Abstract
Background:
An external cue can markedly improve gait initiation in people with Parkinson’s disease (PD) and is often used to overcome freezing of gait (FOG). It is unknown if the effects of external cueing are comparable if the imperative stimulus is triggered by the person receiving the cue (self-triggered) or an external source.
Objective:
Two experiments were conducted to compare the effects of self- versus externally triggered cueing on anticipatory postural adjustments (APAs) during gait initiation in people with PD.
Methods:
In experiment 1, 10 individuals with PD and FOG initiated gait without a cue or in response to a stimulus triggered by the experimenter or by the participant. Experiment 2 compared self- versus externally triggered cueing across three groups: healthy young adults (n = 16), healthy older adults (n = 11), and a group with PD (n = 10).
Results:
Experiment 1: Externally triggered cues significantly increased APA magnitudes compared to uncued stepping, but not when the same cue was self-triggered. Experiment 2: APAs were not significantly improved with a self-triggered cue compared to un-cued stepping in both the PD and healthy older adult groups, but the young adults showed a significant facilitation of APA magnitude.
Conclusion:
The effectiveness of an external cue on gait initiation in people with PD and older adults is critically dependent upon whether the source of the trigger is endogenous (self-produced) or exogenous (externally-generated). These results may explain why cueing interventions that rely upon self-triggering of the stimulus are often ineffective in people with PD.
INTRODUCTION
Impairment in the capacity to initiate movement is a prominent feature of Parkinson’s disease (PD). Initiation impairment is characterized by reduced movement-related cortical potentials [1, 2], prolonged reaction times [3, 4], fractionation of muscle activation [5–7], increased time to generate force [8] and reduced movement speed [9]. Movement initiation is most impaired when the action is self-initiated, that is, when movement is generated endogenously and not in direct response to an external sensory stimulus [9]. Yet, when the same movement is initiated in response to an external stimulus (e.g., auditory tones, surface translation under the foot), the quality of the movement is usually substantially improved [10–12]. In some individuals, the improvements in movement performance when cued by an external sensory stimulus in the off-medication status are equivalent to, or better than the improvements produced by levodopa [10, 11].
A common and dramatic manifestation of impaired movement initiation is the phenomenon of freezing of gait (FOG). Approximately one-half of people with PD will develop FOG [13], which is characterized by profound difficulties in the initiation of gait or turning, despite the intention to walk [14, 15]. In healthy adults, gait initiation is preceded and accompanied by anticipatory postural adjustments (APAs) that function to accelerate the body center of mass forward and laterally toward the initial stance limb prior to toe-off. Acceleration of the center of mass is accomplished by a stereotypical sequence of increased loading of the step leg, unloading of the stance leg and a shift of the center of pressure posterior and laterally toward the step foot [16–18]. In people with PD, APAs are often reduced or absent, resulting in disrupted gait initiation and slower initial gait velocity [10, 19]. Hypometric APAs are not a requisite event preceding a FOG episode, since freezing can occur even when weight shifts during self-initiated (uncued) stepping are large [20], nonetheless, interventions that facilitate APA generation may result in a more reliable, stable and faster transition from standing to walking.
External sensory cues have been shown to significantly improve APAs and stepping in people with and without FOG [10, 22]. For this reason, an external sensory cue is frequently used as a method to improve gait initiation, maintain gait, as well as overcome FOG episodes. Yet, when external cueing strategies are translated to the in-home environment, efficacy is often greatly diminished or short-lived [23].
A key requirement of most interventions used to deliver external cues in the in-home setting is that the user has to self-trigger the delivery of the cue (e.g., pressing a button on a cane to activate a laser generated line on the floor). Self-triggering allows the user to control when the external cue is applied when experiencing a FOG episode, otherwise they would have to rely on an external stimulus provided by the caregiver (e.g., a foot to step over) or a device that senses the FOG event. Accordingly, self-triggering of an external cue requires a sequence of actions: self-initiation of the motor action required to deliver the cue, attention to the cue, and initiation of the desired movement (e.g., gait initiation). As described above, self-initiation of action is significantly impaired in people with PD, thus deficits in the initial action (delivery of the cue) may compromise performance of subsequent actions (i.e., attention to cue and initiation of response). Given the overwhelming evidence that external cues can significantly improve movement initiation in people with PD, an important and, as of yet, unanswered question is, can the benefits of cueing be retained when the person initiating the action to deliver the cue must also respond to the sensory stimulus generated by the cue?
This paper describes two experiments designed to test if the APAs of gait initiation in people with PD can be improved with a self-triggered external cue. The first experiment tested the hypothesis that the capacity to initiate gait would be significantly impaired when the cue was self-triggered, compared with an external trigger, in people with PD and FOG. Based on the findings of the first experiment, a second experiment was designed to test the hypothesis that the effectiveness of a self-triggered cue for gait intiation is significantly reduced in people with PD compared with healthy young and older adults.
METHODS
Experiment 1 participants
Ten participants with PD and freezing of gait symptoms were recruited for this experiment (Table 1). The study was performed at the University of Illinois Urbana-Champaign (UIUC) and the University of Minnesota (UMN). Institutional Review Board approvals were obtained at both institutions and all participants signed informed consent forms for the study. Participants included in the study were diagnosed with idiopathic PD, 45 + years of age, Hoehn & Yahr rating scale of 2.5–4, and had a history of start hesitation and freezing episodes based upon the Freezing of Gait Questionnaire (FOG-Q) [24]. A person was classified as having FOG if they reported at least one weekly FOG episode (score of > 1 on item 3 of the FOG-Q) and there was confirmation of freezing based on report from the referring neurologist or observation during the laboratory visit. Furthermore, the participants had to be capable of ambulation without an assistive device (e.g., cane, walker) in the off-medicated state, be free of other neurological disorders, and had no musculoskeletal disorders that affected movement of lower limbs. Participants were excluded if they had clinically significant reductions in vision (when corrected), hearing, or cutaneous sensation to the feet, a history dementia or cognitive impairment (Mini-Mental Score < 26 or Montreal Cognitive Assessment < 22), a high level of tremor when off-medication (> 2 on questions 20 and 21 of the UPDRS), or had a history of neurosurgery to treat PD (e.g., deep brain stimulation). Testing was conducted in the OFF-medication state following overnight (12 hour) withdrawal from PD mediations.
Summary of participant demographics for Experiments 1 and 2
PD, Parkinson’s disease; H&Y, Hoehn & Yahr Staging; HYA, healthy young adults; HOA, healthy older adults; nFOG-Q, new freezing of gait questionnaire; F, female; M, male; UPDRS III, Unified Parkinson’s Disease Rating Scale Section III (motor) (*UPDRS III total score, **MDS-UPDRS III total score; conducted off parkinsonian medications), ∧data were unavailable for 2 participants, +FOG-Q was used for two participants (scores were 9 and 15); N/A, not applicable.
Experiment 2 participants
Twenty-six individuals participated in Experiment 2 : 16 healthy young adults, 11 older healthy adults and 10 older adults with PD (6 had FOG) (Table 1). Each participant performed six gait initiation trials across five conditions with their preferred stepping leg. The inclusion/exclusion criteria for the participants with PD were the same as Experiment 1. The young participants were included if their age was between 21 and 45 years and they had no history of musculoskeletal or neurological disorders affecting the lower limbs. Older adults were included who were age-matched to the group with PD and had no history of significant musculoskeletal or neurological disorders affecting the lower limbs. Testing in the PD subjects was conducted in the OFF medication state (same withdrawal protocol as Experiment 1). All data were collected at the University of Minnesota. Procedures were approved by the Institutional Review Board and informed consent was obtained prior to participation in the study.
