Abstract
Background
The motor reserve estimates (MRes) derived from a residual approach correlate with motor severity in Parkinson's disease (PD), leaving the independent effect of motor reserve on clinical outcomes unclear.
Objective
Investigate the independent influence of motor reserve on the long-term outcome.
Methods
Using the Parkinson's Progression Markers Initiative (PPMI) and Pusan National University Hospital (PNUH) datasets, we investigated the association of MRes with progression of motor severity as well as risk of phenoconversion to Hoehn & Yahr (H&Y) stage 3. Two MRes types were calculated: (1) original MRes, negative conversion of standardized residuals from a generalized linear model (GLM) between putaminal dopamine transporter (DAT) levels and motor severity, and (2) modified MRes, standardized residuals from the GLM between original MRes and motor scores.
Results
Original MRes correlated with baseline motor scores, while modified MRes did not. Modified MRes in both cohorts were associated with a rapid increase in motor severity (linear mixed effect model, interaction between MRes and disease duration; PPMI, Movement Disorder Society sponsored Unified Parkinson Disease Rating Scale part III [MDS-UPDRS III], estimate 2.248 × 10−3, p < 0.001; PNUH, UPDRS III, estimate 0.027, p = 0.014) In both cohorts, Kaplan-Meier plots showed high modified MRes indicated higher risk of progression to H&Y stage 3. Mediation models using original MRes agreed that high MRes were associated with an accelerated increase in motor scores.
Conclusions
Independent of baseline motor severity, MRes were associated with rapid motor deterioration and high risk of progression to H&Y stage 3.
Plain language summary
Motor reserve is a resilience mechanism that helps manage the degenerative process of Parkinson's disease. It has been suggested that patients with a strong motor reserve may experience milder symptoms. However, the independent influence of motor reserve on symptoms remains unclear, as motor reserve estimates (MRes) are closely linked to symptom scores. We observed that higher MRes scores were associated with faster progression of motor symptoms. The benefit of motor reserve appears to lessen as the disease advances.
Introduction
The motor features of Parkinson disease (PD) are known to appear when the striatal dopaminergic deficit reaches 30—50% of healthy individuals.1,2 Recent studies have proposed the concept of motor reserve that the resilience mechanism coping with neurodegenerative process of PD may exist, and suggested that efficiency of motor reserve would cause symptomatic variability among patients.3,4
Several studies employed the residual approach from the generalized linear model (GLM), with putaminal dopamine transporter (DAT) levels as the predictor and motor severity as the responder. 3 The motor reserve estimates (MRes) at the diagnosis of PD are shown to be associated with education attainment, premorbid exercise, the predominant body side affected by parkinsonian features, and further risk of dementia.3,5–7 However, the association of MRes with long-term motor outcomes in PD remains unclear.
Residuals in a linear model with a low explained variance may correlate with a responder variable,8,9 and this is also the case for MRes. 10 While a statistical test may show a significant association with MRes and clinical features, it remains unclear whether the reserve function has an actual influence on outcome variables. It is still possible that a statistical significance could be mediated by motor severity, especially in case where outcome variables are associated with motor severity. Collinearity between two variables in a single statistical model can be problematic, leading to imprecise estimates or sign flipping. 8 Consequently, statistical models examining the association between MRes and long-term outcomes of PD may be biased by the inherent correlation between MRes and motor severity.
In the present study, we calculated the modified MRes from the linear model between originally published MRes (original MRes) and motor scores to obtain estimates independent of baseline motor severity. In addition, mediation models including baseline motor severity as a mediator were employed to explore the direct effects of original MRes. The association of MRes with motor progression was investigated using two independent cohorts: the Parkinson's Progression Markers Initiative (PPMI) and Pusan National University Hospital (PNUH, Busan, South Korea) datasets.
Methods
Participants
Participants in the study were included from the following two cohorts: the PPMI and Pusan National University Hospital (PNUH). The PPMI is an on-going observational study aimed at identifying biomarkers of PD progression. The inclusion criteria for the PPMI were as follows: (1) diagnosis of PD within 2 years, (2) baseline H&Y stage 1 or 2, (3) striatal dopaminergic dysfunction on DAT scan, and (4) no previous dopaminergic medication before inclusion. 2 Participants of the PPMI were scheduled to visit every 3 months during the first year, and every 6 months during the subsequent years. The data used in this study were downloaded in August 2023. We included patients who were followed up more than 4 years but patients with pathogenic variants in LRRK2 (n = 6), GBA1 (n = 8) or both LRRK2 and GBA1 (n = 1) were excluded. Finally, 323 PD patients were included in the study.
The PNUH cohort consisted of 73 PD patients who were followed up for more than 4 years. Diagnosis of PD was made according to the Movement Disorder Society diagnostic criteria for PD. 11 Exclusion criteria were as follows: (1) disease duration > 3 years or H&Y stage ≥ 3 at the initial visit, (2) any medical illness interfering with activity of daily living during follow-up, (3) administration of anti-dopaminergic medication, and (4) any structural lesion influencing the measurement of striatal DAT uptake.
