Abstract
Background:
Parkinson’s disease (PD) is a complex and debilitating condition that requires care from a multispecialty team. The Integrated Parkinson Care Network (IPCN) is an innovative pragmatic care model that focuses on integrated care, self-management support and technology-enabled care.
Objective:
This study aims to estimate the costs of the IPCN and assess whether benefits gained from the intervention offset its costs based on a single center experience.
Methods:
We conducted a return on investment (ROI) analysis of the IPCN from a societal perspective. The ROI for the IPCN was estimated as a ratio of the net savings and the intervention cost. The intervention cost was calculated as a sum of set-up and implementation costs. Cost savings was measured as the absolute reduction in the societal costs realized by PD patients. A positive ROI indicated that savings generated from the intervention offset its cost.
Results:
The total cost of the IPCN for 100 PD patients was C$135,669, or C$226 per patient per month. IPCN was associated with the reduction in societal cost of C$915 per patient per month (95%CI: –2,782, 951). The ROI per PD patient per month for the IPCN was 3.08 (95%CI: –0.60, 22.93), suggesting that for every C$1 invested in the IPCN, C$4.08 is gained through reduction in societal costs. The returns were greater among advanced PD patients.
Conclusion:
The IPCN has the potential to offer a good return on investment for PD patients, and its value for money is higher among advanced PD patients.
Keywords
INTRODUCTION
Parkinson’s disease (PD) is one of the most common neurogenerative disorder worldwide, with a prevalence of 0.2%in Canada [1]. The prevalence of PD, however, increases with age, ranging between 1%to 2%and 4%to 5%for persons over age 65 and over 85, respectively [2].
PD patients present a variety of motor and non-motor symptoms that adversely affects health-related quality of life. Due to its progressive nature and associated disability, PD poses a significant economic burden for patients and their families, health care systems, and the society. In 2010, the economic burden of PD was approximately $US14 billion in United States and C$181 million in Canada [3, 4].
Due to an aging population, the need for treatments for PD is expected to increase over time. The Integrated Parkinson’s disease Care Network (IPCN) is an innovative pragmatic care network build on the principles of integrated care, self-management support and technology-enabled care. The IPCN is inspired by the Expanded Chronic Care Model [5] structured around four areas of focus: 1) personal skills and self-management support, 2) delivery system design, 3) decision support, and 4) information systems, which are then integrated within the healthcare organization and expanded community linkages as a care network that focuses on personal skills and self-management support, delivery system design, decision support, and information systems.
Understanding the costs and benefits of the IPCN is crucial as these data could help prioritize the allocation of limited health care resources for PD patients and their families. This study aimed to estimate the costs of the IPCN program for PD patients and compare these to benefits accrued from the program.
METHODS
Study design
We conducted a return on investment (ROI) analysis for the IPCN intervention based on a before-and-after clinical study of 100 PD patients who received care at the Parkinson’s disease and Movement Disorders Clinic at The Ottawa Hospital, a Canadian tertiary care center for PD [5].
The study cohort consisted of newly diagnosed PD patients (time since clinical diagnosis < 1 year, n = 25) and advanced PD patients (time since clinical diagnosis > 8 years, or Hoehn and Yahr≥3 in the off state, and not living in a chronic care facility, n = 75) [5].
The clinical study collected information on patient characteristics, direct health resource use, out of pocket expenses, informal care hours, and absence from work due to PD at various time points from the enrollment into the cohort. Data on health resource use, out of pocket expenses, medical costs covered by private insurers, informal care hours, and absence from work as a result of PD in the previous three months was collected at baseline and 6 months from enrollment.
Intervention
The IPCN program consisted of an initial visit with a clinical care integrator (CCI) to define a personal care plan for the participant and provide education about living with PD and healthy living. The CCI enabled streamlined access to health care resources included in the personal care plan. At one month, the CCI reviewed the implementation of the personal care plan and provided further support, if required, over a phone call. At 3 months, an optional in-person visit was offered. At six months, there was a close-out visit to evaluate the degree of completion of the personal care plan. In this study, the set-up phase of the IPCN intervention lasted for 12 months, and the implementation phase for 100 PD patients covered 6 months.
