Abstract
Background:
Parkinson’s disease (PD) non motor symptoms may present early in the disease course and worsen with advancing disease. Respiratory changes can affect individuals to remain physically active, contributing to a reduction of functionality and quality of life.
Objective:
The aim of this systematic review is to synthesize evidence of respiratory disorders in patients with PD.
Methods:
An electronic search was performed up to November 2020 on PubMed-MEDLINE, Embase, Web of Science, Lilacs, Cinahl, and Cochrane using the following keyword combination: [(“Parkinson disease”) AND (“respiratory function tests” OR “evaluation”) AND (“respiratory system” OR “respiration disorders” OR “respiratory muscles”)].
Results:
The electronic search resulted in 601 references in English or Portuguese. The selection process and data extraction were made by two independent reviewers. We selected 19 studies including cross-sectional studies that investigated the respiratory disorders in patients with PD through pulmonary function, respiratory muscle strength, or physical capacity evaluation. We excluded studies that considered patients with other diseases. Eighteen studies evaluated the pulmonary function in patients with PD, eleven studies verified the influence of PD on respiratory muscle strength, and three studies assessed the physical capacity through functional tests.
Conclusion:
The evidence showed that PD patients have higher chances to present a pulmonary dysfunction, either obstructive or restrictive, when compared to healthy subjects. In addition, these patients present lower respiratory muscle strength and a consequent decrease in physical capacity in endurance exercises. The respiratory impairment in PD seems to be directly related to the progression of the disease.
Keywords
INTRODUCTION
Parkinson’s disease (PD) is a neurodegenerative disease firstly described in 1817 [1], characterized by a progressive degeneration of substantia nigra neurons, resulting in striatal dopamine depletion [2, 3]. The most prevalent PD symptoms are motor function impairments, such as bradykinesia, muscular rigidity, rest tremor and postural instability [4]. Moreover, secondary disorders can be observed, such as gait impairment and falls [5], leading to a loss of independence in activities of daily living [6]. However, when the first motor symptoms are clinically identified, 60% of dopaminergic neurons located in the substantia nigra have already been lost [7].
The non motor characteristics of PD have attracted attention, as they can significantly affect functionality and quality of life [8]. Moreover, these symptoms can present early, or in advancing disease, and largely do not improve on dopaminergic treatment. According to Braak’s model of PD staging, symptoms such as sleep behavior disorders [9, 10], olfactory loss [11], and depression [12] can start before motor impairments [13] and could play a crucial role in an early diagnosis [14]. Furthermore, other non-motor symptoms can increase throughout the progression of the disease, such as dementia, urinary incontinence, constipation, pain, fatigue, and respiratory disorders [15–18].
Several assessments are used to quantify respiratory dysfunctions in adults, focused on the evaluation of subjective symptoms, such as dyspnea, tachypnea, or other self-reported clinical features [19], or objective signs, including pulmonary function [20, 21], respiratory muscle strength [22], and functional capacity [23].
Compromised respiratory function in PD represents a risk for developing other pneumo-functional comorbidities [24], increasing the risk of hospitalization [25]. A review by Macleod et al. showed that the mortality index in PD is 50% higher than the control mortality, and there is a decrease in survival of 5% per year of follow up [26], in which pneumonia is the most common cause of death in PD patients [27–29]. Despite these epidemiology data about PD’s complications and its comorbid disorders, respiratory impairments, its mechanisms and evolution are poorly understood. Therefore, the aim of this systematic review is to synthesize evidence regarding respiratory disorders in PD throughout the progression of the disease, and to identify the gaps in the literature regarding this subject to guide future research.
METHODS
Protocol and registration
This systematic review was developed according to the Cochrane Collaboration guidelines [30] and the report followed the Prisma Statement [31]. The study was registered at PROSPERO under reference number CRD42018084545.
