Abstract
Background:
Delirium is a serious acute neuropsychiatric condition associated with altered attention and arousal.
Objective:
To evaluate simple bedside tests for attention and arousal to detect delirium in those with and without Parkinson’s disease (PD) and dementia.
Methods:
Participants from two prospective delirium studies were pooled comprising 30 with PD without cognitive impairment, 24 with Lewy body cognitive impairment (PD dementia or dementia with Lewy bodies), 16 with another dementia and 179 PD and dementia-free older adults. Participants completed standardised delirium assessments including tests of attention: digit span, Memorial Delirium Assessment Scale (MDAS) attention and months of the year backwards; and arousal: Glasgow Coma Scale (GSC), Observational Scale of Level of Arousal (OSLA), Modified Richmond Agitation Scale and MDAS consciousness. Delirium was diagnosed using the DSM-5 criteria.
Results:
On their first admission, 21.7%participants had prevalent delirium. Arousal measures accurately detected delirium in all participants (p < 0.01 for all), but only selected attention measures detected delirium in PD and dementia. In PD and dementia-free older adults, impaired digit span and OSLA were the optimal tests to detect delirium (area under the curve [AUC] = 0.838, p < 0.001) while in PD and dementia the optimal tests were MDAS attention and GCS (AUC=0.90 and 0.84, respectively, p < 0.001 for both).
Conclusion:
Simple bedside tests of attention and arousal at a single visit could accurately detect delirium in PD, dementia and PD and dementia-free older adults; however, the optimal tests differed between groups. Combined attention and arousal scores increased accuracy, which could have clinical utility to aid the identification of delirium neurodegenerative disorders.
INTRODUCTION
Delirium is an acute, neuropsychiatric disorder affecting approximately 15%of older people in hospital [1]. It is characterised by inattention, deranged level of arousal and fluctuating cognitive impairments [2]. It is highly distressing, expensive and associated with poor outcomes, including increased mortality, institutionalisation and cognitive decline [3–5]. However, delirium remains under-diagnosed and poorly documented, resulting in worse outcomes [1, 7].
People with neurodegenerative disorders, such as Parkinson’s disease (PD) [8] and dementia [9] may be at increased risk of delirium. The prevalence of delirium in those with PD admitted to hospital is high at 34%[10], and it is thought that PD with and without dementia confers a vulnerability to delirium [11]. Dementia is a syndrome characterised by cognitive decline and impaired functional independence and activities of daily living [2]. Dementia is a major risk factor for delirium [9]. The prevalence of delirium superimposed on dementia (DSD) is estimated to be 22–89%depending on the setting (e.g., in inpatients across specialties, in nursing homes or in the community) [12], population [9], and outcomes following DSD are worse than in delirium alone [12–14].
It is vital to differentiate delirium from underlying PD or dementia, as delirium may be the first or only symptom of a potentially life threatening, but treatable, underlying cause. Additionally, delirium itself has been shown to be preventable in some cases [15]. However, there is considerable uncertainty, regarding how best to diagnose DSD [16]. Currently, DSD is evaluated using tools designed for delirium alone. No studies have evaluated the efficacy of delirium diagnostic tools in PD or PD with dementia (PDD) [8]. This is problematic given that delirium and PD share many overlapping features, including sleep disturbance, daytime somnolence, impaired attention, hallucinations, and delusions [8, 17], making delirium in PD difficult to identify. Additionally, many of the tools used test multiple cognitive domains and so may be abnormal in someone with PD or dementia, even when delirium is not present. This means that a delirium diagnosis may be missed or incorrectly diagnosed in people with PD and dementia [8].
Demonstrating impaired attention and altered levels of arousal are essential components of Diagnostic and Statistical Manual of Mental Disorders fifth edition (DSM-5) criteria [2], required for a diagnosis of delirium, and both have been shown to be relatively specific to delirium [18–20]. Recent evidence suggested that altered arousal and inattention may be associated with increased mortality [21]. There is consensus that they should be considered together, with the inability to engage in cognitive testing due to abnormal arousal (above the level of coma) considered to be severe inattention for the purposes of delirium diagnosis [22]. A combined assessment of attention (letter vigilance “S-A-V-E-A-H-E-A-R-T” test) and arousal (Observational Scale of Level of Arousal, OSLA) has previously been shown to accurately identify DSD and provides a simple, brief bedside test for delirium, which can be administered with minimal training [17]. However, few studies have evaluated the performance of these tests in people with other neurodegenerative conditions, and none in PD [8]. PD, PDD, and dementia with Lewy bodies (DLB) have a number of intrinsic non-motor, neuropsychiatric manifestations, including inattention and fluctuating arousal [8, 24]. Given the considerable overlap in their clinical phenotypes, it is not known whether common bedside tests for attention and arousal will correctly identify delirium in patients with PD with or without dementia.