Sensory cues and cue presentation
Two types of sensory cueing modalities were tested: an acoustic stimulus (Experiments 1 and 2) and a mechanical assistance cue delivered to the right ankle (Experiment 1 only). The acoustic cue consisted of a tone (80 dB, 1 kHz tone, 500 ms) and the mechanical assistance cue was provided by a portable powered ankle-foot orthosis (PPAFO) [25, 26]. The PPAFO is a capable of providing a sequence of dorsiflexor and plantarflexor torques about the ankle through a bi-directional rotary pneumatic actuator. The sequence of dorsi-plantar flexors torques were designed to facilitate (but not replace) the muscle activation patterns generated during the APA in healthy adults [12]. The dorsiflexor torque (330 ms in duration) was tuned to hold the suspended participant’s foot in neutral position relative to the shank (∼3–5 Nm at 30–50 psig). The plantarflexor torque (83 ms in duration) was set to be 9–10 Nm based on 90 psig air pressure. The PPAFO was fit to the participant and worn on the right limb. The participant’s personal walking shoe was worn on the left foot.
Experiment 1 protocol
Four cueing modality conditions were tested: 1) self-initiated [No-Cue] trials during which the subjects wore in the passive PPAFO in an unpowered mode, 2) an acoustic go-cue with PPAFO in passive unpowered mode to assess the effect of an acoustic cue alone [Acoustic-Passive], 3) mechanical assistance from the PPAFO alone to assess its efficacy as a standalone cue [Assist], and 4) an acoustic go-cue with simultaneous mechanical assistance from the PPAFO to assess the combined effects of both cue modalities [Acoustic-Assist]. Each cueing modality condition was performed using two different strategies for triggering the imperative “go” cue: 1) triggering was self-initiated by the participant by pressing a hand-held button [Self-Triggered], and 2) triggering was initiated externally using an experimenter-controlled output [Externally-Triggered]. During the Self-Triggered trials, a “ready” cue (80 dB, 500 Hz tone, 500 ms) was presented to let the participant know that the trials had begun. They were then instructed to “Wait approximately 2–3 seconds after the ready cue and press the switch in your hand to receive a cue.” During the Self-Triggered No-Cue trials the participant still pressed the switch, but no cue was delivered. This provided a control for the act of pressing the button while stepping across all conditions. For Externally-Triggered trials, an instructed-delay paradigm was used consisting of an initial acoustic warning cue (80 dB, 500 Hz tone, 500 ms) presented 2.5 seconds before the go cue [22, 26]. For all conditions, participants were asked to take two steps forward “as quickly as possible” starting with the right foot for all trials. Blocks of five trials were performed for each combination of trigger modality (self- vs. externally-triggered) and cueing (No-Cue, Acoustic, Assist, Acoustic-Assist) condition (total of 40 trials per participant). Trials with a freezing episode were discarded then repeated. Trial order was block randomized for each condition and trigger modality. Customized software (Code Composer v5, Texas Instruments, Dallas, TX) was used to control all cues (ready cue, acoustic go-cue, and actuation of the PPAFO).
Experiment 2 protocol
Five cueing conditions were tested (Fig. 1). Condition 1 (no warning cue, no go-cue; NWNC); no warning cue was provided and subjects were asked to wait 3 to 5 seconds after hearing the verbal instruction “anytime” before initiating gait. Condition 2 (external warning cue, no go-cue; EWNC); the acoustic warning cue was provided (80 dB, 500 Hz tone, 500 ms) and subjects were instructed to wait 3 to 5 seconds before initiating gait. Condition 3 (external warning cue, external go-cue; EWEC); the acoustic warning cue was provided, followed by a 2.5 second delay prior to the onset of an externally triggered acoustic go-cue. Condition 4 (external warning cue, self-triggered go-cue; EWSC); the acoustic warning cue was provided and subjects were instructed to wait 2 to 3 seconds before self-triggering (via button press) the external cue to initiate gait. Condition 5 (external warning cue, self-triggered sham go-cue; EWSCs); an acoustic warning cue was provided and subjects were instructed to wait 2 to 3 seconds before pressing the button, however, in this condition, the button press did not trigger the go cue. Subjects were instructed to initiate gait when they pressed the button. This latter condition controlled for the potential use of the button press as an internal cue. The initial step was performed with the individual’s preferred leg in all conditions.
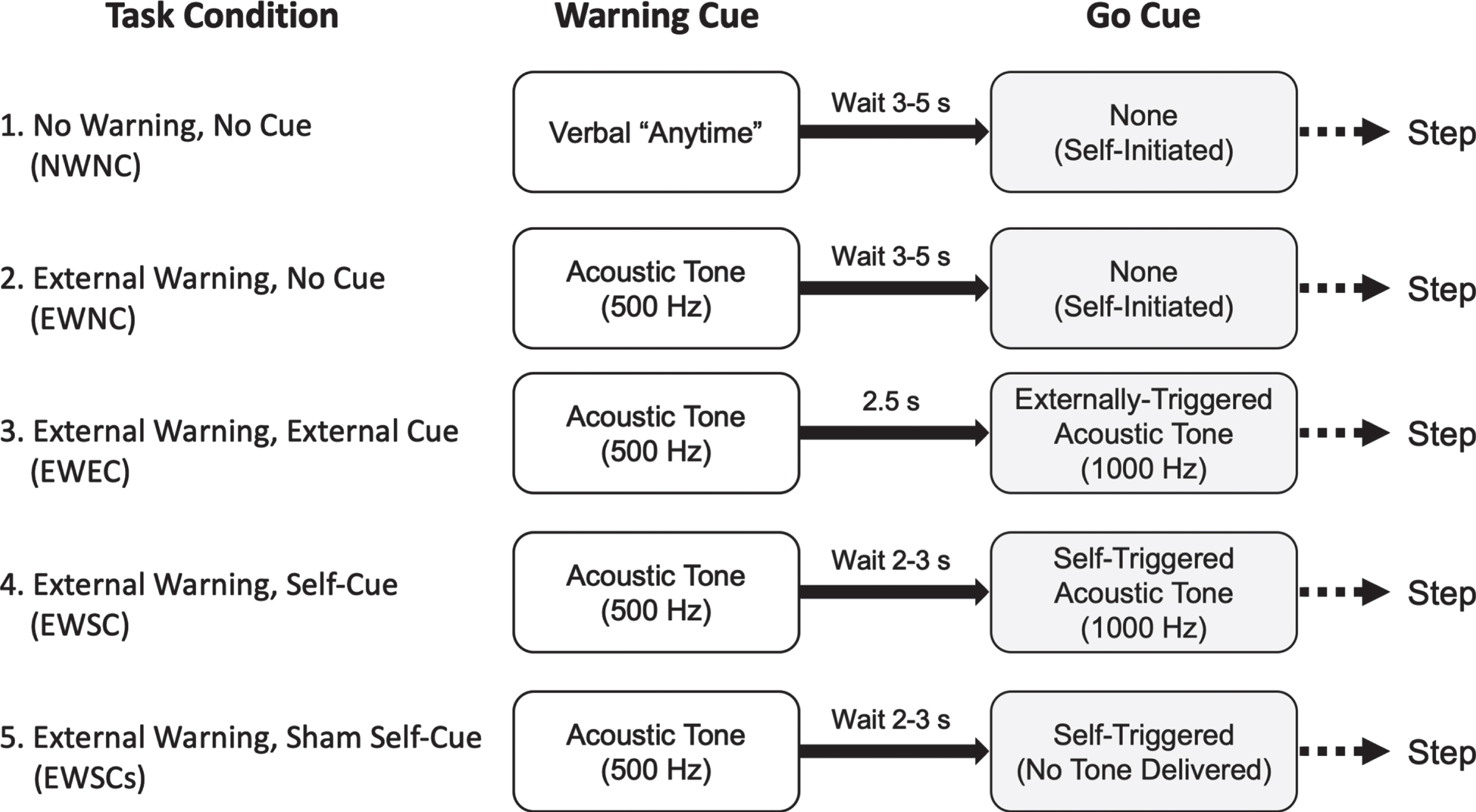
Task conditions for Experiment 2.
