Abstract
Background:
Social functioning is crucial for the determinants of Parkinson’s disease (PD) with dementia; however, there is no social functioning scale applicable to PD.
Objective:
This study aimed to develop a social functioning scale specific to PD (PDSFS) and provide a cut-off score to improve diagnosis accuracy.
Methods:
The items were developed through literature, interview patients, and PD expertise. After the pilot study, one hundred fifty-seven patients and 74 healthy participants were enrolled and completed the Mini-Mental State Examination, Clock Drawing Test, Activities of Daily Living, Neuropsychiatric Inventory, Adaptive Behavior Assessment System–Second Edition (ABAS–II) and part III of the Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS–UPDRS).
Results:
The final PDSFS has 23 items. The exploratory factor analysis revealed three factors, including “Family Life, Hobbies and Self-Care”, “Interpersonal Relationship and Recreational Leisure”, and “Social Bond”. The internal consistency coefficient was 0.883, and the test-retest reliability was 0.774, respectively. The total score of the PDSFS was significantly related to the total score of ABAS–II (r = 0.609, p < 0.001), and was not correlated with the third part of MDS–UPDRS (p = 0.736). A significant intergroup difference was found (p < 0.001), and the healthy controls had the highest PDSFS score, followed by non-demented PD and PD dementia. The optimal cut-off score for PD patients with dementia was 39 (sensitivity: 0.735; specificity: 0.857).
Conclusions:
PDSFS is a practical and psychometrically sound tool to access the social functioning of the PD population.
INTRODUCTION
Parkinson’s disease (PD) is a frequent neurodegenerative disease. As the population ages, the number of patients suffering from PD is expected to double by 2030 [1]. Patients with PD also have non-motor symptoms [2] and may happen in the early stage, even before the onset of the disease, and because of the fatality and morbidity caused by dementia, let it be the most considerable non-motor symptom [3].
Human beings are social species and cannot live alone. The “social functioning” is crucial for measuring the individual’s function and it is an overlooked aspect that determines the quality of life. Investigators suggested that daily life function was an essential aspect of social functioning [4], and studies had found that the decline in social functioning increased the risk of dementia in individuals [5], as well as accelerated the process of dementia [6]. Although social functioning is not easy to measure, clinicians are advised to pay attention to the changes in the social functioning in patients with dementia to facilitate treatment strategies [7]. Weissman pointed out that social functioning should include independence performance, social engagement, friendship/interpersonal functioning, daily activities [8]. Tyrer and Casey (1993) defined the social functioning as “the ability of individuals to connect and interact with their environment throughout society” [9]. Wykes (1998) believed that social functioning consists of three levels [10]. The first level was social attainments, which showd the achievements of a person’s life. The second level was a social role, providing an understanding of the performance of specific roles, such as being parents. The third level was instrumental behavior, such as social skills. McNamara et al. (2010) defined social functioning as “the ability to communicate ideas and emotions between people involved in social interactions” [11]. Based on those opinions and the prevailing social functioning scales, the “social functioning” defines an individual’s ability to fulfill their role within such external environments as social activities and relationships with family.
The pathology of Alzheimer’s disease (AD) is not the same as PD-D with the loss of dopaminergic neurons and the lack of cholinergic. The cognitive impairment profile in PD-D is mainly based on executive dysfunction [12]. Nevertheless, studies of cognitive impairment in PD used the tools developed for AD, which were not sensitive to the vulnerable cognitive domain of non-AD [13], and even many tools excluded PD during development, for example, the Clinical Dementia Rating [13]. Therefore, the social functioning scale is not universal, and it is not appropriate to use an appraisal device for AD to assess PD patient’s clinical features, and tailor-made tools for patients with PD are urgently needed.
We aim to develop a social functioning scale specific for patients with PD, establish its psychometric properties, and find the optimal cut-off score for determining the impaired social functioning in patients with PD.
