Abstract
Background:
Premorbid educational attainment is a well-known proxy of reserve, not only with regard to cognition, but also to motor symptoms.
Objective:
In the present study, we investigated the relationship between educational attainment and long-term motor prognosis in patients with Parkinson’s disease (PD).
Methods:
We analyzed 466 patients with de novo PD without dementia who underwent dopamine transporter (DAT) scans and were followed up more than 2 years. Patients were divided into three groups: low education (years-of-education ≤6, n = 125), intermediate education (6 <years-of-education < 12, n = 108), and high education (years-of-education ≥12, n = 233). The effects of educational attainment on the development of levodopa-induced dyskinesia (LID), wearing-off, and freezing-of-gait, and longitudinal increase in levodopa-equivalent doses (LEDs) were assessed.
Results:
Multiple regression analysis showed that higher education was associated with milder parkinsonian symptoms after adjusting for DAT availability in the posterior putamen. Survival analysis showed that the rate of LID was significantly lower in the high education group than in the low education group (HR = 0.565, p = 0.010). A linear mixed model showed that the high education group had lower LED than the low education group until a period of 30 months; however, this difference in LED was not observed thereafter.
Conclusion:
The present study demonstrated that premorbid educational attainment has protective effects on the development of LID in patients with PD and has sparing effects on LED during the early treatment period. These results suggest that high educational attainment has a beneficial effect on motor outcomes in patients with PD.
INTRODUCTION
The concept of cognitive reserve (CR) accounts for differences between individuals in their susceptibility to pathologies related to Alzheimer’s disease (AD) [1]. There are several well-known proxies of CR such as educational and occupational attainment and leisure activities [1]. Patients with high CR may exhibit fewer cognitive deficits despite having a similar level of AD-related pathologies [1].
Parkinson’s disease (PD) is a prevalent neurodegenerative disease that affects the elderly [2] and imparts significant motor compensation [3]. The severity of motor deficits may differ among patients with similar levels of striatal dopamine depletion. Therefore, similar to the concept of CR in AD, motor reserve in PD may explain individual differences in parkinsonian motor deficits despite similar levels of PD-related pathological changes [4]. Previously, we found that a high level of exercise engagement, high level of educational attainment, and dominant side-onset of motor symptoms are associated with enhanced motor reserve in PD; patients with high motor reserve showed lower levels of motor deficits despite similar levels of striatal dopamine depletion, compared to those with low motor reserve [5–7]. In addition, a recent study demonstrated that enhanced motor reserve may have a beneficial influence on long-term motor prognosis in PD, such as a lower risk of levodopa-induced dyskinesia (LID) and freezing of gait (FOG), and lower levodopa-equivalent doses (LED) during follow-up [8].
Educational attainment is a well-known proxy for both motor and cognitive reserve; patients with high educational attainment show less cognitive dysfunction in AD as well as less severe motor deficits in PD, despite similar levels of disease-specific pathologies [1, 6]. A recent longitudinal study demonstrated that higher education is associated with better baseline motor/cognitive function in PD, but not with motor decline during follow-up [9]. In the present study, we investigated the influence of premorbid educational level on long-term motor prognosis in PD, such as the development of LID and FOG, and increase in LED.
MATERIALS AND METHODS
Participants
Study subjects were selected from the Yonsei Parkinson Center database (consecutive patients sampled from April 2009 to September 2015), who fulfilled the following selection criteria: 1) drug-naive PD patients with documented education history; 2) patients who underwent dopamine transporter (DAT) imaging using [18F] N-(3-fluoropropyl)-2β-carbonethoxy-3β-(4-iodophenyl) nortropane PET (FP-CIT PET) scans; 3) those who showed intact cognition (a score of 24 or higher in the Korean version of the Mini-Mental Status Examination [K-MMSE]) [10]; 4) those who had been followed-up for 2 years or longer. PD was diagnosed according to the clinical diagnostic criteria of the UK PD Society Brain Bank [11], the presence of appropriate DAT defects on FP-CIT-PET scans [12], and the presence of PD drug responses during follow-up. Part III of the Unified PD Rating Scale (UPDRS-III) was used to assess PD motor deficits in each patient at baseline and available in 399 patients. All enrolled patients were followed up for a mean 6.08±2.08 years, and two movement disorder experts (Y.H.S. and P.H.L.) confirmed responsiveness to dopaminergic medication and carefully assessed atypical parkinsonism during the follow-up period. Information on attrition in the cohort over time is shown in Fig. 1. We categorized the patients into three groups according to their levels of education: patients with 6 or less years of education were classified as the low education group; patients with 12 or more years of education were classified as the high education group; and patients with education levels between those of low and high education were classified as the intermediate education group. These 6- and 12-year cut-offs correspond to the schooling system in Korea for elementary and high school graduation, respectively. The ethics committee of our hospital reviewed and approved this study with the exemption of informed consent because of the retrospective nature of this study.