Data collection
Participants stood with each foot on separate force plates embedded in an instrumented treadmill at UIUC (Bertec Corporation, Columbus, OH) or a slightly raised walkway at UMN (Kistler Instrument Corporation, Novi, MI). Ground reaction force and center of pressure (CoP) data were sampled at 1000 Hz. Force data were filtered using a low-pass Butterworth filter with a cut-off frequency of 20 Hz. The net total body CoP under both feet in the medial-lateral (ML) and anterior-posterior (AP) directions were derived from the ground reaction force (GRF) and moment data obtained from the force platforms.
Data analysis
Eleven APA magnitude and timing parameters were derived from the vertical GRF and AP-CoP and ML-CoP data for each test condition (Fig. 2). Timings and magnitudes of vertical GRF (both stance and stepping leg) as well as ML-CoP were quantified (GRFstepPk, tGRFstepPk, GRFstancePk, tGRFstancePk, COPML-Pk, and tCOPML-Pk). Vertical GRF was normalized as a percentage of the participant’s body weight. Two posterior CoP excursion peaks were marked; the first peak occurring near the time of peak vertical GRF “loading-unloading” (CoPAP-Pk1, tCoPAP-Pk1) and the second peak occurring near toe-off (CoPAP-Pk2, tCoPAP-Pk2). The time from APA onset to toe-off of the stepping foot (ttoe-off) was also analyzed. Toe-off time was defined as the time when the stepping foot vertical GRF (normalized by body weight) went below 0.1 % BW. The primary outcomes variables used to test the hypotheses were the GRFstepPk, GRFstancePk, COPML-Pk, and CoPAP-Pk1, reflecting the capacity to load and unload the step and stance legs, and shift pressure lateral and backward to the step foot respectively. Trials with multiple APAs and no APAs were included in the analyses. For multiple APAs, the onset of the first APA was marked as the start of the movement and the peak magnitudes of the largest APA generated were marked. For trials with no APAs, the peak APA magnitudes were the same as baseline and timing data was recorded as “NaN, not a number”.
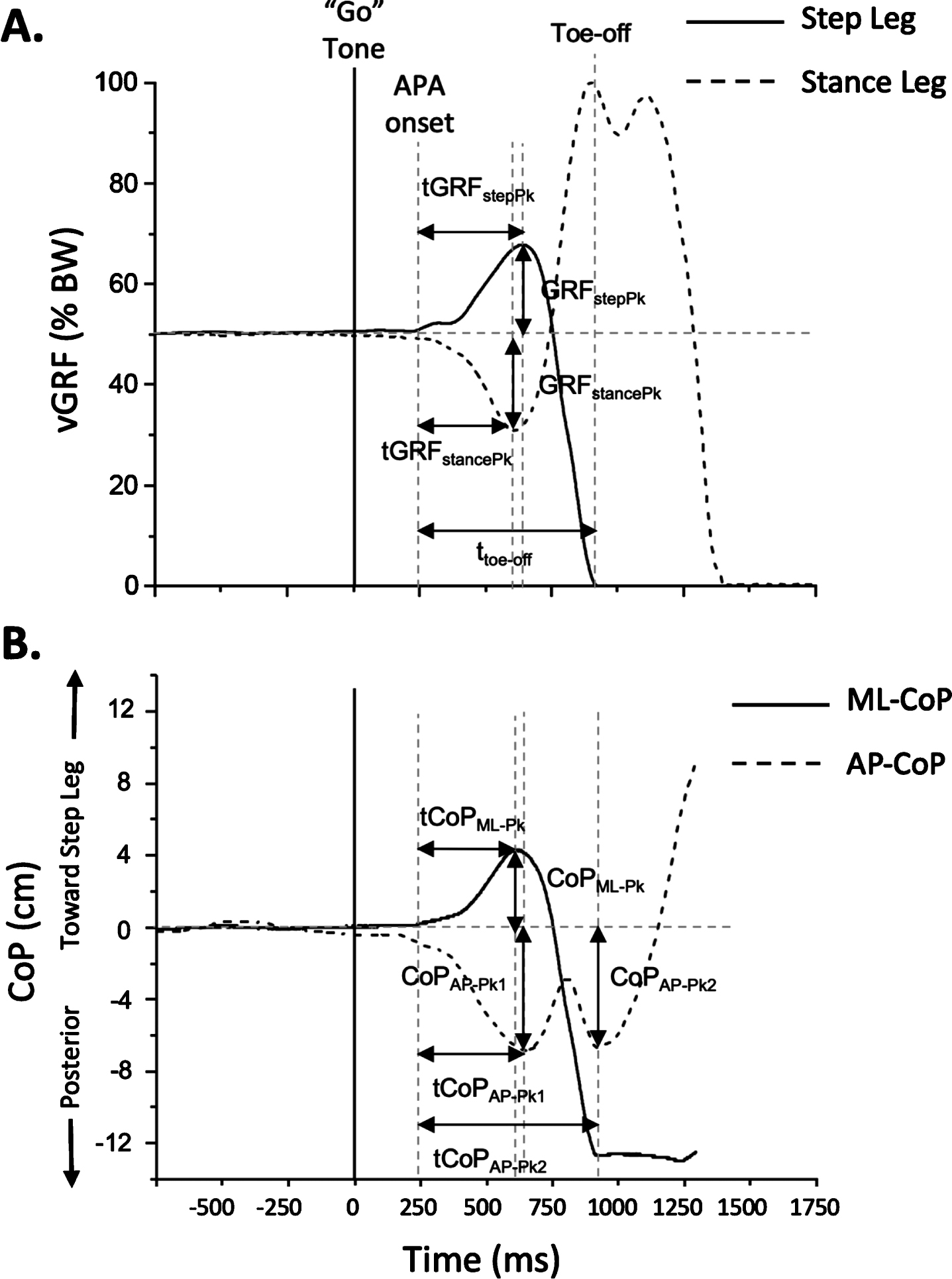
Representative (A) vertical ground reaction force (vGRF) under each foot and (B) the total body’s center of pressure (CoP) profiles during the anticipatory postural adjustment (APA) phase of gait initiation in a healthy adult. The “go” cue is shown by the large vertical line at 0 ms. Eleven APA parameters were quantified including the peak amplitude and timing of the vertical ground reaction force (GRFstepPk, tGRPstepPk, GRFstancePk, tGRFstancePk), lateral excursion of the center of pressure (COPML-Pk, tCOPML-Pk), first and second posterior excursion of the center of pressure (COPAP-Pk1, tCOPAP-Pk1; COPAP-Pk2, tCOPAP-Pk2), and time of step leg toe-off (ttoe-off). All timing parameters were calculated relative to APA onset.
