Abstract
Background
Psychotic symptoms are common in patients with Parkinson’s Disease Dementia and Dementia with Lewy Bodies, known collectively as the Lewy Body Dementias (LBD). It is important to identify these symptoms early and accurately. However, these symptoms are challenging to identify and quantify in clinical practice. The Psychosis and Hallucinations Questionnaire (PsycH-Q) was developed as a self-report tool using patient-friendly language to assess hallucinations and related phenomena and has been previously validated in Parkinson’s Disease patients without dementia and their caregivers.
Objectives
This study aimed to evaluate the utility of the PsycH-Q in patients diagnosed with a Lewy Body Dementia.
Methods
A total of 33 LBD patients, assisted by their caregivers, completed the PsycH-Q, along with the Scales for Outcomes in Parkinson’s Disease-Psychiatric Complications (SCOPA-PC) and the Movement Disorder Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS). Items relating to hallucinations and psychotic phenomena from the SCOPA-PC and MDS-UPDRS were then compared with analogous ratings on the PsycH-Q.
Results
Scores on the PsycH-Q were significantly correlated with SCOPA-PC Questions 1 and 3, as well as the MDS-UPDRS question 1.2 (Spearman’s rho of 0.65, 0.73, and 0.47, respectively; p < 0.01 for all three correlations).
Conclusions
These results support the PsycH-Q as a patient-friendly questionnaire that accurately identifies hallucinatory phenomena and their features in LBD patients via self-report completed by patients with their caregivers. Future use of the PsycH-Q in clinical practice and research could improve early detection of psychotic symptoms in LBD patients, leading to improved management.
Keywords
Introduction
Lewy body dementia (LBD) is an umbrella term that includes patients with Parkinson’s Disease Dementia (PDD) and Dementia with Lewy Bodies (DLB), which share similar clinical and neuropathological characteristics. Both conditions are characterised by the presence of prominent psychotic features, such as hallucinations and delusions. Previous work has identified that the emergence of these psychotic features in LBD represents the primary driver for a loss of independence and nursing home placement. 1 Psychosis is associated with a significant reduction in quality of life and an increase in caregiver distress,2–4 which is compounded by the limited benefits afforded by current treatment options. 5
Psychotic features in LBD are complex in nature and range from ‘minor’ (less troublesome) symptoms, including visual misperceptions, passage and presence hallucinations, through to well-formed visual and non-visual hallucinations, as well as distressing, often paranoid delusions.6,7 Further work has demonstrated that these psychotic features are commonly associated with a range of other clinical features, such as differential cognitive deficits across attentional, executive and visuo-perceptive domains; mood (depression and anxiety); motor phenotype (non-tremor), which might suggest shared neurobiological underpinnings.8,9
While screening tools have been developed to help recognise the diagnosis of DLB10,11 and its early symptoms the available instruments for detecting and monitoring psychotic symptoms in LBD have shown limited effectiveness in this challenging population. Given their adverse outcomes, the early and accurate identification of these psychotic symptoms is crucial for the management of patients with LBD. However, their identification in clinical practice remains challenging. Unlike motor features, these symptoms are not self-evident and clinicians must carefully question patients and, where possible, their caregivers to identify these features. The terminology used to describe psychotic symptoms in Lewy body dementia (LBD) can be challenging for patients to understand due to its complex and technical nature. Terms like “visual hallucinations” ,“delusions,” and “misidentification syndromes” may be unfamiliar or confusing to individuals experiencing these symptoms. The cognitive impairment associated with LBD can further complicate comprehension of such specialized medical language. Patients may struggle to accurately describe or identify their experiences using these clinical terms, potentially leading to underreporting or mischaracterization of symptoms. This difficulty in understanding and communicating about psychotic symptoms could impact the accuracy of self-reported measures and potentially affect diagnosis and treatment planning. Previously, most of the rating scales used have typically been developed for more general psychotic disorders and do not necessarily capture the unique profile of symptoms seen in LBD.12,13 The International Parkinson’s and Movement Disorders Society sought to evaluate and compare the utility of many of the available tools for the identification of psychosis in Parkinson’s Disease (PD) and determined that none of the available scales were capable of adequately evaluating the incidence, prevalence or severity of these symptoms. 12 The group recommended that an optimal scale should encompass the spectrum of symptoms observed in Lewy body diseases whilst also assessing their severity and impact on both the patient and their caregivers. 12
The Psychosis and Hallucinations Questionnaire (PsycH-Q) was developed in response to these recommendations. It was intended to provide a self-report tool that deliberately uses non-scientific, patient-friendly language to identify not only psychotic features but also other associated clinical symptoms (eg, attention, REM sleep behaviour disorder (RBD)). The development and validation of this questionnaire has been described in a previous publication.