Attrition rate in the cohort.
Quantitative analyses of FP-CIT PET data
To assess striatal dopamine depletion, we obtained DAT scans using a GE PET-CT DSTe scanner (GE Discovery STE; GE Healthcare; Milwaukee, WI, USA) to acquire FP-CIT PET scans. Quantitative analyses of PET data were performed according to a previously published methodology [12, 13]. Image processing was performed using SPM8 (Wellcome Department of Imaging Neuroscience, Institute of Neurology, UCL, London, UK) with MATLAB 2013a for Windows (MathWorks, Natick, MA, USA). Quantitative analyses were performed on volumes of interests (VOIs), which were based on a previous study [14]. Briefly, the striatum was divided along the anterior-posterior commissure line on the transverse plane into dorsal and ventral portions. The ventral portion comprised two subregions: the ventral putamen and ventral striatum. Subsequently, the dorsal portion was divided along the coronal anterior commissure plane into the following anterior and posterior subregions: the anterior caudate, posterior caudate, anterior putamen, and posterior putamen. These VOIs were adjusted through a minor translation using our in-house editing software ANIQUE [14]. DAT availability was calculated using the non-displaceable binding potential, which was defined as follows: (mean standardized uptake value of the striatal subregional VOI –mean standardized uptake value of the occipital VOI)/(mean standardized uptake of the occipital VOI) [15, 16].
Assessment of long-term motor prognosis
After PD diagnosis, patients visited our outpatient clinic at 3- to 6-month intervals, and two movement disorder specialists (PHL and YHS) assessed the presence of LID, wearing-off, or FOG based on a history and direct neurological examination at every visit. The presence of LID was assessed through a history from either patients or caregivers, or by direct inspection of patients at the clinic. The onset of wearing-off was defined as the time point when either patients first complained of end-of-dose deterioration or when we first decided to increase the dosing schedule of levodopa from three times to four times a day. Nocturnal or early morning off symptoms were not regarded as indicative of wearing-off. FOG was defined as an unintentional and temporary phenomenon where an individual’s foot failed to move forward, despite an intention to walk [17]. We inspected the patients’ gait carefully at the clinic and specifically asked the patients at every visit whether they experienced the characteristic sensation of their feet becoming “glued to the floor”. Survival duration was defined as the time from treatment initiation to the occurrence of either LID or wearing-off (for patients with LID or wearing-off), the time from parkinsonian symptom onset to the occurrence of FOG (for patients with FOG), or the time to the last clinical visit (for patients without these events). Based on the patients’ medical records, medications for PD prescribed at each visit were checked and LED was calculated according to a previously defined methodology [18, 19].
Statistical analysis
To compare the baseline demographic characteristics between groups, one-way ANOVA with post-hoc Bonferroni correction was used for continuous variables, and Pearson’s χ2 tests were used for categorical variables. We examined the association of education with the baseline UPDRS-III score using linear regression after adjusting for age at symptom onset, sex, disease duration, and baseline DAT availability in the posterior putamen in 399 patients who had baseline UPDRS-III score. The time from the initial visit to the onset of motor complications was assessed using Kaplan–Meier estimates. Pair-wise log-rank tests were used to compare Kaplan–Meier plots between the groups. If pairwise log-rank tests were significant in the Kaplan–Meier plots, the Cox regression model was used to estimate the hazard ratio (HR) and 95%confidence intervals (CIs) while adjusting for age at symptom onset, sex, disease duration, baseline DAT availability in the posterior putamen, and LED per body weight at onset of motor complications in patients with motor complications or at the last visit to the outpatient clinic in those without motor complications.