Outcome measures
Demographic characteristics, including age, sex, disease duration and education period were recorded at baseline. We included MDS-UPDRS III and H&Y stage in the PPMI dataset, and UPDRS III and H&Y stage in PNUH dataset. In the PPMI dataset, subscores for each parkinsonian motor deficit were calculated as follows: (1) bradykineseia – sum of MDS-UPDRS items 3.2, 3.4, 3.5, 3.6, 3.7, 3.8 and 3.14; (2) tremor – sum of items 3.15. 3.16, 3.17 and 3.18; (3) rigidity – sum of item 3.3; and (4) axial motor deficits – sum of items 3.9, 3.10, 3.11, 3.12 and 3.13. In the PNUH dataset, subscores of UPDRS III were calculated as follows: (1) bradykinesia – sum of UPDRS items 19, 23, 24, 25, 26, and 31; (2) tremor – sum of items 20 and 21; (3) rigidity – sum of item 22; and (4) axial motor deficits – sum of items 27, 28, 29 and 30. The levodopa equivalent daily dose (LEDD) was calculated using formula suggested by Jost et al. 12
Dopamine transporter scan and image processing
For the PPMI cohort, the single-photon emission computed tomography (SPECT) images were acquired 3–4 h after injecting 123I-N-3-fluoropropyl-2-β-carboxymethoxy-3β-(4-iodophenyl) nortropane (FP-CIT) and were reconstructed with an iterative method. Using PMOD (PMOD Technologies, Zurich, Switzerland), reconstructed images were corrected by attenuation, smoothed with 6 mm full-width at half-maximum (FWHM) of Gaussian filter, and finally normalized to the Montreal Neurologic Institute (MNI) space. Eight consecutive axial slices around the slice with the highest striatal uptake were averaged to create a single axial slice and then regions-of-interests (ROIs) for each side of the caudate and putamen were applied to measure the regional uptake. Additionally, an ROI for the occipital cortex was applied to measure an uptake value for the reference region. 2
For the PNUH cohort, 18F-FP-CIT PET was performed 180 min after injection of 18F-FP-CIT with Siemens Biograph 40 Truepoint PET/CT (Siemens Healthcare, Knoxville, Tennessee, USA). Continuous spiral CT was obtained with a tube voltage of 120kVp. PET scan was obtained in a 3-dimensional mode and reconstructed using an ordered-subset expectation maximization algorithm. The automated processing tool Magia 13 (https://github.com/tkkarjal/magia) was used to process brain PET scan. Processing began with the co-registration of the PET and MR images. Magia uses FreeSurfer 7.3.0 (http://surfer.nmr.mgh.harvard.edu/) to generate anatomical parcellations for defining ROIs. FreeSurfer assigns an anatomical label to each brain voxel, and ROIs thus consist of all the voxels with the same anatomical label. 14 Then, the MRI is segmented into gray and white matter probability maps for spatial normalization. This step of Magia pipeline was validated and documented in previous publication. 13 Brain PET scans were spatially normalized to MNI-space and finally smoothed using a Gaussian kernel. After the extraction of specific binding of each ROI, specific binding ratios (SBRs) were calculated as follows with a reference of cerebellum; SBR = (target– cerebellum)/cerebellum.)
Calculation of the MRes
The original MRes were derived by taking the negative conversion of standardized residuals from a GLM using age, disease duration, and log-transformed putaminal DAT levels as predictors and baseline motor scores (MDS-UPDRS III or UPDRS III) as a responder variable. The residuals represent a difference between predicted and measured values of motor scores (observed values – predicted values); therefore, lower original MRes indicates a greater motor severity than the predicted. 15 In addition, we calculated additional MRes in the PNUH dataset using DAT levels in the posterior putamen. In this manuscript, MRes-WP denotes MRes calculated with DAT levels in the whole putamen, whereas MRes-PP refers to estimates based on the posterior putamen.
Our datasets reproduced significant correlations between motor scores and original MRes (Supplemental Figure 1A, D, G) as previously reported. 10 To minimize a potential bias from the inherent correlation with baseline motor scores, we applied the modification of MRes calculating standardized residuals from the GLM using baseline motor scores and log-transformed putaminal DAT levels as predictors and the original MRes as a responder 8 (original MRes ∼ baseline motor scores + log-transformed DAT levels). The modified MRes represent the motor reserve when predictors are zero; therefore the modified MRes explain a distribution of original MRes independent of motor severity. The modified MRes showed no correlations with motor scores and DAT availability at baseline (Supplemental Figure 1B, E, H), but exhibited significant correlations with original MRes (Supplemental Figure 1C, F, I).
Statistical analyses
Continuous variables were compared by independent t-test or Mann-Whitney U test according to a distribution of variables. Frequencies of categorical variables were compared by the chi-square test. To explore an effect of modified MRes on the motor scores, we composed the linear mixed effect (LME) models with random slopes and intercepts using the modified MRes, age, sex, disease duration, LEDD, baseline motor scores as fixed effects. The interaction term between disease duration and modified MRes was also included in fixed effects. To obtain Gaussian distribution of the dependent variable in the LME model, we underwent square-root transformation of MDS-UPDRS III total scores in PPMI dataset. The UPDRS III scores in PNUH cohort were included in the LME model without transformation. To demonstrate a categorical difference in risk of progression to H&Y stage 3, we classified subjects into low and high modified MRes using the mean of modified MRes as the cut-off value. Log-rank tests were employed to compare a risk of progression to H&Y stage 3 in Kaplan-Meier plots. Finally, we conducted the mediation analyses to evaluate the direct effect of original MRes on the mean values and change rates of motor scores (estimates from linear models using disease duration as a predictor and motor scores as a responder) during study period. The original MRes were included in the models as a predictor and baseline motor scores were employed as a mediator. Statistical analyses were conducted using the R software (version 4.2.2; r-project.org) and packages including lme4 (for LME model; version 1.1.35.3) and lavaan (for mediation models; version 0.6.18). Results with a two-tailed p-values < 0.05 were considered statistically significant.
