Abstract
Background:
REM-sleep behaviour disorder (RBD) is a parasomnia and a common comorbidity in Parkinson’s disease (PD). There is evidence that the presence of RBD is associated with more severe PD. The differences in the clinical manifestations and the natural history are likely to imply underlying differences in the pathophysiology among PD patients with and without RBD. The increasing number of neuroimaging studies support this notion.
Objective:
Our primary objective was to review the current evidence regarding the brain neuroimaging findings in PD patients with RBD (PDRBD).
Methods:
A systematic review of articles, published in PubMed between January 1, 2000 and September 23, 2020 was performed. We evaluate previous studies that assessed PD patients with RBD using various brain structural and functional magnetic resonance imaging (MRI) techniques and brain nuclear medicine imaging.
Results:
Twenty-nine studies, involving a total of 3,347 PD subjects among which 912 subjects with PDRBD, met the selection criteria and were included. The presence of RBD in PD patients is associated with structural and functional alterations in several brain regions, mainly in brainstem, limbic structures, frontotemporal cortex, and basal ganglia, raising the hypothesis of a PDRBD neuroimaging phenotype.
Conclusion:
The current review provides up-to-date knowledge in this field and summarizes the neurobiological/neuroimaging substrate of RBD in PD.
Keywords
INTRODUCTION
Rapid eye movement (REM) sleep behavior disorder (RBD) is a parasomnia with abnormal nocturnal dream enactment during REM sleep. During REM sleep, the normal sleeping person is mentally active and dreaming, while the skeletal muscles are paralyzed. In RBD, however, REM atonia is altered, resulting in dream enactment that may vary from slight hand movement to loud vocalization and potentially injurious behavior [1, 2]. RBD is very common among patients with Parkinson’s disease (PD), already from the early PD stages [3] and recent evidence suggests that the presence of RBD is associated with a more diffuse malignant PD subtype, mainly with differences in the spectrum of clinical symptoms, in natural history and prognosis of PD [4], which are likely to imply underlying differences in the pathophysiology and possibly the distribution of neurodegeneration among PD patients with and without RBD. Indeed, RBD in PD patients is associated with more motor [5] and non-motor symptoms, in particular psychiatric manifestations, cognitive alterations and sleep-wake disturbances [6, 7]. These data support the notion that RBD should no longer be considered a complication but a biomarker or even a driving force for neurodegeneration [8]. However, the pathophysiological mechanisms leading to RBD in PD are not fully elucidated.
In recent years, an increasing number of structural and functional magnetic resonance imaging (MRI) as well as nuclear medicine imaging studies reported specific brain alterations in PD patients with RBD (PDRBD). Our primary objective was to review the current evidence and provide up-to-date knowledge regarding the brain neuroimaging findings in PDRBD. Secondly, we discuss pathophysiological hypotheses to explain the association between RBD and neurodegeneration in PD, in respect to brain neuroimaging findings.
METHODS
Extraction of articles was performed by two independent observers (R.M. and P.B.) and was evaluated by the rest of the authors. Discrepancies in selection were resolved by discussion. A systematic literature search in Pubmed/Medline via Ovid was conducted for articles published between January 1, 2000 and September 23, 2020. We used MeSH-term (medical subject heading) search as well as the search terms (“Parkinson’s disease” or Parkinson) AND (“rapid eye movement sleep behavior disorder” or “rem sleep behavior disorder”) AND (“imaging” OR “MRI” OR “magnetic resonance imaging” OR “DTi” or “diffusion tensor imaging” OR “diffusion-tensor” OR “SPECT” OR “single photon emission tomography” OR “DatScan” OR “PET” OR “positron emission tomography” OR “spectroscopy” OR “magnetic resonance spectroscopy”.
The inclusion of studies was based on the following inclusion criteria: 1) original studies (reviews, editorials, case reports etc. were excluded), 2) full text was written in English 3) human subjects were involve, 4) aim of the study was to describe structural or functional neuroimaging findings in PD and RBD patients.
The main scope of the study was to assess the brain neuroimaging findings in PD with RBD or in iRBD that converts to PD. To best serve this scope, we excluded from the analysis, studies that reported on other synucleinopathies (i.e., Lewy body dementia).
