Abstract
Background:
Hypomimia is a clinical feature of Parkinson’s disease (PD). Based on the embodied simulation theory, the impairment of facial mimicry may worsen facial emotion recognition; however, the empirical results are inconclusive.
Objective:
We aimed to explore the worsening of emotion recognition by hypomimia. We further explored the relationship between the hypomimia, emotion recognition, and social functioning.
Methods:
A total of 114 participants were recruited. The patients with PD and normal controls (NCs) were matched for demographic characteristics. All the participants completed the Mini-Mental State Examination and the Chinese Multi-modalities Emotion Recognition Test. In addition to the above tests, the patients were assessed with the Movement Disorder Society-Unified Parkinson’s Disease Rating Scale and Parkinson’s Disease Social Functioning Scale (PDSFS).
Results:
Patients with PD with hypomimia had worse recognition of disgust than NCs (p = 0.018). The severity of hypomimia was predictive of the recognition of disgust (β= –0.275, p = 0.028). Facial emotion recognition was predictive of the PDSFS score of PD patients (β= 0.433, p = 0.001). We also found that recognizing disgust could mediate the relationship between hypomimia and the PDSFS score (β= 0.264, p = 0.045).
Conclusion:
Patients with hypomimia had the worst disgust facial recognition. Hypomimia may affect the social function of PD patients, which is related to recognizing the expression of disgust. Emotion recognition training may improve the social function of patients with PD.
INTRODUCTION
Parkinson’s disease (PD) is the most common neurodegenerative disease; its prevalence is only less than that of Alzheimer’s disease [1]. PD has motor and non-motor symptoms [2]. The impairment of social cognition in PD has been brought to public attention in recent years [3]. Emotion recognition, a social cognition ability, is the foundation of social behavior, and it may have multiple modalities, including facial and prosodic [4]. Recognizing facial emotion plays a significant role in social interaction. Understanding facial emotion recognition is essential for patients with PD and their caregivers, to improve their interaction and quality of life [5].
Some studies have indicated that facial emotion recognition in patients with PD is intact [6, 7]; however, most studies have reported that facial emotion recognition is defective in patients with PD, especially for negative emotion recognition, including anger, fear, sadness, and disgust [8–13]. Impairments of positive emotion recognition have also been reported [14]. The reasons may include the differences in stimulus intensity, stimulus type, task type, emotion type, and heterogeneity of patients with PD based on differences in their motor symptoms, disease duration, severity, medication, comorbidity, and cognitive function [4, 6].
Hypomimia (masked face) is a specific motor characteristic of PD, and it reduces spontaneous and voluntary facial expressions, including facial emotion expression [15, 16]. It makes others think that PD patients have no interest in others or the surrounding environment [15, 17]. Studies have indicated that facial emotion expression may be related to emotion recognition [18–20]. According to the embodied simulation theory, the basic mechanism for understanding the minds and emotions of others is not conceptual reasoning but simulation through mirror mechanisms [21]. People can simulate the facial expressions of others to trigger the sensorimotor neuron processes, stimulate proprioceptive feedback, generate a corresponding emotional state, and prompt emotion recognition [21, 22]. Brain regions, including the basal ganglia and mirror neuron systems, are active during facial mimicry [23]. If facial mimicry is limited, emotion recognition ability may decrease [24].
According to the embodied simulation theory, the process of simulation in patients with PD may be overt, covert, or both [25, 26]. The overt account of embodied simulation refers to the deterioration of voluntary facial muscle control in PD, resulting in the inability to imitate the emotional expressions of others, thereby contributing to impaired emotion recognition. The covert account of embodied simulation refers to the function of the mirror neuron-related motor system involved in voluntary muscle control, and facial expression mimicry is impaired due to neurodegeneration. Therefore, imitating the facial expressions of others cannot stimulate the corresponding emotional state, which impairs emotion recognition [25, 27].
