Abstract
Background:
The identification of reliable biomarkers in Parkinson’s disease (PD) would provide much needed diagnostic accuracy, a means of monitoring progression, objectively measuring treatment response, and potentially allowing patient stratification within clinical trials. Whilst the assessment of total alpha-synuclein in biofluids has been identified as a promising biomarker, conflicting trends in these levels across patient plasma samples relative to controls has limited its use. Different commercially available assay platforms that have been used to measure alpha-synuclein may contribute to different study outcomes.
Objective:
To compare different platform immunoassays for the measurement of total alpha-synuclein using the same plasma samples from 49 PD patients and 47 controls.
Methods:
Total plasma alpha-synuclein concentrations were assessed using the BioLegend, MesoScale Discovery, and Quanterix platform in plasma samples from PD patients and matched controls.
Results:
A significant increase in total plasma alpha-synuclein was observed in PD patients using the Biolegend (10%), Mesoscale Discovery (13%) and Quanterix (39%) assays. The Mesoscale Discovery and Quanterix assays showed the strongest correlations (
Conclusion:
All three immunoassays were sensitive enough to detect group level differences between PD patients and controls, with the largest effect size observed with the Quanterix assay. These results may help inform assay choices in ongoing clinical trials.
INTRODUCTION
Mutations in the alpha-synuclein protein are a cause of inherited Parkinson’s disease (PD) [1], and alpha-synuclein misfolding and aggregation in brain tissue is considered as the pathological hallmark of disease [2]. Consequently, there is substantial interest in the measurement of alpha-synuclein as a PD biomarker. However, measurement of alpha-synuclein is complicated as it can exist as a monomer, in higher order complexes (e.g., tetramer), in post translationally modified forms (e.g., phosphorylated, nitrosylated), and in aggregated pathogenic forms (oligomers of different sizes and fibrils) [3–6]. While there is merit in detecting different forms of alpha-synuclein, assays to detect the post translationally modified and aggregated forms of alpha-synuclein in human biofluids remain largely in development (e.g., [7–20]), and/or have not yet been cross-validated by multiple research groups.
In contrast, assays to detect total alpha-synuclein are established and cross-validated [21]. Many studies have measured the concentration of total alpha-synuclein in cerebrospinal fluid (CSF), with the general finding that alpha-synuclein levels are reduced in PD patient CSF [22–28], leading to the inference that this results from increased accumulation of alpha-synuclein in PD brain tissue. However, the invasive nature of CSF collection, as well as limited accessibility in clinical settings, particularly in the context of a longitudinal study, has led to a desire for blood-based biomarkers [29]. Indeed, alpha-synuclein is present in plasma [30] and has been reported as higher in PD patient plasma using the newer technology ultrasensitive single molecule array (SIMOA) platform [31]. Longitudinal assessment of total alpha-synuclein also suggests its levels increase over time and it may be useful as a biomarker of disease progression [32]. Furthermore, changes in serum alpha-synuclein have been used to demonstrate efficacy in early clinical trials of alpha-synuclein antibody therapies indicating clinical utility in monitoring disease progression [33]. However, conflicting trends in total alpha-synuclein levels in PD patients relative to controls have been reported [19, 29–34]. At least one possibility for the discrepancies observed across different studies may be the sensitivity and accuracy of the assay platforms that are used to measure alpha-synuclein.
In addition to assay sensitivity, pre-analytical factors, limited age and sex matching and consideration of hemoglobin levels, have been identified as cofounders to the measurement of total alpha-synuclein [29, 38], complicating method comparisons between different cohort studies. Red blood cells, in particular, are a major source of endogenous alpha-synuclein, accounting for more than 99% of alpha-synuclein in whole blood [44]. Any red blood cell lysis during blood collection and/or processing can therefore lead to artificially elevated plasma alpha-synuclein levels. Controlling for potential red blood cell lysis, as assessed by hemoglobin levels, has therefore been suggested, yet not mentioned in most studies [29]. Therefore, the present study aimed to compare the ability of three independent assays of increasing sensitivity, namely the BioLegend sandwich ELISA, the Mesoscale Discovery (MSD) electrochemiluminescence ELISA, and Quanterix single molecule counting (SIMOA) assay respectively, to determine plasma levels of total alpha-synuclein in a common sample set. We hypothesised that increasing assay sensitivity would increase group level differences in total alpha-synuclein between PD and control samples.