The reference lists of articles that met the eligibility criteria were further screened to identify additional studies that may fall within the scope of this review.
Quality assessment
To assess risk of bias in the included studies, we applied nine quality criteria as previously published in by Carey et al. [9]: (1) demographics; (2) imaging procedure; (3) spatial normalization method; (4) determination of ROIs; (5) reproducibility of the analyzes; (6) statistical tests used to substantiate the results; (7) correction for the multiple testing problem; (8) figures and tables; (9) quality control measures. A summary of the quality assessment is presented in the Supplementary Material.
RESULTS
The initial search identified 376 records. After exclusion of duplicates, we identified 238 potentially eligible studies. We additionally included 3 relevant studies found in the reference list of review articles (in total 241 studies). Of these, 29 studies, with 3,347 subjects in total and 912 subjects with PDRBD, met all above-mentioned selection criteria and were included in the systematic review (Fig. 1). Among the included studies, 14 used polysomnography for the diagnosis of RBD while 15 studies used RBD questionnaires.
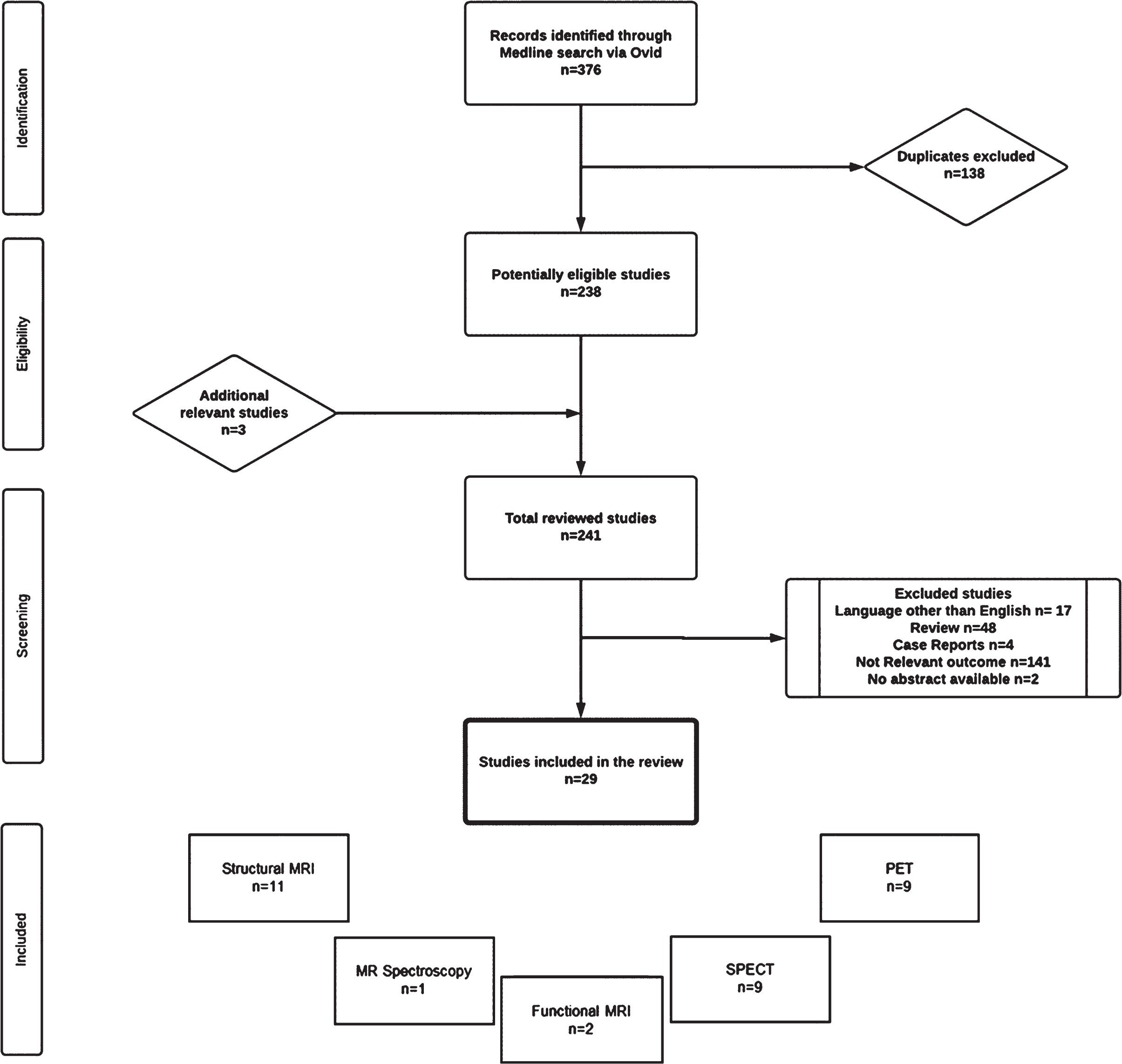
Flow diagram demonstrating the process of article selection for systematic review.