Contrary to the embodied simulation theory, Bologna et al. (2016) used a three-dimensional optoelectronic system to record the facial movements and used the Ekman 60 Faces test to measure emotion recognition performance. Their results indicated no correlation between facial emotion expression abnormalities and emotion recognition deficits in patients with PD, suggesting that these two abnormalities result from different pathophysiological mechanisms [28]. However, some studies support the embodied simulation theory [27, 30]. Marneweck et al. (2014) indicated that emotion discrimination and recognition (including happiness, anger, sadness, disgust) were positively correlated with the voluntary control of facial muscles after controlling for disease severity and age [27]. Some researchers use electromyography to assess facial muscle activities (i.e., zygomaticus major, corrugator, and orbicularis oculi) when PD patients perform emotion recognition tasks [29, 30]. These studies found that patients with PD demonstrated facial mimicry when recognizing facing emotional expressions. The zygomaticus major in PD was almost inactive when recognizing happiness, and greater activity of the zygomaticus major muscle region was associated with a quicker identification of happiness [30]. Another study found a significant decrease in facial mimicry for happiness [29]. Facial mimicry decreased with the reduction of the activities of the zygomaticus major and orbicularis oculi muscles when recognizing happiness [29]. The association between impairments of facial mimicry in PD and emotion recognition has not been established.
Furthermore, emotion recognition is the foundation of social interaction; people interact with others by understanding the emotions of others and adjusting to behave appropriately. Impairments of emotion recognition may damage social functioning [31], decrease quality of life, and increase the burden on caregivers [32].
Therefore, based on the embodied simulation theory, the present study aimed to explore decreased emotion recognition ability by PD-related hypomimia. Furthermore, we investigated the effect of emotion recognition impairments on social functioning and the relationship between hypomimia, emotion recognition, and social functioning.
MATERIALS AND METHODS
Demographic and clinical characteristics
All participants were interviewed to facilitate an understanding of their demographic and clinical characteristics. We used the Mini-Mental State Examination (MMSE) [33] to assess global cognitive ability. We recorded the onset age and disease duration and calculated the LED according to the recommendations of Tomlinson et al. (2010) [34] for the patients with PD. We quantified the motor symptoms and disease severity using the Movement Disorder Society–Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) [35] and Hoehn & Yahr staging criteria. We used item 3.2 of the MDS-UPDRS to evaluate the severity of hypomimia in patients with PD.
Emotion recognition
The Chinese multi-modalities emotion recognition test (CMERT) [36]
The CMERT is an emotion recognition test that includes nine subtests and 198 questions, and it involves presenting the stimulus by a tablet. The CMERT has several modalities (i.e., facial, prosodic, and cross-modal tests), and each modality has comprehensive emotions (i.e., neutral, happiness, sadness, anger, disgust, fear, and surprise).
In subtest 1, the participants were required to determine whether the two faces were for the same person. Subtest 1 was used to examine the basic facial recognition abilities of participants. In addition, we used subtests 3 and 4 to explore participants’ facial emotion recognition ability—the two subtests were targeted at different task types. (i.e., emotion identification and emotion discrimination tasks).
Each item of subtest 3 had one photograph conveying different emotions (i.e., neutral, happiness, sadness, anger, disgust, fear, and surprise) and seven emotion labels on the tablet screen. The participants were asked to choose one of the seven emotion labels that best fit the emotions expressed by the people in the picture. Each item of subtest 4 had one emotion label and seven faces with various emotions on the tablet screen. The participants were asked to choose one of the seven photographs conveying different emotions that best fit the emotion label.
Social functioning
Parkinson’s disease social functioning scale (PDSFS) [37]
The PDSFS is a 23-item scale that assesses the social functioning of patients with PD. This scale assesses three factors: family life, hobbies, and self-care; interpersonal relationship and recreational leisure; and social bond.
The family life, hobbies and self-care items covered the management of the primary needs of people (e.g., dining, clothing, medication, transportation) and their participation in production activities (e.g., doing housework). It also assessed their ability to interact with others (e.g., running an errand, shopping, and volunteer by oneself). The interpersonal relationship and recreational leisure items covered subjective social interaction skills (e.g., appropriate reaction when interacting with others, maintain good relationships with others). Last, the social bond items included the frequency of gatherings, chatting and participating in activities with others, and meetings with relatives, friends, or neighbors.