Search strategy
The electronic search was performed up to November 2020 on PubMed-MEDLINE, Embase, Web of Science, Lilacs, CINAHL, and Cochrane Library, using the following keyword combination: [(“Parkinson disease”) AND (“respiratory function tests” OR evaluation) AND (“respiratory system” OR “respiration disorders” OR “respiratory muscles”)]. Two independent reviewers selected the retrieved titles, excluding those that were clearly unrelated to this systematic review. The abstracts of the selected titles were evaluated, and those that met the inclusion criteria were selected for a final assessment of their full texts. Likewise, the relevant full texts were included in this review, and their reference lists were checked to potentially identify relevant studies not identified in the electronic search. Disagreements during the selection process were solved through consensus between the two reviewers, and a third reviewer was consulted when consensus was not achieved. Analysis and selection processes of the primary studies were performed using the reference manager software called START (State of the Art through Systematic Review), version 2.0.
Inclusion criteria
This systematic review included primary studies which: 1) assessed patients diagnosed with PD at any stage of the Hoehn and Yahr scale [32] without any other respiratory or neurological diseases; 2) investigated respiratory impairments through different measurements of respiratory function, respiratory muscle strength or physical capacity; 3) compared the results of the PD group to a healthy control group (CG) or to predicted values previously described in the literature; and 4) presented a cross-sectional study design.
Studies not related with the topic of this systematic review were excluded. Titles obtained from abstracts published in congress or workshops were excluded.
Data extraction
Two independent reviewers used a data extraction form, considering: 1) population aspects such as number of participants, comparison groups, age, gender, and the Hoehn & Yahr stage of PD; 2) type of evaluation and assessed outcomes; and 3) observed results based on the evaluation of interest. The information was collected by two independent reviewers.