We aimed to evaluate the ability of simple bedside tests used by non-specialists in hospital settings for attention and arousal to detect delirium in those with and without PD and dementia. We also tested for the optimal cut-offs for each test for the different neurodegenerative disease groups. We hypothesized that simple bedside tests would be sensitive to identifying delirium in PD and dementia, but that different cut-offs would be required compared to older adults without underlying neurodegenerative diseases.
METHODS
Participants and recruitment
All participants from the Identifying Delirium in People with Parkinson’s Disease (DETERMINE-PD) [10] and Delirium and Cognitive Impact in Dementia (DECIDE) [25] studies were included in the analysis. Both studies were approved by Research Ethics Committees (DETERMINE-PD REC reference: 17/YH/0402; DECIDE REC reference: 15/NE/0353).
The DETERMINE-PD study [10] was a pilot study which aimed to identify delirium in inpatients with PD. Patients who attended outpatient clinics in Newcastle Hospitals for the management of their PD and admitted to hospital between 26 March and 25 July 2018 were invited to take part in the study. Inclusion criteria comprised a diagnosis of PD according to UK Brain Bank Criteria [26] made by a movement disorder specialist, and a hospital admission during the recruitment period.
The DECIDE study was nested within the Cognitive Function and Ageing Study II –Newcastle cohort (CFAS II-Newcastle). CFAS II is a large, population-based cohort of people aged≥65 years from three geographical areas in the UK including Newcastle upon Tyne [25, 27]. From 5th January 2016 to 5th January 2017, we invited participants from CFAS II-Newcastle to participate in DECIDE on admission to hospitals in Newcastle.
We were alerted to admissions of participants in both CFAS II-Newcastle and DETERMINE-PD by a Recurring Admission Patient Alert (RAPA) attached to the participants’ electronic records. For both studies, if participants lacked capacity to give written informed consent, an appropriate personal consultee was requested to provide written confirmation of willingness to participate. A formal capacity assessment informed by the UK Mental Capacity Act [28] was performed by a trained member of the research team. Participants were excluded if they lacked capacity to consent and no personal consultee was available, they were receiving end of life care, they were being isolated for infection control reasons or they were expected to be in hospital for fewer than 24 hours. In DETERMINE-PD, participants were also excluded if they had a diagnosis of non-idiopathic PD or had insufficient English to complete the assessments.
In DETERMINE-PD, participants were assessed in a single research visit while in hospital as soon as possible after admission. In DECIDE, participants were assessed as soon as possible after admission to hospital (visit 1) and repeatedly assessed daily, as far as possible, during their hospital admission. To facilitate comparisons and data integration with the DETERMINE-PD study, only DECIDE visit 1 data were used.
Measures
Baseline data included age, sex, years of education, frailty (Clinical Frailty Score, CFS) [29] and comorbidities (including dementia or Parkinson’s disease diagnosis) [30]. In PD participants, an existing diagnosis of mild cognitive impairment (PD-MCI) or dementia (PDD) were recorded where available.
We ascertained delirium on admission to hospital using a standardised approach based on DSM-5 criteria used in the DECIDE study (Supplementary Table 1), which is described in full elsewhere [25]. In brief, the criteria included assessments, questions or information extracted in the following order: objective testing (bedside tests of attention and awareness, memory, orientation, language, visuospatial ability, and perception), participant observations, collateral history from a next of kin, information from nursing staff and medical notes. The collateral history was taken from participants’ next of kin to determine whether symptoms were an acute change. Items from the Memorial Delirium Assessment Scale (MDAS) were completed at the end of assessments; where there was overlap in assessments (i.e., orientation, short term memory, digit span), test performance as part of the DSM-5 criteria were used to rate MDAS items. We also recorded performance on several tests of attention and arousal (Table 1). The delirium assessments took approximately 10–15 minutes to complete.