Measures
Program costs
The costs of the IPCN were equal to the sum of set-up and implementation costs. Set-up costs consisted of resources expended before the actual implementation of the program. Implementation costs consisted of resources expended during the actual operation of the program.
Set-up costs covered the costs of infrastructure required for the program, personnel costs for planning, training, creation of database of healthcare resources for PD patients, and setting-up of infrastructure. Implementation costs included personnel costs for program operation, and ongoing costs of office supplies and maintaining infrastructure. Personnel costs during implementation included personnel time for patient consultations, ongoing patient communication, creation of patient care plans, technical support, outreach, and coordination.
Program costs were estimated using a gross costing technique, where costs are calculated as a product of resource use and unit cost. Information on resource use to set-up and implement the IPCN intervention, salary estimates for program personnel were collected from program staff through direct interviews.
Costs
We measured costs and savings from a societal perspective. Costs for each patient were calculated at baseline and at six months from enrollment. The societal cost for each patient was calculated as the sum of health system costs, and costs of medication, medical aids, home care and nursing, productivity losses, informal caregiving, and out of pocket expenses incurred by patients and caregivers as a result of PD. Health system costs for each patient included costs of primary care physician visits, specialist visits, emergency department visits, day surgery visits, inpatient hospitalization, and rehabilitation incurred in the previous three months.
Costs were calculated as a product of resource use and its respective unit cost. Data on health resource use was obtained from the before-and-after study. Unit costs of primary care physician and specialist visits were extracted from the Ontario Schedule of Benefits [6]. Per day costs of inpatient hospitalization, cost of an emergency department visit and a day surgery visit as a result of PD were abstracted from the Ontario Case Costing Initiative (OCCI) [7].
Productivity losses due to absenteeism were calculated using the human capital approach. We obtained the median annual income in Canada for 2020 from Statistics Canada [8]. A median daily wage was estimated by dividing the median annual wage by 249 days. Cost of productivity loss per patient was estimated by multiplying the median daily wage and number of days taken off from work due to PD.
Costs of informal care were estimated as the cost value of time spent by family members or volunteers in assisting patients with activities of daily living as a result of PD. Informal care costs for each patient were calculated as a product of total hours of informal care received as a result of PD and the average hourly caregiver wage in Canada [9]. Cost data are presented in 2020 Canadian Dollars.
Analysis
Cost analysis
We estimated the total cost of the IPCN intervention as the sum of set-up and implementation costs for 100 PD patients. The intervention cost per PD patient was calculated by dividing the total intervention cost by the number of patients enrolled in the IPCN program, i.e. 100 patients. We also calculated the intervention cost per PD patient per month by dividing the intervention cost per patient by the follow up period for a patient enrolled in the study, i.e., 6 months. Key drivers of set-up and implementation costs were also described.
In addition, we classified program costs into fixed and variable costs. A fixed cost is a cost that does not depend on the number of patients enrolled, while a variable cost is associated with each additional patient enrolled in the program.
Return on investment analysis
The return on investment (ROI) for the IPCN program was calculated as follows:
A positive ROI (> 0) indicates that the savings in societal costs incurred by a PD patient as a result of the IPCN is greater than the intervention cost. Savings were estimated by subtracting the mean monthly societal costs per patient at the baseline from the mean monthly societal costs at the end of the study follow-up. We used a non-parametric test (Mann-Whitney U test) to assess the statistical difference in the mean monthly societal cost incurred by a PD patient after enrolment in the IPCN compared to baseline.
Furthermore, we performed a probabilistic sensitivity analysis using the Monte Carlo simulation technique. We varied program costs over±25%of their base-case values and cost savings over their 95%confidence interval (CIs), and repeated the ROI analysis over 1,000 iterations. Of these 1,000 iterations, we estimated the probability that that IPCN would provide a good return on investment (i.e., ROI > 0). We performed a scenario analysis where we excluded patients who incurred high costs at baseline (outliers).