APA onset was calculated based on a monotonic change of greater than three standard deviations in relation to the baseline signal (mean of 1000 ms prior to the go-cue). For the No-Cue conditions, the mean signal 100–300 ms before vGRF onset was used to calculate the baseline signal. A trial was considered to have “no-APA” behaviors (peak magnitude was set to zero and time was not recorded) if no clear monotonic increase was observed in a signal. Using visual inspection, all parameters were verified by a trained researcher.
Statistical analysis
For Experimental 1, a 2×4 repeated-measures univariate analysis of variance (ANOVAs) was conducted to assess the effect of triggering strategy (self vs. external) and cueing condition (No-Cue, Acoustic, Assist, Acoustic-Assist) across the eleven APA parameters. When the distribution of data violated the Mauchly’s test of sphericity, the Greenhouse-Geisser corrected p-values were used. Post-hoc analyses of significant interaction effects were restricted to comparisons between triggering strategies within a cueing condition (e.g., externally triggered acoustic cue vs. self-triggered acoustic cue) since these are the comparisons that tested the primary hypothesis, and were conducted using Tukey’s Honest Significant Difference (HSD) test with a significance threshold of p < 0.05. Statistical analyses were conducted using SPSS statistical software (Version 20, IBM Corp, Armonk, NY).
For Experiment 2, a 3×5 repeated measures ANOVAs was used to test for differences between groups (HYA, HOA, PD) and across cueing conditions (NWNC, EWNC, EWEC, EWSC, EWSCs). Post-hoc tests of main and interaction effects were conducted using Tukey’s Honest Significant Difference (HSD) test (significance threshold of p < 0.05).
RESULTS
Experiment 1: APA magnitude remained diminished and prolonged when cues were self-triggered for people with PD and FOG
Overall, external triggering was associated with increases in the magnitudes of the step (GRFstepPk) and stance (GRFstancePk) vertical GRF and lateral shift of the body’s CoP (COPML-Pk) toward the stepping leg relative to the No-Cue condition (Fig. 3 and Table 2). In contrast, when the same cue modalities were self-triggered, the APA amplitudes showed little change relative to the No-Cue condition. There were significant main effects of triggering strategy (F > 5.82, df = 1, p < 0.020), cueing condition (F > 21.34, df = 3, p < 0.002) and an interaction effect for GRFstepPk, GRFstancePk, and COPML-Pk (F > 4.68, df = 1,3, p ≤ 0.009). Post-hoc analyses showed that the magnitudes of the GRFstepPk, GRFstancePk and COPML-Pk were significantly larger for external versus self-triggering for the Assist and Acoustic-Assist cueing conditions (p < 0.024). There were significant trigger strategy×cue condition interaction effects for the times to peak APA magnitude and toe off (F > 4.92, df = 1,3, p < 0.008). Post-hoc analyses showed that only the time to peak GRFstancePk and CoPAP-Pk1 were significantly longer (p < 0.028) for the self-triggered versus externally triggered strategy during the Assist cue condition.
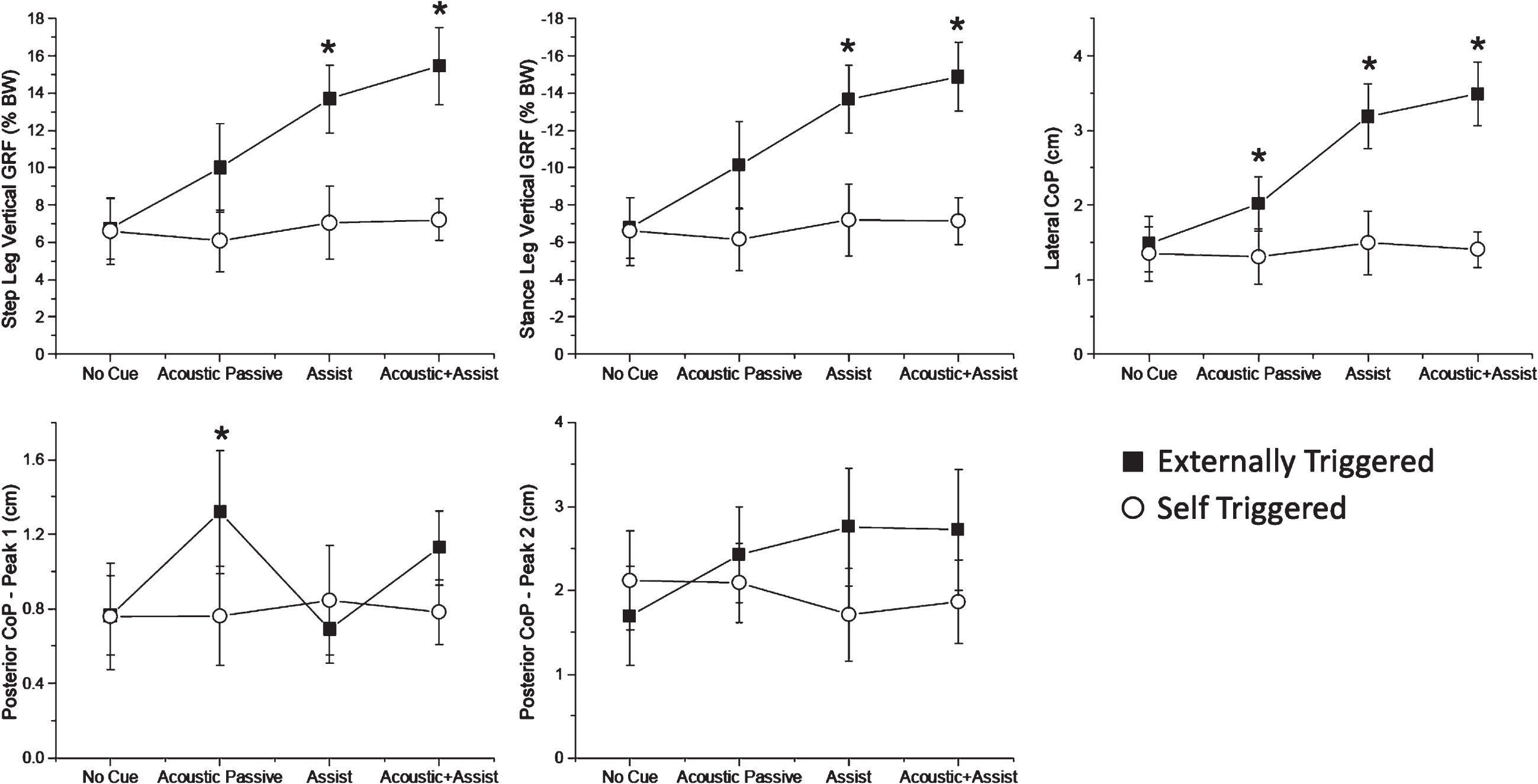
Magnitude results for Experiment 1. Summary of the mean (±1 standard error) peak vertical ground reaction forces (GRF) for step and stance legs and center of pressure excursions (CoP) in the lateral and posterior directions across subjects for the external- (black squares) and self-triggered (white circles) conditions. An asterisk indicates a significant difference between trigger modalities for the given cueing condition (p < 0.05). BW, body weight.