Tests of attention and arousal recorded for all participants
Statistical analysis
Statistical analyses were performed using SPSS software (Version 24.0; SPSS, Armonk, NY: IBM Corp). Data were examined for normality of distribution with visual histograms and Kolmogorov-Smirnov’s test. Comparisons of means between two groups were performed using independent t-tests, Mann-Whitney U tests or Pearson χ2 tests, depending on distribution. Comparison between more than two groups were performed using ANOVA or Kruskal-Wallis tests, as appropriate. Groups comprised participants with and without delirium, PD and dementia-free older adults, PD without cognitive impairment, PD with cognitive impairment (PD-MCI and PDD) and those with another dementia. A small number of participants had a diagnosis of DLB (n = 4); therefore, they were included with the PD with cognitive impairment group to form a Lewy body cognitive impairment (LB-CI) group.
The accuracy of measures of attention and arousal were determined using the area under the receiver operating characteristic (ROC) curves (AUC) with 95%confidence intervals (95%CIs). Sensitivity and specificity scores were calculated and Youden’s index was used to identify optimal cut-offs. To aid interpretation, the m-RASS was modified so that negative scores were multiplied by -1, thus all scores were positive (0–5). All statistical tests were two-sided with statistical significance set at 0.05.
Data availability statement
Unidentifiable data may be shared on request.
RESULTS
In total, 249 participants accounting for 371 admissions were included in this analysis (Fig. 1). Fifty-four (21.7%) participants had a Lewy body disease comprising 30 (12.0%) participants with PD without cognitive impairment, 24 (9.6%) with LB-CI (PD-MCI, PDD, or DLB) and 16 (6.4%) with another dementia. Of these, 21.7%(n = 54) had prevalent delirium (delirium present on first assessment) on their first admission. Participants in the DETERMINE-PD study were significantly younger, had completed more years of education, were frailer and had a higher proportion of male participants compared to DECIDE (p < 0.05 for all, Supplementary Table 2).
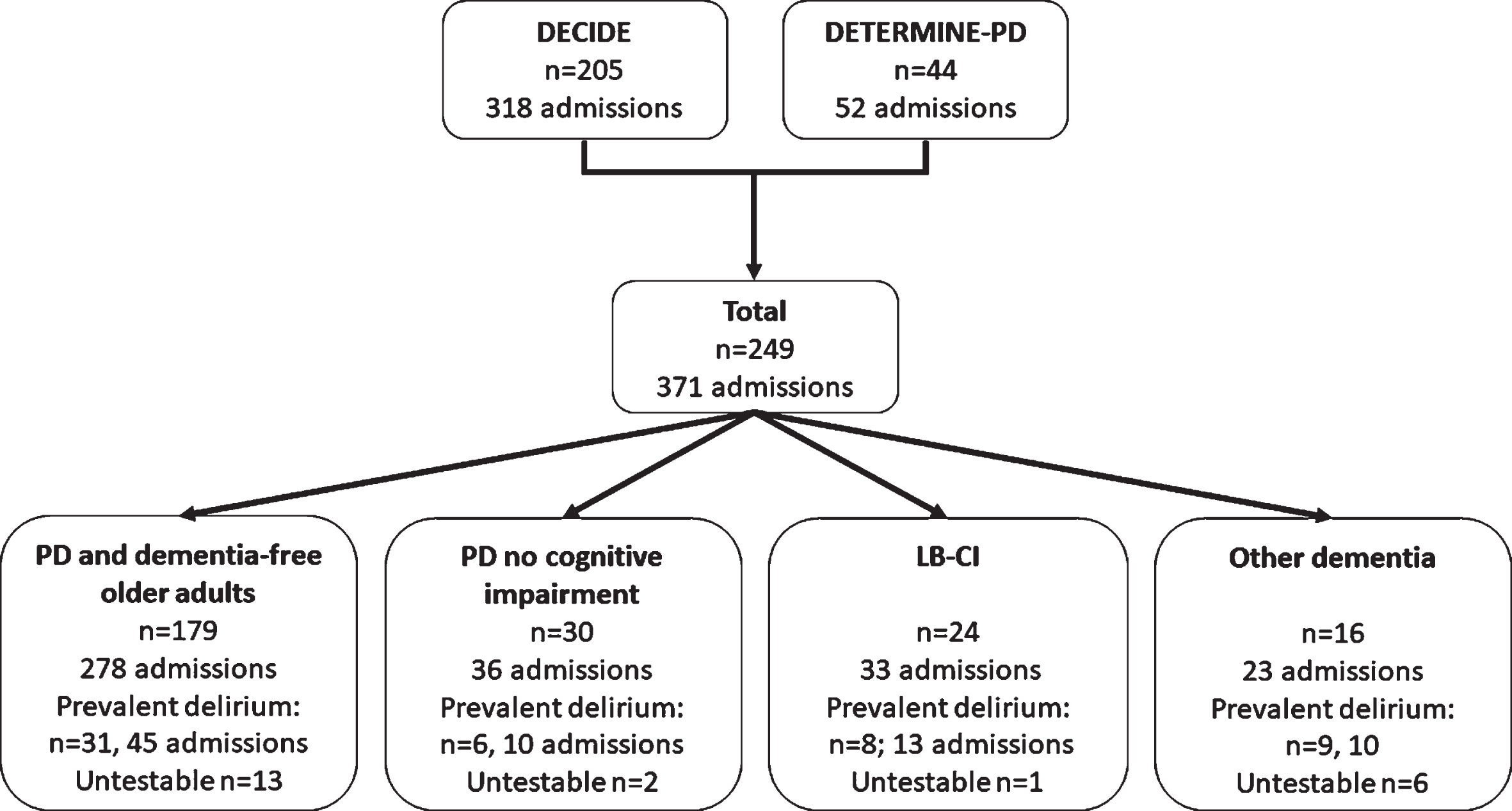
Pooled participants from the DECIDE and DETERMINE-PD studies. PD, Parkinson’s disease; LB-CI, Lewy body cognitive impairment group; PD-MCI, Parkinson’s disease with mild cognitive impairment; PDD, Parkinson’s disease dementia; DLB, dementia with Lewy bodies, untestable, participants with abnormal arousal levels where it was not possible to complete tests of attention.