MATERIALS AND METHODS
Scale development
Item generation
We develop the social functioning items through literature review [8–11] and reference to other scales [14–18]. We referred to social functioning-related scales that have been applied in other disease population, such as The Social and Occupational Functioning Assessment Scale (SOFAS) [16], The Personal and Social Performance Scale (PSP) [17], Social Functioning in Dementia (SF-DEM) [18], The Stroke Impact Scale (SIS) [19], and Mini-ICF-Rating für Aktivitäts- und Partizipationsstörungen bei psychischen Erkrankungen (Mini-ICF-APP) [20]. The target subjects, examiner, and the scoring method of the referred scales were all analyzed and considered in the generation of social functioning items. According to the literature review and existing scales, we generated questions from seven aspects: social activities, social support, social connection, interpersonal communication, leisure activities, daily activities, and occupational function.
Ten patients with PD and their families were recruited to join in the pretest procedure to check the relevance and readability of these items. We simultaneously invited a clinical neuropsychologist and a neurologist to critique and modify the scale to establish expert validity. After the pretest of patients and caregivers, we added an item “talked less than before”, and we combined using transportation and taxi into one item “manage transportation”, using cooking tools and preparing fruit into one item “use the appliance to prepare food”. According to PD experts’ comments, we revised the item to make it more specific and readable. Besides, we combined some similar items into one and added five questions (e.g., “working hours”, “the duration of work”, “clean rooms or living space”, “maintain self-cleaning”, and “repair stuff”) to the scale. This scale had a total of 36 questions, named as “Parkinson’s Disease Social Functioning Scale (PDSFS)”, and is a self-report scale. If the patient was illiterate, it could be filled in by the family or caregivers.
Item scoring
The PDSFS was mainly based on the Likert four-point scale (always/very agree = 3 points, often/agree = 2 points, occasionally/disagree = 1 point, rarely/very disagree = 0 points), only one reverse question. To standardize the reference value, we defined “always” as at least once a day, “often” as at least twice a week, “occasionally” as at least once a week, and “rarely” as less than once a week. According to the normal distribution of the sample, the monthly scores of meeting relatives, friends, and neighbors were 0 points within three persons, 1 point in 4 to 15 persons, 2 points over 16 persons. The higher the scale scores, the better the social functioning of the patient.
Measurement
We followed the diagnostic procedures for PD-D [21] and used the Mini-Mental State Examination (MMSE) [22], Clock Drawing Test (CDT) [23], Activity of daily living scale (ADLs) [24], and Neuropsychiatric Inventory [25] to discriminate patients with non-demented Parkinson’s disease (PD-ND) and PD-D.
The Adaptive Behavior Assessment System–Second Edition (ABAS–II)–Adult Edition [26] and its subscales (i.e., the family life, social, and leisure subscales) were used to establish the convergent validity. The part III of the Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS–UPDRS) [27] was used to establish discriminative validity and to exclude the effects of motor symptoms on the social functioning.
Participants
Two hundred and thirty-one subjects participated in the current study; seventy-four healthy participants were enrolled from the community or the patients’ spouses. One hundred fifty-seven outpatients diagnosed with idiopathic PD were recruited from northern and southern Taiwan, and the diagnosis of PD was according to the UK PD Society Brain Bank Clinical Diagnostic Criteria [28]. The PD-D group was based on diagnostic procedures that were proposed by the MDS Task Force [21], and the patients with PD were divided into the PD-ND and PD-D groups. Participants were excluded from the history of psychiatric illness (e.g., depressive disorders, anxiety disorders, etc.), history of substance abuse, other neurological diseases, severe systemic diseases, and traumatic brain injury. The participants completed the informed consent before enter into the study and the ethical standards were draw up in the 1964 Declaration of Helsinki. The study protocols were confirmed by the ethical research committee of hospitals.
Statistical analyses
We used Statistical Package for Social Science version 20 (SPSS 20) (SPSS Inc., Chicago, IL, U.S.A.) for all statistical analyses, and all hypothetical tests for inferential statistics set a significant level of 0.05.