All eleven APA parameters (average±s.e.m) across conditions and trigger modalities (ET – External-Triggering, ST – Self-Triggering) for Experiment 1. P-values of the triggering strategy×cueing condition interaction effect from the repeated measures ANOVA are shown in the far right column (significant values are bolded). Significant differences within conditions (between trigger modalities) are indicated with ‡ (p < 0.05)
GRF, vertical ground reaction force of step leg; COPML, medial-lateral center of pressure; CoPAP, anterior-posterior center of pressure; Pk, peak; tCOPML-Pk, time of peak lateral excursion of ML-CoP; tCoPAP-Pk1, time of first posterior peak of CoP excursion; tCoPAP-Pk2, time of second posterior peak of CoP excursion; ttoe-off, time of step leg toe-off; BW, body weight.
FOG episodes were relatively rare during the data collection and seen in 4 of the 10 individuals tested. The number of FOG episodes was higher during self-triggered (Baseline-Passive = 0, Acoustic-Passive = 3, Assist = 4, Acoustic-Assist = 0) compared to externally-triggered (Baseline-Passive = 0, Acoustic-Passive = 0, Assist = 1, Acoustic-Assist = 2) conditions.
Experiment 2: APA magnitudes remained diminished in healthy older adults and people with PD, but not healthy young adults
The results of Experiment 2 are summarized in Figs. 4–6. Significant main effects of group (F > 102.4, df = 2, p < 0.001) and cueing condition (F > 16.4, df = 4, p < 0.001) were observed for all APA magnitude variables (Fig. 4). The peak loading (GRFstepPk) and unloading (GRFstancePk) of vertical GRFs, COPML-Pk, CoPAP-Pk1, and CoPAP-Pk2 shifts were significantly larger in the young and older adult groups compared with the PD group (p < 0.05). The external warning-external cue (EWEC) condition was associated with significantly larger GRFs and CoP shifts compared with the NWNC and EWNC conditions (p < 0.001).
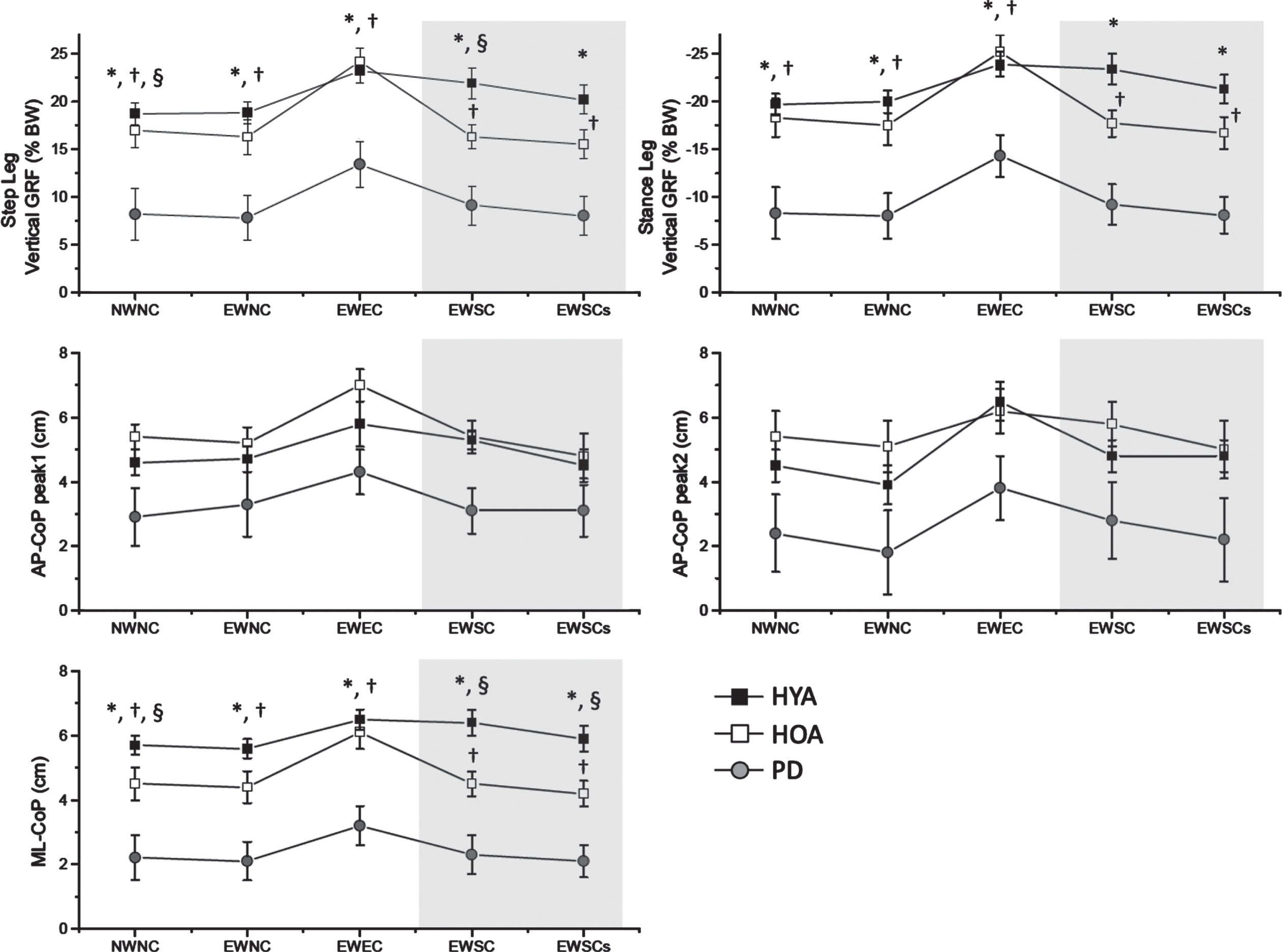
Magnitude results for Experiment 2. Summary of the mean (±1 standard error) peak vertical ground reaction forces and center of pressure excursions for each group across subjects and cueing conditions. The gray zone of each plot highlights the data during the self-triggered conditions. NWNC, no warning cue, no go-cue; EWNC, external warning cue, no go-cue; EWEC, external warning cue, self-triggered go-cue; EWSC, external warning cue, self-triggered go-cue; EWSCs, external warning cue, self-triggered sham go-cue; ML-CoP, medial lateral center of pressure; AP-CoP, anterior-posterior center of pressure; HYA, Healthy Young Adults; HYO, Healthy Older Adults; PD, Parkinson’s disease; BW, body weight. Significant differences (p < 0.05) are indicated by *(HYA > PD), †(HOA > PD), §(HYA > HOA). The gray zone of each plot highlights the data during the self-triggered conditions.
