2 to –0.66) [62]. Volpe et al. [28] observed non-significant changes for COP sway with eyes closed in the aquatic therapy group in comparison to participants who underwent land-based physiotherapy (45.4±64.9 vs. 6.9±45.3, p = 0.05).
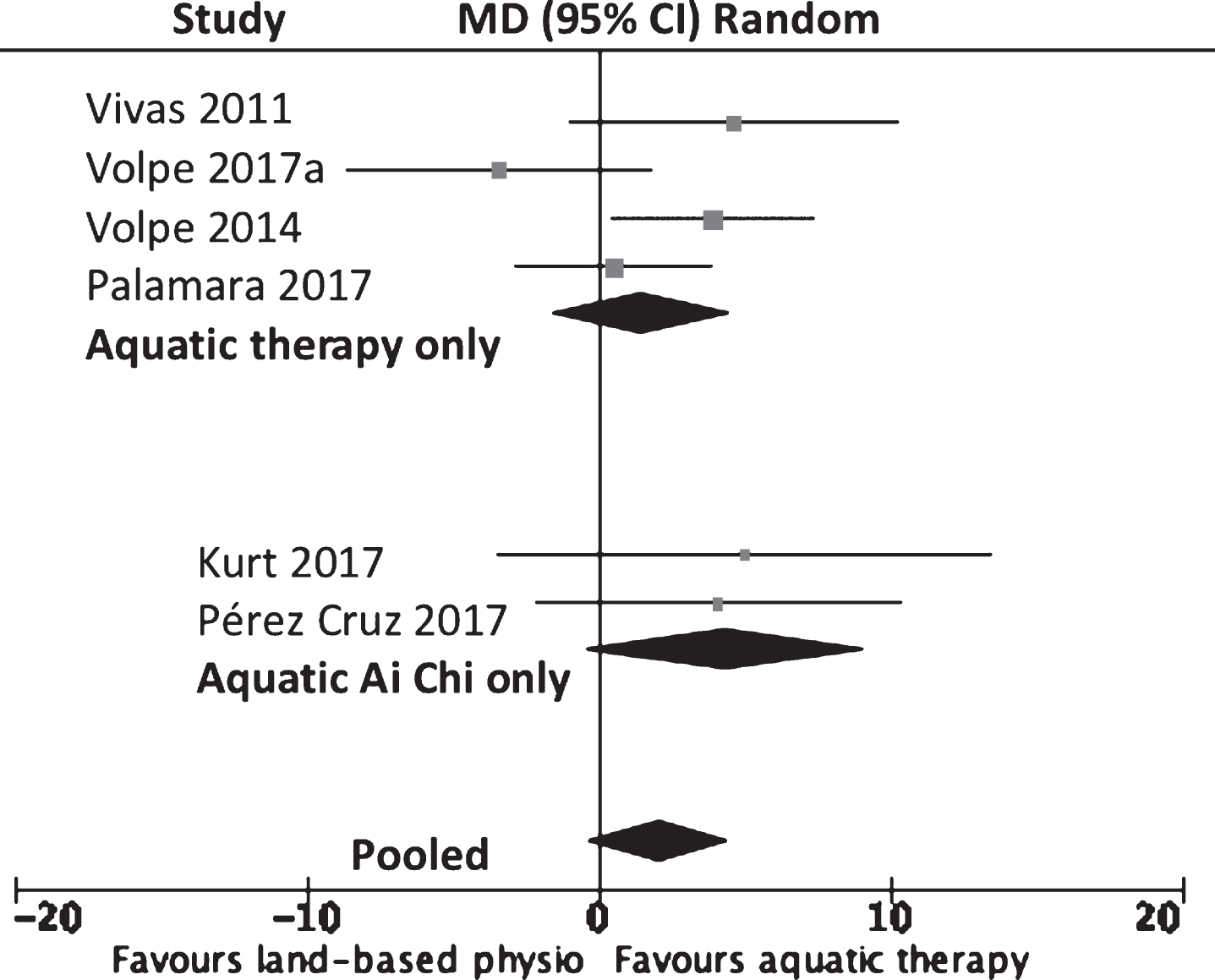
MD (95% CI) of effect of aquatic therapy immediately after 4 to 8 weeks of intervention on balance (Berg balance scale) compared to land-based physiotherapy. Data pooling for six studies (n = 173).