Results
Demographics and clinical characteristics at baseline
Demographics and clinical characteristics at baseline are provided in Table 1. In the PPMI cohort, 149 patients (46.1%) progressed to H&Y stage 3 during a mean (± SD) follow-up period of 106.7 ± 106.6 months (median 117; range 48 ∼ 153 months). In the PNUH dataset, 27 patients (37.0%) showed phenoconversion to H&Y stage 3 during a follow-up period of 84.4 ± 84.4 months (median 84; range 48 ∼ 124 months; Supplemental Figure 2). LME models using disease duration as the fixed effect exhibited annual increase of 1.20 in MDS-UPDRS III in the PPMI cohort, while showing annual increase of 0.85 in UPDRS III in the PNUH dataset. (Supplemental Figure 3) Although there were significant differences between the PPMI and PNUH datasets in sex ratio, age at onset, disease duration at baseline, and education level (Table 1), the frequency of progression to H&Y stage 3 did not significantly differ between the two groups (χ2 0.322, p = 0.570). There were no significant associations of the original and modified MRes with education years and sex.
Demographic and clinical characteristics at baseline.
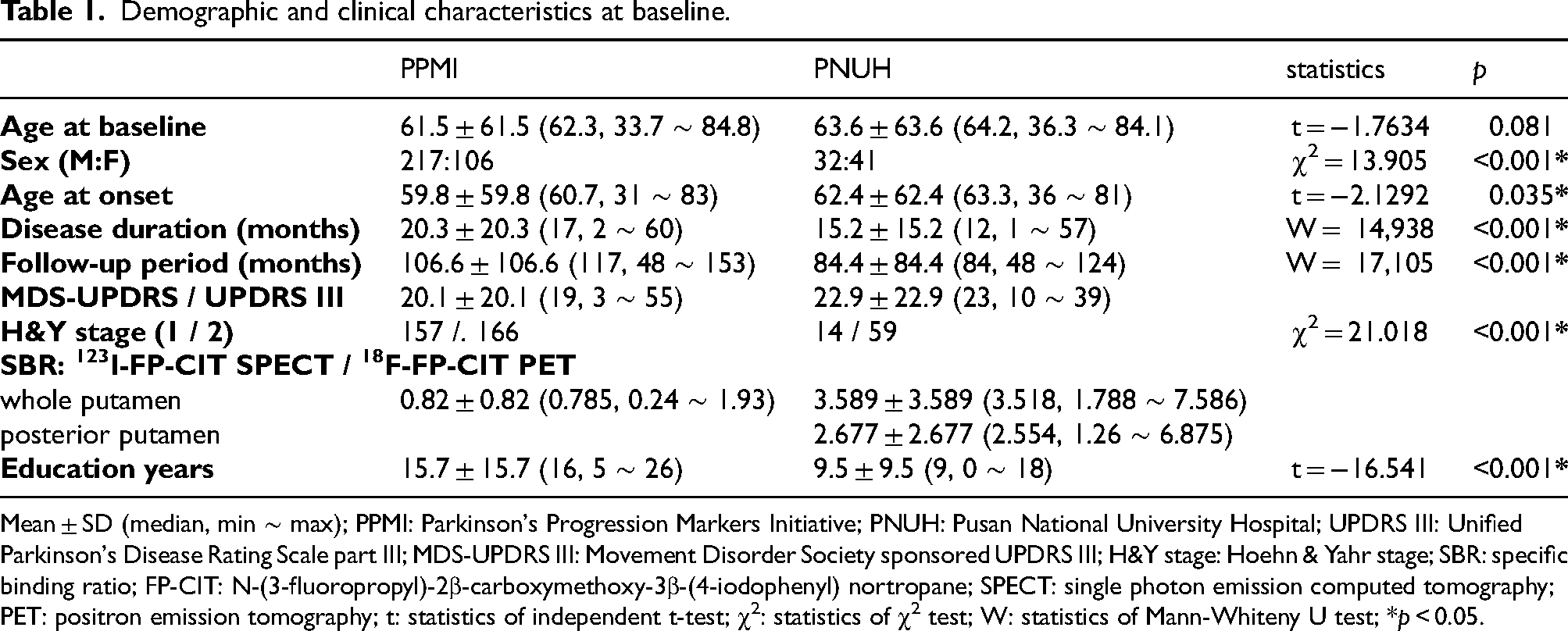
Mean ± SD (median, min ∼ max); PPMI: Parkinson's Progression Markers Initiative; PNUH: Pusan National University Hospital; UPDRS III: Unified Parkinson's Disease Rating Scale part III; MDS-UPDRS III: Movement Disorder Society sponsored UPDRS III; H&Y stage: Hoehn & Yahr stage; SBR: specific binding ratio; FP-CIT: N-(3-fluoropropyl)-2β-carboxymethoxy-3β-(4-iodophenyl) nortropane; SPECT: single photon emission computed tomography; PET: positron emission tomography; t: statistics of independent t-test; χ2: statistics of χ2 test; W: statistics of Mann-Whiteny U test; *p < 0.05.