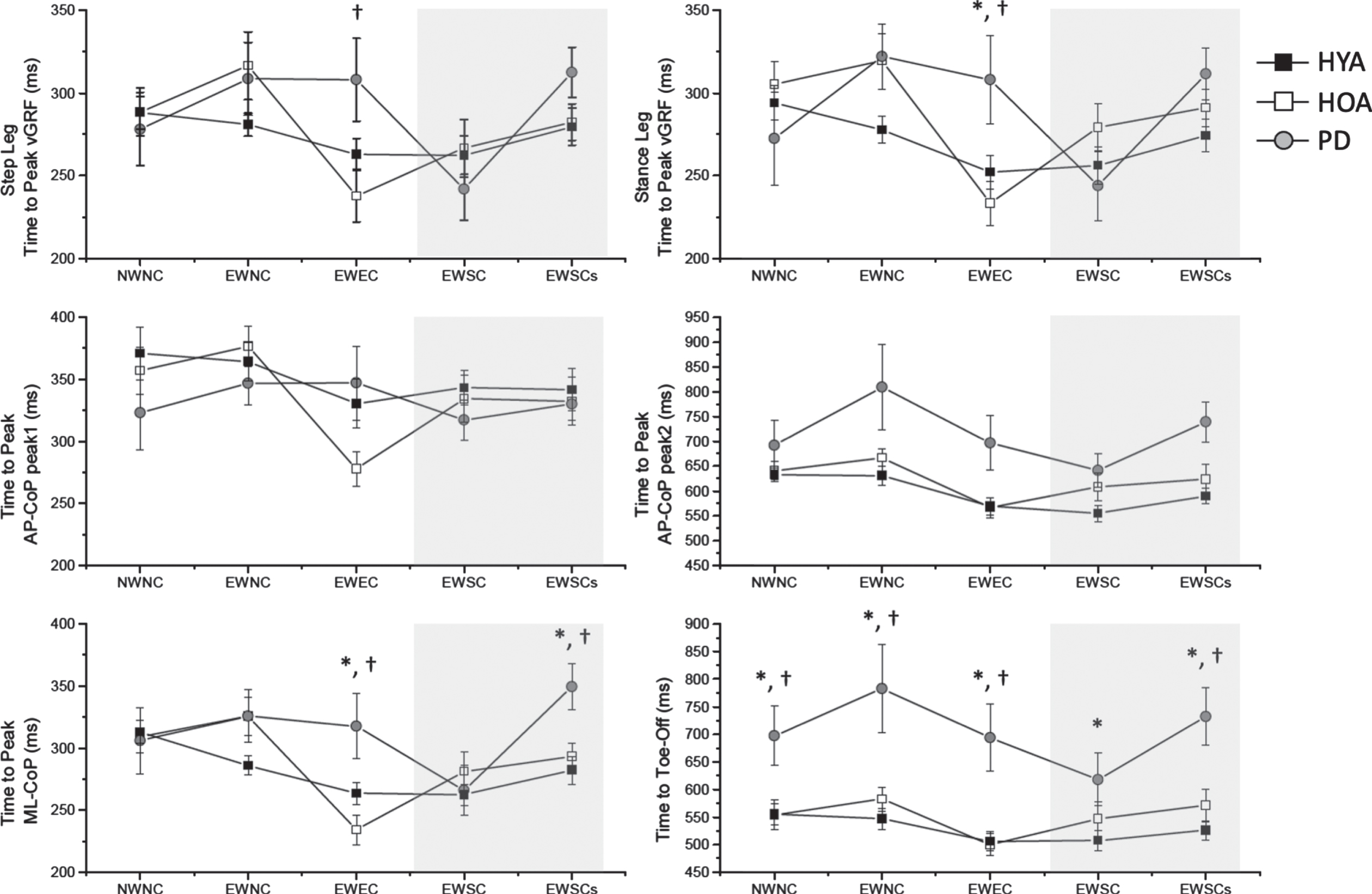
Timing results for Experiment 2. Summary of the mean (±1 standard error) peak timings of the vertical ground reaction forces and center of pressure excursions for each group across subjects and cueing conditions. See prior figure captions for descriptions of terms.
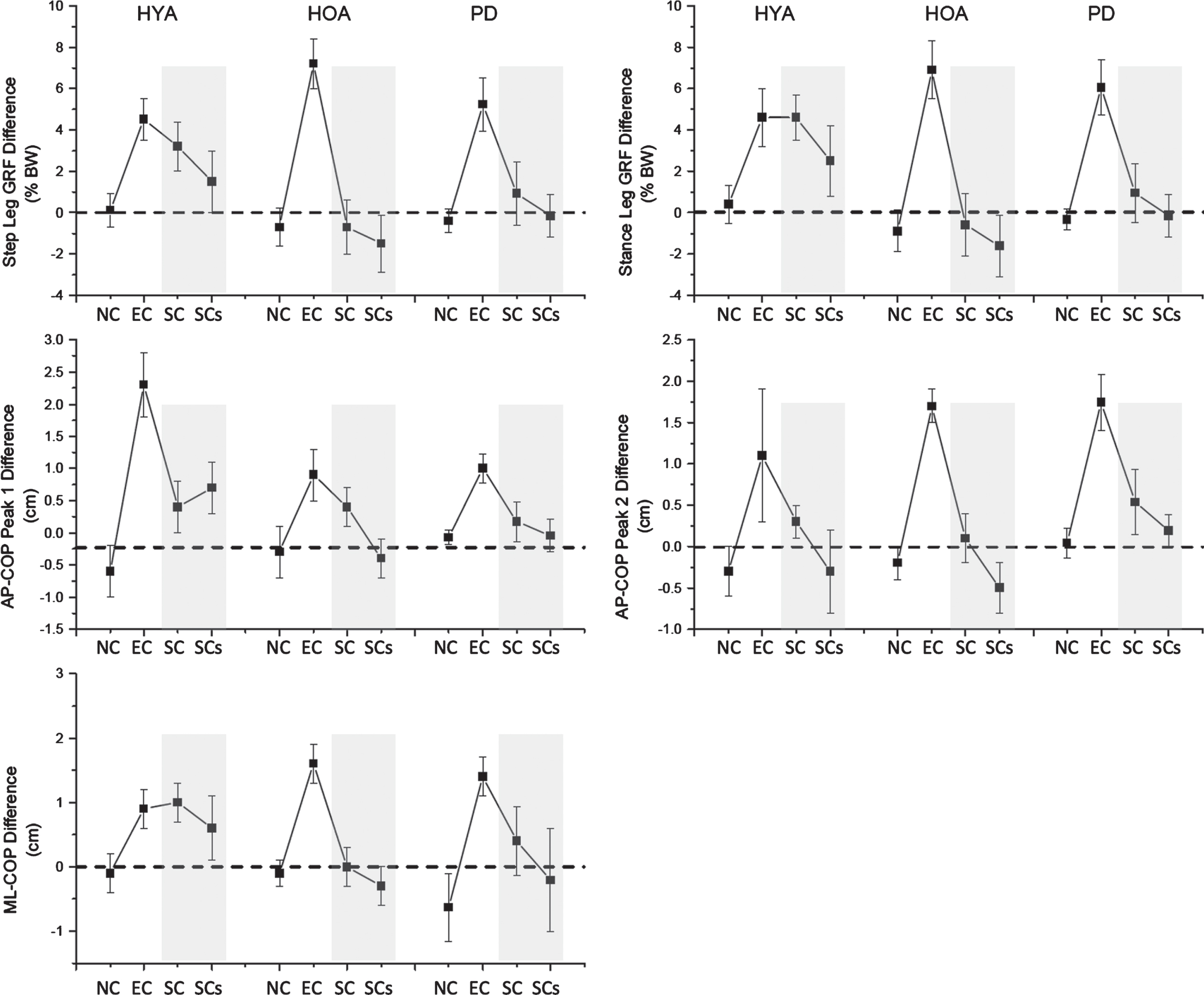
Experiment 2. Summary of the mean (±1 standard error) change in the peak amplitude of the vertical ground reaction forces and center of pressure excursions relative to the no warning cue, no go-cue condition (NWNC) for each group across subjects and cueing conditions. See prior figure captions for descriptions of terms.