Participants
Our study had 114 participants. Fifty-seven PD patients (i.e., 15 without hypomimia (masked face) [PD-MF(–)] and 42 with hypomimia (masked face) [PD-MF(+)]) and fifty-seven healthy participants, as normal controls (NCs), were recruited and matched for gender, age, and education. All the PD patients were diagnosed using the United Kingdom Parkinson’s Disease Society Brain Bank clinical diagnostic criteria [38]. The patients were divided into two groups based on the presence or absence of hypomimia. We used item 3.2 of the MDS-UPDRS to evaluate hypomimia in PD. The PD patients with scores of 0 and 1 were allocated to the PD without hypomimia group, and those with scores of 2, 3, and 4 were allocated to the PD with hypomimia group. The exclusion criteria for the PD group included atypical parkinsonism, illiteracy, history of brain injury, brain operation, psychiatric disorders (e.g., depression or anxiety), and other severe systemic diseases. The NCs were recruited from the learning centers and communities of senior citizens. The exclusion criteria for the NCs group were the same as those for the PD group, but the NCs with MMSE scores lower than 24 were excluded to rule out the possible cognitive impairment problem. In addition, we excluded all participants with CMERT subtest 1 scores lower than 19 to prevent confounding by the effects of poor ability to distinguish basic facial features on facial emotion recognition.
Statistical analyses
All the data were tested for the normality of distribution using the Kolmogorov-Smirnov test. The differences between the groups were compared using the t-test, one-way analysis of variance (ANOVA) with the Tukey posthoc test (p < 0.05), and analysis of covariance (ANCOVA) with the Bonferroni posthoc test (p < 0.05) for parametric variables. The nonparametric variables were compared using the chi-squared, Mann–Whitney U, and Kruskal–Wallis tests.
To understand the relationship between the severity of hypomimia and emotion recognition and the relationship between emotion recognition and social functioning, we used Pearson’s and Spearman’s rank correlation analyses depending on the normality of the distribution of data. Simple linear regression and hierarchical regression analyses with adjustment for clinical characteristics other than the severity of hypomimia were performed. The CMERT subtest and seven emotion scores were considered independent variables for the simple linear regression analyses. The dependent variables were the PDSFS total score and the “family life, hobbies and self-care,” “interpersonal relationship and recreational leisure,” and “social bond” scores. The severity of hypomimia and other clinical characteristics were independent variables for the hierarchical regression, and the CMERT subtest score and seven emotion scores were considered dependent variables.
We used mediated regression analyses to understand the relationship between hypomimia, emotion recognition, and social functioning. The severity of hypomimia was considered the independent variable, the emotion recognition score was considered the mediator, and the PDSDS score was considered the dependent variable.
RESULTS
Demographic and clinical characteristics
The demographic data are summarized in Table 1. No significant differences in gender, age, education years, and MMSE score were found between NCs and PD.
Demographics and clinical characteristic in the study groups
NCs, normal controls; PD, Parkinson’s disease; PD-MF(–), Parkinson’s disease without masked face (hypomimia); PD-MF(+), Parkinson’s disease with masked face (hypomimia); SD, standard deviation; CMERT, Chinese Multi-modalities Emotion Recognition Test; LED, levodopa equivalent dosage; MDS-UPDRS, Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale; PDSFS, Parkinson’s Disease Social Functioning Scale; FLHSF, Family Life, Hobbies and Self-Care; IRRL, Interpersonal Relationship and Recreational Leisure. acomparison between NCs and PD; bcomparison between NCs, PD-MF(+) and PD-MF(–); cchi-square. dt test. eMann-Whitney U test. fANOVA; one-way analysis of variance. gKruskal-Wallis test. **p < 0.01, ***p < 0.001.
All the patients with PD were divided into two groups (i.e., PD without hypomimia and PD with hypomimia). There were significant gender-related differences among the three study groups. Significant differences were also observed in the motor symptoms (part III score of the MDS–UPDRS) between the two patient groups. Nevertheless, no significant differences were found in the other demographic variables between the three study groups. No significant differences were found in the onset age, disease duration, Hoehn & Yahr stage, or levodopa equivalent dosage between the two PD groups.