A linear mixed model was used to compare the rates of longitudinal LED changes between the groups. The linear mixed model was expressed as follows:
where covariates included age at symptom onset, sex, disease duration, and DAT availability in the posterior putamen. Because most of the increase in LED occurred within the first 6 months, and the doses of dopaminergic medications were adjusted every 3 to 6 months thereafter, time was treated as a categorical variable with a 6-month interval, up to 72 months (when > 40%of the patients (n = 221) were followed up). The effects of the PD group on longitudinal changes in LED were tested using a group×time interaction term. Statistical analyses were performed using SPSS (version 25.0; IBM Corporation, Armonk, NY, USA) and R (v4.0, http://www.r-project.org). Statistical significance was set p < 0.05.
Data availability
The de-identified data that support the findings of this study are available from the authors upon reasonable request.
RESULTS
Baseline demographic characteristics
The demographic and clinical characteristics of the enrolled patients are summarized in Table 1. The age at symptom onset was lower in the high education group than in the low education group. There were significantly fewer women in the high education group than in the intermediate and low education groups. The high education group showed higher K-MMSE scores than the other groups. Other variables such as disease duration, BMI, and DAT availability in the posterior putamen were comparable among the groups.
Demographic characteristics (follow up≥2 years)
Values are expressed as mean±standard deviation or number (percentage). p-values were obtained from analyses of variance, independent sample t-test, χ2 tests, or Fisher exact tests, as appropriate. aSignificantly different in comparison between low and middle education groups. bSignificantly different in comparison between low and high education groups. cSignificantly different in comparison between middle and high education groups. BMI, body mass index; DAT, dopamine transporter; PD, Parkinson’s disease; UPDRS III, Unified Parkinson’s Disease Rating Scale Part III.
Although baseline UPDRS-III scores were comparable among the groups, higher education was associated with lower UPDRS motor scores after adjusting for baseline DAT availability in the posterior putamen (continuous, β= –0.258, standard error [SE] = 0.104, p = 0.014; categorical, β= –2.613, SE = 1.107, p = 0.019; Table 2). Models with additional adjustment for age at symptom onset, sex, and disease duration showed similar results (continuous, β= –0.299, SE = 0.109, p = 0.006; categorical, β= –3.023, SE = 1.142, p = 0.008; Table 2).
Association between education level and UPDRS
Analyses were performed using linear regression, and regression coefficients (β) are displayed together with their standard error (SE). *Adjusted for DAT availability in the posterior putamen. **Adjusted for age at symptom onset, sex, disease duration, and DAT availability in the posterior putamen. DAT, dopamine transporter; UPDRS III, Unified Parkinson’s Disease Rating Scale Part III.
Long-term motor prognosis among the groups
During the follow-up period, LID developed in 50 (21.5%) of the 233 patients in the high education group (follow-up duration, 5.94±2.02 years), 27 (25.0%) of the 108 patients in the intermediate education group (follow-up duration, 6.02±2.13 years), and 46 (36.8%) of the 125 patients in the low education group (follow-up duration, 6.39±2.14 years). Wearing-off and FOG developed in 71 (30.5%) and 48 (20.6%) patients in the high education group, 29 (26.9%) and 28 (25.9%) patients in the intermediate education group, and 36 (28.8%) and 34 (27.2%) patients in the low education group, respectively. Kaplan–Meier analyses revealed that the high education group had a lower risk of developing LID than the low education group (ploghboxrank =0.016, Fig. 2). This difference was maintained after adjusting for potentially confounding variables, such as age at symptom onset, age at symptom onset, sex, disease duration, DAT availability in the posterior putamen, and LED per body weight (HR, 0.565; 95%CI, 0.367–0.871; p = 0.010, Table 3). When treating years of education as a continuous variable, higher education level was associated with a lower risk of developing LID (HR, 0.935; 95%CI, 0.894–0.977; p = 0.003, Table 3). There was no significant difference in the development of wearing-off and FOG between the groups.