RESULTS
Data for 98 study participants were included in the analysis. Two participants were excluded from the study as they did not enter the IPCN program. Missing data was less than 5%for all variables and was managed using the listwise deletion approach.
The mean age of patients was 67.8 years (SD 8.83), and the majority were male (66%). In our study, 39 patients (40%) had motor fluctuations, 32 patients (33%) had cognitive impairment, 35 patients (36%) had treatment induced dyskinesia and 21 patients (21%) had a history of recurrent falls as a result of PD. 73 patients (75%) had advanced PD, and 25 patients (26%) were newly diagnosed with PD. Table 1 presents the baseline characteristics of PD patients in our study.
Baseline characteristics of PD patients
SD, standard deviation; PD, Parkinson’s disease; HY, Hoehn Yahr.
Program costs
The total cost of the IPCN for 100 patients was C$135,669. The set-up cost for the IPCN intervention was C$49,215 (36%), and the implementation cost for 100 PD patients over a follow-up period of six months was C$ 86,454 (64%). The intervention cost per PD patient was C$1,368 and the intervention cost per PD patient per month was C$226.
The primary cost drivers during the set-up phase were development of the electronic data capture system (56%), followed by personnel time for creation of a database of healthcare resources for PD patients (31%) (Table 2). Costs of personnel time for coordination (32%) and patient consultations (28%) accounted for majority of implementation cost (Table 2). We found that 42%of program costs were incurred upfront, and 58%recurred throughout the follow-up period, i.e. these costs increased with each additional patient enrolled in the program. We predicted that the intervention cost per PD patient would decrease with increased number of patients enrolled in the program and plateau at a program coverage of approximately 500 patients (Fig. 1).
Breakdown of the program costs for IPCN