Emotion recognition performance
First, we compared the emotion recognition performances of the NCs and PD groups, as shown in Table 2. A significant difference was observed in the CMERT-subtest 4-disgust (p = 0.010). We divided all the patients with PD into two groups according to the presence or absence of hypomimia. The comparison of emotion recognition performances among the three groups is summarized in Table 2. After the covariate of the gender effect, a significant difference was observed in the CMERT-subtest 4-disgust (p = 0.018), and the posthoc test indicated that the NCs performed better than the PD-MF (+).
The performances of the Chinese Multi-modalities Emotion Recognition Test in the study groups
See Table 1 for abbreviations; acomparison between NCs and PD; bcomparison between NCs, PD-MF(+) and PD-MF(–) (after covariate the gender effect); ct test. dMann-Whitney U test. eanalysis of covariance (ANCOVA) with the Bonferroni posthoc test. *p < 0.05, **p < 0.01.
Relationship between hypomimia and emotion recognition
Correlation analyses and hierarchical regression analyses were conducted to facilitate a better understanding of the relationship between the severity of hypomimia and emotion recognition without the effects of gender and other clinical variables in patients with PD. As shown in Table 3, only two emotions were significantly related to the severity of hypomimia. The CMERT-subtest 3-sadness score was negatively correlated with hypomimia (ρ=–0.384, p = 0.003), gender (ρ= –0.261, p = 0.049), Hoehn & Yahr stage (ρ= –0.334, p = 0.011), and onset age (ρ= –0.383, p = 0.003). The CMERT-subtest 4-disgust was negatively associated with hypomimia (ρ= –0.261, p = 0.049) and the Hoehn & Yahr stage (ρ= –0.336, p = 0.011).
The correlations between the severity of hypomimia, gender, clinical variables, and the Chinese Multi-modalities Emotion Recognition Test score
See Table 1 for abbreviations; aSpearman correlation. bPearson correlation. *p < 0.05 **p < 0.01.
We further performed hierarchical regression analyses to determine the predictive value of hypomimia for the performances for CMERT-subtest 3-sadness and CMERT-subtest 4-disgust after controlling for gender and other clinical variables (Table 4).
The hierarchical regression model about the prediction of the hypomimia to the Chinese Multi-modalities Emotion Recognition Test
See Table 1 for abbreviations; *p < 0.05 **p < 0.01.
First, the gender, onset age, and Hoehn & Yahr stage accounted for 26.4%of the CMERT-subtest 3-sadness scores (F[3,53] = 6.322, p = 0.001); after adjusting for gender, onset age, and Hoehn & Yahr stage, hypomimia was not predictive of the CMERT-subtest 3-sadness scores (β= –0.205, p = 0.141).
Second, the Hoehn & Yahr stage accounted for 12.7%of the CMERT-subtest 4-disgust scores (F[1,55] = 8.012, p = 0.006); after adjusting for the Hoehn & Yahr stage, hypomimia increased the CMERT-subtest 4-disgust score by 7.5%(F[1,54] = 5.097, p = 0.028). Hypomimia was predictive of the performance for the CMERT-subtest 4-disgust (β= –0.275, p = 0.028), indicating that a higher hypomimia score was associated with a lower CMERT-subtest 4-disgust score, as shown in Table 4.
Relationship between emotion recognition and social functioning
To understand the relationship between emotion recognition and social functioning without the effect of other variables, such as gender, age, and clinical characteristics in the patients with PD, correlation analyses and simple regression analyses were conducted.
As shown in Table 5, hypomimia was significantly associated with the total PDFSF score (ρ= –0.309, p = 0.020) and the family life, hobbies, and self-care score (ρ= –0.343, p = 0.009). The PDSFS social bond score was positively correlated with the total score of the CMERT-subtest 3 (ρ= 0.286, p = 0.031) and the neutral score (ρ= 0.344, p = 0.009). The PDSFS score of family life, hobbies and self-care was positively correlated with the CMERT-subtest 4-disgust score (ρ= 0.263, p = 0.048).