Effects of modified MRes on the progression of parkinsonian motor deficits
In the PPMI dataset, modified MRes were associated with lower square-root transformed MDS-UPDRS III total scores during the follow-up period (Table 2, main effect; estimate −0.333, SE 0.056, p < 0.001). Diagnostic plots evaluating the assumption of LME model are presented in Supplemental Figure 4. The interaction between disease duration and modified MRes had a positive estimate, indicating that high modified MRes was linked to a more rapid deterioration of motor features (Figure 1(a); Table 2, interaction with disease duration; estimate 2.248 × 10−3, SE 3.248 × 10−3, p < 0.001). In addition, modified MRes were correlated with lower severity of bradykinesia, rigidity and axial motor deficits but were associated with a rapid increase of subscores. We observed similar effects of modified MRes in dataset that included MDS-UPDRS III scores measured during “off-state” (>12 h after last medication). Modified MRes was associated with higher tremor scores; however, this association was no longer significant in the dataset using off-state MDS-UPDRS III scores. When patients in the PPMI cohort were classified to low and high modified MRes (below or above the mean of modified MRes), the participants with high modified MRes had significantly greater original MRes compared to those with low modified MRes. (−0.174 ± 1.053 vs. 0.218 ± 0.890, independent t-test, p < 0.001) Kaplan-Meier plots of low and high modified MRes groups showed that PD patients with high modified MRes had a higher risk of progression to H&Y stage 3 compared to those with low MRes (Figure 2(a)).
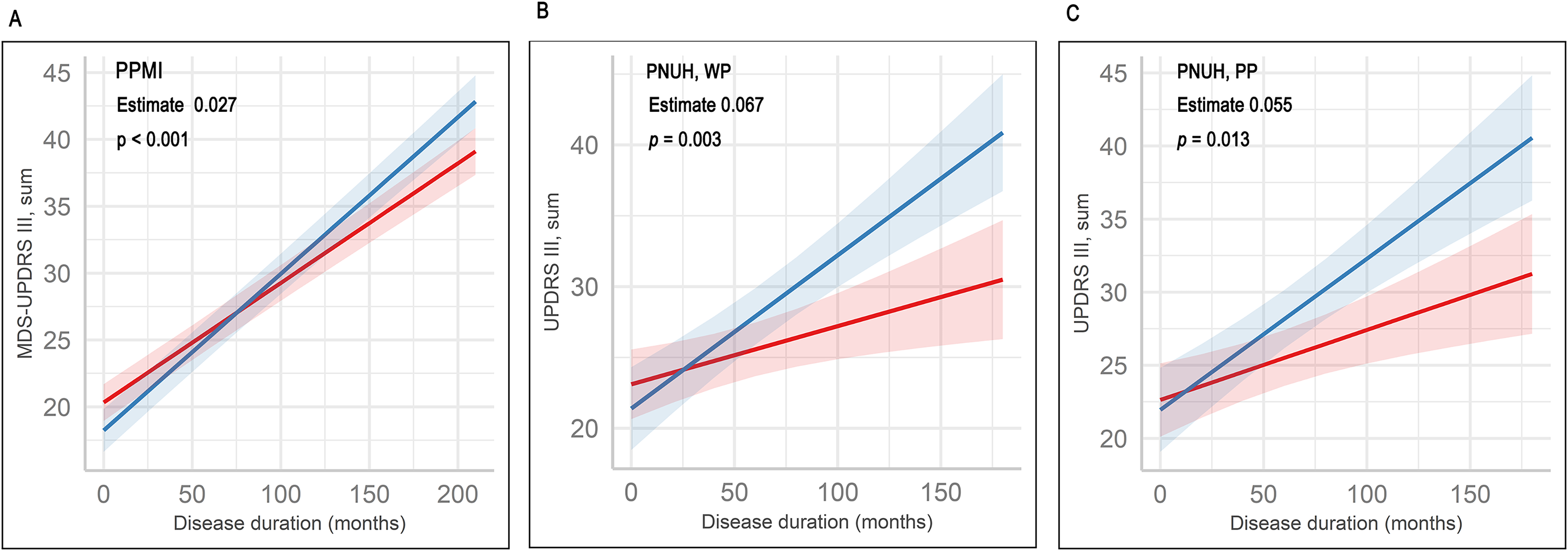
Predicted motor scores (lines) and confidence intervals (shaded areas) in the PPMI (a) and PNUH (b, c) datasets. The MDS-UPDRS III total scores in PPMI dataset were not transformed for interpretability. Predicted values were obtained from linear mixed effect (LME) models using disease duration, motor reserve estimates (MRes) and interaction between disease duration and MRes. Subjects were categorized into low (below the mean of MRes) and high MRes (above the mean of MRes) groups. The estimates and p-values in the panels represent statistics of interaction term. The MRes in the PNUH dataset were calculated using dopamine transporter availability in the whole putamen (WP, B) and posterior putamen (PP, C).