Demographics of participants from their first admission (Table 2) to hospital show that cases with prevalent delirium were significantly older and frailer (p < 0.05). Seventy-eight (31.3%) participants had ≥2 admission to hospital. Cases with prevalent delirium were more likely to have a dementia (23.1%vs. 6.5%, p < 0.01) or PD (29.5%vs. 14.3%, p < 0.01).
Baseline demographics characteristics of cases with and without prevalent delirium
Data presented are mean (SD) unless otherwise stated. Significant results are highlighted in bold. CFS, Clinical Frailty Score; PD, Parkinson’s disease; LB-CI, Lewy body cognitive impairment comprising PD-MCI, PDD, and DLB; PD-MCI, Parkinson’s disease with mild cognitive impairment; PDD, Parkinson’s disease dementia; DLB, Dementia with Lewy bodies.
Between group differences for attention and arousal
Differences in attention and arousal scores for each disease group (PD without cognitive impairment, LB-CI, other dementia and PD and dementia-free older adults) in cases with and without delirium are shown in Table 3. Due to low arousal levels, n = 22 participants were unable complete attention tests. In older adults without PD or dementia, cases with delirium scored significantly worse on all attention and arousal tests (p < 0.001 for all) apart from the m-RASS (p > 0.05). In PD without cognitive impairment, cases with delirium scored significantly worse on all measures apart from digit span forwards and m-RASS (p > 0.05 for both). Conversely those with LB-CI scored poorer for all arousal measures, but only poorer for MDAS digit span and MDAS attention (p < 0.05 for all) in terms of attention tests. Only MDAS attention and arousal as measured by the GCS and modified m-RASS were significantly poorer in cases with dementia (LB-CI and other dementias) and delirium compared to cases without delirium (p < 0.05).
Between group differences in attention and arousal tests at admission in cases with and without prevalent delirium
Data presented are mean (SD) unless otherwise stated. Significant results are highlighted in bold. PD, Parkinson’s disease; LB-CI, Lewy body cognitive impairment group comprising PD-MCI, PDD, and DLB; PD-MCI, Parkinson’s disease with mild cognitive impairment; PDD, Parkinson’s disease dementia; DLB, Dementia with Lewy bodies; MDAS, Memorial Delirium Assessment Scale; MOTYB, months of the year backwards; GCS, Glasgow Coma Scale; OSLA, Observational Level of Arousal; m-RASS, Modified Richmond Agitation Scale.