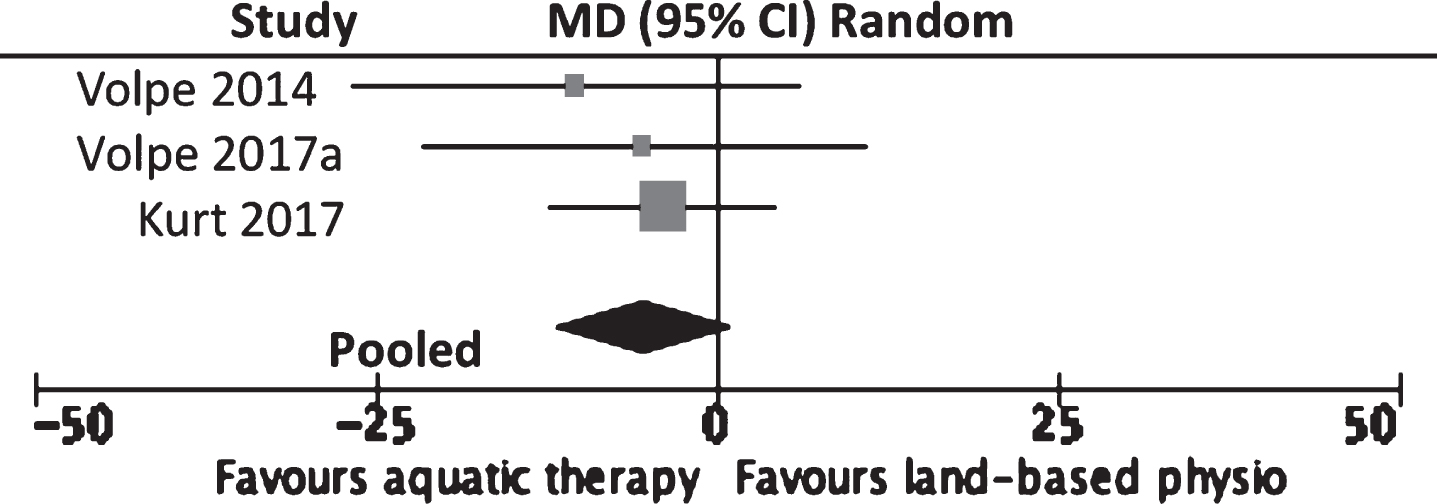
Motor disability
Five trials with a total of 162 participants reported data for the Unified Parkinson’s Disease Rating Scale motor subsection III (UPDRS III) with no difference between the aquatic therapy and aquatic Ai Chi groups compared to land-based interventions (–0.32 points, 95% CI –2.36 to 1.75; p = 0.75, I2 = 0%). (Refer to Fig. 3, or for a more detailed forest plot, see the Supplementary Material: Appendix II, Supplementary Figure 3). Carroll et al. [29] reported a significant improvement in UPDRS III scores in the aquatic therapy group (p < 0.01) in comparison with a control group who received medication alone. Small and non-significant effects were observed in favor of the aquatic Ai Chi groups compared to land-based rehabilitation [30, 63]. Two trials identified no significantly greater benefits when aquatic therapy was provided in addition to land-based movement rehabilitation [31, 66].