The correlations between the Chinese Multi-modalities Emotion Recognition Test and the Parkinson’s Disease Social Functioning Scale
See Table 1 for abbreviations; *p < 0.05 **p < 0.01.
Then, we performed simple regression analyses to determine the predictive values of the CMERT-subtest 3-total score and the CMERT-subtest 3-neutral score for the PDSFS social bond score; the predictive values of the CMERT-subtest 4-disgust score for the PDSFS family life, hobbies, and self-care score.
The CMERT-subtest 3-total score was predictive of the PDSFS social bond score (F[1,55] = 5.110, β= 0.292, p = 0.028), indicating that a higher CMERT-subtest 3-total score was associated with a higher PDSFS social bond score. The CMERT-subtest 3-neutral score was predictive of the PDSFS social bond score (F[1,55] = 5.666, β= 0.306, p = 0.021), indicating that a higher CMERT-subtest 3-neutral score was associated with a higher PDSFS social bond score. In addition, the CMERT-subtest 4-disgust score was predictive of the PDSFS score for family life, hobbies, and self-care (F[1,55] = 12.680, β= 0.433, p = 0.001), indicating that a higher CMERT-subtest 4-disgust score was associated with a higher PDSFS score for family life, hobbies, and self-care, as shown in Table 6.
The simple regression model about the prediction of the Chinese Multi-modalities Emotion Recognition Test to Parkinson’s Disease Social Functioning Scale
See Table 1 for abbreviations; *p < 0.05 **p < 0.01.
Relationship between hypomimia, emotion recognition, and social functioning
We found that the hypomimia was predictive of the performance of the CMERT-subtest 4-disgust score after adjusting for the Hoehn & Yahr stage (Table 4). We also found that the CMERT-subtest 4-disgust score was predictive of the performance of the PDSFS score for family life, hobbies, and self-care (Table 6). Therefore, we further explored the relationship of hypomimia, the CMERT-subtest 4-disgust score, and the PDSFS score for family life, hobbies, and self-care. To test for mediation, we estimated the following three regression equations [39]:
After adjusting for the Hoehn & Yahr stage, the hypomimia was predictive of the performance of the CMERT-subtest 4-disgust score (β= –0.275, p = 0.028). Condition one was established, as shown in Model 2 of Table 7.
After adjusting for the Hoehn & Yahr stage, the hypomimia was predictive of the PDSFS score for family life, hobbies, and self-care (β= –0.301, p = 0.014). Condition two was established, as shown in Model 4 of Table 7.
After adjusting for the Hoehn & Yahr stage, considering hypomimia and the CMERT-subtest 4-disgust score simultaneously, the CMERT-subtest 4-disgust score was predictive of the PDSFS score for family life, hobbies and self-care (β= 0.264, p = 0.045). Hypomimia was not predictive of the PDSFS score for family life, hobbies, and self-care, and complete mediation was established, as shown in Model 6 of Table 7.
The regression model of hypomimia, the CMERT-subtest 4-disgust, and the PDSFS-“Family Life, Hobbies and Self-Care”
The values in the table are standardized regression coefficients. See Table 1 for abbreviations; df, degree of freedom; *p < 0.05 **p < 0.01 ***p < 0.001.
After adjusting for the Hoehn & Yahr stage, the CMERT-subtest 4-disgust score completely mediated the relationship between hypomimia and the PDSFS score for family life, hobbies, and self-care, showing that the hypomimia can affect the PDSFS family life, hobbies, and self-care by recognizing the expression of disgust. A higher score for hypomimia was associated with a lower CMERT-subtest 4-disgust score and a lower PDSFS score for family life, hobbies, and self-care, as shown in Fig. 1.