Most importantly, there was a significant group×cueing interaction effect (F > 2.2, df = 2,4, p < 0.035) for GRFstepPk, GRFstancePk, and COPML-Pk. Post-hoc analysis found that the HYA group had significantly increased vertical GRF and ML-CoP shifts (GRFstepPk, GRFstancePk, and COPML-Pk) for both the externally triggered (EWEC) and self-triggered (EWSC) conditions compared with the NWNC condition (p < 0.009 for EWEC; p < 0.026 for EWSC). In the PD and HOA groups, the EWEC condition was associated with increases in vertical GRF and ML-CoP shifts (GRFstepPk, GRFstancePk, and COPML-Pk) compared with the NWNC condition (p < 0.003), but there was no significant facilitation of the APAs when the cue was self-triggered (EWSC vs. NWNC) (p > 0.573). While the peak vertical GRF and ML-CoP shifts were comparable in magnitude between the HYA and HOA groups when externally triggered (p > 0.491), significant differences were observed between groups for the self-triggered condition (GRFstepPk, and COPML-Pk, p < 0.015). Self-triggering of the cue had no significant effects on the amplitude of the first or second posterior shifts of the CoP in any group (CoPAP-Pk1, CoPAP-Pk2, p > 0.58).
Experiment 2: APA timings were prolonged in the PD group compared healthy older and younger adults
Along with APA magnitudes, APA timing was also influenced by the cue condition (Fig. 5). There were significant main effects of cueing condition for all APA timing variables. Group effects were observed for the time to the second posterior peak of the CoP (tCoPAP-Pk2) and time to toe-off (ttoe-off, p < 0.007). For both variables, the times were significantly increased in the PD group relative to both the HYA and HOA groups (p < 0.05). Significant group×cueing condition interactions were seen for the time to peak vertical GRFs (tGRFstepPk, tGRFstancePk), peak ML-CoP (tCOPML-Pk) and toe-off (ttoe-off, p < 0.009). The tGRFstepPk, tGRFstancePk, and tCOPML-Pk interactions were due to significantly prolonged times in the PD group compared with both the HYA (with the exception of tGRFstepPk) and HOA group for the EWEC condition (p < 0.032). Step leg toe-off times (ttoe-off) were significantly prolonged in the PD group for all conditions except the EWSC (self-triggered) condition. No freezing events were observed during Experiment 2.
DISCUSSION
There are two main findings from these experiments. First, the ability of cues to facilitate gait initiation was significantly diminished or lost in the participants with PD and FOG when the external sensory stimulus was self-triggered (Experiment 1), and these findings were replicated in a cohort of people with PD with and without FOG (Experiment 2). Second, a diminished self-triggering capacity was also seen in healthy older adults but not the young adults (Experiment 2). The latter finding suggests that factors related to aging may contribute to deficits in self-triggering.
External triggering of a cue facilitates gait initiation
Both experiments recapitulated the findings from many previous studies supporting that the provision of an externally applied sensory cues can significantly improve APAs that precede and accompany gait initiation [10, 22]. Burleigh-Jacobs et al. [10] found that applying electrical stimulation to the earlobe as an imperative stimulus evoked significant improvements in the magnitude of APAs. Moreover, the effectiveness of the cue in the OFF medication state was comparable to the APAs observed in the ON medication state. Similar findings have been reported for acoustic, visual and vibrotactile cues [22]. External cueing has also been shown to improve the peak velocity of upper limb ballistic movements in the OFF medication state to levels comparable to the ON medication state [11]. The results of the first experiment in the present study found that an acoustic tone, mechanical assistance, and combined acoustic mechanical assistance cue, when applied by an external trigger, were associated with average improvements in the vGRF of 64%, 93%, and 115%, respectively. The mechanical assistance provided by the PPAFO [26] served two purposes that likely contributed to improved APAs and stepping. First, the actuator of the PPAFO was designed to facilitate the sequence of dorsi-flexor, then plantar-flexor, torques generated by the anterior and posterior muscles of the lower leg during gait initiation. Thus, the device was designed to facilitate the actions of the ankle musculature due to the reduced force generation capacity in PD [26]. Second, the ankle actuation imposed by the PPAFO induced a proprioceptive and cutaneous stimulus that could act as a cue to initiate movement. The combined facilitation of force about the ankle in conjunction with intersensory facilitation from the acoustic, exteroceptive and proprioceptive inputs [27] likely explain the improvement in APAs produced during the combined Acoustic-Assist condition.
Impaired self-triggering in Parkinson’s disease
The key finding from experiment 1 was the observation that APAs in people with PD and FOG were significantly increased in magnitude when the sensory cue to initiate movement was triggered by an external source (i.e., experimenter), but not when the same stimulus was triggered by the individual receiving the cue (self-triggered). For example, external triggering of the combined acoustic and mechanical cue resulted in an average 2.4-fold increase in the magnitude of the vGRF, yet when the same combined stimulus was self-triggered there was no appreciable increase. These results demonstrated that the effectiveness of an imperative sensory stimulus on the initiation of gait in people with PD is critically dependent upon whether the source of the trigger is endogenous (self-produced) or exogenous (externally generated). These results raised two important follow-up questions. First, is inability to facilitate APAs during gait initiation with a self-triggered cue a phenomenon that is restricted to PD? Second, are the effects of an external cue attenuated when the cue is self-triggered in healthy adults (young and old), or is it a phenomenon associated with aging and not PD alone? The second experiment was designed to address these questions by comparing the efficacy of a self-triggered cue for gait initiation in a group of healthy young adults, healthy older adults, and older adults with PD.
Young adults can self-trigger
The results of experiment 2 demonstrated that self-triggering was ineffective in both the PD and healthy older adult groups, but the healthy young adults retained the capacity to improve gait initiation with self-triggering. The average improvements in the magnitude of the stepping leg vGRFs with externally triggered and self-triggered cueing were 25% and 24%, respectively, relative to the no cue condition in the healthy young adult group. Improvements were also seen in the stance leg vGRF and initial lateral shift of the CoP excursion. The first and second posterior peaks of the CoP were not improved with self-triggering in the HYA group.