PD, Parkinson’s disease; IPCN, Integrated Parkinson Care Network; IT, information technology.
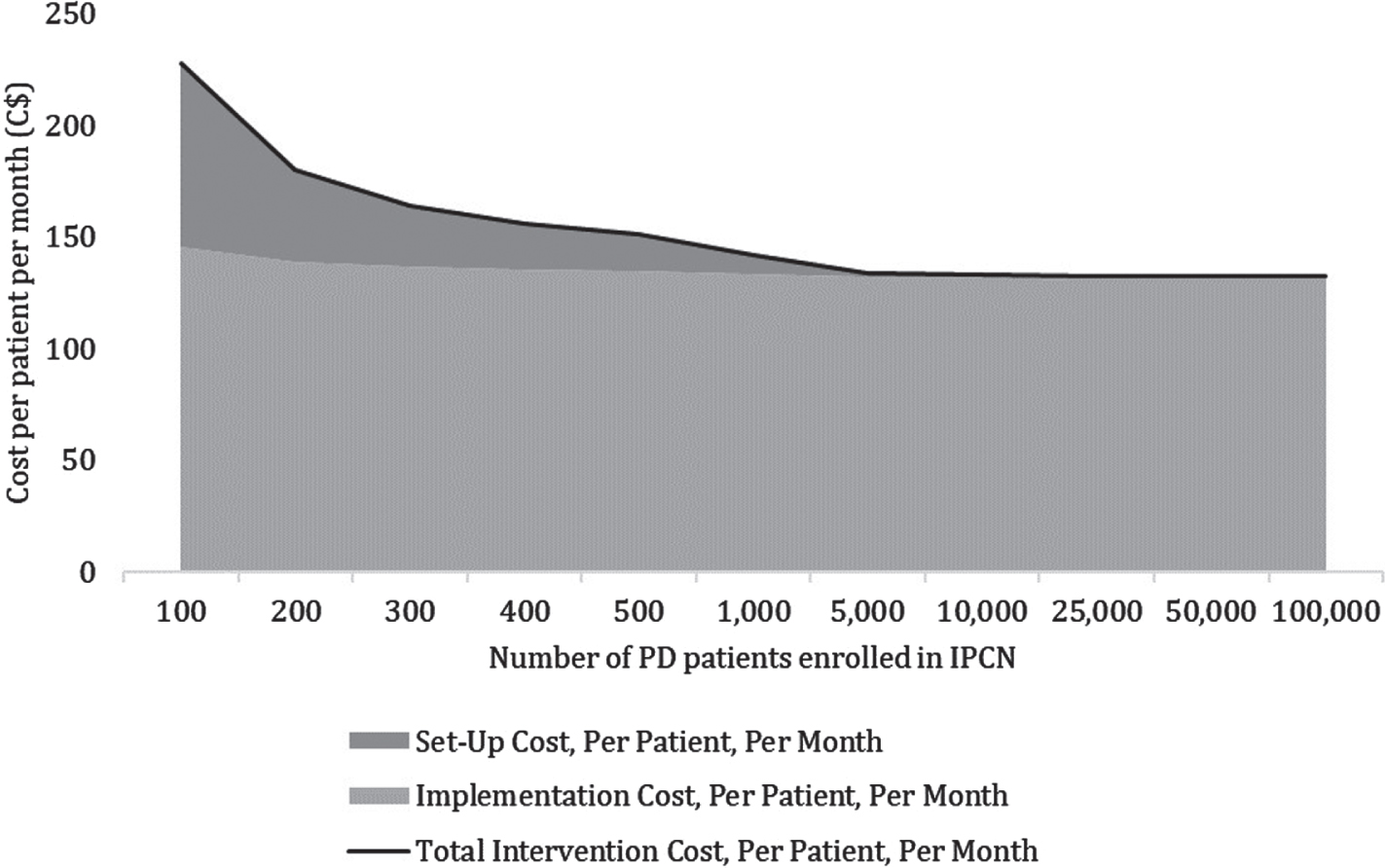
Intervention cost per PD patient per month by program coverage. PD, Parkinson’s disease; IPCN, Integrated Parkinson Care Network.
Outcomes of the IPCN intervention
PD, Parkinson’s disease; IPCN, Integrated Parkinson Care Network; SD, standard deviation; CI, confidence interval.
Societal costs
IPCN was found to be associated with a non-significant reduction in the mean monthly societal costs per PD patient compared to baseline (C$ –915; 95%CI: –2,782, 951; p = 0.721). Reduction in societal costs was largely driven by the reductions in inpatient costs, adaptation costs, and out of pocket costs incurred by PD patients and caregivers. Supplementary Table 1 presents a breakdown of pre- and post-intervention societal costs incurred by PD patients, by service category. Supplementary Tables 2 and 3 present a breakdown of pre- and post-intervention societal costs for each subgroup of patients with advanced and newly diagnosed PD, by service category.
Reduction in monthly societal costs per PD patient as a result of the IPCN was more pronounced for advanced PD patients (C$ –1,277; 95%CI: –3,772, 1,218; p = 0.712). However, we observed a slight increase in the societal costs among newly diagnosed PD patients who received the IPCN (C$ 115; 95%CI: –446, 676; p = 0.992).
Return on investment
The ROI per PD patient per month for the IPCN was 3.08 (95%CI: –0.60, 22.93). The ROI per PD patient per month for the IPCN among advanced PD patients was 4.61 (95%CI: –0.47, 28.60) and that among newly diagnosed PD patients was –1.51 (95%CI: –12.38, –1.00). Results from probabilistic sensitivity analysis showed that the probability of IPCN being a good return on investment (ROI > 0) at a program coverage of 100 PD patients was 75.22%(Fig. 2). Furthermore, the probability of the IPCN being a good return on investment increases if more patients are enrolled to the program.