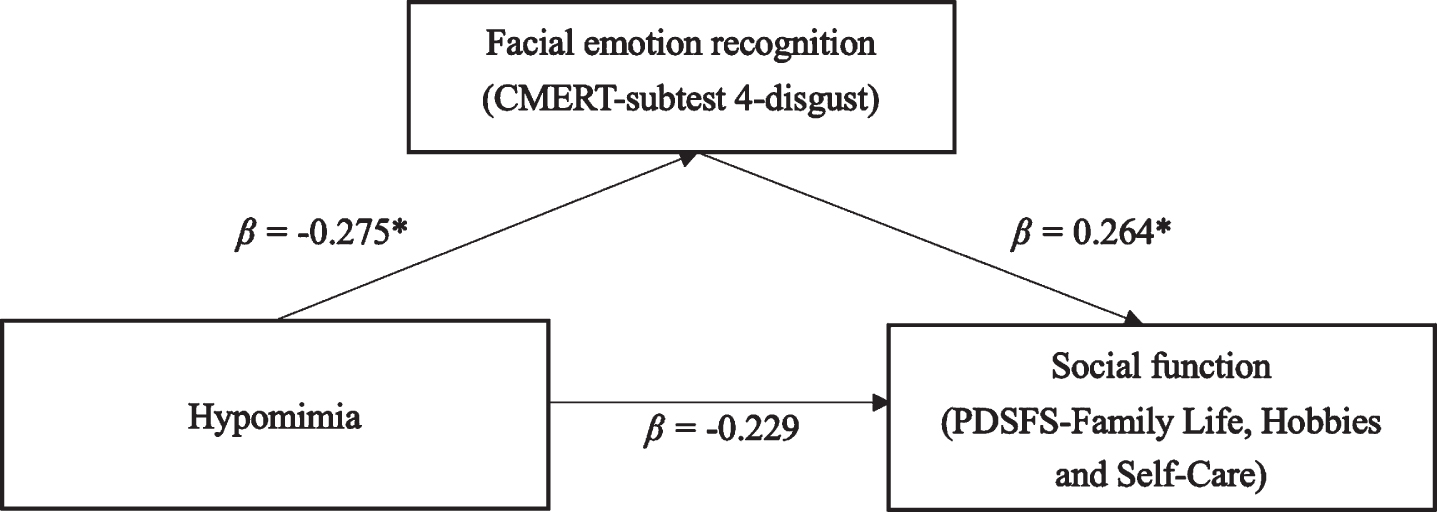
The mediation model of the hypomimia, the facial emotion recognition, and the social function. This mediation model showed that after the Hoehn-Yahr stage was adjusted, disgust recognition ability completely mediated the relationship between hypomimia and social functioning (i.e., PDSDS-“Family Life, Hobbies and Self-Care” performance). The values in the figure were standardized regression coefficients. See Table 1 for abbreviations.
DISCUSSION
This study explored the influence of hypomimia in patients with PD on emotion recognition based on the embodied simulation theory. We found that 1) PD patients, especially those with hypomimia, did not recognize disgust as healthy participants did. 2) More severe hypomimia was associated with worse facial emotion recognition, especially disgust recognition. 3) Facial emotion recognition ability affects the social functioning of PD patients, especially social bond and family life, hobbies, and self-care. We further found that hypomimia can affect family life, hobbies, and self-care by compromising the recognition of disgust.
These results revealed that hypomimia might affect the simulation processes based on the embodied simulation theory, making it difficult for patients with PD to recognize the disgust of others through simulation. The problem may further result in the inability of patients with PD to exhibit appropriate social behaviors when interacting with others, which may worsen social functioning.
PD patients with hypomimia had the worst performance in disgust recognition
In the current study, we found that patients with PD performed worse than healthy older adults in disgust recognition. These findings were consistent with those of previous studies [11–13]. Suzuki et al. (2006) suggested that the ability of disgust recognition in patients with PD is associated with a deficit in the basal ganglia neural circuit [13]. Pell and Leonard (2005) also found that there was a tendency toward the statistical significance of the relationship between the increased severity of motor symptoms and decreased ratings for the facial expression of disgust [6], emphasizing that the deficit of the dopamine circuit of the basal ganglia in patients with PD may negatively affect disgust recognition. These studies showed that the motor symptoms associated with basal ganglia neural circuit deficits could worsen disgust recognition. Hypomimia, which is a motor symptom, also results from dopamine depletion in the basal ganglia neural circuit [15].