Impaired self-triggering in Parkinson’s disease and aging
Self-triggering of an external cue did not significantly improve gait initiation in the participants with PD and the healthy older adults. Yet, in both groups, external triggering of the cue produced significant improvements in APAs, which demonstrates that the capacity to improve gait initiation is retained in both PD and aging. This is consistent with previous work showing that people with PD and matched controls (older adults) retained the capacity to prepare the spatial and temporal features of the appropriate APA well in advance of the imperative “go” cue and that the sequence could be rapidly released when either a startling or non-startling acoustic stimulus was presented [12]. In those experiments, an instructed delay paradigm was used (3.5 seconds between the external warning and go cues) to allow time for participants to prepare the motor plan in advance. A similar experimental paradigm was used in the present experiment and the improvements in the APAs with externally triggered cues were comparable in magnitude to those of Rogers et al. [12] suggesting that subjects may have benefited from early movement planning. This raises the question of whether the lack of significant improvement in gait initiation with self-triggered cues was due to reduced movement planning. It has been shown that movement-related cortical potentials that precede self-initiated movements, including gait initiation, are reduced in people with PD [1, 29]. Reduced preparatory cortical activity preceding self-initiated movements in PD in the OFF medication state has been predominantly ascribed to attenuated activity in the supplementary motor area resulting from disordered output from basal ganglia thalamocortical pathways [1]. Accordingly, the deficits in motor planning and preparation that precede self-initiated gait (NWNC and EWNC conditions) may also translate to the motor actions required to press the button to receive the external cue. Since oral dopamine replacement therapies have been shown to increase movement-related cortical activity [29], one can speculate that impaired self-triggering may be improved in the optimally medicated state.
Dual task deficits in PD and aging
Another factor that may have contributed to deficits in the efficacy of self-triggering in the PD and older adult groups was the dual-task nature of the self-triggering paradigm. The instructions for the EWSC and EWSCs conditions required participants to wait 2 to 3 seconds following the external warning cue, press the button to receive the external cue, and then initiate gait when they heard the tone. Nonetheless the sequence of events, with instruction and execution components close in time (press button and step) likely provided a challenge to those with deficits in dual-task performance. Dual-tasking has been shown to deteriorate the performance of steady state gait and gait initiation in people with PD when in the off medication state, particularly in those with FOG [30–34]. The detrimental effects of increasing cognitive load, such as dual-tasking, on gait and gait initiation in people with PD have been attributed to a putative “posture second strategy” [35]. This hypothesis posits that, due to reduced cognitive resources, cognitive demands are prioritized over resource needed to appropriately maintain posture and locomotion. In the context of the present experiment, the demands of pressing the button to deliver the external cue may have been prioritized over the planning and execution of the first step. Particularly in experiment 1, disruption or delays in the timing between the button press and activation of the PPAFO may have contributed to attenuated APAs. Temporal alignment of the cue with the early phase of the APA with dorsiflexion actuation is necessary to benefit from mechanical assistance [36–38]. Dual-task interference is also a common feature of aging, particularly when the motor and cognitive tasks engage the same circuits [39]. The performance of balance, gait and gait initiation tasks deteriorates during cognitive-motor dual tasking paradigms in healthy older adults compared with younger adults [40]. These changes are associated with age-related alterations in anterior white and gray matter, and putative compensatory changes in prefrontal cortex circuits [39].
Deficits in self-triggering: impaired sensory prediction?
Self-generated actions produce predictable sensory feedback. The integration of sensory feedback with internal forward models of the sensory consequences of movement (efference copy) are proposed to provide the capacity to differentiate between self- and externally-caused events [41, 42]. During self-generated actions, accurate feedforward predictions lead to sensorimotor attenuation such that that sensory consequences of the action are perceived to be weaker compared to when the same sensory input is provided by an external source. The best example of this phenomenon is the marked attenuation of cutaneous sensations when a person tries to tickle themselves; i.e., you can’t tickle yourself [43]. Prior to and during the initiation of self-generated movements, when task-related sensory afference is absent, sensory afferent signals are normally attenuated. This reduction in exteroceptive and proprioceptive precision has been proposed as a mechanism that allows higher-level feedforward predictions to drive the movement (e.g., from motor and premotor regions of cortex) [44]. Wolpe et al. have shown that aging is associated with an increased reliance on sensorimotor prediction, as reflected in an increased attenuation of perceived sensations from self-generated versus externally-generated actions [45]. Moreover, aging-related increases in sensorimotor attenuation correlated with reductions in sensory sensitivity, suggesting that there is a shift over time from a reliance on exteroceptive or proprioceptive feedback to the use of internal predictive models. This shift is associated with structural and functional alterations in frontostriatal pathways [45]. In people with PD, increasing disease severity is associated with reduced sensorimotor attenuation, suggesting that the progressive loss of nigrostriatal neurons is associated with deterioration of sensorimotor predictions [46]. Thus, in PD, deficiencies in higher-level sensorimotor predictions, in conjunction with reduced precision and gating of sensory signals, may result in a conflict between action and perception that impairs movement initiation [46]. In the context of the present experiments, deficient prediction of the consequences of the button press, in concert with abnormal gating of the combined exteroceptive (cutaneous input from the hand) and external sensory input may explain the absence of facilitation of gait initiation in the PD group. Unfortunately, precise sensory testing was not conducted on the participants in the study, so the influence of reduced exteroceptive or proprioceptive sensitivity could not be evaluated.
CONCLUSIONS
The results of this study demonstrate that the movement facilitation benefits of an external cue are lost in people with PD and older adults when the imperative cue is delivered by the individual receiving the stimulus (i.e., self-triggered). Yet, younger adults retain the capacity to improve gait initiation with a self-triggered external cue. The deficits in self-triggering in the PD and older adults might be explained by the dual-task demands and increased cognitive load of the self-triggering paradigm or alterations in sensorimotor prediction and afferent feedback. It is important to note that these findings pertain to when the imperative external cue is delivered in conjunction with the timing of the intended step. Future investigations are needed to see if individuals can benefit from external cues when there is a delay between the self-trigger (button press) and initiation of the intended movement.
Footnotes
ACKNOWLEDGMENTS
The authors would like the participants in this study as well as the Human Dynamics and Controls Lab, the Movement Disorders Laboratory, Chiahao Lu, Danielle Berres, Daniel Bergevin-Smith, Hannah Bergeson, Sarah Lund, Rebecca George, and Kristin Seely.
This work was supported by the NSF under Grant No. 0903622 and partial funding from the Center of Compact and Efficient Fluid Power (CCEFP) Grant No: 0540834, NIH grants RO1 NS070264 and P50 NS09857, the University of Illinois at Urbana-Champaign Dissertation Travel Grant, the MnDRIVE Brain Conditions Fellowship, and the Parkinson Study Group and the Parkinson’s Disease Foundation’s Advancing Parkinson’s Treatments Innovations Grant.
CONFLICT OF INTEREST
Elizabeth Hsiao-Wecksler is a co-inventor of a US patent for the PPAFO device. This patent has not been licensed to any company or organization.
Hsiao-Wecksler, E. T., K. A. Shorter, V. Gervasi, D.L. Cook, R. Remmers, G.F. Kogler, W.K. Durfee, “Portable active pneumatically powered ankle-foot orthosis” United States Patent (Pub. No.: US9480618 B2). Filing date: Mar 14, 2012. Publication date: Nov 1, 2016