Attention and arousal accuracy
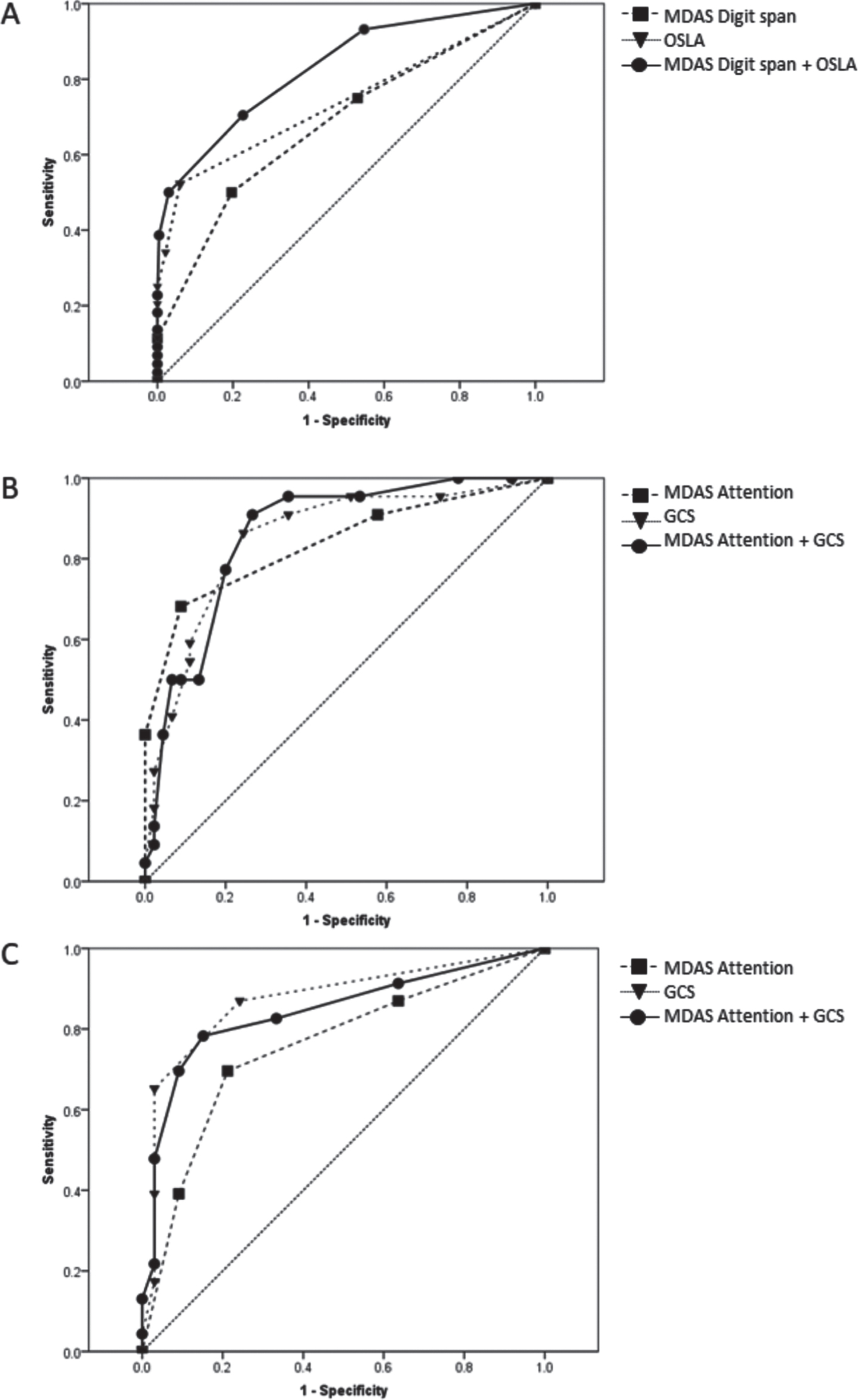
ROC curves for (A) older adults without PD or dementia, (B) participants with PD/LB-CI, and (C) participants with dementia. PD, Parkinson’s disease; LB-CI, Lewy body cognitive impairment comprising PD-MCI, PDD, and DLB; PD-MCI, Parkinson’s disease with mild cognitive impairment; PDD, Parkinson’s disease dementia; DLB, Dementia with Lewy bodies; MDAS, Memorial Delirium Assessment Scale; GCS, Glasgow Coma Scale; OSLA, Observational Level of Arousal. To facilitate comparison, GCS scores were reversed.