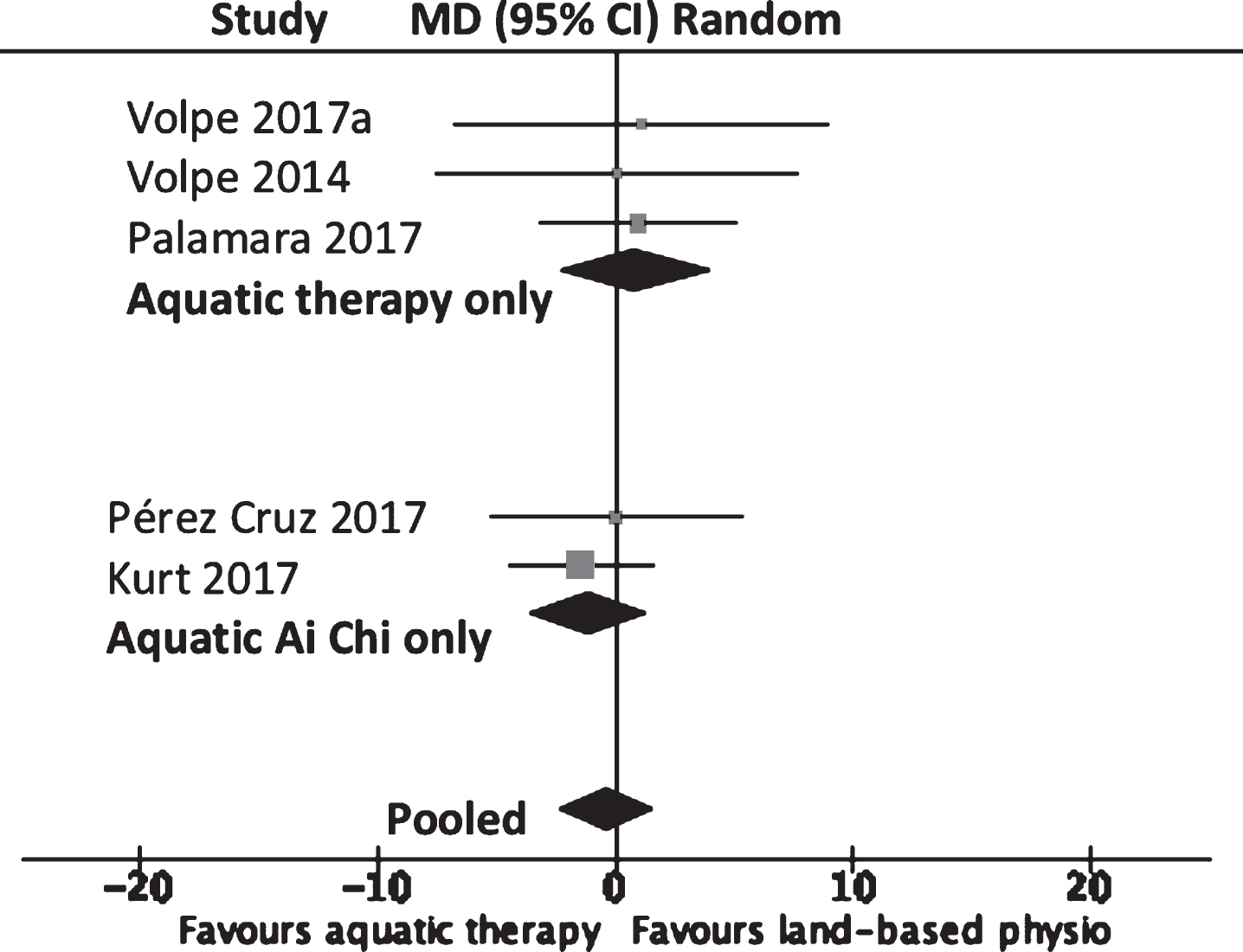
MD (95% CI) of effect of aquatic therapy immediately after 5 to 8 weeks of intervention on motor disability (UPDRS III) compared to land-based physiotherapy. Data pooling for five studies (n = 162).
Functional mobility
Following short-term aquatic therapy, statistically significant improvements (–1.5 s, 95% CI –2.68 to –0.32; p = 0.01, I2 = 13%) were seen for the Timed Up and Go (TUG) when the results were pooled for six of the 14 studies [28, 70] (refer to Fig. 4, or for a more detailed forest plot, refer to the Supplementary Material: Appendix II, Supplementary Figure 4).