MATERIALS AND METHODS
Plasma samples
Plasma aliquots from a prior study conducted by our group to investigate glucocerebrosidase activity in PD patients were used [39]. Participants were recruited with ethical approval from the University of Sydney Human Research Ethics Committee (#2016/363) and with informed consent. Venous blood was collected into 8 ml CPT vacutainers (BD Biosciences) in a non-fasted state, and centrifuged at 1800×g for 20 min at room temperature as previously described [39]. Plasma was then collected, snap-frozen into aliquots and stored at –80°C.
Participant details
PD cases were classified according to clinically established criteria [40], whereas controls were age and sex matched participants with no neurological, psychiatric or immunological conditions, and with no first-degree relatives diagnosed with PD. Demographic and clinical data are shown in Table 1.
Demographic and clinical details. Demographic and clinical details of the participants who donated the plasma samples used in this study. Data are mean±standard error, with the range shown in parentheses. Disease severity was recorded using the Hoehn and Yahr (H&Y) scale. LEDD, L-Dopa equivalent daily dose; Hgb, hemoglobin; NA, not applicable
Assessment of hemoglobin
Plasma hemoglobin (Hgb) levels were determined using a Human Hemoglobin ELISA Kit (Abcam; cat# ab157707) as per manufacturer instructions using a 1:1000 dilution of plasma.
Assessment of total alpha-synuclein using the BioLegend assay
We previously measured total alpha-synuclein levels using the LEGEND MAXtrademark Human
Characteristics of the assays used to measure plasma alpha-synuclein levels. Available details are provided to allow comparison of the different assays used to measure alpha-synuclein
Assessment of total alpha-synuclein using the MSD assay
Total plasma alpha-synuclein was additionally assessed using the U-PLEX Plus Human alpha-synuclein kit (Mesoscale Discovery; Cat. No. K151WKP-2), according to the manufacturer instructions. In brief, MSD GOLDTM Small Spot Streptavidin plates were coated with a biotinylated capture antibody and incubated for 1 h at room temperature (with shaking at 700 rpm). Plates were then washed three times with provided wash buffer, and a detection antibody conjugated with electrochemiluminescence (ECL) label (MSD GOLD SULFO-TAGtrademark) was added. Immediately after, diluted samples (1:8) were added, and the mixture was incubated for 2 h at room temperature with shaking at 700 rpm. Plates were then washed three times, diluted Read Buffer was added, and the plate was read using a MesoQuick Plex SQO 120 (Mesoscale Discovery). An 8-point calibration curve and 3 quality controls, provided in the kit were added to each plate. The calibrator ranged from 0–10,500 pg/mL; concentrations of alpha-synuclein in samples and controls were interpolated from the calibration curve using a 4-parameter logistic curve fit (1/Y2 weighted). All calibration points, samples and quality controls were performed in duplicate. Further details are provided in Table 2.
Assessment of total alpha-synuclein using the Quanterix assay
Total plasma alpha-synuclein was further assessed using the ultrasensitive single molecule array (SIMOA) Human Alpha-Synuclein DISCOVERY KIT (Quanterix; Cat. No. 102233) and Quanterix HD-1 Analyzer (Quanterix). Plasma aliquots were sent to GeneWorks (Adelaide, SA) on dry ice with the Quanterix assay performed on a fee for service basis following the manufacturer instructions. Briefly, anti-alpha-synuclein monoclonal antibody coated paramagnetic beads were incubated with thawed plasma samples (1:10 dilution) and biotin labelled detection antibody to allow the simultaneous binding of plasma alpha-synuclein molecules with antibody coated beads and detection antibody. Following a wash with Discovery Bead Reagent added to Bead Stock, streptavidin beta-galactosidase (SBG) was added and mixed, to allow binding of the detection antibody to the immunocomplex on beads. After a second wash, beads were resuspended in a resorufin beta-D-galactopyranoside (RGP) substrate solution, loaded onto the array, and sealed with oil. In the presence of beta-galactosidase, RGP substrate is hydrolysed into a fluorescent product that provides the signal for measurement. A 7-point calibration curve and two quality control samples were included. The calibration curve ranged from 0–10,000 pg/mL; concentrations of alpha-synuclein in samples and controls were determined from the calibration curve using a cubic curve fit (1/Y2 weighted). All calibrator points, samples and controls were performed in duplicate. Further details are provided in Table 2.