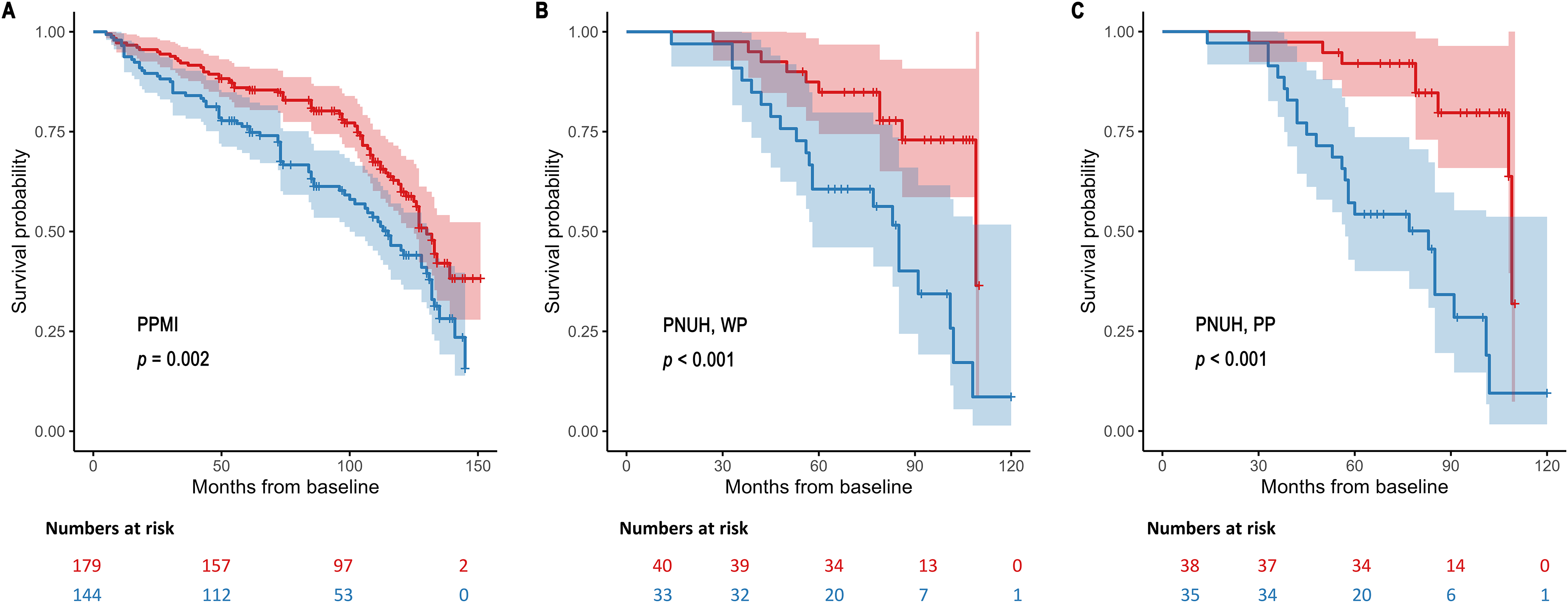
Kaplan-Meier plots for risk of PD progression to H&Y stage 3 in the PPMI (a) and PNUH (b, c) datasets. Shaded areas represent confidence interval. Subjects were categorized into low (below the mean of MRes) and high MRes (above the mean of MRes) groups. The MRes estimates in the PNUH dataset were calculated using dopamine transporter availability in the whole putamen (WP, B) and posterior putamen (PP, C). p-values = p-values from log-rank tests.
Effect of modified MRes on the progression of motor scores.
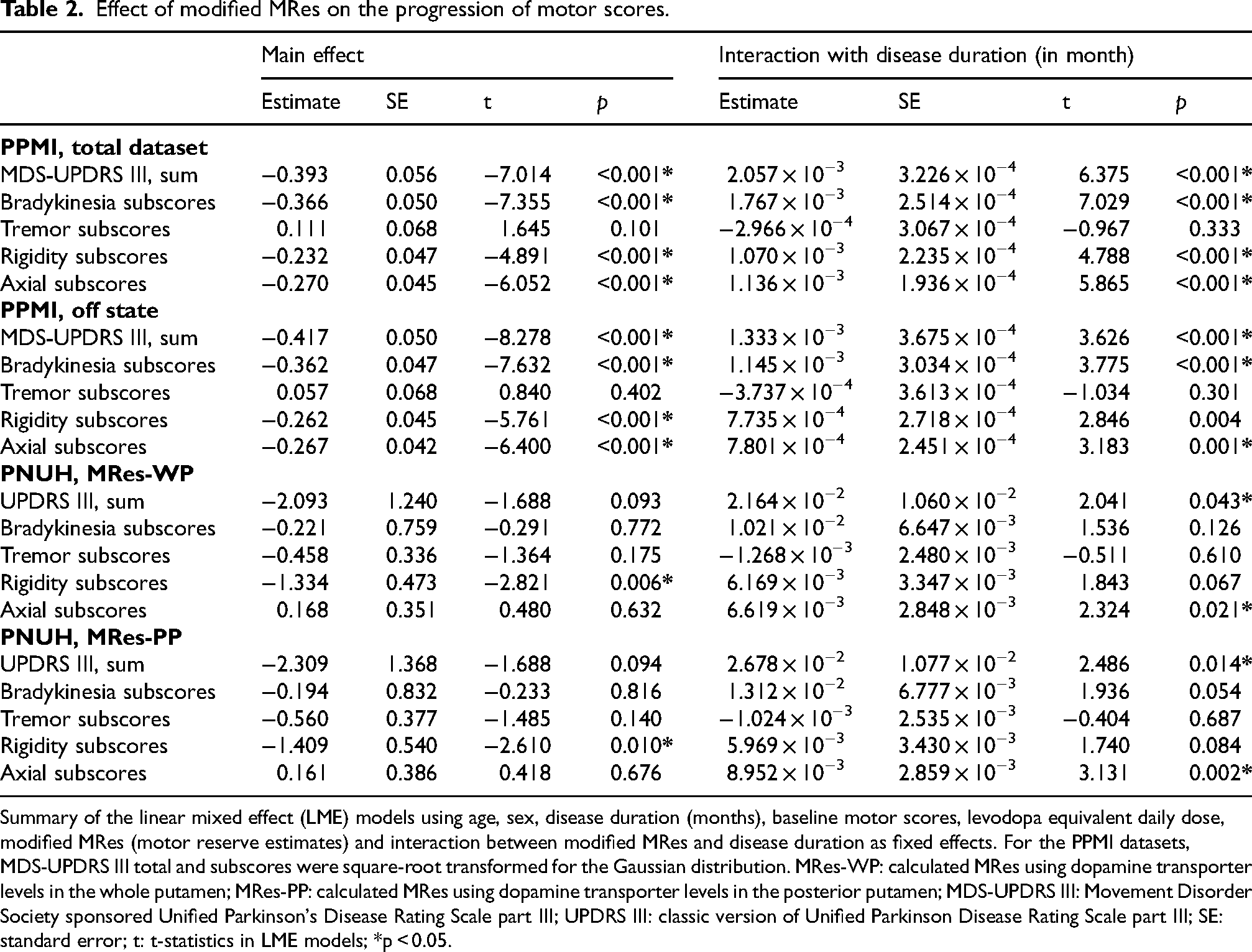
Summary of the linear mixed effect (LME) models using age, sex, disease duration (months), baseline motor scores, levodopa equivalent daily dose, modified MRes (motor reserve estimates) and interaction between modified MRes and disease duration as fixed effects. For the PPMI datasets, MDS-UPDRS III total and subscores were square-root transformed for the Gaussian distribution. MRes-WP: calculated MRes using dopamine transporter levels in the whole putamen; MRes-PP: calculated MRes using dopamine transporter levels in the posterior putamen; MDS-UPDRS III: Movement Disorder Society sponsored Unified Parkinson's Disease Rating Scale part III; UPDRS III: classic version of Unified Parkinson Disease Rating Scale part III; SE: standard error; t: t-statistics in LME models; *p < 0.05.
In the PNUH dataset, quantile-quantile plots of LME models exhibited several outliers, however, plots showing residuals against fitted values displayed no specific pattern (Supplemental Figure 4). Modified MRes-WP showed a similar effect to that in the PPMI cohort (Figure 1(b)), although the LME model adjusting for age, sex, disease duration, and LEDD had a statistically marginal significance (Table 2). Modified MRes-PP were associated with a rapid increase in UPDRS III total scores (Table 2, interaction with disease duration; estimate 2.678 × 10−2, SE 1.077 × 10−2, p = 0.014; Figure 1(c)). Both of MRes-WP and -PP were linked to a rapid increase in subscores for axial motor deficits. In addition, statistically marginal significances were observed between MRes-WP and change rates of rigidity subscores (Table 2, interaction with disease duration; estimate 