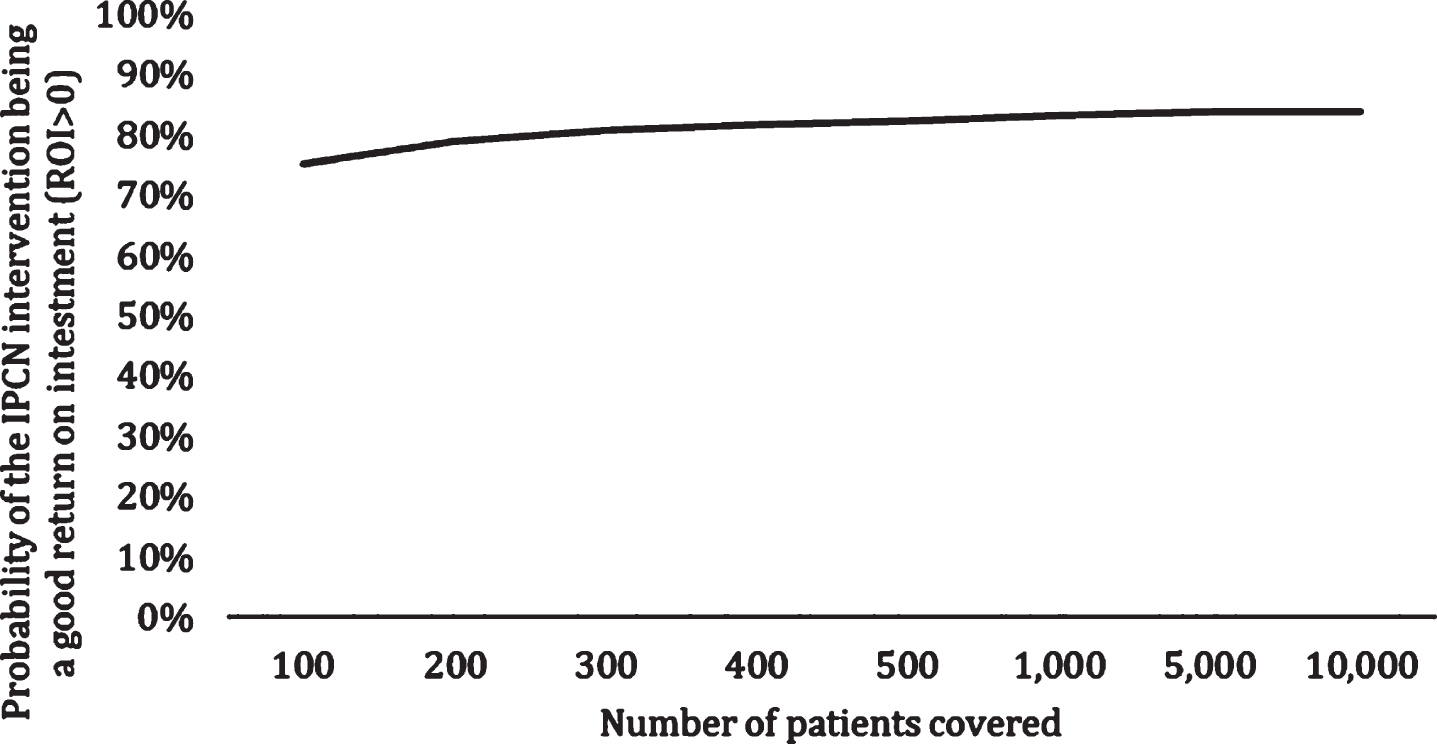
ROI Acceptability Curve by Program Coverage.
A scenario analysis that excluded two patients with high costs of inpatient stay and adaptation due to PD at baseline showed that the ROI per PD patient per month for the IPCN intervention was reduced to –1.02. The ROI per PD patient for advanced PD patients was –0.86 and that for newly diagnosed PD patients was –1.49.
DISCUSSION
Our study shows that the total cost of the IPCN was C$135,669. One-time activities required to set-up the technical infrastructure and create database for PD patients were the key cost drivers. Moreover, a significant proportion (42%) of intervention costs were fixed, suggesting that costs would not increase significantly if the IPCN were to expand to more PD patients. Furthermore, activities such as identification of local health care resources and care coordination would require fewer resources with the recent development of web-based referral platforms such as Caredove, which is an online database of health and community support services with direct referral booking potential.