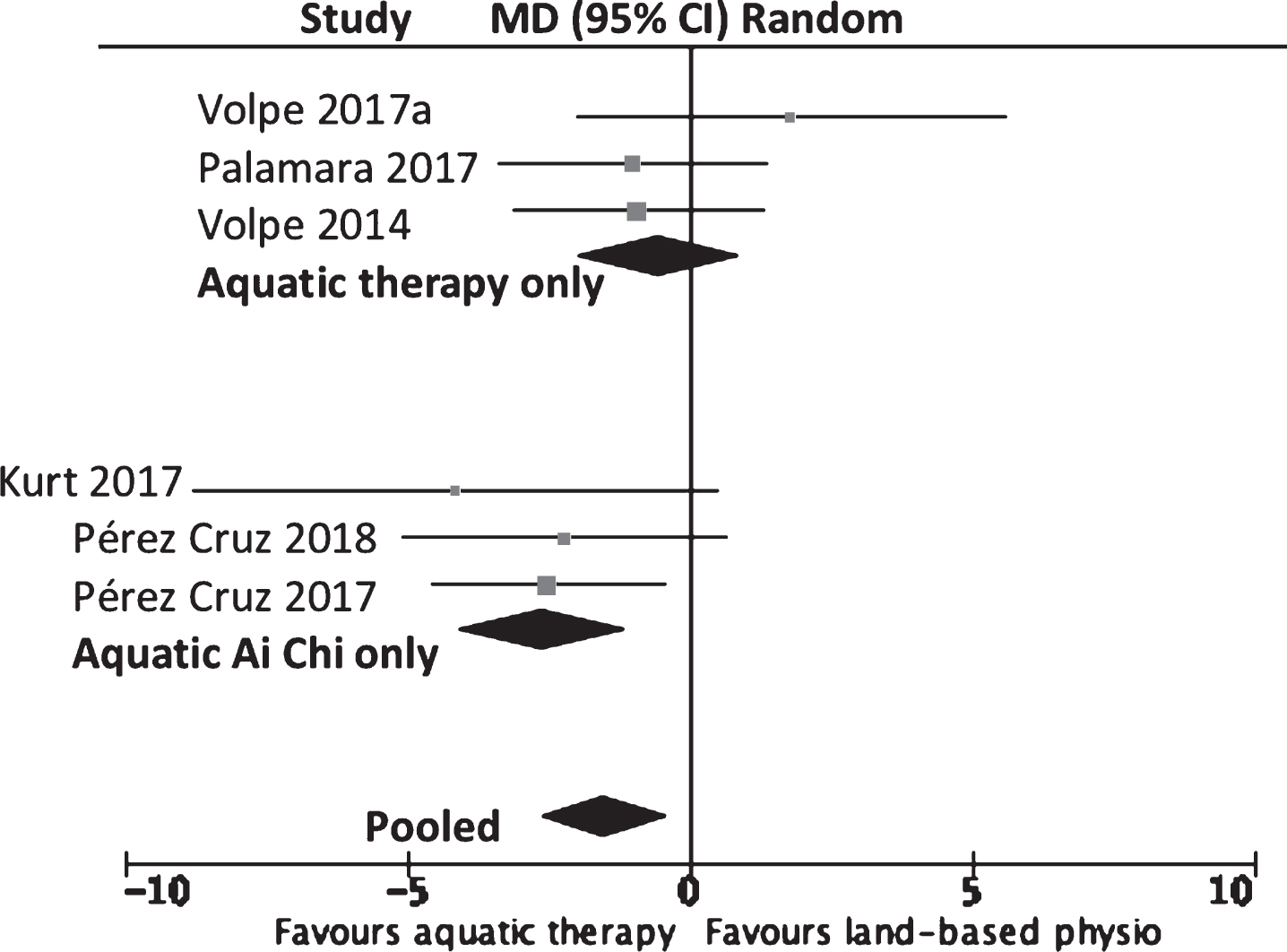
MD (95% CI) of effect of aquatic therapy immediately after 4 to 11 weeks of intervention on functional mobility (Timed Up and Go) compared to land-based physiotherapy. Data pooling for six studies (n = 191).
Falls
Two studies evaluated the outcome of aquatic therapy on falls using the Falls Efficacy Scale (FES) [28, 65] and a falls diary [28]. Both reported a significant reduction in falls following aquatic therapy and Volpe et al. [28] found significant differences on the FES (–5.9 SD 4.8 vs. –1.9 SD 1.4; p = 0.003) and falls diary (–2.4 SD 2.2 vs. –0.4 SD 0.5; p = 0.001) in favor of aquatic therapy.