6.169 × 10−3, SE 3.347 × 10−3, p = 0.067), as well as between MRes-PP and the progression of bradykinesia subscores (Table 2, interaction with disease duration; estimate 1.312 × 10−2, SE 6.777 × 10−3, p = 0.054). As shown in the PPMI dataset, there were significant differences of the original MRes between high and low modified MRes groups (low vs. high MRes groups, independent t-test; MRes-WP −0.318 ± 0.888 vs. 0.385 ± 1.019, p < 0.001; MRes-PP −0.255 ± 0.893 vs. 0.275 ± 1.060, p < 0.001) In addition, the MRes-WP and -PP were associated with a high risk of phenoconversion to H&Y stage 3 (Figure 2(b) and (c)).
Effects of the original MRes on the progression of parkinsonian motor deficits
In the mediation analysis using the baseline MDS-UPDRS III scores as a mediator and original MRes as a predictor, the total effect of original MRes was associated with lower mean values of MDS-UPDRS III in the PPMI dataset over the study period (Supplemental Table 1, total effect of MRes; estimate – 4.983, SE 0.404, p < 0.001), as similar to findings in the LME model using modified MRes. However, the direct effect of original MRes had an opposite direction (Table 3 and Supplemental Table 1; estimate 3.694, SE 1.157, p = 0.001), suggesting that the direction of total effect was driven by a robust indirect effect (Supplemental Table 1, indirect effect; estimate – 8.678, SE 1.109, p < 0.001). In contrast, an additional mediation model using the change rates of MDS-UPDRS III total scores over time as an outcome variable revealed that original MRes had a significant direct effect on the rapid increase in MDS-UPDRS III total scores (Table 3 and Supplemental Table 1; estimate 0.071, SE 0.025, p = 0.005), indicating that the association of original MRes with a rapid deterioration of motor severity was not mediated by baseline MDS-UPDRS III scores. For subscores of MDS-UPDRS III, the original MRes had significant direct effects on the mean values and change rates of subscores for bradykinesia and axial motor deficits (Table 3 and Supplemental Table 1). The association between original MRes and tremor subscores were no longer significant when including off-state MDS-UPDRS III.
Direct effects of original MRes on the motor scores.