Methodological quality of included studies
The methodological quality of the studies was accessed by the JBI Critical Appraisal Checklist for Analytical Cross-Sectional Studies [33]. Its score ranges from zero to eight points, and all studies with a score higher or equal to 5 points (62.5%) were considered as high methodological quality.
RESULTS
Search strategy
The electronic search resulted in 601 references in English or Portuguese. The selection process was carried out through consensus between two independent reviewers, resulting in 17 selected studies. A manual search was performed based on the analysis of reference lists of the selected studies resulting in the selection of 16 additional papers, of which three studies matched the inclusion criteria. The final selection process resulted in the inclusion of 19 studies (Fig. 1).
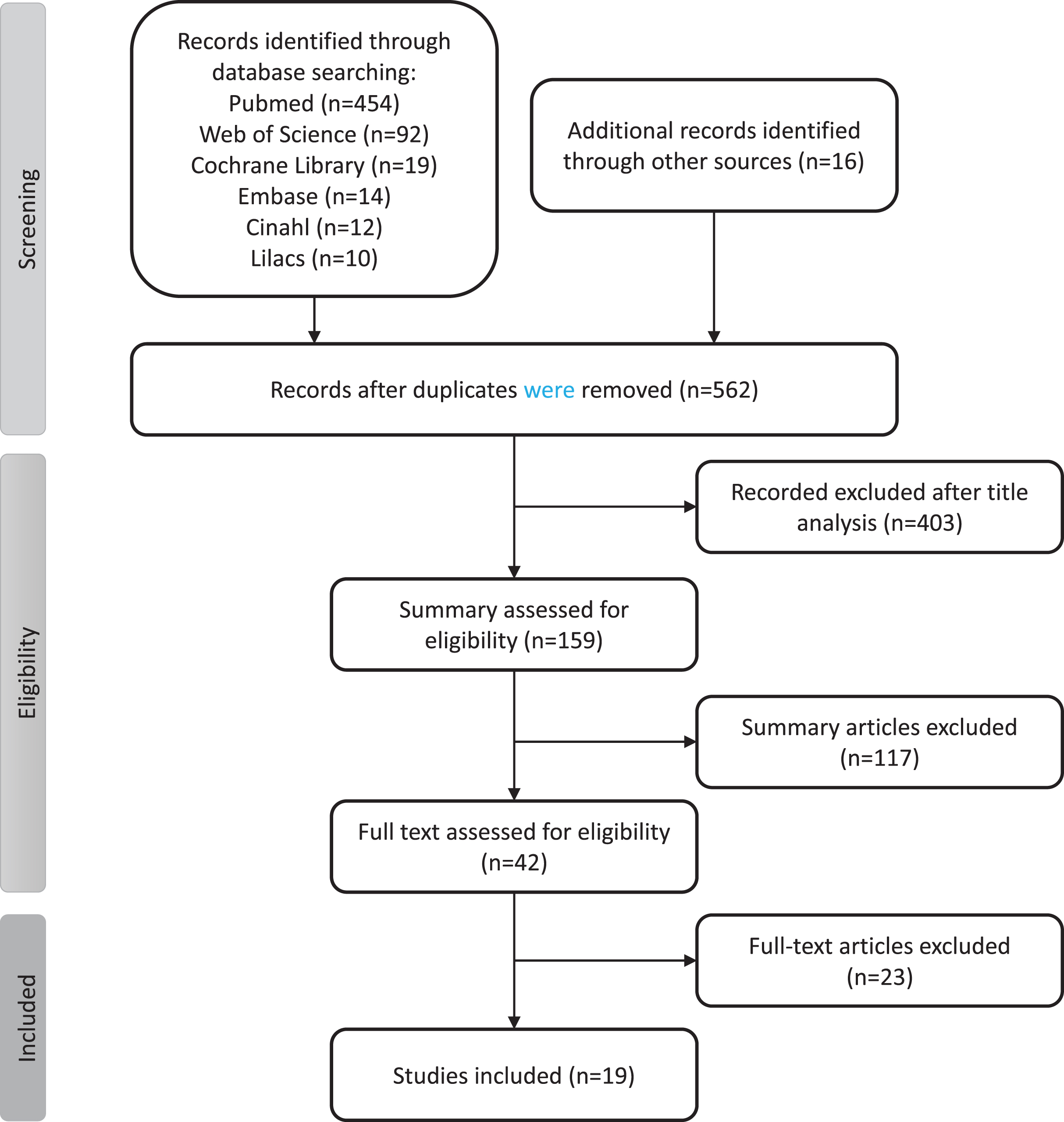
Trial flow diagram of the study's selection process.
Methodological quality of included studies
Among the nineteen relevant studies, fifteen showed high methodological quality (score higher or equal to 5 points) according to the JBI Critical Appraisal Checklist for Analytical Cross-Sectional Studies [33]. Three studies did not reach the score for high methodological quality (Fig. 1).
Characteristics of studies
Table 1 presents the selected characteristics of the studies, including demographics, recruitment, evaluations of interest and the score for the JBI Critical Appraisal Checklist for Analytical Cross-Sectional Studies. Three different evaluations were selected for analysis in this systematic review: pulmonary function, respiratory muscle strength and physical capacity.
Characteristics of the studies
H&Y, Hoen & Yahr Scale; JBI, JBI Critical Appraisal Checklist for Analytical Cross-Sectional Studies; PD, Parkinson’s disease; *age-matched controls; **laboratory volunteers.
Eighteen studies evaluated the pulmonary function in patients with PD, comparing different outcomes in these patients to a control group or predicted values available in the literature [34–51]. The main outcomes are presented in Table 2.