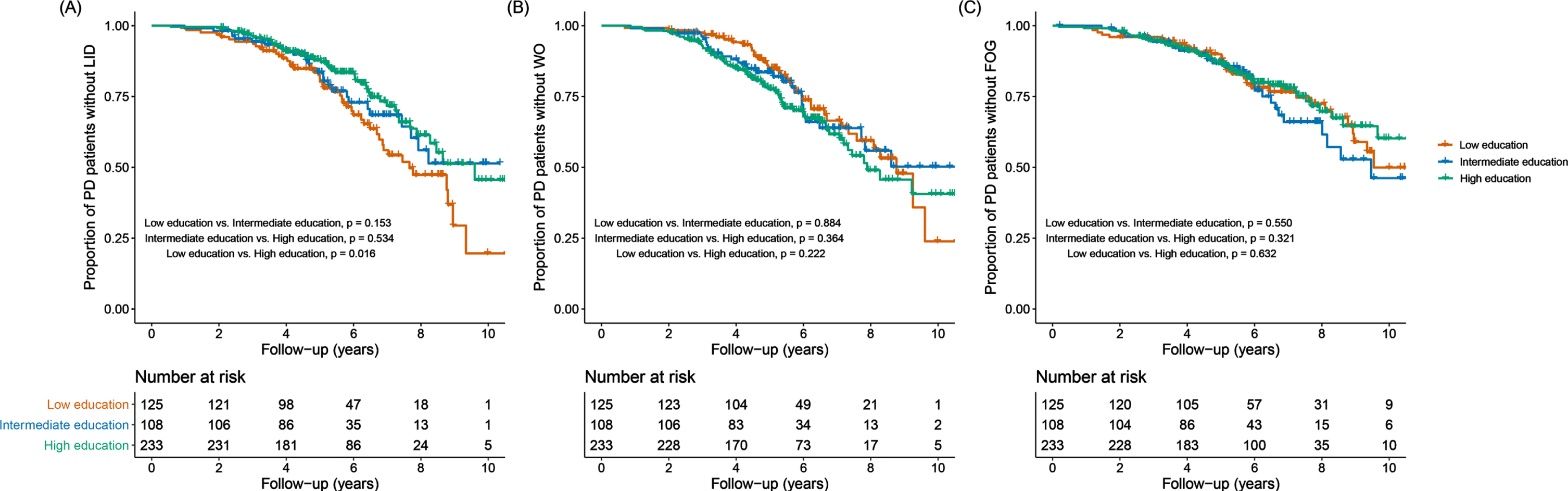
Kaplan-Meier survival curves for motor complications. Curves of Kaplan-Meier estimates of the onset of levodopa-induced dyskinesia (A), wearing-off (B), and freezing of gait (C) after initial visit in the high (green), intermediate (blue), and low education (red) groups. The high education group had a lower risk of early development of levodopa-induced dyskinesia than the low education group (ploghboxrank =0.016). A risk of development of wearing-off or freezing of gait was comparable among the groups. FOG, freezing of gait; LID, levodopa-induced dyskinesia; PD, Parkinson’s disease; WO, wearing-off.
Multivariate Cox Hazard models for prediction of the development of LID
Results of Cox regression analyses for the development of levodopa-induced dyskinesia after controlling for age at symptom onset, sex, disease duration, baseline DAT availability in the posterior putamen, and LED per body weight. DAT, dopamine transporter; LED, levodopa-equivalent dose.
There was a significant group×time interaction in the mixed model (p < 0.001) after adjusting for age at symptom onset, sex, disease duration, baseline DAT availability in the posterior putamen, time, and group×time, indicating that the pattern of longitudinal changes in LED differed among the groups. Throughout the follow-up period, the difference in the estimated mean of the LED between the high and low education groups was significant until 30 months; thereafter, the estimated mean LED of the two groups were comparable thereafter (Table 4 and Fig. 3).
Longitudinal changes in levodopa-equivalent doses in patients with Parkinson’s disease
Values are expressed as the estimated mean (standard error) for levodopa-equivalent doses after controlling for age at symptom onset, sex, disease duration, baseline DAT availability in the posterior putamen, time, and group×time. p values were calculated using a linear mixed model analysis. aSignificantly different in comparison between low and intermediate education groups. bSignificantly different in comparison between low and high education groups. DAT, dopamine transporter.