Magnetic resonance imaging
Structural MRI studies
We identified 11 studies [10–20] that reported findings from structural MRI methods, including voxel-based morphometry (VBM), DTI, deformation based morphometry, neuromelanin-sensitive MRI and diffusion MRI connectometry. Three studies used PSG and 8 studies used questionnaire to diagnose RBD (Table 1). Four fMRI studies comprised 217 participants, of whom 174 were PD patients and 43 were HCs.
Structural MRI studies in PDRBD
PDRBD+, Parkinson’s disease’s patients with RBD; PDRBD–, Parkinson’s disease’s patients without RBD; HC, healthy controls; RBDQ, REM Sleep Behavior Disorder Questionnaire; PSG, polysomnography; DBM, deformation-based morphometry; VBM, voxel-based morphometry; SCN, structural correlation network; DTI, diffusion tensor imaging; QA, quantitative anisotropic; FA, fractional anisotropy; MD, mean diffusivity; DMRIc, diffusion MRI connectometry; NM-MRI, neuromelanin-sensitive MRI.
Morphometry studies
The 7 morphometry studies [11–13, 19] comprised 1,231 participants, of whom 265 were PDRBD+ patients, 622 were PDRBD–patients and 344 were HCs (Table 1).
Six studies used VBM, and one study used defor-mation-based morphometry (DBM) to perform volumetric analyse grey matter analysis.
All studies reported on regional measures and 2 studies [15, 16] reported on global network measures or whole-brain analyses as well. One study reported on the correlation between RBD severity and structural changes [16].
In VBM studies comparing PDRBD+ and PDRBD–groups, RBD was associated with de-creased grey matter volume in thalamus, medial temporal cortex, left posterior cingulate, hippocampus and putamen.
In VBM studies comparing PDRBD+ groups and HC, the PDRBD+ patients exhibited increased grey matter volume in thalamus, precuneus, cuneus, medial frontal gyrus, postcentral gyrus, inferior parietal lobule, right pars orbitalis superior frontal gyrus, left fusiform, superior temporal gyrus, angular cuneus and caudate and decreased grey matter volume in cerebellum, fusiform gyrus, inferior frontal gyrus, superior frontal gyrus and putamen.
There was a negative correlation between RBD symptom severity and volume of putamen, thalamus and hippocampus [16].
Two VBM studies found no significant differences between PDRBD+ and PDRBD–groups [12] or among PDRBD+, PDRBD–and HC groups [13].
The only deformation-based morphometry study reported significantly smaller volume in pontomesencephalic tegmentum, medullary reticular formation, hypothalamus, thalamus, putamen, amygdala and anterior cingulate cortex in PD patients with probable RBD compared to PD patients without RBD and to HC [11].
Diffusion MRI studies
The 4 fMRI studies comprised 217 participants, of whom 174 were PD patients and 43 were HCs.
The 6 diffusion MRI studies [10, 18] comprised 345 participants, of whom 159 were PDRBD+ patients, 142 were PDRBD–patients and 44 were HC (Table 1).
All studies reported on anisotropy (fractional anisotropy (FA) or quantitative anisotropic) and 2 studies [12, 17] reported on mean diffusivity (MD) as well. None of the studies reported on the correlation between RBD severity and structural changes.
In DTi studies comparing PDRBD+ and PDRBD–groups, RBD was associated with reduced connectivity (either FA or MD) in fornices, cingulum pathways, corpus callosum, inferior longitudinal fasciculus, inferior fronto-occipital fasciculi, corticospinal tracts, and the middle cerebellar peduncles in three other studies [10, 18].