Pulmonary function evaluation
PD, Parkinson’s disease; CG, control group; PNV, predicted normal value; TLC, total lung capacity; FEV1, forced expiratory volume in one second; FVC, forced vital capacity; VC, vital capacity; PEF, peak expiratory flow; PIF, peak inspiratory flow; MEFV, maximal expiratory flow volume; MIF50, maximal inspiratory flow at 50%; MEF75, maximal expiratory flow after 25% of FVC; MEF50, maximal expiratory flow after 50% of FVC; MEF25, maximal expiratory flow after 75% of FVC; FIV1, forced inspiratory volume in one second; MVV, maximal voluntary ventilation; PEFR, peak expiratory flow rates; PIFR, peak inspiratory flow rates; MMEFR, maximum mid-expiratory flow rate; MMIFR, maximum mid-inspiratory flow rate; IC, inspiratory capacity; FRC, functional residual capacity; RV, residual volume; FEF25–75 %, forced expiratory flow during middle half of FVC; FEF50, forced expiratory flow at 50% of VC; RAW, airway resistance; DLCOcSB, carbon monoxide diffusion capacity.
Eleven studies used manovacuometry to evaluate the maximal inspiratory and expiratory pressures, verifying the influence of PD on respiratory muscle strength [34, 50–52]. The results are summarized in Table 3.
Respiratory muscle strength evaluation
PD, Parkinson’s disease; CG, control group; PNV, predicted normal value; MIP, maximal inspiratory mouth pressure; MEP, maximal expiratory mouth pressure; SNIP, sniff nasal inspiratory pressure.
Three studies assessed the physical capacity through functional tests, such as the six-minute walk test, the ten-meter walk test and the cycle ergometer exercise test [36, 52]. The results are presented in Table 4.
Physical capacity evaluation
PD, Parkinson’s disease; CG, Control Group; PNV, Predicted Normal Value; H&Y, Hoen & Yahr Scale. *approximated values based on Figure 4.
DISCUSSION
In this systematic review, nineteen included studies used different methodologies to evaluate pulmonary function, respiratory muscle strength, and the physical capacity of 677 subjects with mild, moderate or severe PD. Eleven studies evaluated the respiratory muscle strength of PD patients through maximal expiratory pressure (MEP) and maximal inspiratory pressure (MIP) measurements. Eight of them showed lower MIP and MEP values in PD patients compared to age-matched CG or predicted normal values [34, 52]. These results show evidence of a respiratory muscle weakness, which starts in the very early stages of PD [34, 51], and increases throughout the progression of the disease [39, 52]. Only three studies did not observe any difference between PD patients and a CG [38, 50], and PD and predicted normal values [37]. In a study conducted by Canning et al. (1997), 13 out of 16 patients related that they regularly performed aerobic exercises, such as walking or cycling, which could explain the good outcome of the PD patients [37].
The respiratory muscles consist of two types of muscular fibers—type I and II—which are categorized according to its type of metabolism and contractile characteristics [53]. Wrede et al. (2012) showed that PD patients often present myopathological changes, such as hypertrophy of type I fibers, combined with a loss of type II fibers, leading to a decreased activation of fast-twitch fibers. The cause of this myopathy is unknown but could be a mechanism behind the respiratory muscle dysfunction observed in this review [54].
We believe that the reduction of the respiratory muscle strength observed in PD could be responsible for secondary respiratory disorders, such as changes in pulmonary function. This affirmation is supported by a review study by van de Wetering-van Dongen [55] that showed that respiratory training presents positive effects on pulmonary function and quality of life of PD patients.
The evaluation of pulmonary function is commonly used to diagnose, monitor or evaluate pulmo-nary impairments [56], as well to determine the type of pulmonary dysfunction, such as obstructive, restrictive or a combination of both impairments [57]. The most relevant measurements include the forced vital capacity (FVC), forced expiratory volume in one second (FEV1), vital capacity (VC), and FEV1/FVC ratio [58]. These parameters can provide important information to guide respiratory training, which has been proved clinically efficient in this population [59].
In this review, 18 studies assessed pulmonary function of PD patients. An obstructive pattern was identified in seven studies [35, 46], and a restrictive dysfunction was shown by four studies [38, 45]. In addition, one study identified that PD patients have both obstructive and restrictive dysfunctions [47]. Four studies did not observe any changes in the pulmonary function of PD patients [34, 51], however in this case only early-stage PD patients were evaluated.