The diagnostic test accuracy of each cognitive measure was assessed using ROC curves (Table 4) in PD and dementia-free older adults, in cases with PD (with and without cognitive impairment and DLB), and those with dementia (including LB-CI). Area under the curve ranged from 0.62-0.89. For measures of attention, only the MDAS attention scores significantly predicted delirium in all groups (AUC range 0.70–0.74, p < 0.001 for all). Forward digit span was significantly associated with delirium in PD and dementia-free older adults (AUC = 0.67, p = 0.01) but not in those with PD or dementia. Backwards digit span, MDAS digit span and MOTYB scores were significantly associated with delirium in PD and dementia-free older adults (AUC = 0.77, 0.77 and 0.74, respectively, p < 0.001) and PD (AUC = 0.70, 0.75 and 0.73, respectively), but not in dementia (p > 0.05). All tests of arousal were significantly associated with delirium in all admission groups (AUC = 0.61–0.89, p < 0.05 for all).
Identifying cut-off points for identifying impaired tests in participants with delirium
Significant results are highlighted in bold. PD, Parkinson’s disease; LB-CI, Lewy body cognitive impairment comprising PD-MCI, PDD, and DLB; PD-MCI, Parkinson’s disease with mild cognitive impairment; PDD, Parkinson’s disease dementia; DLB, Dementia with Lewy bodies; MDAS, Memorial Delirium Assessment Scale; MOTYB, months of the year backwards; GCS, Glasgow Coma Scale; OSLA, Observational Level of Arousal; m-RASS, Modified Richmond Agitation Scale.
Identifying optimal cut-offs
Optimal cut-offs of significant measures of arousal and attention were calculated using sensitivity and specificity scores and Youden’s index (J) (Table 4). In PD and dementia-free older adults, the optimal test for identifying impaired attention was the MDAS digit span with cut-off of 3/4 (AUC = 0.77, sensitivity 61.1%, specificity 79.4%, J = 0.41), while the optimal test for arousal was the OSLA with a cut-off of 0/1 (AUC = 0.74, sensitivity 52.3%, specificity 94.0%, J = 0.46).
In PD, LB-CI and dementia, the optimal test for attentional dysfunction in delirium was the MDAS attention item with a cut-off of 0/1 (PD and LB-CI: AUC = 0.84, sensitivity 68.2%, specificity 92.9%, J = 0.61; dementia; AUC = 0.78, sensitivity 68.1%, specificity 83.9%, J = 0.52). The optimal test for arousal was the GCS in both groups, but the optimal cut off differed, with 13/14 identified in PD/LB-CI participants (AUC = 0.89, sensitivity 73.9%, specificity 90.5%, J = 0.6.4) and a cut-off of 14/15 in dementia participants (AUC = 0.89, sensitivity 88.0%, specificity 75.8%, J = 0.64).
Combined attention and arousal scores
Combining optimal measures of attention and arousal scores (MDAS attention and GCS, reversing GCS scores for compatibility to yield a max score of 18, with higher scores indicting greater impairment) correctly classified 87.0%of PD and LB-CI participants (cut-off 1/2, AUC = 0.899, p < 0.001, sensitivity 82.6%, specificity 89.1%, J = 0.717) and 85.7%of dementia participants (cut-off 1/2, AUC = 0.871, p < 0.001, sensitivity 78.3%, specificity 90.9%, J = 0.69). Combining optimal test scores in older adults without PD or dementia, (MDAS digit span and OSLA, range 0–18 with higher scores indicting greater impairment) using a cut-off of 0/1 correctly classified 73.7%(AUC = 0.838, p < 0.001, sensitivity 84.1%, specificity 71.8%, J = 0.56).