In DTi studies comparing PDRBD+ groups and HC, PDRBD+ patients exhibited reduced FA mainly in frontal areas and widespread reduced mean diffusivity [17], increased FA in the tegmentum of the midbrain and rostral pons [13] and increased apparent diffusion coefficient in the midbrain (cerebral pendunculus and substantia nigra) [13].
Three DTi studies found no significant differences between PDRBD+ and PDRBD [12, 17].
None reported on the correlation between RBD symptom severity and FA or other DTi parameter.
Neuromelanin-sensitive MRI
The 2 neuromelanin-sensitive MRI studies [13, 20] comprised 97 participants, of whom 40 were PDRBD+ patients, 26 were PDRBD–patients and 31 were HC. Both studies reported reduced signal intensity in the coerueleus/pons region in the PDRBD+ group compared to the PDRBD–and HC groups [13, 20]. In one study, the reduction of the perceived signal correlated with the degree of REM atonia loss in polysomnography [13] (Table 1).
Functional MRI studies
Two resting state functional MRI (rs-fMRI) studies [21, 22] comprised 130 participants, of whom 56 were PDRBD+ patients, 30 were PDRBD–patients and 44 were HC (Table 2).
Functional MRI studies in PDRBD
PDRBD+, Parkinson’s disease’s patients with RBD; PDRBD–, Parkinson’s disease’s patients without RBD; HC, healthy controls; PSG, polysomnography; RS-fMRI, resting state-functional MRI; ALFF, amplitude of low frequency fluctuations; PPN, pedunculopontine nucleus; SMA, supplementary motor area; MPFC, medial prefrontal cortices; ACC, anterior cingulate cortices.
Magnetic Resonance Spectroscopy studies in PDRBD
PDRBD+, Parkinson’s disease’s patients with RBD; PDRBD–, Parkinson’s disease’s patients without RBD; PSG, polysomnography; 1H-MRS, 1H-MR Spectroscopy.
Gallea et al. studied the functional connectivity and found reduced connectivity between the pedunculopontine nucleus (PPN) with the anterior cingulate cortices (ACC) in both PDRPD+ patient groups compared with the PDRBD–patients and HCs. In addition, the presence of postural instability in PDRBD+ patients was associated with defective connectivity between the PPN and the supplementary motor area (SMA) and medial prefrontal cortices areas [21].
The other rs-fMRI study by Li et al. investigated baseline brain activity and reported amplitude of low frequency fluctuations (ALFF). In PD patients, the presence of RBD was associated with significantly reduced amplitude of ALFF values in primary motor and in the premotor cortex. In addition, RBDPD+patients had decreased ALFF values in caudate and putamen and increased ALFF values in prefrontal cortex compared to HC [22].
No studies reported on the correlation between RBD symptom severity and rs-fMRI parameters.
Magnetic resonance spectroscopy
We found only one study using magnetic resonance spectroscopy. The study did not show any statistical significance in respect to proton magnetic resonance spectroscopy (1H-MRS) parameters (mainly the ratios of metabolites relative to creatinine in brainstem were calculated: N-acetylaspartate/creatinine, choline/creatinine, myo-inositol/creatinine) between 12 PDRBD+ and 12 PDRBD–(Table 3).
Nuclear medicine imaging
Single photon emission computed tomography (SPECT)
The nine SPECT studies [19, 24–31] comprised 1,194 participants, of whom 336 were PDRBD+ patients, 707 were PDRBD–patients, 48 were HC and 103 were iRBD (Table 4).
SPECT studies in PDRBD
PD, Parkinson’s disease; PDRBD+, Parkinson’s disease’s patients with RBD; PDRBD–, Parkinson’s disease’s patients without RBD; HC, healthy controls; iRBD, idiopathic REM Sleep Behavior Disorder; RBDQ, REM Sleep Behavior Disorder Questionnaire; RBDSQ, REM Sleep Behavior Disorder Screening Questionnaire; PSG, polysomnography; SBR, specific binding ration; 123I-FP-CIT, 123I-2β-carbomethoxy-3β-(4-iodophenyl)-N-(3-fluoropropyl)nortropane; SPECT, single-photon emission computed tomography; DAT, dopamine transporter
Seven studies compared PDRBD+ (N = 375) vs. PDRBD–(N = 540) patients [19, 29–31], two studies compared PDRBD+ vs. HC, one study followed the phenoconversion from iRBD to PDRBD+ [27] and one study reported in a non-controlled study on dopaminergic transporter (DAT) binding changes related to the presence of RBD in PD patients [28]. All studies reported on DAT binding using 123I-FP-CIT-SPECT.
