Abstract
Background:
Even though a significant fraction of Parkinson’s disease (PD) patients presents with only minor or no motor asymmetry, the motor symptoms in PD typically start on one side of the body and worse symptoms on the side of the disease onset usually persist long after the disease has become clinically bilateral. The asymmetric presentation of PD has been studied over the years, with some studies showing slower progression in PD subjects with asymmetric disease presentation. In other studies, however, it was not possible to relate the asymmetry to disease progression.
Objective:
The main objective of the present study was to assess the effect of asymmetry at disease onset on disease progression.
Methods:
Using the data available in the Parkinson’s Progression Markers Initiative (PPMI) database, at baseline, 423 subjects with
Results:
There was no correlation between neither clinically, nor DatSCAN defined asymmetry and non-motor, motor, and disability progression in the
Conclusion:
Asymmetry at disease onset does not predict progression of PD. Further studies are needed to investigate whether early detection of asymmetry on clinical grounds could successfully distinguish between PD and symmetric types of atypical parkinsonism in the early stages of the disease.
INTRODUCTION
An important clinical feature of Parkinson’s disease (PD) is the asymmetric distribution of the severity of motor symptoms [1]. Namely, in PD motor symptoms typically begin on one side of the body, and worse symptoms on the side of onset often persist long after the disease has become clinically bilateral [2], although a significant fraction (up to 20–40%) of PD patients presents with only minor or no motor asymmetry [2–5]. The asymmetric presentation of PD has been studied over the years, with some studies showing slower progression [6], better quality of life [7], and better mental state [8] in PD subjects with asymmetric disease presentation. However, other studies found no association between asymmetry and PD progression [1]. In addition, recent studies have shown a possible asymmetry in the presentation of some non-motor symptoms in PD, such as auditory dysfunction [9], or pain [10].
A major methodological challenge in the investigation of asymmetry is its definition, and thus the measurement of asymmetry. For example, different scales for measuring the motor symptoms of the disease, such as the Hoehn and Yahr scale [5], SCOPA-Motor scale [1], and Unified Parkinson’s Disease Rating Scale (UPDRS) [11–14], have been used to define asymmetry mostly as the absolute difference between one and the contralateral side of the body. The definition of asymmetry in PD is therefore still arbitrary, which at least partially explains the inconsistency of results between studies. In addition, the mechanisms of asymmetric presentation of the disease are still unclear, although studies suggest earlier involvement of the substantia nigra pars compacta and the adjunct basal ganglia contralateral to the more affected side of the body [2].
The main objective of the present study was to assess the effect of asymmetry on the progression of PD using data available in the Parkinson’s Progression Markers Initiative (PPMI) database. We hypothesized that asymmetry would significantly influence disease progression, so that the more asymmetric the disease, the less progressive the disease progression, and vice versa, the more symmetric the disease, the faster the disease progression.
METHODS
Study design
This is a retrospective analysis based on prospectively collected data within the PPMI database.
Subjects
The data used for the preparation of this article were taken from the PPMI database. At baseline, 423 (146 females)
The demographic data of the patients (number of patients - N, mean age in years ± standard deviation –SD and gender distribution) as well as the mean values ± SD of the Movement Disorders Society- Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) I, II and III at baseline, and 1-, 3-, and 5-year follow-up are presented in the table
Procedure