Obstructive disorder is characterized by a greater reduction in FEV1 compared to FVC, resulting in a reduced FEV1/FVC ratio below the lower normality limit [60]. In PD, the reduction of the respiratory muscle strength, associated with laryngeal muscle dysfunction [61] and autonomic dysfunction [62] can lead to a reduced ventilatory capacity and upper airway obstruction, increasing respiratory work.
The restrictive pattern is marked by a reduced FVC in the absence of an obstructive pattern [63]. Restrictive disorders in PD can be caused by stiffness, chest wall deformity and respiratory muscle weakness, which decreases mobility thoracic, lung volumes and capacities [19, 65].
Furthermore, some of the included studies showed a correlation between the pulmonary impairments and the disease stage. It is known that the worsening of pulmonary function is associated with the severity of the disease, and it is expected that patients with a higher score on the HY scale will have significantly lower parameters compared to those in the early stages of PD [35, 46].
Ultimately, three studies assessed the influence of PD on physical capacity, through walk tests (six-minute walk and ten-meter walk tests) and cycle ergometer tests. Patients with PD presented a reduced walk distance when compared to an aged-matched control group during the 6MWT [36]. In addition, PD showed a lower velocity than the predicted normal values during the ten-meter walk test at fast speed [37], and a positive correlation between the PD subjects' performance and an increased values of mouth pressures at the cycle ergometer test [52] and 6MWT [36]. These results suggest that PD patients have physical capacity impairments, which could be a result of the characteristic respiratory dysfunction, resulting in impairments of their daily living functions.
Moreover, Canning et al. showed that PD patients present normal values during the cycle ergometer test and the ten-meter walk test at normal speed, and related that the regular exercise habits of the included subjects may have contributed to maintaining normal levels. The same study found differences in the test results between PD active and sedentary subjects [37].
In summary, this review presented common respiratory disorders in PD, which is extremely important to guide clinical practice. This systematic review revealed that the respiratory muscle weakness can be observed from early stages of PD, and that other dysfunction such as obstructive and restrictive patterns, and physical capacity impairments are mostly observed in moderate and advanced subjects.
With that in mind, we believe that the respiratory weakness is an important marker to be considered during patient care, and that early respiratory training could be effective in the treatment of respiratory dysfunctions, delaying the appearance of secondary disorders. There is some evidence showing that respiratory training can improve respiratory parameters in PD [55, 66], but we did not find any study that investigated its effects on preventing the progression of respiratory disorders in PD.
Future studies should investigate the cause of the observed respiratory symptoms, establishing the following: 1) what are the mechanisms behind the respiratory symptoms?; 2) is pulmonary dysfunction secondary to respiratory muscle weakness?; 3) is early respiratory training effective to prevent the appearance of pulmonary dysfunction?
Study limitations
This systematic review gathered evidence of the common respiratory disorders in PD patients. However, these findings need to be interpreted in the context of its limitations. The included studies presented a transversal evaluation of PD patients; however, a longitudinal follow up would provide more specific information regarding the progression of symptoms. Some included studies presented a low number of subjects per group, especially when comparing the results among different disease stages. Moreover, the heterogeneity of the number of patients, control population, disease duration and mea-sured values makes the comparison among the studies difficult.
CONCLUSION
In conclusion, this systematic review showed that respiratory disorders are a common feature in PD. Respiratory muscle weakness is observed from early PD stages and increases throughout the progression of the disease. The pulmonary dysfunction, such as obstructive and restrictive patterns, and the physical capacity impairments are mostly observed in moderate and advanced subjects, and we believe that it could be a direct result of the decreased muscle strength. Furthermore, larger studies should address the remaining questions discussed in this systematic review, by using standardized measurements and long-term follow up.
Footnotes
ACKNOWLEDGMENTS
This research was supported by CAPES finance code 001 and FAPESP (process 2017/24879-2).
CONFLICT OF INTEREST
We have no conflict of interest to declare.