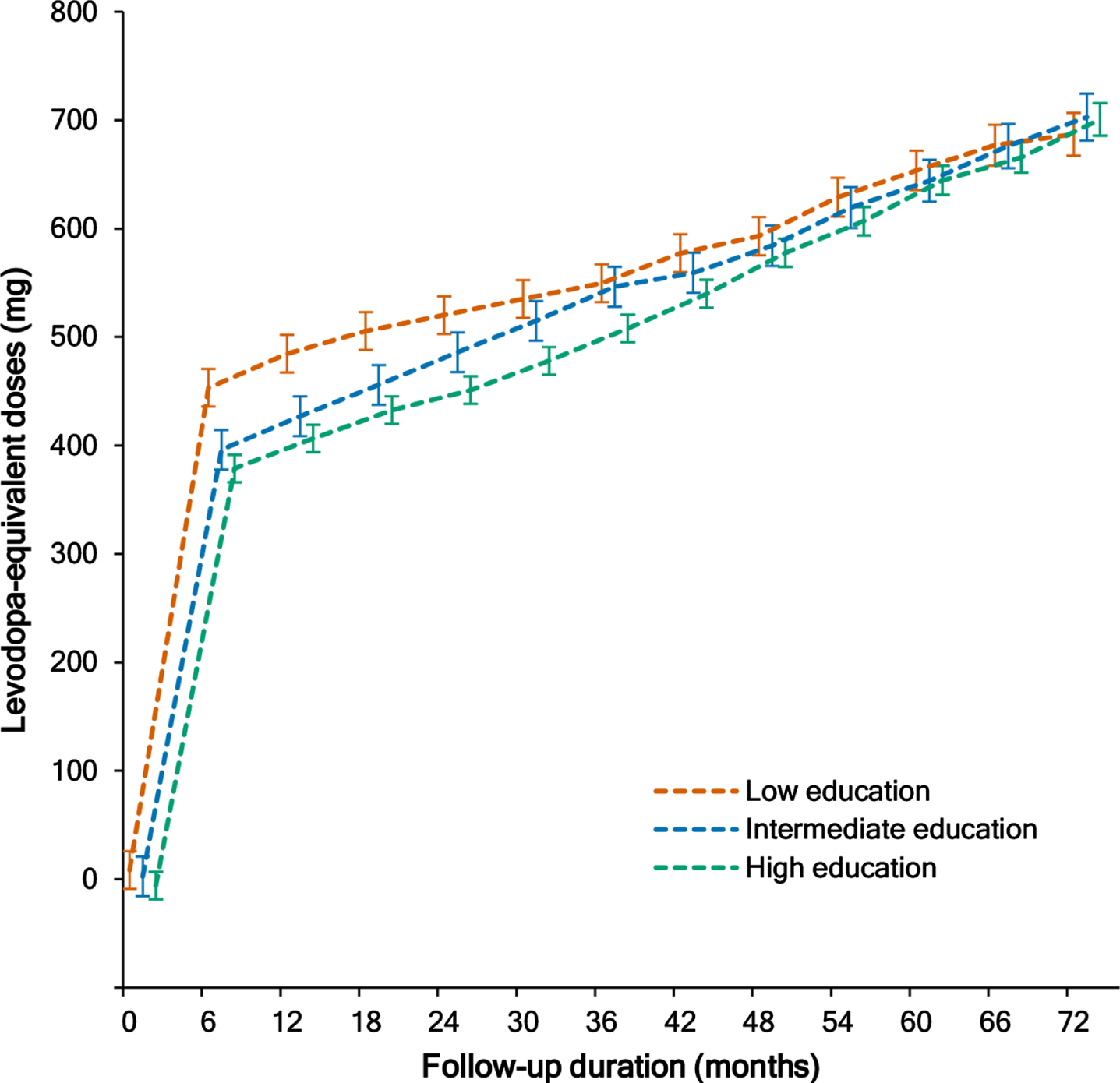
Longitudinal increases in levodopa-equivalent doses. A linear mixed model analysis showed longitudinal changes in levodopa-equivalent doses in the high (green), intermediate (blue), and low education (red) groups. Error bars represent standard errors of the estimated means.