Statistical analysis
Where applicable, concentration values for Hgb and alpha-synuclein were log transformed to achieve a normal distribution as assessed by Shapiro-Wilk test (
RESULTS
Measurement of total alpha-synuclein levels in plasma samples from PD patients and controls using three independent assays
Analysis of the demographic data showed that the PD and control participants were matched for age and sex. There was also no significant difference in the Hgb levels between the groups (Table 1). An independent samples
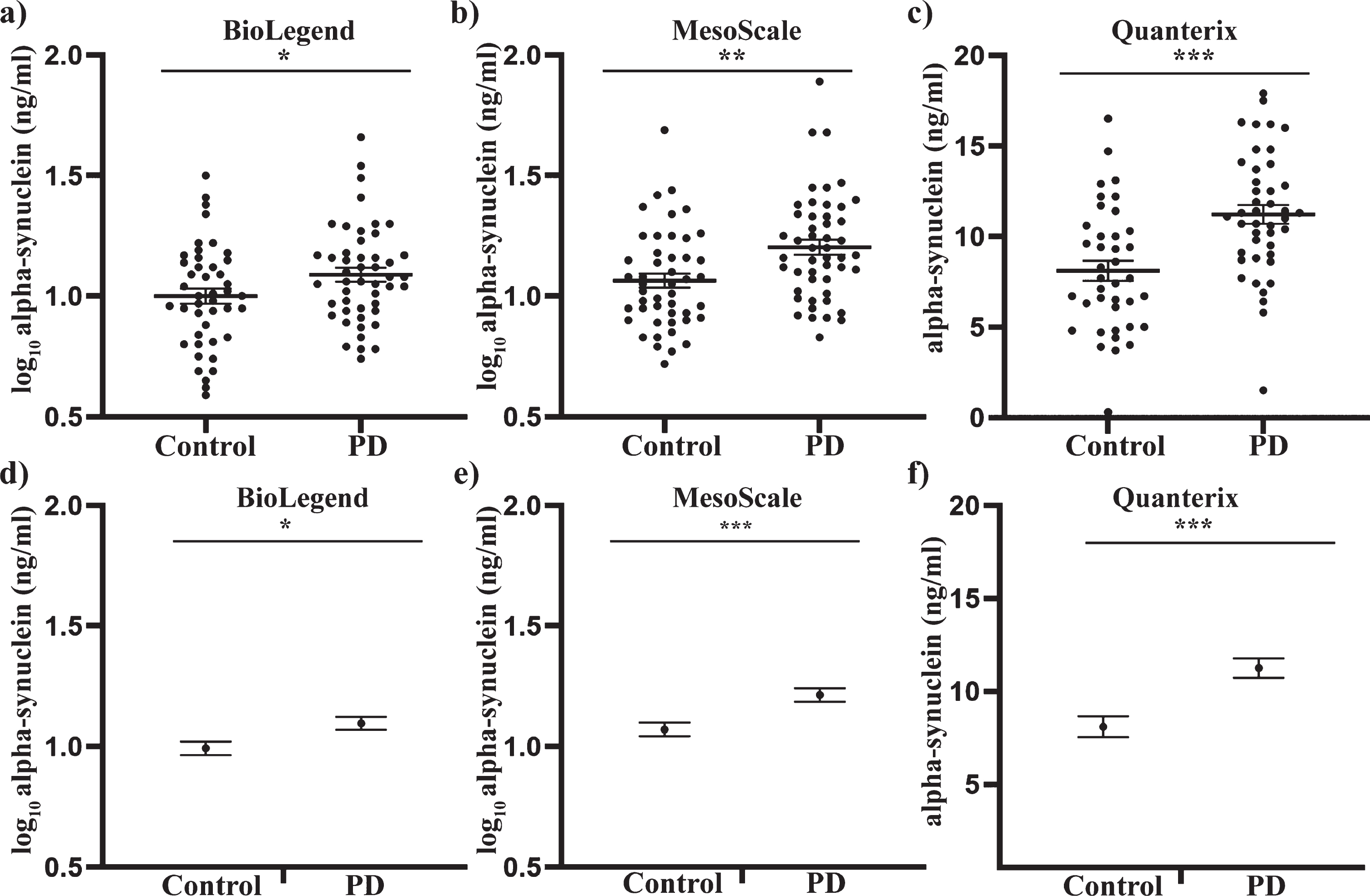
Total alpha-synuclein concentrations in plasma samples of PD patients and healthy controls, using three independent assays. Where applicable, data were transformed to a log scale to achieve normality. An independent sample
Comparison of total alpha-synuclein levels in plasma samples as determined using BioLegend, MSD and Quanterix assay