In 123I-FP-CIT-SPECT studies comparing PDRBD+ and PDRBD–groups, RBD was associated with low specific binding ratio (SBR) at caudate [24–26, 29], in putamen [26, 29] and in ventral putamen [32].
One study reduced DAT uptake was more prominent in the less affected side [25] Interestingly the interhemispheric asymmetry in DAT binding is less profound as disease progresses [26].
In 123I-FP-CIT-SPECT studies comparing PDRBD+ and iRBD groups, PDRBD+ patients exhibited lower DAT uptake in the putamen [24, 31]. In 123I-FP-CIT-SPECT studies comparing PDRBD+ groups and HC, PDRBD+ patients exhibited lower DAT uptake, especially in the putamen [19, 31] and caudate [19]. Two 123I-FP-CIT-SPECT study found no significant differences between PDRBD+ and PDRBD–.
One study [27] examined 20 iRBD patients at baseline and in two intervals of 1.5 years to examine the association between the development of PD in iRBD with 123I-FP-CIT uptake. The patients who developed PD (N = 3) had generally the poorest 123I-FP-CIT uptake at baseline and a decrease in the left putamen(32%., the right putamen (30%., the left caudate nucleus (27%., and in the right caudate nucleus (24%. after 3 years compared with HCs [27].
One study reported a significant negative correlation between RBD symptoms (assessed by RBDSQ) and striatal SBR in 123I-FP-CIT-SPECT [28].
Positron emission tomography studies
The nine PET studies [20, 32–38] comprised 401 participants, of whom 213 were PDRBD+ patients, 185 were PDRBD–patients, 224 were HC and 131were iRBD (Table 5).
PET studies in PDRBD
PDRBD+, Parkinson’s disease’s patients with RBD; PDRBD–, Parkinson’s disease’s patients without RBD; HC, healthy controls; iRBD, idiopathic REM sleep behavior disorder; RBDQ, REM Sleep Behavior Disorder Questionnaire; PSG, polysomnography; VOIs, volume of interest; DVRs, distribution volume ratios; PET, positron emission tomography; 11C-MeNER, (S,S)-11C-2-(α-(2-methoxyphenoxy)benzyl)morpholine; 18F-FDG, 2-deoxy-2-[fluorine-18]fluoro- D-glucose; DAT, dopamine transporter; 18F-FP-CIT, N-(3-18F-fluoropropyl)-2-carbomethoxy-3-(4-iodophenyl) nortropane; [11C]PMP AChE, [11C]methylpiperidyl propionate acetylcholinesterase; DTBZ, [11C]dihy-drotetrabenazine; VMAT2, Vesicular Monoamine Transporter type 2; DASB, 11C3-Amino-4-(2-dimethylaminomethyl-phenylsulfaryl)-benzonitrile; SERT, serotonin transporter.
Six PET studies compared PDRBD+ vs. PDRBD–patients [20, 36] six studies compared PDRBD+ vs. iRBD and HC [20, 38], one study followed the phenoconversion from iRBD to PDRBD+ [35].
Several tracers/radioligands have been reported including 11C-methylpiperidyl propionate acetylcho-linesterase, dopamine transporter tracers, (S,S)-11C-2-(α-(2-methoxyphenoxy)benzyl)morpholine (11C-MeNER PET), Fluorodeoxyglucose-18F (18F-FDG) and serotonin tracers.
In PET studies comparing PDRBD+ and PDRBD–groups, RBD was associated with several findings. First, a reduced neocortical, limbic cortical and thalamic cholinergic innervation in 11C-methylpiperidyl propionate acetylcholinesterase tracer [36]. Second, lower mean values of distribution volume ratios (SVR) in all imaged brain areas, especially in brainstem [20], and a profound decrease in mean values of the volume of interest in the precentral and postcentral gyri [33] in the 11C-MeNER PET. Finally, an increased metabolism in anterior cortical regions, a decreased metabolism in posterior cortical regions in 18F-FDG-PET [25], and a poorer DAT uptake in the putamen (mainly ventral putamen) in 18 FP-CIT-PET [32].