14
Briefly, the PsycH-Q is a 20-item questionnaire divided into two sections (Figure 1). Section I consists of three subscales: visual misperceptions (first subscale: Questions 1 to 6) to assess visual hallucinatory phenomena; sensory misperceptions (second subscale: Questions 7 to 10) to assess the presence of misperceptions and hallucinations in other sensory modalities (eg, auditory, touch, olfactory and gustatory); and disordered thought (third subscale: Questions 11 to 13) that probe for the presence of thought disorder and delusions. Section II consists of seven questions divided into two subscales (attentional dysfunction and sleep impairment) and was designed to explore deficits in attention and sleep, both of which have been associated with hallucinatory phenomena in LBD.8,9,15 Structure of Psych-Q questionnaire.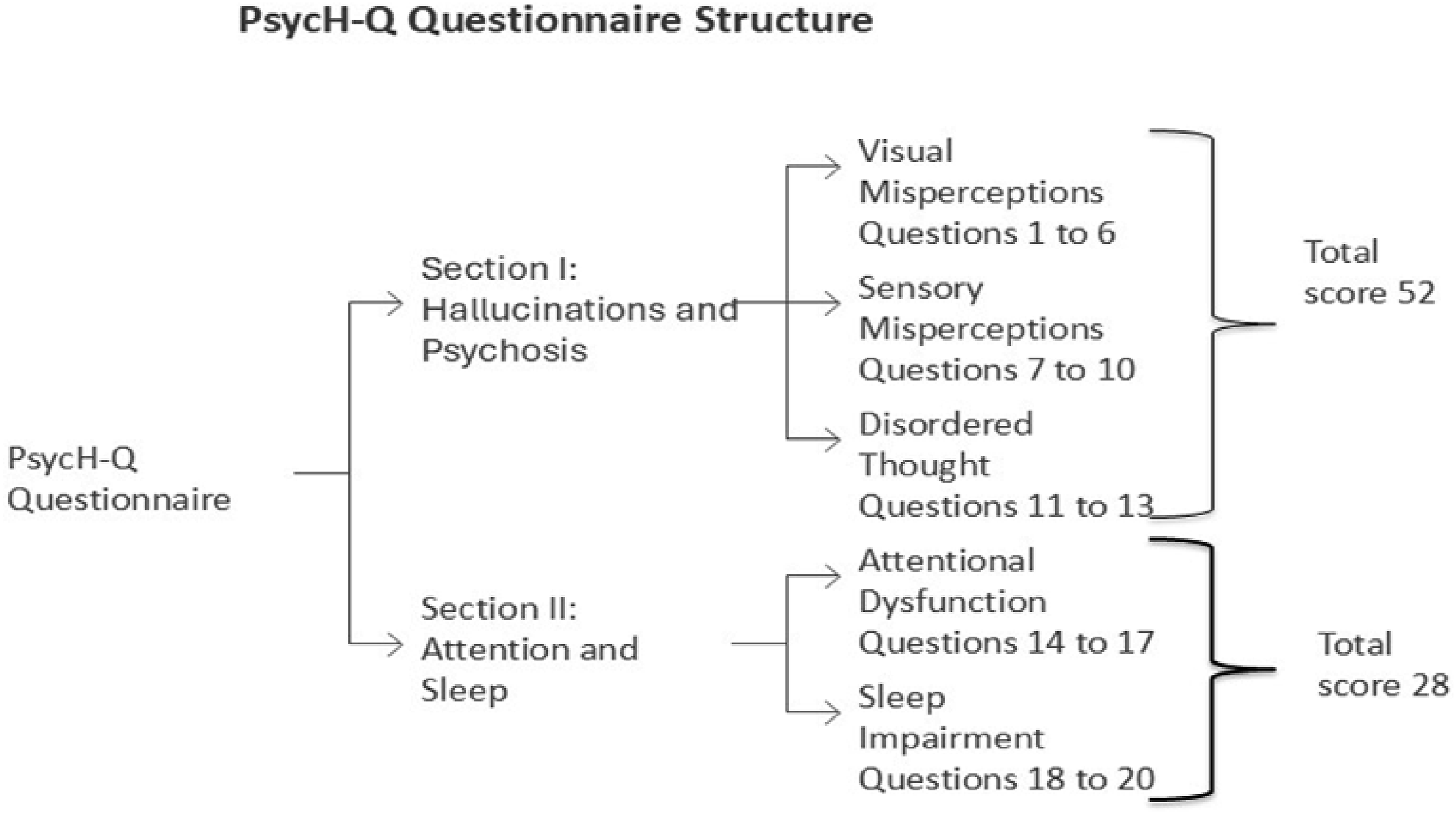
Each question uses a 5-point Likert scale to rate the frequency of symptoms (Never, Less than once per week, Weekly, Most days of the week and Daily). Additionally, subjects are also asked to rate the severity of symptoms on a 4 point Likert separately (None, Mild, Moderate, and Severe distress), which is not included in the final score. The total from Section I (maximum score 52) is utilized as a screening tool for hallucinations and psychosis, whilst Section II (maximum score 28) provides a basis for evaluating symptoms associated with psychosis in LBD (sample questionnaire – Appendix 2).
A validation study of the PsycH-Q conducted in PD patients without dementia demonstrated excellent validity when compared to analogous items across three existing scales (the Neuropsychiatric Inventory Questionnaire (NPI-Q), the SCales for Outcomes in PArkinson’s disease-Psychiatric Complications (SCOPA-PC) and the Parkinson Psychosis Questionnaire (PPQ)), as well as a high level of internal consistency. 14 A second study of the PsycH-Q, investigated the effectiveness of incorporating informant reports from caregiver dyads, alongside the patient self-assessment. 