Discussion
This is the first study to evaluate simple bedside tests of attention and arousal in PD and dementia compared with PD and dementia-free older adults, and to identify optimal cut-offs. We found that all tests were sensitive to detecting delirium in older adults without PD or dementia during a single study visit. Although all arousal measures could accurately detect delirium in participants with PD and dementia, only selected attention measures were able to identify delirium in these groups. Furthermore, we identified that stricter cut-offs for attention measures may be needed to accurately detect delirium in those with PD or dementia. In PD and dementia-free older adults, impaired digit span and OSLA were the optimal tests to detect delirium, while in PD and dementia the optimal tests were MDAS attention and GCS. Consistent with findings by Richardson et al. [17], a single combined score summing the two tests (attention plus arousal) performed better than the two tests individually in all groups.
Consistent with previous studies in other populations, we found that brief measures of attention and arousal are able to detect delirium in PD and dementia-free older adults with high levels of sensitivity and specificity [17, 20]. Dysfunction in attention and arousal are essential criteria for the diagnosis of delirium, and in the absence of an existing biomarker, simple bedside diagnostic tests are used to identify delirium in clinical practice [31]. However, few studies have evaluated the psychometric properties of scales or measures used to determine their accuracy in identifying dysfunction. In addition, it is important to identify whether these tools differentiate delirium from important differential diagnoses with overlapping phenotypes, including PD [8] and dementia [17].
We found that the forward digit span test was only sensitive to detecting delirium in PD and dementia-free older adults, while backward digit span, MDAS digit span and MOTYB were sensitive to detecting delirium in PD and dementia-free older adults and PD and LB-CI, but not those with non-Lewy body dementia. Previous studies have reported that impaired MOTYB [32–34], letter vigilance test [17, 33] and digit span tests [33] were significantly associated with delirium in older adults and dementia participants, although accuracy differs between studies, and compromise between sensitivity and specificity decrease with existing cognitive impairment [35]. Performance on the forwards and backwards digit span tests rely on different cognitive processes [36]; forward digit span relies on attention while backwards digit span relies on executive function. It has been suggested, therefore, that these tests should not be combined into a single score, as per the MDAS digit span item [37]. Our results highlight that forward and backward digit span tests should not be used interchangeably, particularly in groups with underlying neurocognitive disorders, such as those with Lewy body disease, where impaired attention and executive function are common in the absence of delirium [38].
Although MOTYB backwards has been used as part of screening tools for cognitive impairment and dementia in PD [23, 35], to our knowledge this is the first study to show it may have clinical utility in detecting delirium in PD and LB-CI patients. Only MDAS attention was significantly associated with detecting delirium in all three groups (PD/LB-CI, other dementia and PD and dementia-free older adults), with stricter cut-offs being applied in those with PD/LB-CI or other dementia. This may be due to a higher proportion of participants with PD and/or dementia being unable to complete attention tests due to underlying cognitive impairment or due to low arousal levels, which is a limitation of most tests of attention [17]. The MDAS attention item, however, is a subjective rating based on the assessor’s evaluation of performance, e.g., whether the participant was distractible or loses track of the conversation, whether questions had to be repeated and how much this disrupted the interview. In the DECIDE and DETERMINE-PD studies, this scale was completed at the end of the assessment, which included several tests of attention. Therefore, the assessor’s subjective impression may be influenced by impairment in specific tests. Thus, the MDAS attention item in isolation without the other attention tests (e.g., digit span) should be used with caution as it may not have the same level of accuracy.
In participants with PD/LB-CI and dementia, level of arousal measures were more accurately able to detect delirium compared to attention tests. A recent systematic review of rapid delirium tests found that measures of level of arousal, specifically the OSLA and RASS, correctly identified the most cases of delirium in PD and dementia-free older adults [39]. As with the MDAS attention measure, the arousal measures are observational and so it is always possible to score in all participants [17]. Consistent with previous studies, the OSLA [17, 39] had good specificity and sensitivity for delirium, even in those with PD and/or dementia, while the m-RASS had good specificity in all groups [19, 39]. However, we found the GCS had overall better accuracy in PD and/or dementia participants. Previous studies have shown that abnormal levels of arousal are strongly associated with delirium [20]. However, patients with PD, LB-CI or dementia may have an overlapping phenotype [10, 16] and present with lower levels of arousal and fluctuations as part of their clinical phenotype (e.g., daytime somnolence) [40]. We found that in participants with PD and/or dementia, the optimal cut-offs for all measures of arousal indicated greater levels of impairment (i.e., poorer scores) in these groups compared to PD and dementia-free older adults. Using stricter cut-offs in these groups and repeated testing may, therefore, reduce the risk of false-positive results in these participants and increase overall accuracy when using these measures.