Health related quality of life
No significant differences between treatment groups (–5.30 points, 95% CI –11.99 to 0.30; p = 0.12, I2 = 0%) were observed for self-reported measures of quality of life on the PDQ-39 in three trials [28, 65] (Refer to Fig. 5 or for a more detailed forest plot, see the Supplementary Material: Appendix II, Supplementary Figure 5). Total scores for the PDQ-39 were not available for one study [69], with no significant differences between aquatic therapy and land-based physiotherapy. Shahmohammadi et al. [62] reported greater improvement in the aquatic therapy group compared to a land-based exercise group for quality of life using the Farsi version of the Parkinson’s Disease Quality of Life (PDQL) questionnaire.
MD (95% CI) of effect of aquatic therapy immediately after 4 to 8 weeks of intervention on quality of life (Parkinson’s disease questionnaire 39) compared to land-based physiotherapy. Data pooling for three studies (n = 98).
DISCUSSION
The studies included in this review provided variable dosage, content and duration of aquatic interventions for people living with PD. Overall, the meta-analysis demonstrated that aquatic therapy had comparable positive outcomes to land-based physiotherapy for gait, balance, motor disability, falls and health related quality of life in the early to mid-stages of disease progression (stages I–III). Greater improvements were identified for functional mobility measured using the TUG (1.5 s). However, these improvements were less than the minimal detectable change (MDC) score of 3.5 s reported for the TUG in people with PD [72]. Arguably, a lack of targeted aquatic exercise interventions for specific movement disorders in PD may have limited the effects of aquatic therapy.
This meta-analysis adds to previous reports [37–39, 73], with some new insights gained. A previous review by Methajarunon et al. [73] was unable to draw firm conclusions as it only included two trials with small sample sizes. Another analysed within group effects only and included non-randomized trials [38]. Our analysis is based on raw data from seven randomized controlled trials and a total of 209 participants. A recent meta-analysis by Cugusi et al. [74] had similar results for functional mobility. In contrast to our findings, that review [74] also reported small to moderate improvements following aquatic therapy for balance, fear of falling and health related quality of life when compared to land-based exercise. Whereas our review calculated the mean difference of pre and post-test values, Cugusi et al. [74] included only mean values for post-test data.
Optimal frequency, intensity, duration, and type of aquatic therapy?
Due to large variations in aquatic therapy interventions and outcome measures, we were unable to ascertain the optimum, intensity, time and type of aquatic therapy suitable for PD. It is likely that the content, dosage, duration, intensity and scheduling of aquatic therapy needs to be tailored to individual needs, considering the person’s stage of disease progression, level of impairment, medications, activity limitations, co-morbidities and general fitness.
Frequency
Findings from high quality studies on people with mild to moderate PD that implemented between three [31] and five sessions per week [28, 64] reported statistically greater improvements in balance, [28, 64] functional mobility, health related quality of life, motor disability [63] and gait [64] in the aquatic therapy group than land-based physiotherapy. This suggests that in order to gain clinically meaningful improvements in movement and wellbeing for PD, a minimum of three sessions per week ought to be conducted. However, participation in three to five sessions every week may not be feasible for all people living with PD. There can be challenges with access to aquatic therapy resources, trained health professionals and supervision in community settings, with a lack of time identified as a barrier to exercise in people with PD [75]. Thus, exploration of land based strategies to augment the benefits gained and meet the frequency recommendations may be useful.
Duration
Variations in the length of time of each session (30 min to 1 h) and duration of aquatic therapy interventions (three to 11 weeks) were noted. These variations require consideration and may have influenced outcomes. The mean aquatic therapy dose was 3.4 sessions of 51.8 min over 6.6 weeks. The European physiotherapy guidelines for exercise in PD [3] recommends a minimum of three, 45-min sessions per week over an eight-week period, which is a total of 24 sessions lasting 1,080 min [3]. Four of the studies included in this review achieved this dose [28, 65], with greater improvements noted in these moderate to high quality trials for balance [28, 63], trunk posture [65], and quality of life [28, 63] in the aquatic therapy groups compared to land-based physiotherapy. Thus, it is recommended that researchers use appropriate guidelines for frequency and intervention dosage in order to enable comparison and repeatability of aquatic therapy for PD.