In PET studies comparing PDRBD+ groups and HC, PDRBD+ patients exhibited lower mean values of distribution volume ratios (SVR) in all imaged brain areas, especially in brainstem [20] in the 11C-MeNER PET. In addition, several studies reported a decreased metabolism in the occipital areas and increased metabolism in the primary motor cortex, SMA, premotor cortex, thalamus, basal ganglia (putamen and globus pallidus), cerebellum, pons, parahippocampal gyrus, rostral and orbital areas of the superior frontal cortex [25, 38]. Finally, one study showed a profound reduction in the DAT density in caudate nucleus, anterior putamen, posterior putamen and substantia nigra [37] in 18F-FDG-PET.
Similar findings were reported in PET studies when PDRBD+ patients were compared with iRBD patients [34, 38]. In addition, one 18F-FDG-PET study [35] followed 20 iRBD patients showing the 4 patients who developed PD in the 4 years of the follow up, had suprathreshold Parkinson’s Disease Related Pattern (PDRP), a pattern that characterizes early PD and may differentiate it from MSA and other atypical parkinsonian disorders, and also a more rapid change in the PDRP score [35].
One 11C-MeNER PET study found no significant differences between PDRBD+ and PDRBD–in respect to cortical thickness [33].
DISCUSSION
To our knowledge, this is the first systematic review that examines and critically discusses available literature on brain neuroimaging findings in PD with RBD. We included studies that used both structural and functional MRI techniques as well as nuclear medicine techniques including SPECT and PET.
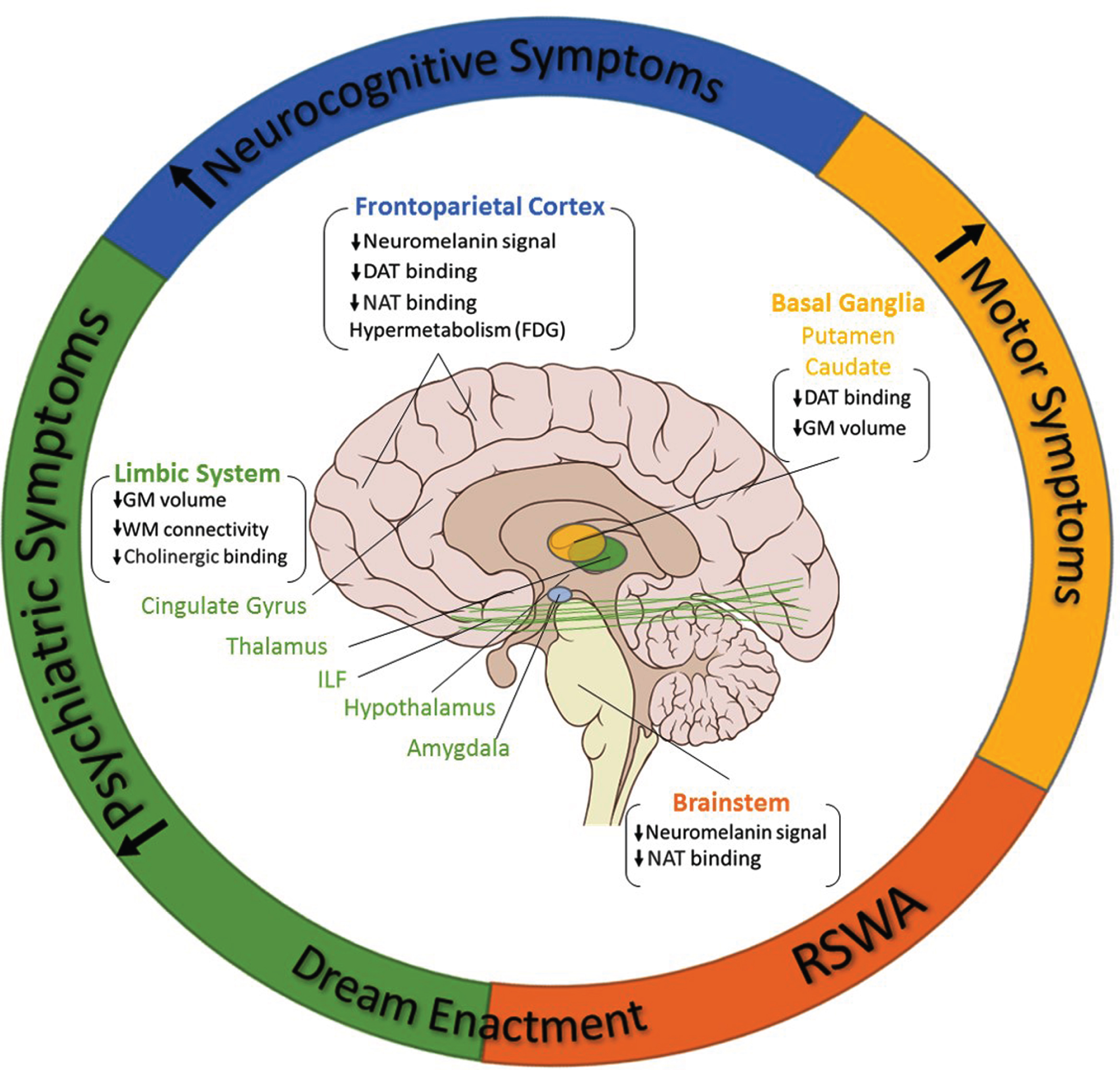
The association of RBD with multiple brain imaging alterations and a more malignant motor and non-motor subtype in PD. Orange area (RSWA): RBD in the early phases of PD is directly related to metabolic alterations (reduced NAT binding) and neurodegenerative processes (lower neuromelanin signal) in several brainstem nuclei which are implicated in the regulation of REM sleep and REM atonia. Green area (psychiatric symptoms): Structural (affecting both grey and white matter) and functional alterations in key limbic