The influence of PD heterogeneity (i.e., hypomimia) on this result is an important finding. We further found that PD patients with hypomimia had the worst disgust recognition after controlling for gender. Our findings verify the embodied simulation theory. According to the embodied simulation theory [21], the brain stimulates the corresponding sensory-motor feedback with a particular emotion during facial mimicry. We experienced the same feelings as others and this further promotes emotion recognition [21, 25]. During facial mimicry, the active brain regions include the basal ganglia and the mirror neuron system [22], which also impair PD [40, 41]. In addition to impairments of the neuronal systems related to facial mimicry, several patients with PD also suffer from hypomimia, which limits facial mimicry [15]. Based on the combination of overt and covert simulation processes [25, 27], PD patients with hypomimia had reduced basal ganglia and mirror neuron system activities. They also had difficulty imitating facial expressions because of hypomimia, making their emotion recognition ability the worst.
Furthermore, the impairments of disgust recognition in PD with hypomimia were only manifested during the administration of subtest 4 of the CMERT, but not in the CMERT-subtest 3. These subtests were different; subtest 3 was the identification task, and subtest 4 was the discrimination task. The type of emotion recognition test that is more sensitive to detecting facial emotion recognition in patients with PD is still inconclusive [4, 5]. Argaud et al. (2018) revealed that identification tasks highlighted facial emotion recognition deficit in patients with PD more frequently because identification tasks require additional knowledge during the categorization process [5]. However, Gary and Tickle-Degnen (2010) found that a more significant facial emotion recognition deficit was demonstrated during discrimination tasks [4]. Our results are consistent with those of Gary and Tickle-Degnen (2010). We suggest that patients with PD are vulnerable during emotion discrimination tasks. In addition, the CMERT-subtest 4 showed one emotion label; participants had to choose one of seven photographs conveying different emotions that best fit the emotion label. However, the CMERT-subtest 3 showed one photo expressing different feelings; participants had to choose one of the seven emotion labels that best fit the photo. According to the embodied simulation theory [21, 25], when patients with PD face emotional faces, it is necessary to have a simulation process to recognize emotions. Nevertheless, patients with PD may experience difficulties in simulations [5, 24]. In addition, patients with PD are supposed to have more simulation processes during the CMERT-subtest 4 because they are shown seven emotion photos instead of one during the CMERT-subtest 3, leading to worse emotion recognition.
The relationship between hypomimia and emotion recognition
The findings of previous studies on the relationship between facial muscle movement and emotion recognition ability in PD are inconsistent [27–30]. This study found that reduced facial movement ability was related to a deficit in recognizing disgust. Our results were partially in line with those of a study by Marneweck et al. (2014), who found that emotion discrimination and recognition (including happiness, anger, sadness, disgust) were associated with voluntary control of the facial muscle; this supports the embodied simulation theory [21]. We further found that after controlling for clinical factors (e.g., disease severity), the severity of hypomimia was predictive of the performance in recognizing disgust. Previous empirical studies emphasized that the lower face presence and mimicry are important in recognizing disgust [42, 43].
Nevertheless, we did not find a relationship between hypomimia and the ability to recognize sadness, anger, and happiness emotion. We supposed that this might be due to differences in methodology. Previous studies have indicated that stimulus intensity, stimulus type, task type, and emotion type may lead to differences in the results [4, 45]. Our study included seven basic emotions. However, we did not distinguish the intensity of emotional stimuli, and the facial stimuli were presented statically. In addition, we evaluated hypomimia when PD was stable rather than dynamic, such as with EMG. The above factors may have caused the differences in the results of the studies.
Relationship between emotion recognition and social functioning in patients with PD
Facial emotion recognition is the basis of social interaction; individuals understand the views and emotions of others by recognizing facial emotions, and they exhibit corresponding behaviors to interact with others. Therefore, emotion recognition impairment may damage the social functioning of patients with PD [31]. In this study, we found that worse facial emotion recognition ability in patients with PD was associated with less face-to-face meetings with relatives, friends, or neighbors; less participation in group activities; less proactive invitations of others to engage in activities together; and less social connection with others.