Scatter plots of alpha-synuclein concentration using the average of the duplicates were used to compare the assays against each other (Fig. 2a-c). Transformed data were used where applicable, with Pearson’s correlation used to determine associations between alpha-synuclein concentrations across all three assays. A significant positive correlation was found between the BioLegend assay and the MSD assay (
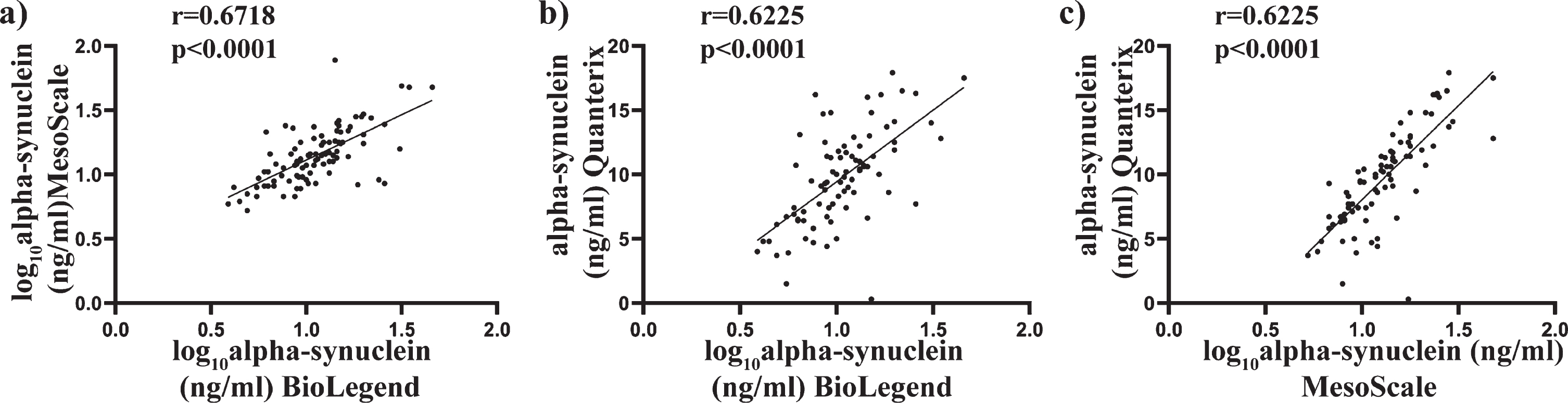
Comparison of total alpha-synuclein levels in plasma samples as determine using BioLegend, MSD and Quanterix assay. Scatter plots showing correlations between total alpha-synuclein (pg/mL) measured in the same patient plasma samples. Where required, data were transformed to achieve normality. Pearson analysis was used to assess significance at the 0.05 level. A significant correlation was observed between the BioLegend and MesoScale assay (a), BioLegend and Quanterix assay (b) and MesoScale and Quanterix assay (c).