regions, including thalamus, hypothalamus, amygdala, cingulate gyrus, and related structures (i.e., ILF) are thought to possibly underlie the increased non-motor profile, in particular psychiatric disturbances, more intense dreaming and dream enactment, that characterizes PD patients with RBD. Blue area (neurocognitive symptoms): RBD-specific structural (lower neuromelanin signal), degenerative (reduced DAT binding in SPECT) and functional (FDG hypermetabolism) cortical changes, in particular in frontoparietal regions, might underly the unique and specific impaired cognitive profile in PD patients with RBD. Yellow area (motor symptoms): Neurodegeneration (reduced DAT binding) and atrophy (reduced grey matter volume) in basal ganglia might be the neural substrate for the increased motor symptoms in PD patients with RBD compared to those without RBD. DAT, dopamine transporter; FDG, Fluorodeoxyglucose (18F); GM, grey matter; ILF, inferior longitudinal fasciculus; NAT, noradrenaline transporter; PD, Parkinson’s disease; RBD, REM sleep behavior disorder; REM, rapid eye movement; RSWA, REM sleep without atonia; WM, white matter. Up arrow for increase, Down arrow for decrease.
MRI studies assessing structural changes in grey matter, report abnormalities and atrophy in limbic structures, such as the thalamus, hypothalamus, cingulate gyrus, amygdala, hippocampus and in basal ganglia mainly the putamen, in PDRBD patients compared to PDRBD–or HC. Similarly, the presence of RBD in PD is associated with white matter abnormalities in limbic connectivity but also in midbrain, brainstem and corticospinal connectivity which are closely involved in locomotion and muscle tone regulation.
In consistence with structural MRI findings, functional MRI studies report abnormal functional activation and connectivity in the motor/premotor cortex, in brainstem regions and in basal ganglia associated with RBD in PD.
Lastly, SPECT and PET studies show functional changes in dopaminergic, noradrenergic, and cholinergic transmission as well as metabolic changes in the fronto-limbic neural circuitry, in brainstem and in basal ganglia in PDRBD compared to PDRBD–and HC.