Similar to a previous study [17], we found that combined attention and arousal testing increased the diagnostic accuracy of identifying delirium across all groups. Combining tests has the advantage of being able to assess patients and research participants when cognitive testing is not possible and may be more appropriate in those with underlying cognitive impairment [39]. These findings have implications for clinical practice. We suggest that in older adults without an underlying neurodegenerative condition, the MDAS digit span (≥2) and OSLA (≥1) should be considered as brief assessments to identify delirium, while in participants with established PD and/or dementia, the MDAS attention measure (≥2) and GCS (≤13) may be more appropriate tests to screen for delirium.
Strengths of this study include a large number of participants from two studies assessed using robust standardised procedures based on the DSM-5 criteria. Although the largest cohort of its kind, the sample size is relatively small, with small numbers of PD, DLB and dementia participants relative to the older adult group. The small numbers in the subgroups may therefore lack statistical power to detect subtle differences in attention or arousal in some measures. There were also demographic differences between the cohorts in terms of age, sex, and years of education; future studies should use age, education, and sex-matched cohorts to compare test performance. We compared several tests of attention and arousal across groups that were common to both studies in this analysis but acknowledge that some commonly used bedside tests that were part of DECIDE (e.g., S-A-V-E-A-H-E-A-R-T and 20-to-1 tests) were not used in the DETERMINE-PD study and so could not be explored. Future studies should consider a wider range of attention and arousal tests. Finally, this analysis is cross-sectional and only includes participants with prevalent delirium on hospital admission and does not consider the fluctuating nature of delirium. Future studies should consider prospective longitudinal assessments to determine changes in attention and arousal measures in those with incident delirium and once delirium resolves.
We found that simple bedside tests of attention and arousal could accurately detect delirium in PD, dementia and PD and dementia-free older adults in hospitals; however, the optimal tests and cut-offs differ between groups. In inpatients with PD and LB-CI, backwards digit span, MOTYB and the MDAS attention item may be useful tests to identify impaired attention associated with delirium, while the GSC, OSLA, m-RASS and MDAS consciousness item may be useful tests to identify altered levels of arousal. Consistent with previous studies, a combined attention and arousal score performed better than the two tests individually. We propose that these tests have clinical utility to aid the identification of delirium in those with neurodegenerative disorders with overlapping presenting phenotypes in inpatients, specifically in PD and dementia. However, these tests should not be used to diagnose delirium in isolation as they do not capture the fluctuating course of delirium and should be used instead within the context of the DSM-5 criteria. Further work is needed to determine their utility in the community and by non-specialists in secondary care. Future studies should consider replicating these findings in a larger cohort across a wider range of neurodegenerative conditions, and evaluating a wider range of cognitive tests longitudinally to identify incident delirium, and developing robust composite tools to aid delirium diagnosis.
Footnotes
ACKNOWLEDGMENTS
We thank the participants, their families, the general practitioners and their staff, and staff at Newcastle upon Tyne Hospitals NHS Foundation Trust for their co-operation and support. We thank Angela Green, the DECIDE research nurse, and Victoria Foster, the DETERMINE-PD Clinical Trails Associate, for their valuable contributions. We thank our funders and the dementias and neurodegenerative disease research network in Newcastle. The views expressed are those of the authors and not necessarily those of the NHS, the NIHR, or the Department of Health.
This work was supported by a Clinical Research Fellowship from the Alzheimer’s Society [grant number 239 (AS-CTF-14-001)] awarded to S.J.R.; CFAS II-Newcastle is funded by the UK Medical Research Council [grant number G0601022] and the Alzheimer’s Society [grant number 294]. The DETERMINE-PD study was sponsored by Newcastle upon Tyne Hospitals NHS Foundation Trust and was funded by Parkinson’s UK (K-1701). The research was supported by the National Institute for Health Research (NIHR) Newcastle Biomedical Research Centre and the Clinical Ageing Research Unit based at Newcastle upon Tyne Hospitals NHS Foundation Trust. L.A. is supported by the National Institute for Health Research Applied Research Collaboration South West Peninsula.
CONFLICT OF INTEREST
None.