Martinet et al. (2018) revealed that caregivers overestimated the ability of their PD patients to recognize emotions. Hence, the caregivers believed that the patient would perform the same interpersonal reaction as expected during the interaction. When the patient could not recognize emotions well, it was challenging to respond to appropriate social behavior, which may have led to interaction problems between the patients and their caregivers, increasing the burden on caregivers [32]. However, we did not find that the impairment of emotion recognition in patients with PD affected their interpersonal relationships and recreational leisure (i.e., part two of PDSFS). Gunnery et al. (2016) suggested that the lack of ability to recognize emotions in patients with PD may protect the relationship between patients and their caregivers. PD may not be able to recognize the negative emotions of caregivers. Therefore, the interaction conflict between PD and caregivers was reduced [46].
The role disgust recognition plays in social functioning
In the present study, we found that worse facial disgust recognition was associated with worse social functioning. Panksepp (2007) highlighted that disgust is a strong emotional feeling involving human instincts, just like hunger and thirst, and it affects the survival of humans [47]. People try to avoid things they do not like, and people who are more sensitive to disgust show less aggressive behaviors [48]. In social situations, disgust plays a role in social avoidance. When someone feels disgusted, their motivation for social interaction decreases [49]. The above studies highlighted that disgust played an essential role in social interaction. If individuals cannot correctly recognize the disgust of others, social interactions may be affected. The PDSFS–family life, hobbies and self-care item reflected the ability to engage the individual’s primary need (e.g., dining, clothing, and medication) and participate in production activities (e.g., doing housework), as well as the ability to interact with others, such as going out to do things, shopping, and volunteering by oneself, among others. These activities involve the ability to interact with others. Therefore, the relationship between disgust recognition and social function revealed that the impaired recognition of the disgust of others by patients with PD may affect their ability to interact appropriately with others, further deteriorating social functioning.
After adjusting for the disease severity, the disgust recognition ability completely mediated the relationship between hypomimia and social function, showing that hypomimia negatively affected social function through the dysfunction of disgust recognition. This result validated the embodied simulation theory [21]. Hypomimia may result in the difficulty of patients with PD in recognizing disgust in others and their inability to exhibit appropriate corresponding social behaviors well when interacting with others, which may worsen social functioning.
Through the results of our research, we hope that a more comprehensive treatment plan, such as emotion recognition training for patients with PD, may be provided in the future. Social functioning and quality of life may be improved in patients with PD and their caregivers by knowing the emotion recognition difficulties and associated consequences. In addition, people recognize other’s emotions through various channels. We suggest that future studies explore emotion recognition via multiple modalities (e.g., prosodic modality or multiple modalities) and methodological modifications (e.g., recruit blind individuals as participants) to clarify the role of facial emotion recognition and the relationship between modalities.
Limitations and future studies
The assessments were performed face-to-face in our laboratory. The willingness of participants to participate in research has been affected by the COVID-19 pandemic, resulting in a small number of samples. Nevertheless, the percentage of patients with or without hypomimia in this study was similar to that reported in a previous study [50]. In addition, most PD patients without hypomimia are women; therefore, we tried to compensate for the effect using statistics when comparing the emotion recognition performances of the groups. We suggest that further studies should include more patients with PD to replicate our results. Second, the PDSFS is a self-reported scale, but patients with PD may lack insight [51]. Self-reporting may have subjective bias. Thus, we suggest that further investigations should consider caregivers’ opinions or use other techniques to collect objective data on social function. Third, our study was a cross-sectional study, and the causal relationship between emotion recognition and social functioning could not be determined. A longitudinal study to explore the casual relationship and long-term effects of emotion recognition or other elements on social functioning is suggested. Finally, emotion recognition may have multiple modalities, not only through the face but also through prosody [4]. Attention should be paid to the multi-modalities of emotion recognition, and further research is recommended for this to achieve a better understanding of human beings’ emotion recognition.
Footnotes
ACKNOWLEDGMENTS
We thank all the participants involved in this study. We are grateful for the grant support from the Ministry of Science and Technology (MOST), Taipei, Taiwan (MOST 110-2628-B-006-020 and 108-2410-H-006-046-) and the Higher Education Sprout Project, Ministry of Education to the Headquarters of University Advancement at National Cheng Kung University (NCKU).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