The internal consistency was calculated by Cronbach’s alpha to examine internal reliability. Thirty patients were invited to complete the scale after 8 to 12 weeks from the baseline, and the intra-class correlation coefficient was performed to confirm the retest reliability.
Before the exploratory factor analysis, Kaiser-Meyer-Olkin (KMO) and Bartlett’s Test of Sphericity would be used to judge whether the data were suitable for factor analysis. The relationship between the items was understood from the extraction commonalities, and the index less than 0.3 were deleted. The Direct Oblimin was selected, and the factors were identified and named. Pearson’s correlation analysis was performed to measure the relationship between the total score of the PDSFS and the ABAS–II and the third part of the MDS–UPDRS score to examine the convergent and discriminative validity, respectively. We used the analysis of covariance to covariate the effect of education and age, and then to explore the differences between groups as a means of verifying the known group validity. Last, the receiver operating characteristic curve (ROC curve) was used to find the optimal specificity and sensitivity cut-off score to distinguish PD with or without dementia.
RESULTS
Demographic and clinical characteristic
The demographic data were summarized in Table 1. The demographic variables and scores of the MMSE, CDT, ADLs and ABAS-II revealed significant differences between the study groups. No significant difference was found in the disease duration and levodopa equivalent dosage between the two PD groups; nevertheless, significant differences were revealed in the onset age, motor symptoms (part III score of the MDS–UPDRS), and disease severity (the Hoehn-Yahr stage) between the two patient groups.
Demographics and clinical characteristic in study groups
HCs, healthy controls; PD-ND, non-demented Parkinson’s disease; PD-D, Parkinson’s disease with dementia; SD, standard deviation; LED, levodopa equivalent dosage; MMSE, Mini-Mental State Examination; MDS-UPDRS, Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale; CDT, Clock Drawing Test; ADLs, Activity of daily living scale; ABAS-II, Adaptive Behavior Assessment System–Second Edition; PDSFS, Parkinson’s Disease Social Functioning Scale.
Factor analysis
The KMO value of this study was 0.791, and Bartlett’s Test of Sphericity was significant (χ2 = 2144.081, p < 0.001), meant the factor analysis could be performed. Thirteen items (e.g., practice physical activities, practice mentally stimulating activities, marital status, residency arrangements, the number of children you will meet at least once a month, etc.) were deleted due to their poor representation variability and low extraction communalities (below 0.3) (Table 2). Three factors were extracted by the principal factor analysis and Direct Oblimin rotation, with factors fixed by three. Table 3 revealed the cumulative contribution, and the value was 52.35%.
Communalities of each items of the PDSFS
PDSFS, Parkinson’s Disease Social Functioning Scale; h2, communalities, the proportion of the variance in each item which is explained by the factors.
Principal components and factor loadings of the PDSFS
PDSFS, Parkinson’s Disease Social Functioning Scale.
Reliability
The Cronbach’s alpha of the PDSFS was 0.883. The first factor was 0.913, which was named “Family Life, Hobbies and Self-Care”, the second factor was 0.752, and it was named “Interpersonal Relationship and Recreational Leisure”, the third factor was 0.745, and it was named “Social Bond”. The mean test-retest days were 81.2 days, and the overall test-retest reliability of the PDSFS was 0.774 (p < 0.001). The test-retest of the factor 1, 2, and 3 were 0.801 (p < 0.001), 0.543 (p = 0.022), and 0.674 (p = 0.002).