Relationship between total alpha-synuclein concentrations and age, Hgb, disease severity, disease duration and L-Dopa equivalent daily dose across three independent assays
Pearson’s correlation was also used to determine any significant associations between alpha-synuclein concentrations and the age and Hgb covariates used across all three assays. No significant correlation with age was observed across all three assays (all
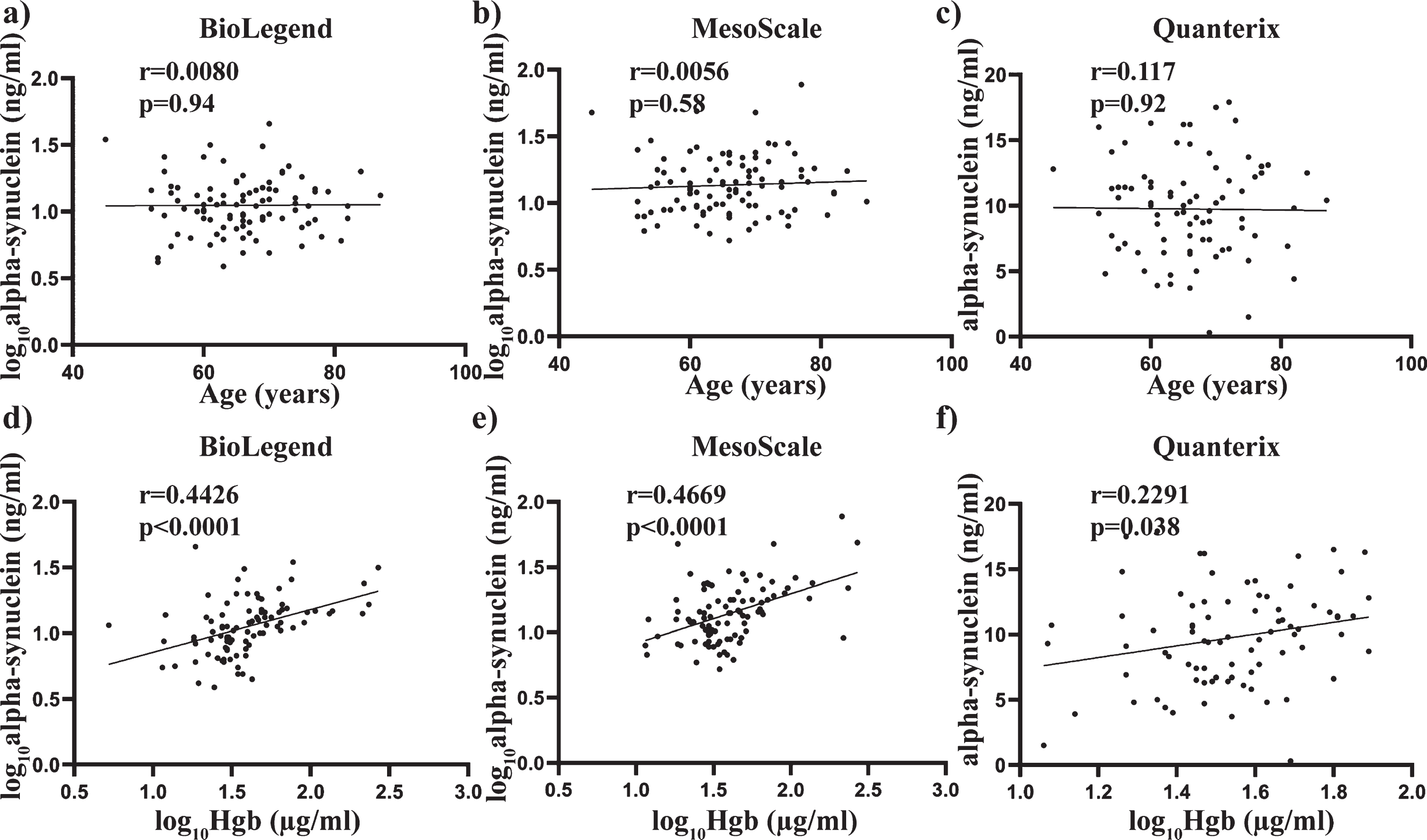
Relationship between total alpha-synuclein concentrations and covariates age and Hgb across all three independent assays. Scatter plots showing correlations between total alpha-synuclein (pg/mL) and covariates age and Hgb (ug/mL). Pearson analysis was used to assess significance at the 0.05 level. No significant correlations were observed between age and total alpha-synuclein measured using the BioLegend (a), MesoScale (b) or Quanterix (c) assay. A significant correlation was observed between plasma levels of hemoglobin and total alpha-synuclein measured using the Biolegend (e), MesoScale (e) and Quanterix (f) assays
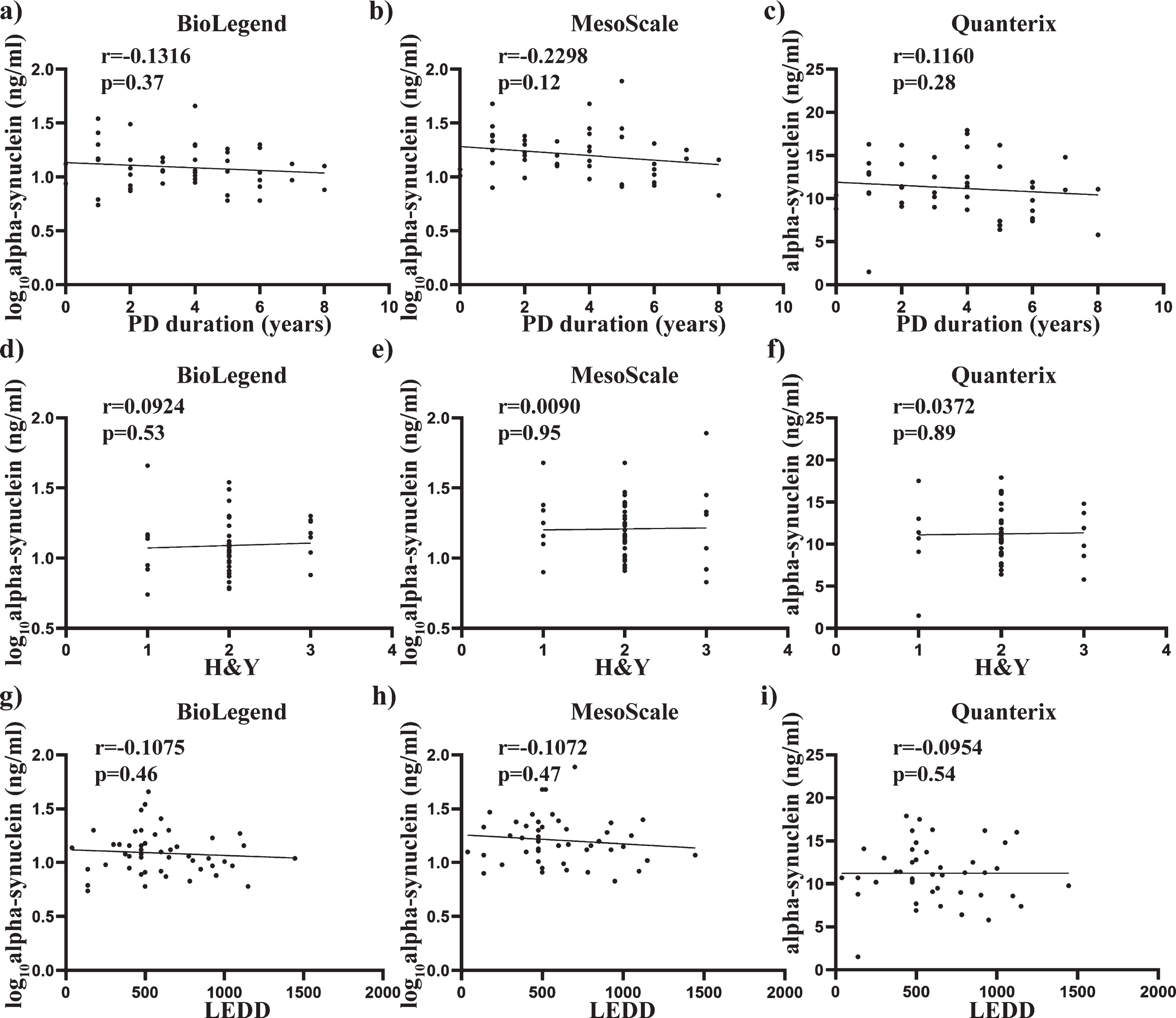
Relationship between total alpha-synuclein concentrations and clinical measures, disease duration and H&Y scores. Scatter plots show correlations between total alpha-synuclein (pg/mL), disease duration (years) and H&Y scores. Spearman analysis was used to assess significance at the 0.05 level. No significant correlations were found between disease duration and total alpha-synuclein measured using BioLegend (a), MesoScale (b) and Quanterix (c) assays. No significant correlations were also found between H&Y scale and total alpha-synuclein measured using BioLegend (d), MesoScale (e) and Quanterix (f) assays. No significant correlations were further found between LEDD and total alpha-synuclein measured using BioLegend (g), MesoScale (h) and Quanterix (i) assays.
DISCUSSION