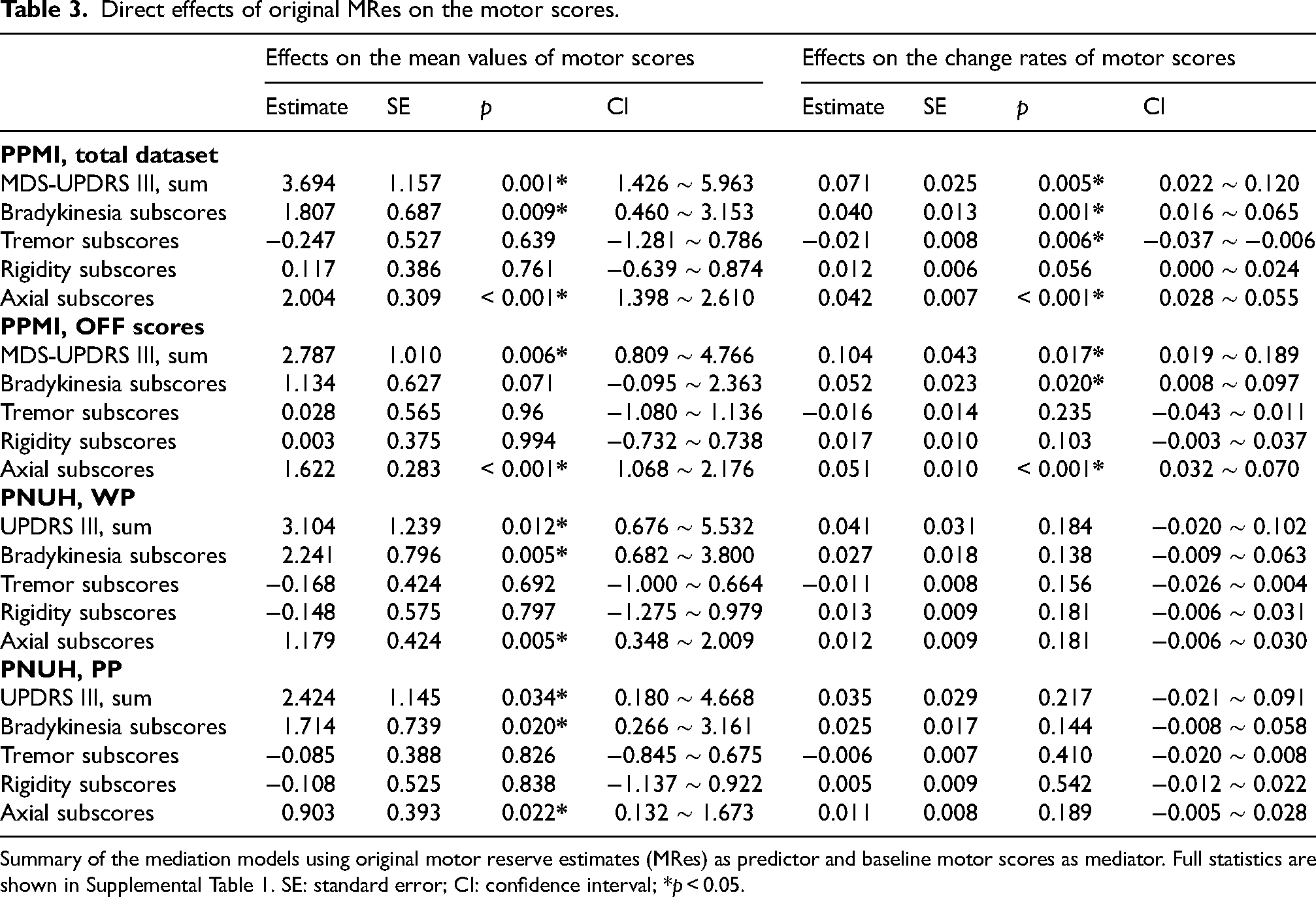
Summary of the mediation models using original motor reserve estimates (MRes) as predictor and baseline motor scores as mediator. Full statistics are shown in Supplemental Table 1. SE: standard error; CI: confidence interval; *p < 0.05.
In the PNUH dataset, the mediation models showed no significant direct effects of original MRes-PP and MRes-PP on the change rates of UPDRS III total scores and subscores. However, the direct effects of original MRes-WP and MRes-PP were associated with greater mean values of UPDRS III total scores and subscores for bradykinesia and axial motor deficits (Table 3 and Supplemental Table 1).
Discussion
The present study assessed the association between MRes, independent of baseline motor severity, and the long-term progression of motor deficits using two independent cohorts. Our modified MRes in the PPMI dataset were associated with a low severity but a rapid deterioration of motor deficits. Although the modified MRes-WP in the PNUH dataset did not show a statistical significance, the MRes-PP were associated with a rapid progression of motor deficits. The modified MRes in both cohorts were associated with a high risk of progression to H&Y stage 3. Mediation models using the original MRes as a predictor showed significant direct effects on the mean values and change rates of MDS-UPDRS III scores in the PPMI dataset. The direct effects of MRes-WP and -PP in the PNUH dataset were associated with a greater mean value of UPDRS-III total scores over the study period. Given that the original MRes-WP and -PP were correlated with low motor scores, these findings may be linked to a rapid increase in motor scores in patients with high original MRes. SPECT and PET scans have different anatomical resolution, 16 thus ROI-based measurements of striatal DAT levels can be varied. However, consistent results in both cohorts strengthen the validity of the findings that high MRes may be associated with an accelerated deterioration of motor dysfunction in PD.
The modified MRes in the present study are independent of motor severity. Therefore, our modified MRes do not intuitively reflect the symptomatic compensation of motor reserve. However, due to the strong correlation between the original MRes and motor severity, a statistical approach using the original MRes may have a circularity issue. It is known that motor severity at an early stage is associated with the long-term motor outcome of PD. 17 Similar to our approach, a mediation model can serve as a statistical alternative; however, a strong negative correlation between a predictor and a mediator may result in a spurious effect, reversing the direction of the direct effect.18,19 In the present study, we employed both types of residual approaches and observed consistent effects of the original and modified MRes on the long-term motor outcomes.