Intensity
Most of the studies included omitted measuring intensity or the level of effort achieved during aquatic therapy. Trials did not always account for rest periods taken during aquatic exercise programmes affecting accuracy in reporting exercise intensity levels [76]. The majority of interventions focused solely on the progression of neuromotor or task specific aquatic exercises and were not directed at achieving a high intensity rate, required to positively influence exercise enhanced neuroplasticity [77]. The inclusion of intensity measurement tools to improve speed and load achieved during future trials for example, the Borg rating of perceived exertion scale [78, 79], an auditory metronome [80], or heart rate polar monitor [81] using the formula HRmax in water = HRmax on land – ΔFC (where ΔFC = bradycardia resulting from water immersion) as described by Graef and Kruel [79], are important in order to establish an optimum intensity target for people with PD exercising in water [76]. To enable people with PD to perform past their self-perceived ability, future research could incorporate introduction sessions where participants are introduced to single-task exercises targeting specific impairments with an emphasis on movement quality [22, 62], and how buoyancy and drag can influence movement in water [82]. Higher intensity neuro-motor exercise training in water, if sufficiently prescribed, may lead to better outcomes over land-based exercise for people with PD.
The limited effectiveness of aquatic therapy over land-based exercise, with the majority of studies unable to achieve a sustained treatment effect, may be due a number of factors such as inadequate clinical application of buoyancy and drag forces during task specific activities in water [83] and failure to educate patients on an optimal speed of movement in water [82]. Heywood et al. [82] identified that without precise guidance on speed of movement required for task-specific exercises, maximal speeds of movements in water will likely be less that on land, due to the impact of drag forces. Thus, in line with previous research findings on speed-amplitude relations in PD, it is recommended that clinician’s focus on movement amplitude as one possible intervention strategy to improve speed-amplitude scaling for people with PD exercising in water [84].
Content of physiotherapy interventions
Our findings provide a preliminary comparison of various aquatic therapy approaches utilized by physiotherapist for people with PD. Variances in aquatic therapy outcomes could contribute to some of the equivocal findings of this review, which may be explained by the wide range of aquatic therapy exercise components adopted across trials. For example, aquatic therapy comprising of “perturbation-based balance training” [28] might be beneficial for some people with PD experiencing loss of balance and falls by exposing them to drag forces and performing exercises against a turbulent flow of water [33] to stimulate enhanced balance reactions.
Ai Chi
Higher quality trials suggested that aquatic Ai Chi [63] can be an effective approach to improving balance, disability, mobility and quality of life in PD. People with PD who participated in the Ai Chi intervention groups, performed a series of repeated exercises standing in shoulder deep water. The exercises progress in difficulty from static to dynamic, symmetrical to asymmetrical, and trunk rotational movements, with additional visual and non-visual challenges also included. It may be hypothesized that this type of aquatic exercise, focusing on amplitude and precision of movement could benefit impairments of bradykinesia and postural control. While the inclusion of conscious breath training along with the physiological benefits of water immersion, could help target non-motor impairments such as anxiety and depression, by regulating the autonomic nervous system [33].
Aquatic obstacle course training and dual-task aquatic exercises
Findings from high quality trials on aquatic obstacle course training [64], involving multi-directional movements up, down, around and over changing obstacles could improve freezing impairments in some people with PD. The pathophysiology of freezing of gait in PD is not clearly understood but one could hypothesize that aquatic obstacle training, utilizing resistance forces in water may stimulate positive coupling of a normal postural preparation for step to the swing phase of a step, by directing attention to the acquisition of specific stepping skills in water [85]. Moderate quality research on dual-task aquatic exercise training incorporated similar multidirectional activities in water and offers a promising aquatic approach for improved functional mobility, balance and gait in PD. It is recommended that for future trials, researchers provide a theory driven rationale for the development of interventions and clearly define the specific characteristics of aquatic therapy [69], which are optimal for targeting varying movement disorders associated with PD (Table 4).