Alpha-synuclein is a key protein implicated in the pathogenesis of PD and has been described as a promising biomarker candidate. Since its identification in peripheral body fluids, there has been much
interest in the measurement of total alpha-synuclein in plasma samples as a favourable alternative to the collection of CSF in clinical settings. In the present study, we aimed to compare the performance of three independent assays of increasing reported sensitivity for the measurement of total alpha-synuclein in plasma samples: the BioLegend, MSD and Quanterix assays respectively. A common sample set was used to ensure that potential pre-analytical cofounders, including sample collection and processing and storage conditions [38], did not interfere with the study objective. All assays were commercially available and designed with specificity for alpha-synuclein, although the exact extent to which different forms of alpha-synuclein can be detected by the different assay platforms is unknown. Post-translational modification of alpha-synuclein may also affect detection in the different assay platforms. However, none of antibody epitope regions overlapped at least with the serine 129 phosphorylation site, the most common disease-associated post-translational modification site in alpha-synuclein. Included quality controls further indicated that all immunoassays performed as expected with a high degree of positive correlation seen across all the assays for the measured levels of alpha-synuclein. These results suggest that despite using different assay chemistries and antibodies, all of the utilised assays were reliably detecting total alpha-synuclein. Interestingly, the
Interestingly, we also noted improved