Validity
After controlled age, education, and gender, a significant difference in the scores between different groups on the PDSFS (F = 19.025, p < 0.001) was found. After pairwise comparisons, we found that the healthy control group scored the highest, followed by the group with PD-ND and PD-D group (Table 4). Because of the significant correlation between the PDSFS and the Hoehn-Yahr stage (r = –0.373, p < 0.001), gender (r = 0.238, p < 0.001), age (r = –0.234, p < 0.001), disease duration (r = –0.241, p = 0.004), we controlled these variables to perform partial correlation coefficients. The total score of the PDSFS was significantly correlated with the total score of ABAS–II (r = 0.609, p < 0.001), the total score of the “Family Life, Hobbies and Self-Care” in PDSFS was significantly related to the scale score of the family life (r = 0.661, p < 0.001) subscale of the ABAS–II, and the total score of the “Interpersonal Relationship and Recreational Leisure” plus “Social Bond” in PDSFS was significantly correlated with Social Domain in ABAS–II (r = 0.509, p = 0.002), which was calculated by leisure and social subscale. The total score of the PDSFS was not correlated with the third part of MDS–UPDRS (p = 0.736).
Analysis of covariance (ANCOVA) on the PDSFS
HCs, healthy controls; PD-ND, non-demented Parkinson’s disease; PD-D, Parkinson’s disease with dementia; df, degrees of freedom.
The total score of the PDSFS had an acceptable ability to distinguish non-demented patients from dementia (the Area under Curve was 0.823). The optimal cut-off score was 39 when the sensitivity was 0.735, and the specificity was 0.857 (Fig. 1).
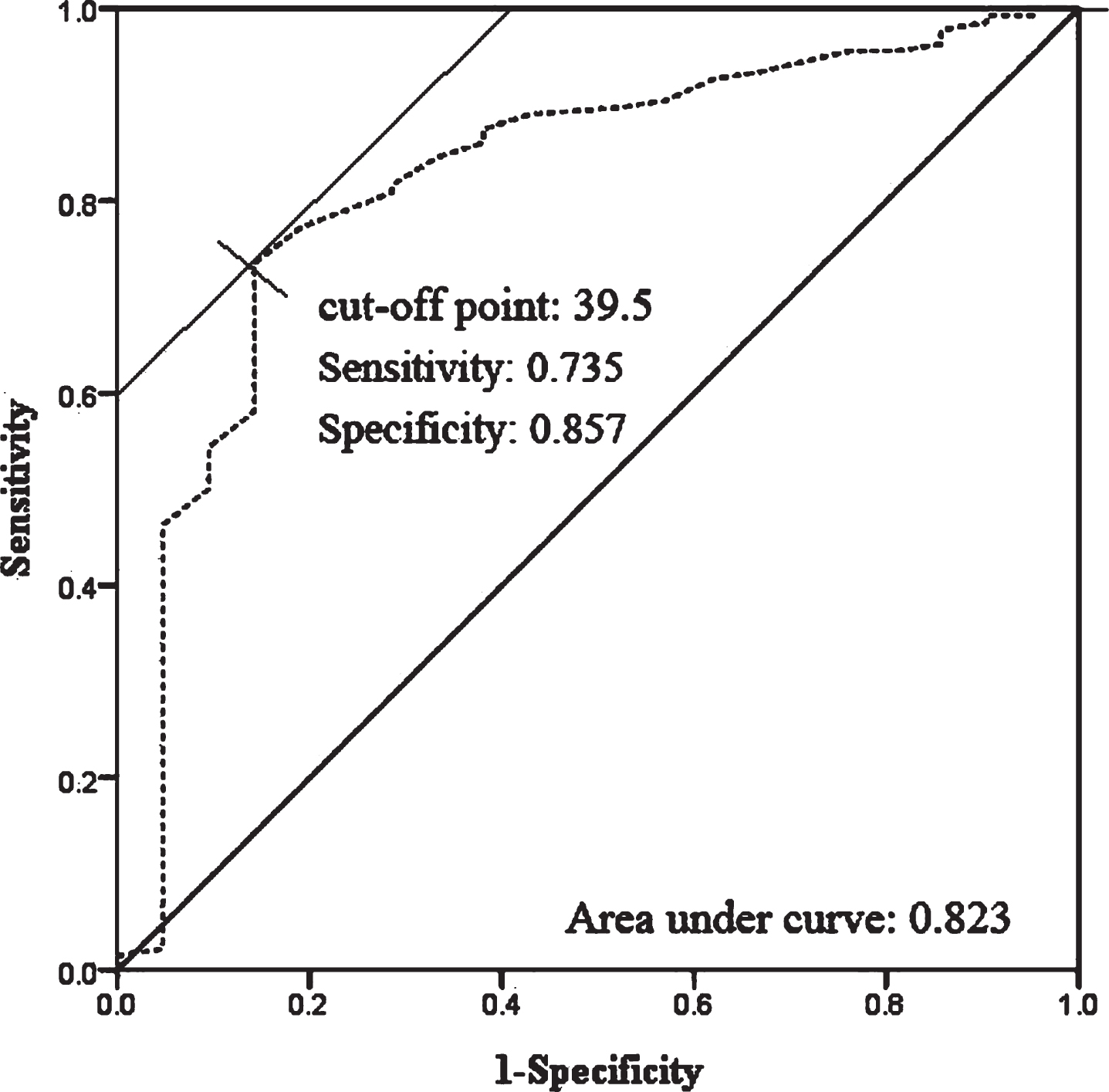
The receiver operating curves (ROC) shows the association between sensitivity and specificity on the PDSFS. The gray dotted line represented the association between sensitivity and specificity on the PDSFS.
DISCUSSION
According to the literature review, the results of the pilot test, and experts’ opinion, we tailored a scale, the PDSFS, for assessing social functioning for patients with PD. The PDSFS scale has 23 items, and three factors were extracted, including “Family Life, Hobbies and Self-Care”, “Interpersonal Relationship and Recreational Leisure”, and “Social Bond”. The PDSFS is a psychometrically sound and the cut-off score, 39, for discriminating patients with PD-D from PD-ND.