DISCUSSION
This study showed that patients in the high education group exhibited lower UPDRS-III scores at similar levels of striatal dopamine depletion, compared to patients in the low education group. This result is consistent with our previous study [6], and supports the notion that high educational attainment enhances motor reserve in PD. The lower number of women in the high education group is also consistent with our previous study [6], indicating that women may have had fewer opportunities for schooling in Korea in the past. The finding of older age at symptom onset in the low education group compared to the other groups was unexpected. A previous epidemiological study showed an association between higher education and increased PD risk [20]; however, this result could be biased by the fact that education can be an indicator of increased medical awareness and access to specialty care [21], Thus, less educated people, particularly those who did not graduate from elementary school, may have a low level of medical awareness, and thus the delayed recognition of their illnesses. On the other hand, this result may simply indicate that older people may have had fewer opportunities for schooling in the past. Although we excluded patients with cognitive impairments, there were subtle, but significant differences in K-MMSE scores among the groups. This difference was fully expected, as shown in our previous study [6].
In terms of long-term motor prognosis, patients in the high education group were less likely to develop LID during follow-up. This result is consistent with a previous observation of an association between higher motor reserve and a lower chance of LID occurrence [8]. Dominant-side onset of motor symptoms is an acknowledged biomarker for enhanced motor reserve in PD [7] and can delay the development of LID; a greater potential of synaptic plasticity in the dominant hemisphere may exert a protective role for the development of LID compared to that in the non-dominant hemisphere [22]. Environmental enrichment from enhanced cognitive activities can enhance adult neurogenesis, synaptogenesis, and synaptic plasticity in experimental animals [23]. In humans, higher education level is associated with greater cerebral volume, stronger white matter integrity, more synaptic plasticity, and more efficient recruitment of brain networks or recovery mechanisms [24–26]. These changes may also exert a protective role for the development of LID, as observed in dominant-side PD [22].
Patients in the high education group exhibited a different pattern of changes in LED during follow-up compared to those in the low education group. In the early period of treatment up to 30 months, the high education group required lower LED to control for their symptoms than did the low education group; however, this difference diminished thereafter and was eventually eliminated at the end of the observed follow-up period, 72 months after treatment. Lower LED in the high education group was associated either with higher baseline motor reserve or with younger age at symptom onset because they were known to be potential contributing factors for LED requirements during early treatment period up to 36 months [8, 27]. As PD progresses, PD drugs become less effective presumably due to the emergence of either levodopa-resistant symptoms or response fluctuations. Therefore, it is fully conceivable that the beneficial effects of enhanced baseline motor reserve, i.e., low LED requirements, cannot be maintained after a certain stage of PD. In AD, individuals with high cognitive reserve showed faster cognitive decline once they reach a critical threshold [1]. Similarly, in this study, patients in the high education group exhibited a faster increase in LED after a 30-month period of treatment. Previous studies have repeatedly demonstrated that higher educational attainment is associated with better motor performance in patients with PD as well as in healthy elderly individuals, but not with PD- or aging-related motor decline [9, 29].
We enrolled a relatively large number of consecutive patients with de novo PD, which may have minimized patient-sampling biases. A relatively long follow-up period could provide more appropriate information about the long-term motor prognosis in PD. However, this study had some notable limitations. First, the patients were subdivided into three groups according to their educational level, which may be arbitrary and act as a bias. There is no consensus on the cut-offs for educational levels, but previous studies have used cut-offs corresponding to their respective schooling system, usually high school graduation if divided into two groups [30, 31]. We modified this classification into the three groups, with elementary school and high school graduates as cut-offs, because a significant number of Korean PD patients have not graduated from elementary school. Second, because of the high proportion of women in the low education group, a finding that has also been previously reported [6, 32], the influence of gender on the present results cannot be completely excluded. Third, this study did not consider traditional risk factors such as excessive alcohol drinking, marital status, co-morbidities, and physical inactivity that may act as confounders of motor prognosis in patients with PD. Fourth, due to the lack of follow-up data on Hoehn and Yahr stages or UPDRS-III scores, we assessed the clinical progression based on the longitudinal changes in LED, which may not accurately reflect the motor status of patients with PD. Finally, the study’s retrospective design that determined LID, FOG, and wearing-off based on medical records precluded definitive conclusions. Prospective observations can provide more reliable information.
In summary, the present study demonstrated that premorbid educational attainment has protective effects on the development of LID in patients with PD and the role of education as a motor reserve proxy fades away as the disease progresses.
Footnotes
ACKNOWLEDGMENTS
This research was supported by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute, funded by the Ministry of Health & Welfare, Republic of Korea (HU21C0053).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