Our finding of elevated alpha-synuclein in PD plasma samples is in agreement with the study of Ng et al. [31], who found a 16% increase in PD plasma using the SIMOA platform and controlling for age and sex as covariates. Additional studies using earlier platforms, including colormetric ELISA assays, have also demonstrated increases in total alpha-synuclein in plasma samples of PD patients relative to controls [37, 48], although conflicting studies using similar platforms also exist [42]. In light of our findings, this suggests that consideration of pre-analytical factors, including Hgb levels and cohort demographics may become even more important when platforms of lower sensitivity are being used. It is, however, noteworthy that even with the inclusion of covariates, the effect size of increased alpha-synuclein in PD plasma is relatively small, suggesting that total plasma alpha-synuclein levels may not make a definitive diagnostic biomarker on their own. Indeed, several lines of evidence have suggested that pathological forms of alpha-synuclein, including oligomeric and phosphorylated species may be better diagnostic aids, and demonstrate heightened sensitivity when normalised to total alpha-synuclein [49–52], although these have not yet been cross-validated by multiple groups. The diagnostic potential of total alpha-synuclein in plasma may therefore lie in its ability to normalise the measurement of toxic alpha-synuclein species, or as part of a biomarker panel, rather than as a stand-alone measure. Additionally, the assessment of alpha-synuclein to promote fibrillar aggregation using real-time quaking induced conversion (RT-Quic) and/or protein misfolding cyclic amplification (PMCA) is currently showing diagnostic promise for PD [12, 53], albeit currently limited to CSF. Finally, the molecular basis of why plasma alpha-synuclein is increased in PD patients remains unclear. In this regard, plasma alpha-synuclein has been suggested to result from efflux from CSF [54]. However, there is also support that plasma alpha-synuclein may originate from peripheral tissues such as the enteric plexus which is also pathologically affected in PD [55, 56].