Study designs
Involvement of key stakeholders in treatment design [86] as well as the inclusion of patient orientated outcomes and goals was only reported in two trials [31, 66]. Participant involvement in the research engages experiential evidence that includes the participant’s perspective, functional difficulties and their preferred treatment needs [87]. The input of people with PD in a study’s protocol design, taking account of their individual opinions on treatment acceptability, may also help to increase the cost effectiveness of trials by directing researchers towards interventions, which are likely to be beneficial and effective for future trials [88, 89].
Recommendations for future research
Further, large scale randomized controlled trials with longer follow-up periods, specified intensity doses are required and would allow better comparisons of aquatic therapy with land-based physical interventions. The characteristics and mechanisms by which aquatic therapy can optimally contribute to impairments present in PD, and the extent to which these improvements may last over time await further investigated. The inclusion of outcome measurement tools evaluating underwater movements, such as 3D motion gait analysis [61] would help to strengthen our understanding of the specific features and benefits of aquatic therapy for PD.
This review included PD stages I–IV with no adverse effects during aquatic therapy intervention found however, group aquatic therapy could be unsafe for people with advanced PD, Hoehn & Yahr stages IV-V and may warrant consensus among expert researchers and clinicians working in the field. Recent research suggests that PD can interfere with swimming and/or floating capacity and may pose a safety risk relating to asymmetric characteristics, poor coordination, and problems executing complex tasks [90].
None of the trials reviewed incorporated an economic assessment on the cost of implementing aquatic therapy, thus little is known about the economic value and cost effectiveness of aquatic therapy for PD. Given the increasing burden of cost associated with PD progression [91], aquatic therapy services need to be deliverable within the financial limitations of modern healthcare systems. Due to a decline in hospital hydrotherapy services for the treatment of chronic conditions in some countries such as the UK [92, 93], as well as increasing pool closures, the costs of implementing aquatic therapy for people living with PD needs to be taken into account when designing trials [92, 93]. A recent retrospective analysis of medical claims [94] provided preliminary evidence that specialized physiotherapy delivered through a community-based programme can reduce overall treatment costs and is associated with less disease related complications. The extent to which specialist aquatic therapy services may reduce costs and limit complications associated with PD related impairments needs to be investigated further.
Strengths and limitations
All randomized controlled trials were comparable in focusing on the effectiveness of aquatic therapy in PD, with the seven studies and outcomes included for meta-analysis demonstrating minimal heterogeneity (I2 <50%) however, there are some limitations of this meta-analysis. Firstly, reporting bias was identified in five studies that failed to indicate time periods over which participants were recruited [30, 69]. Relevant data such as loss of patients to follow-up and the inclusion of intention-to-treat analysis was omitted in six trials [29, 65–67]; thus, outcome reporting bias in these studies may have founded misleading, positive results of the effectiveness of aquatic therapy. Trials included were small with only seven studies [30–32, 66–68] including a power and sample size calculation, while five studies were documented pilot studies [60, 69].
The limited effects of aquatic therapy over land-based physiotherapy for gait variables such as gait variability, step amplitude, turn time and cadence may have been associated with an insufficiently powered trial [29, 60]. Only one low quality study [60], which conducted testing during the “OFF” medication phase was suitable for inclusion in the meta-analysis making it difficult to establish if there was an overall difference in results when comparing to “ON” medication testing. Finally, the Berg Balance Scale was included in the meta-analysis as it was used in a number of studies (n = 6). However, research has shown [95] that it has modest sensitivity and high ceiling effects and thus may not be the most appropriate measure to detect a change in balance during the early stages of PD. Other measures, such as the Mini-BESTest may have been more suitable to demonstrate differences especially where balance was targeted [96].
Conclusion
Aquatic therapy has comparable positive outcomes to land-based physiotherapy, showing benefits for gait, balance, motor disability, mobility, falls and quality of life in people in the early to middle stages of disease progression in PD. Aquatic therapy had superior outcomes for gait, balance and mobility for trials that delivered at least 3–5 sessions per week. The optimal frequency, intensity, duration, and type of aquatic exercise for people living with PD could not be confirmed from this review due to comparatively low dosages of therapy delivered and variations in therapy content. Group-based aquatic therapy is likely to be contra-indicated in those with advanced PD due to safety issues associated with severe movement disorders and co-morbidities.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