The contents of the first factor include the ability to manage the individual’s primary need (e.g., food, clothing, and transportation) and engaging in production activities (e.g., grading, doing chores, volunteer for hospital). In addition to the personal field, the communication quality with others is considered to be one component of social functioning. Moreover, the third factor includes the frequency of meeting, chatting, and engaging in activities with relatives, friends, and neighbors. Fratiglioni and colleagues (2000) suggested that deprived and limited social networks increase the likelihood of cognitive impairment [29]. Our study indicated that considering the residential habit of Taiwan, neighbors play an important role in life and should, therefore, be included in the “Social Bond”. The lack of social connection with others is the risk for mental disability; that is, individuals living alone or have no friends may have an increased risk of developing cognitive impairment.
During the development procedure, we deleted some items because of their weak variability. We removed “Doing things with others such as watching television/exhibitions, listening to concerts, dining at restaurants, or traveling.” A possible explanation for the homogeneous responses is that most of our participants live with their families; therefore, there are naturally many things in life that involve “others”. We also deleted “When you feel happy, sad, scared or angry, you will express your emotion.” The possible reason for the weak variability of this item may be the fact that Chinese people are not used to expressing their own emotions to others. In Eastern countries, most people tend to have the “interdependent self-construal” which means that a person’s behavior depends on other’s opinions and actions, and they also place great emphasis on interpersonal relationships [30]. It is not surprising that people tend not to express themselves to others to avoid offending others. The items measure the frequency of practicing physical activities (e.g., walking, climbing, or swimming), and mentally stimulating activities (e.g., writing, painting, or reading newspapers) were also deleted. The homogeneity of our participants’ responses might be due to that our participants were all elderly and have deteriorated vision and physical ability, and they tend to give up activities that require excellent motor skills or precision vision.
We also deleted marital status, residency arrangements, and the item to measure how many children the patient will meet at least once a month. Chinese people are influenced by Confucian culture; therefore, most children live with their parents and take care of them [31], representing the “filial piety”. This phenomenon is different from western culture [32]; thus, cultural factors should be included when determining one’s social functioning. Besides, we have asked participants about their occupational functions, including length and stability of employment. However, the variability of those items is weak, and possibly due to that, most of our participants were retired.