16 This work highlighted important discrepancies in the levels of agreement between these two sources with patients consistently reporting a higher frequency of hallucinations compared to informant accounts. Furthermore, clinical interviews alone, resulted in a significant number of false-negative outcomes. 16 The findings from this study highlighted the necessity of adopting a comprehensive approach that integrates informant input and self-assessments in patients with PD without dementia. 16 To extend the potential utility of the PsycH-Q, the current study sought to investigate whether this self-report tool administered to patients with input from caregivers, could identify and quantify the presence of psychotic symptoms in more severely affected patients with LBD.
Methods
This study evaluated participants with LBD recruited through the Parkinson’s Disease Research Clinic, Sydney. The study protocol was approved by the Human Research Ethics Committee of Macquarie University (HREC #16842) and all study participants provided written informed consent prior to their participation.
Participants were initially assessed by qualified clinicians (SJGL, EM, CWG) and were evaluated by trained clinicians using several scales including the International Parkinson and Movement Disorder Society (MDS)-sponsored revision of the UPDRS (MDS-UPDRS), 17 the Scales for Outcomes in PArkinson’s disease-Psychiatric Complications (SCOPA-PC) 18 and the Montreal Cognitive Assessment (MOCA). 19 Trained clinicians were available to provide clarification if needed. We did not formally evaluate the time required to complete the questionnaire, nor did we objectively record the level of additional assistance that was necessary given that it was minimal. Finally, we did not collect demographic data from those caregivers who might have offered assistance with completion of the PsycH-Q.