These neuroimaging findings are consistent with the hypothesis that RBD in PD is not simply the result of a brainstem pathology affecting the REM sleep regulating nuclei but may represent a certain subtype of PD, with unique progression and prognosis and strongly related to specific mechanisms, patterns and localization of PD-related pathology [39] (Fig. 2). Indeed, according to the Braak’s hypothesis, abnormal a-synuclein accumulation in PD follows a specific pattern, from neurons of the nasal cavity and in the gut to the brainstem and pontine regions (Stage 1–2), to limbic structures and later to substantia nigra and basal ganglia (Stage 3–4), and finally to the neocortex and frontoparietal lobes (Stages 5–6) [40]. In agreement with this a-synuclein pathology staging scheme [40], it has been suggested that RBD in the prodromal and early phases of PD is directly related to neurodegeneration in several brainstem nuclei, including the cholinergic PPN, the noradrenergic locus coeruleus and the serotonergic raphe nuclei, which are implicated in the regulation of wakefulness, the generation of REM-sleep circuit, the sleep staging and the generation of REM atonia [41]. As degeneration progresses, damage to key limbic regions, such as hypothalamus, amygdala, thalamus, is thought to possibly underlie the increased psychiatric/emotional disturbances and a more severe non-motor parkinsonian phenotype of PDRBD compared to PDRBD–as well as dreaming and dream enactment that PDRBD patients experience already from the early stages of the disease [41–43]. Lastly, RBD-specific cortical changes, in particular in frontoparietal regions, might underly the unique and specific impaired cognitive profile in PD patients with RBD [44].
Further support for the hypothesis that the presence of RBD might define a specific PD subtype and that PDRBD progresses along a disease continuum from iRBD, derives from increasing number of neuroimaging studies showing specific structural and functional brain abnormalities in the group of iRBD patients that convert to PDRBD [45]. Indeed, iRBD patients that developed PD, showed poor 123I-FP-CIT uptake in basal ganglia areas [27] and hypometabolism in parieto-occipital and premotor cortices concomitant with metabolic preservation or hypermetabolism in cerebellum, pons, thalamus and lentiform nucleus in FDG-PET [35] (the PDRP), all consisting early neuroimaging markers of PD. These observations are consistent with clinical studies demonstrating that patients with iRBD present non-motor symptoms and signs also found in PD. However, further appropriate studies following a greater number of patients are needed to decipher this hypothesis.
Limitations
With the exception of some morphometry and very few nuclear medicine studies, the majority of studies included small sample sizes. Furthermore, studies often have included patients with different types of PD, in various disease and treatment stages (i.e., newly diagnosed/drug naïve vs. medically treated), therefore the results between the studies were not always comparable. The lack of a HC group in some of the included studies presents another limitation. 15 studies used RBD questionnaires instead of polysomnography, thus compromising the diagnostic accuracy of RBD. The presence of RBD in PD is associated with more severe non-motor symptoms which might consist important confounders for several neuroimaging findings (i.e., depression or apathy for abnormalities associated with limbic regions or cognitive deficits for abnormalities associated with cortical regions). The majority of studies assessed clinical/neuropsychological/neurocognitive symptoms in the comparison groups; however, only 4 studies [13, 28] performed a clinico-radiological correlation analysis and only two studies grouped PD patients based on a second clinical characteristic (depression [14] and postural instability [21]), apart from RBD, in order to identify clinical confounders in their neuroimaging findings. Finally, most of the studies were cross-sectional, which implies difficulties in concluding about temporal or causal relations. In the studies with a longitudinal follow-up design a very small number of PDRBD patients has been included.
Another limitation of our systematic approach was that the period over which the reviewed studies were published spanned 20 years. During this time, the precision of the performed techniques and the diagnostic criteria for RBD have changed. Finally, there is a possibility of selection bias based on language criteria due to the exclusion of articles that were not written in English and of publication bias since we only included papers published on PubMed.
CONCLUSIONS
In this systematic review, we showed that RBD was associated with a widespread structural and functional brain impairment mainly involving limbic, brainstem and basal ganglia neural circuits. This could be attributed to a unique pattern of neurodegeneration of an ongoing condition in different stages including iRBD, iRBD with non-motor symptoms and PDRBD. This is consistent with the increasing evidence that, apart from the characteristic nocturnal behaviors, PD patients with RBD experience more non-motor symptoms than PD patients without RBD. Further studies are needed to better elucidate the pathophysiology and neuroanatomical substrate of RBD in PD.