There was a significant relationship between the PDSFS and the ABAS–II, suggesting that PD with better social functioning might also have better adaptive behaviors. It is believed that individuals who have better social functioning also have better adaptive behaviors. Besides, the PDSFS was not related to the third part of MDS–UPDRS, representing that the PDSFS may not interfere with the patient’s motor symptoms and the discriminative validity of the PDSFS has been confirmed. We believe that the elimination of the effects of the motor symptoms is crucial in the assessment of mental or social functioning in PD patients. However, there were still words in the items that may involve motor functioning, although we attempted to remind patients to exclude the effects of motor symptoms. It is recommended that future research be careful when designing the scale; for example, “can repair items in the house” can be modified into “can repair items in the house or know how to repair items in the house”.
In the current study, a significant group effect was revealed. The lowest PDSFS scores in the PD-D group, confirming that the patients with PD-D have impaired social functioning. Besides, we found that the individuals in the PD-ND group have a significantly lower social functioning than those in the healthy control group. That is, although the social functioning loss in our patients with PD-ND is not as severe as PD-D, the patients with PD-ND’s social functioning has its limitation. The above finding raises an essential issue that although PD patients who not yet go into the dementia process might have a declined social functioning. The heterogeneity among the PD-ND group may be one of the explanations for the above issue. The PD-ND group may include PD patients with mild cognitive impairment (MCI) and intact cognitive ability, and we believe that the two patient groups may have various social functioning. Pirogovsky and colleagues’ study showed compared to the healthy group, the patients with PD-MCI scores lower on drug and financial management ability, while cognitively intact PD patients did not show difficulties in the superior ability [33]. It seems to be in line with our opinion. Furthermore, we found out that the optimal cut-off score of the PDSFS for the PD-D is 39 points. If patients score less than or equal to 39 points, it is very likely to suffer from a social functioning impairment; that is, the patient may have entered the course of dementia and is recommended to further comprehensive examination.
The first limitation of this study was that we did not use comprehensive neuropsychological tests to assess the cognitive function in various domains, so it may be too brief when distinguishing patients from PD-D or PD-ND, and unable to clarify the heterogeneity in the PD-ND group, such as PD-MCI group. Therefore, further research is needed to elucidate the social functioning in PD patients with MCI and elucidate the difference between PD-ND, PD-MCI, and PD-D. Second, the assessment of the social functioning may be culturally different; further studies are suggested to collect the cross-cultural and larger samples to revalidates our results. Third, most of our patients live at home; therefore, further studies need to explore the applicability of this scale to patients living in health centers or institutes. Fourth, the PDSFS is suitable for retired patients as the majority of patients with PD were elderly and retired; however, the occupational function and work dimension are crucial and may influence the individual’s social function for younger or patients who still be working. Further study was needed to elucidate this issue. Last but not least, the sample sizes in PD-D group was relatively small, and the severity of dementia was mild. This scale is suitable for the PD patients with normal cognition or mild dementia. It is challenging to test too severe dementia patients, and their clinical symptoms were obvious, allowing clinicians or family directly infer the patient’s social dysfunction, while milder PD-D patients were often in an ambiguous state and need further examination for judgment.
In summary, with the increase in the prevalence of PD in the aging society, the accuracy of the diagnosis and early detection of patients with PD-D become vital. In the current study, the PDSFS was developed, and its psychometric properties were established, as well as its cut-off score was provided. The result might benefit the clinical practice of population aging and contribute to the detection of cognitive decline and dementia. Also, the PDSFS is handy, and the time required for the PDSFS is short (around 10 minutes), which can be self-reported without an additional trainer. That is, the PDSFS has clinical utility.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
We thank all the subjects who participated in this study, and we are grateful to the grant support from the Ministry of Science and Technology (MOST), Taipei, Taiwan (MOST 106-2410-H-006 -039 -MY2 and MOST 108-2410-H-006-046 -).