All DLB participants were diagnosed according to the 4th DLB consensus criteria. 20 The diagnosis of dementia was defined as progressive cognitive decline of sufficient magnitude to interfere with normal social or occupational functions, or with usual daily activities. 20 Core features of DLB were assessed through clinical interview and examination.
Diagnosis of PDD was based on the diagnostic criteria by the Movement Disorder Society in 2007. 21 The participants with PDD were selected from two groups both of whom participated in the research clinic. The first group were patients under the care of SL who were reviewed regularly in the outpatient setting allowing a clinical diagnosis of dementia to be made. The second group of participants were only assessed annually in the research clinic. The diagnosis of dementia in these participants was confirmed by a score of 3 or more on the MDS-UPDRS question 1.1 (Cognitive impairment). In addition, all PDD participants scored 24 or lower on the MoCA 22 with involvement across at least two cognitive domains (attention, executive function, visual construction, and memory impairment).
All patients underwent routine cranial imaging using either magnetic resonance imaging or computed tomography to exclude alternate diagnoses.
Statistical Analysis
All statistical analyses were conducted using R version 4.4.1. We compared Section I of the PsycH-Q with the relevant subsections of SCOPA-PC (Question 1: Hallucinations, Question 2: Illusions and Misidentification of persons and Question 3: Delusions), along with Item 1.2 from the MDS-UPDRS (hallucinations and delusions). The total of the frequency score for Questions to 1-10 of the PsycH-Q [Maximum score of 40] were analysed for correlations with the sum of SCOPA-PC Question 1 and 2, as well as the MDS-UPDRS Item 1.2 (hallucinations and delusions). Similarly, the composite scores from Questions 11 to 13 of the PsycH-Q [Maximum score of 12] were examined for correlations with Question 3 of SCOPA-PC (paranoid ideation). Bivariate Spearman’s rank-order correlation analysis was conducted for all correlations to assess these relationships.
Internal consistency was analysed separately for frequency and severity using Cronbach’s alpha. “Alpha if item omitted” tests were measured for each item’s contribution to the subscale and section of interest. Statistical significance was assessed using Wilcoxon Signed-Rank test for categorical variables and Fisher’s exact test for continuous variables.
Results
Demographics
Relevant data were available for 33 participants with Lewy body dementia (26 [79%] Male, 7 [21%] Female). Of these, 12 participants had PDD and 21 were diagnosed with DLB. All DLB participants met criteria for Probable DLB due to having two core features.
The mean age of the DLB cohort at diagnosis was 72 y ± 5.8, whereas the PDD cohort were initially diagnosed with their PD at a mean age of 61 y ± 8, transitioning to PDD at a mean age of 73 y ± 6.6. Thus, there was no significant difference between the age at diagnosis of dementia onset between DLB and PDD (72 y ± 6 v 73 y ± 6.6; P = 0.5). Participants were evaluated at an average age of 74 ± 6 years (DLB: 73 y ± 6; PDD: 75 y ± 7; P = 0.3) with an average of 1.5 y ± 1.8 following their diagnosis of dementia, with no significant difference observed between DLB and PDD groups (1.3 y ± 1.8 years vs 1.6 y ± 1.5; P = 0.5).
Demographic and Clinical Data of the Participants.

Summary of Neuropsychiatric Scores.