Our study showed that the IPCN was numerically associated with a reduction in societal costs incurred by PD patients. The reduction in societal costs was driven by a decrease in inpatient costs and out of pocket expenses incurred by patients and caregivers. We observed a reduction in societal costs attributable to the IPCN among advanced PD patients, but not among newly diagnosed PD patients. The ROI per PD patient for the IPCN was 3.08 (95%CI: –0.60, 22.93). This suggested that for every C$1 invested in the IPCN, C$4.08 is gained through reduction in societal costs. A substantial uncertainty in the ROI estimate was partly due to a small sample size of PD patients enrolled in our study. Despite the high uncertainty, our probabilistic sensitivity analysis suggested that the chance that the IPCN program is economically attractive was 75.22%. Results from sub-group analyses indicated that return on investment of the IPCN was larger among advanced PD patients but the program did not offer good value for money for newly diagnosed PD patients. Advanced PD patients likely have established care needs and access to same care resources. The initial evaluation showed an improvement in health-related quality of life after completing the IPCN program in this group of patients [5]. The current results suggest that this is accomplished with a significant return of investment margin and reduction in societal costs. Patients with newly diagnosed PD observed an increase in healthcare costs after enrollment in IPCN. This may indicate improved access to health care resources after enrollment in the IPCN for patients who had been diagnosed with PD in the previous year and were previously healthy. For example, newly diagnosed PD patients may be recommended to participate in exercise programs and physiotherapy as part of early management, access to which the IPCN program may have facilitated.
Findings from our study are in line with previous research. A study by Bloem et al. described costs and savings associated with an integrated care and network-based initiative for the treatment of PD patients in Netherlands called ParkinsonNet [10]. The initiative involves training and building evidence-based guidelines for professionals, informing and engaging PD patients through educational programs, and facilitating multidisciplinary regional networks of allied health professionals to deliver integrated care for PD patients. The set-up cost for ParkinsonNet was US$3.45 million (C$4.8 million) and was driven by personnel costs to set-up a coordination center to support regional networks (27.5%), develop evidence based guidelines (22.5%), and develop technical infrastructure (10%). The per patient implementation cost for ParkinsonNet was based on the potential annual volume of PD patients in Netherlands (41,000 patients) and was estimated to be US$29 (C$41). Implementation costs for ParkinsonNet were primarily driven by personnel costs for coordination and support (23%), providing trainings (23%) and maintenance of technical infrastructure (15%). The study reported cost savings associated with ParkinsonNet between US$439–US$1,613 (C$621–C$2,280) per PD patient. Cost savings attributable to IPCN estimated in our study were within the range of that reported for ParkinsonNet. Costs of developing technical infrastructure accounted for a significant proportion of set-up costs for both interventions. At a similar program coverage of 41,000 PD patients, the per patient implementation cost for IPCN would be C$131.
Our study has certain limitations that must be acknowledged. Firstly, direct and in-direct health resource utilization was based on self-reported data and may be subject to recall bias. We attempted to minimize this bias by reducing the recall period to three months. Secondly, data on direct health resource use in the study was collected for all inpatient and outpatient visits in the previous three months. Therefore, it was difficult to ascertain the proportion of resource utilization that was attributable to PD. However, the use of before-and-after design can mitigate the impact of factors unrelated to PD that could have caused the observed changes in health care utilization. Lastly, a small sample size and the lack of control group in our clinical study made it difficult to attribute the observed changes in the cost and ROI estimates to IPCN. Future experimental studies are required to show the cause-and-effect relationship of the IPCN and outcomes.
Despite these limitations, our study is one of the few existing studies that provide a comprehensive description of costs of a pragmatic integrated care model for PD patients and provide information on the potential return on investment for such a program. A recent published study [5] showed that IPCN was associated with a numerical improvement in the patient-reported outcomes (Parkinson’s Disease Questionnaire–8: 2.7; 95%CI: 0.4, 5.0). This may suggest that the IPCN could potentially be a cost-effective option; however, a full economic evaluation is needed to assess whether the attributable clinical and patient benefits of the IPCN justify its costs. Findings from our study could help inform the decision to adopt or expand such an initiative as the IPCN for PD patients in Canada and its dissemination in Canada and other countries.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