As stated above, a strong inverse correlation between variables may lead to an increase in the indirect effect, potentially converting the direction of the direct effect.18,19 It is possible that the direct effects of original MRes might be influenced by significant indirect effects, however, our findings could also represent a true causal relationship. Assuming two PD patients with identical motor scores but different original MRes, the patient with a high MRes might have a lower striatal DAT level than the other. Studies employing DAT scans have reported associations between low striatal DAT levels and further balance deficits, as well as an increased risk of progression to a higher H&Y stage.20,21 Another analysis of the PPMI dataset found an association between low striatal 123I-FP-CIT availability at baseline and a higher risk of gait freezing. 5 Therefore, it remains possible that motor reserve in PD might be linked to a high risk of balance deficits and further risk of progression in disease stage. Indeed, our modified MRes independent of baseline motor scores also demonstrated the association between motor reserve and the rapid progression of parkinsonian motor deficits.
It is unclear whether the efficiency of resilience mechanisms coping with neurodegenerative process of PD may be maintained along with disease progression. A study of 205 PD patients observed that higher original MRes correlated with slower increases in LEDD, suggesting that motor reserve at early stage may be associated with a slow disease progression. 10 In contrast, another study showed a correlation between original MRes and the local volume of the right anterior putamen, a region linked to a rapid increase in longitudinally measured LEDD. 22 Although the present study is unable to provide a definite conclusion, our results suggest that the efficiency of resilience mechanisms may not be maintained as PD advances. Motor symptoms in PD progress despite a slow decrease in DAT availability in the posterior putamen over the 4 years following diagnosis. 23 Event-related functional MRI (fMRI) studies have suggested that parieto-precentral connectivity in PD patients may serve as a cortical compensation for bradykinesia,24,25 however, activity in the parieto-precentral region on fMRI diminishes with PD progression. 26 Patients with an efficient motor reserve may have better function in early stage but could experience rapid deterioration as the efficiency of resilience mechanism decays. 24 Likewise suggested in Alzheimer's disease, patients with high reserve might have more advanced pathology at the onset of symptoms, shorter duration until they reach point when pathology overwhelms function, and thus a more rapid rate of decline. 27
Our study has several limitations. The validation dataset of the PNUH cohort had a small sample size and did not contain records measured during off-state. Both cohorts had significant differences in several demographic characteristics. Therefore, the association of MRes with motor outcomes in this study requires further validation by studies including an appropriate sample size. However, in our datasets, sex and education periods were not associated with the original and modified MRes, and frequencies of conversion to H&Y stage 3 were comparable between PPMI and PNUH cohorts. A fundamental limitation to the MRes method is that its ability to capture motor reserve is contingent upon the accuracy of the measurement. If the DAT scan or motor examinations do not sufficiently reflect the neurodegenerative process of PD, the resulting MRes become less accurate. 9 DAT availability in the posterior putamen may not reliably indicate the extent of nigrostriatal dopaminergic degeneration when nigral cell loss surpasses 50%, 28 although there are no alternative surrogate markers that can replace DAT scans currently. 10
In summary, PD patients with high MRes had favorable motor scores at baseline; however, they showed a rapid deterioration of motor deficits and a higher risk of progression to H&Y stage 3 compared to those with low MRes. The paradoxical effect of MRes may result from the diminishing efficiency of the resilience mechanism along with disease progression. Further studies are required to investigate the neurobiological underpinning of MRes and figure out a potential intervention to maintain the effectiveness of resilience mechanisms.
Supplemental Material
sj-pdf-1-pkn-10.1177_1877718X251324413 - Supplemental material for Is motor reserve associated with a rapid progression of Parkinson disease?
Supplemental material, sj-pdf-1-pkn-10.1177_1877718X251324413 for Is motor reserve associated with a rapid progression of Parkinson disease? by Myung Jun Lee, Kyoungjune Pak, Jae-Hyeok Lee, Seok Jong Chung and Min Seok Baek in Journal of Parkinson's Disease
Footnotes
Acknowledgments
Parkinson's Progression Markers Initiative (PPMI) - a public-private partnership - is funded by the Michael J. Fox Foundation for Parkinson's Research and funding partners, including AbbVie, Allergan, Amathus Therapeutics, Avid, Biogen, BioLegend, Bristol-Myers Squibb, Celgene, Denali, GE Healthcare, Genentech, GlaxoSmithKline, Golub Capital, Handl Therapeutics, Insitro, Janssen Neuroscience, Lilly, Lundbeck, Merck, MesoScaleDiscovery, Pfizer, Piramal, Prevail Therapeutics, Roche, Sanofi Genzyme, Servier, Takeda, TEVA, UCB, Verily, and Voyager Therapeutics.
Ethical approval
The PPMI study is registered at ClinicalTrials.gov (NCT01141023). Each PPMI site received approval from an ethics committee on human experimentation prior to study initiation. The Institutional Review Board of PNUH also approved the present study for cross-validation (2310-014-132).
Consent to participate
Written informed consent for the research was obtained from all individuals in the PPMI and PNUH cohorts.
Consent for publication
Not applicable.
Funding
This research was supported by Biomedical Research Institute Grant (20210274), Pusan National University Hospital.
Declaration of conflicting interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