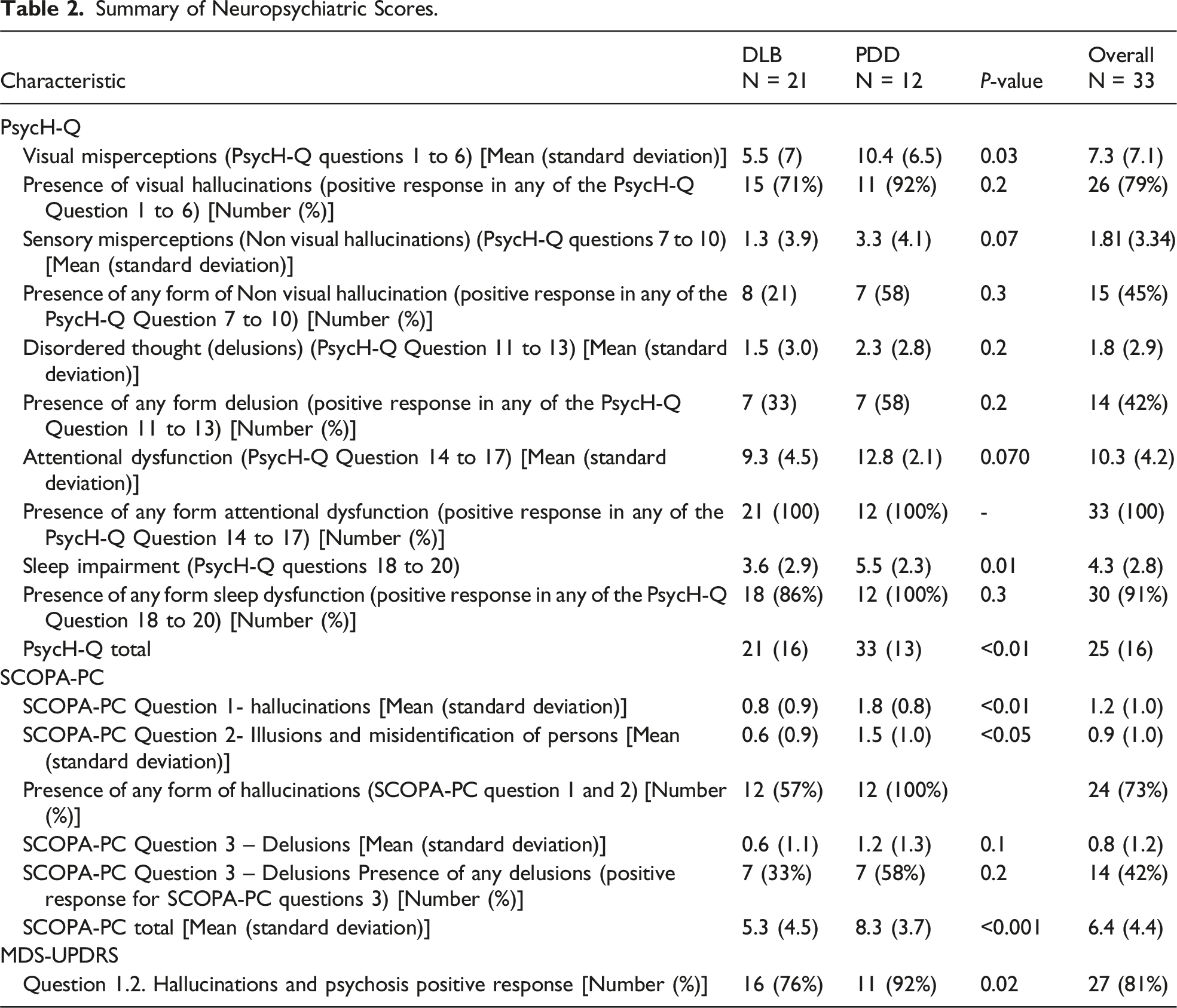
The PDD cohort exhibited more severe sleep and attentional symptoms compared to those with DLB, as evidenced by higher mean scores in these sections of the PsycH-Q. Notably, these two symptoms were prevalent in this cohort, with attentional dysfunction observed in all 33 participants and sleep impairment reported in 30 (91%) of the cases.