When assessed against clinical measures, no significant findings were identified with either disease duration or H&Y score across all three immunoassays. These findings could be attributed to the very small range of patients within these measures in our study as H&Y score has previously been correlated with plasma alpha-synuclein levels using the SIMOA platform [12]. In particular, an increase in alpha-synuclein levels were observed in milder stages of disease (H&Y 1-2), with a decrease in detectable total alpha-synuclein observed across later disease stages (H&Y 3-4). The authors proposed that this observation might be due to increased c-terminal truncation and aggregation in the later stages, which would escape detection in the current assays targeting the c-terminal of alpha-synuclein [31]. These findings support the longitudinal study by Foulds et al., who reported total alpha-synuclein as a potential marker of disease progression in milder stages of PD (H&Y 1-2) [32]. Increased levels of alpha-synuclein have also been measured in exosomes derived from the plasma of PD patients, and found to correlate with motor progression [57]. In contrast to the limited longitudinal studies using plasma samples, several groups have reported longitudinal assessment of total alpha-synuclein in CSF samples using different PD patient cohorts. From this work, most studies report no significant change in total alpha-synuclein in CSF overtime, suggesting this is not a marker of disease progression [58–61], although conflicting studies again exist suggesting an increase in CSF alpha-synuclein with PD [18, 62], or even a significant decrease [63]. Importantly however, total alpha-synuclein levels in CSF do not appear to correlate with total alpha-synuclein levels in corresponding plasma samples [64], suggesting that conclusions drawn from longitudinal studies using CSF may not directly apply to the ability of plasma alpha-synuclein to serve as a disease progression biomarker for PD. Thus, further longitudinal assessment of plasma alpha-synuclein is of merit, and an obvious limitation in our current cross-sectional work.
To the best of our knowledge, this is the first study to directly compare total plasma alpha-synuclein levels using the BioLegend, MSD and Quanterix assays. While the Quanterix assay was superior in detecting the highest group level difference of total alpha-synuclein PD and control plasma samples, the MSD assay performed similarly and may represent a suitable alternative in discriminating total alpha-synuclein plasma levels in PD patients from controls in settings where the SIMOA platform may not be feasible and/or accessible. Limitations to the current study include that we did not assess inter or intra assay variation to compare assay performance between batches or over time. The study also did not address the specificity of increased alpha-synuclein for PD, and this could be done by comparing to other neurodegenerative diseases such as Alzheimer’s disease. Whilst our study suggests that all three assays were sensitive enough to discriminate PD patients from controls, a larger PD cohort with a wider clinical range and data with a greater range of scores (such as MDS UPDRS motor scores and cognitive assessments) would have expanded the study by enabling the assessment of each immunoassay according to its progressive biomarker potential.
Footnotes
ACKNOWLEDGMENTS
This project received funding from the Australian Government as part of Australian Parkinson’s Mission, a collaboration between the Garvan Institute of Medical Research, The University of Sydney, The Cure Parkinson’s trust, The Michael J Fox Foundation and the Shake It Up Australia Foundation. SJGL is supported by an NHMRC Leadership Fellowship (#1195830). GMH holds a NHMRC senior leadership fellowship (#176607). The Dementia and Movement Disorders Laboratory is supported by ForeFront, a collaborative research group dedicated to the study of non-Alzheimer disease degenerative disorders, funded by NHMRC grants (#1037746, #1095127 and #1132524).
CONFLICTS OF INTEREST
The authors have no conflicts of interest to declare