Correlations
Significant statistical correlations were detected between the corresponding items of the SCOPA-PC and the PsycH-Q. An analysis of Hallucinations indicated a strong correlation (Spearman’s Rho = 0.65; P < 0.001) comparing the SCOPA-PC (aggregate of questions 1 and 2) with the aggregate of PsycH-Q questions 1 to 6 (Section I - Visual misperceptions). A strong significant correlation for Delusions (Spearman’s Rho = 0.73; P < 0.001) was also seen in a comparison between SCOPA-PC question 3 and the composite score for PsycH-Q questions 11-13 (Section I - Disordered thought). Furthermore, a moderate strength correlation was identified between the PsycH-Q questions 1 to 6 (Section I - Visual misperceptions) and the MDS-UPDRS question 1.2 on psychosis (Spearman’s Rho = 0.47; P < 0.01). As expected, there was also a significant correlation between Sections I and II of the PsycH-Q (Spearman’s Rho = 0.49; P < 0.01), confirming the association between attentional dysfunction, sleep disorders and the presence of psychosis in LBD.
Internal Consistency
Cronbach’s alpha for Section I demonstrated excellent reliability (α = 0.93, 95% CI = 0.90 -0.96], indicating strong cohesion among the questionnaire items for measuring the underlying construct. The standardized alpha (0.93) and average inter-item correlation (r = 0.42) further supported the instrument’s robust internal consistency. The item total correlations ranged from 0.35 to 0.55 with items demonstrating good discriminative properties. Sensitivity analysis revealed that the removal of any single item from the PsycH-Q did not substantially improve the overall reliability coefficient (Appendix 1). The distress scale also demonstrated excellent internal consistency with a Cronbach’s alpha of 0.90. Inter-item correlation analysis revealed moderate to high correlations among items, and sensitivity analysis confirmed that no single item disproportionately influenced the scale’s reliability.
Discussion
This study supports the utility of the PsycH-Q as a highly reliable self-report questionnaire, demonstrating excellent validity and internal consistency in this cohort. The PsycH-Q effectively identified a broader range of psychotic phenomena in both groups encompassing hallucinatory and thought disorders. The importance of this is further highlighted by prevalence of both attentional, sleep related issues and non-visual hallucinations captured by the PsycH-Q. Furthermore, the PsycH-Q demonstrated excellent internal consistency with a Cronbach’s Alpha of 0.93 for both the frequency and severity sections of the questionnaire.
Available tools for detecting and monitoring psychotic symptoms in LBD have demonstrated limited utility in this challenging population. The PsycH-Q was specifically designed to offer a self-report framework that is sensitive to important clinical features for patients with LBD and their caregivers. The PsycH-Q also offers a comprehensive approach to assessing the hallucinatory phenotype by providing a detailed profile that encompasses both the frequency of symptoms and the associated distress levels. This dual-faceted evaluation allows clinicians to gain a more nuanced understanding of the patient’s experience, moving beyond mere symptom occurrence to consider the impact on the individual’s daily life. By highlighting the most burdensome symptoms, the PsycH-Q enables health care providers to prioritise treatment strategies, which could be incorporated into clinical trials, ultimately leading to improved patient care and better outcomes. Moreover, the PsycH-Q’s ability to offer prognostic information serves as a valuable tool for clinicians in their interactions with patients and caregivers. Features such as psychosis are closely linked to carer stress, poorer outcomes and mortality.4,23 Detecting these clinical features could facilitate more informed discussions about prognosis, guide treatment options and inform long-term management outcomes. By providing a clearer picture of the patient’s likely trajectory, clinicians can set realistic expectations, tailor interventions more precisely, and engage in shared decision-making with patients and their support networks.
Although it did not impact on the ability of the PsycH-Q to identify psychotic features in LBD, we would highlight that both the total PsycH-Q and SCOPA-PC scores were higher in the PDD cohort compared to DLB, with the PDD group also scoring lower on the MoCA. This observation was made despite the absence of a discernible difference in the duration of their dementia, but it is important to note that accurately determining the exact duration of such deficits is challenging due to both the lack of prospective, longitudinal data and the likelihood of poor recall or recall bias. Furthermore, the differences in both the degree of psychotic features and cognitive impairment may also reflect a selection bias in the recruitment of DLB participants who were likely less severe than their PDD counterparts, as these were generally diagnosed earlier into their illness (mean disease duration of 1 year). This difference in severity was mirrored by the greater motor symptoms observed in the PDD compared to DLB participants reported here. Future prospective studies comparing these two groups will be necessary to confirm the aetiology of these differences.
The validation of the PsycH-Q in non-demented participants has been previously documented. 14 As anticipated, a higher proportion of the current cohort reported hallucinations compared to the non-demented participants in our original publication (76/197 [39%] in non-demented vs 26/33 [79%] in demented participants). Additionally, non-visual hallucinations were more prevalent among demented participants (6/197 [3%] in non-demented vs 15/33 [45%] in demented participants). Similarly, scores for visual misperceptions (non-demented: 4.5 ± 5 vs demented: 7.3 ± 7.1), sensory misperceptions (non-demented: 1.8 ± 3.3 vs demented: 1.4 ± 2.5), and delusions (non-demented: 0.5 ± 1.5 vs demented: 1.8 ± 2.9) were all higher in the demented cohort.
Both attentional deficits and sleep disturbances have been closely associated with hallucinations in LBD.8,9 Therefore, the correlations observed between Sections I and II of the PsycH-Q reported here further strengthen the likelihood of shared neurobiological underpinnings that exist between psychotic phenomena and both attentional dysfunction and sleep disruption. Thus, the PsycH-Q may offer a framework for future studies utilising neuroimaging and neurophysiological approaches to identify the neural correlates of these relationships. In addition, the tool could potentially be used as an outcome measure in future clinical trials targeting these symptoms.
The limited sample size of this study, coupled with its single-center design, limits the generalizability of these findings. Furthermore, we did not have the opportunity for repeat testing to assess the test-retest reliability of the PsycH-Q in these participants, which should be evaluated in future studies. Additionally, we did not formally evaluate the extent of supervision required by participants from their caregivers to complete the questionnaire nor did we collect any demographic data from those caregivers that might have offered assistance with completion of the PsycH-Q. However, we did receive positive feedback about the ease with which the questionnaire could be completed . Furthermore, we did not formally evaluate our participants for the presence of other sleep-wake disorders such as Restless Legs Syndrome or Obstructive Sleep Apnoea, which may also be contributing to sleep disruption, and this could certainly be considered in future studies. We would assert that the collection of such information should be included in future studies.
It is hoped that the future use of the PsycH-Q in both clinical practice, prospective research studies and clinical trials could potentially improve the early detection and monitoring of patients with LBD. Further research is necessary to evaluate the relationship between PsycH-Q subsection scores and outcomes, as well as to compare its performance with other widely utilized psychosis questionnaires in LBD.
Footnotes
Acknowledgements
The authors would like to thank the participants involved in this study for their ongoing support.
Author Contributions
(1) Research Project: A. Conception, B. Organization, C. Execution; (2) Execution; Statistical Analysis: A. Design, B. Execution, C. Review and Critique; (3) Manuscript: A. Writing of the First Draft, B. Review and Critique. C.W.G.: 1B,1C,2A,2B,3A. S.J.G.L.:1A,1C,2C,3B. E.M.:2C,3B. P.B.:3B.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: E.M. is supported by a National Health and Medical Research Council Emerging Leadership Fellowship (2008565) and the United States Department of Defence Congressionally Directed Medical Research Program (PD220061). S.L. is supported by a National Health and Medical Research Council Leadership Fellowship (1195830) and has received research funding from The Michael J. Fox Foundation and the Australian Research Council, as well as consulting for Pharmaxis.
Ethical Considerations
The Human Research Ethics Committee of Macquarie this study ((HREC #16842) written, informed consent was obtained from all participants involved in the study. We confirm that we read the Journal’s position on issues involved in ethical publication and affirm that this work is consistent with those guidelines.