Clinical data as measured by MDS-UPDRS parts I, II, and III [15], and imaging data (DatSCAN) were used in the study. To minimize the confounding effects of dopaminergic medication, the analysis of the motor MDS-UPDRS part III in this study was limited to scores recorded OFF medication, with a minimum of six hours from the last dose of the dopaminergic medication.
Two asymmetry indices were used in the study. One asymmetry index was based on the MDS-UPDRS part III score at baseline (
The total score of MDS-UPDRS part I, which measures the non-motor aspects of experience of daily living, was used to assess the
Statistical analysis
Mean age in years±standard deviation and frequency distribution by gender was used to demographically describe the study participants. We used Pearson’s
The coefficient of determination,
RESULTS
We did not observe linear correlation between MDS-UPDRS III asymmetry index and non-motor progression, disability progression, and motor progression of the disease (Fig. 1A–C) at one- (
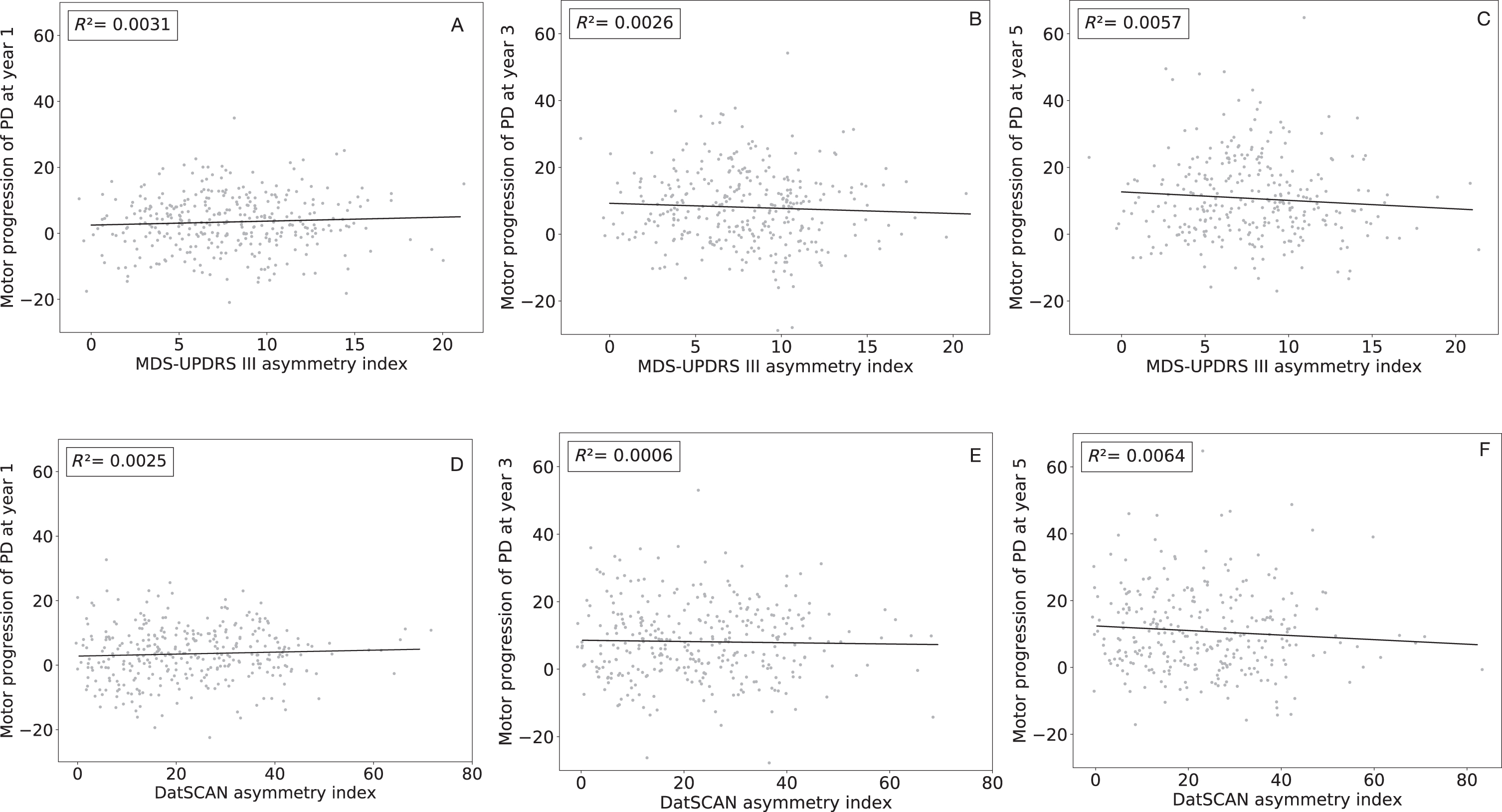
Linear regression of the Movement Disorders Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) asymmetry index and DatSCAN asymmetry index (x-axis) and the motor progression of the disease at one- (1A and D respectively), three- (1B and E respectively), and five-year (1C and F respectively) follow-up (y-axis). The coefficient of determination (
DISCUSSION
The main finding of this study is that motor asymmetry was not correlated with either motor, or non-motor progression of the disease, or progression of disability. Furthermore, DatSCAN asymmetry index was also not correlated with disease progression.
The precise cause of asymmetry of presentation of PD are not known, and it has been suggested that the choice of side of predominance in PD could be of a stochastic nature [2]. Our results are in line with a recent study on the importance of motor asymmetry in PD [1], in which the association of the symmetric symptom distribution in PD with poorer performance in activities of daily living, depression, sleep, and autonomic functions, was fully explained by the confounding effect of age and disease duration in the symmetric (sub)group of patients. However, in this study the extent of asymmetry negatively correlated with age, age at disease onset and disease duration, which was already shown in a previous study [14]. This indicated that the older the age (at onset) and the longer the disease duration the more symmetric the presentation of the disease is. Moreover, in this study, age exerted a stronger effect on reducing asymmetry than disease duration. According to the α-synuclein origin and connectome (SOC) model of PD [16], the symmetric presentation of motor symptoms relates to the symmetric propagation of α-synuclein via the vagal nerves to both brain hemispheres through both dorsal motor nuclei of the vagal nerve. Therefore, these patients have a larger, symmetric burden of pathological changes involving α-synuclein, which promotes faster disease progression, including faster cognitive decline. On the contrary, the pathological changes in patients presenting with asymmetric motor signs of PD, the pathology starts in the brain, they have less α-synuclein burden at disease onset and the progression is slower. In another study [17], using cluster analysis based on clinical data, three disease clusters were identified—motor/slow progression, intermediate, and diffuse/malignant disease cluster; the diffuse/malignant cluster of patients had in general more severe motor and non-motor symptoms and signs and lower asymmetry score [13] indicating less asymmetric disease. At disease onset, fewer patients in this cluster had unilateral onset of motor symptoms. There were no differences in age and disease duration between the cluster of this study. The mechanism for the differences between the clusters is not clear, but it might include relative vulnerability of substantia nigra (e.g., mainly motor subtype patients having more vulnerable nigral neurons and hence presenting earlier with pure motor symptoms), or even variable propensity for the spread of α-synuclein from one region to another.
In addition to using the absolute difference between sides based on UPDRS [11–14] as asymmetry index, the Webster and Hoehn and Yahr Scale [5] as well as the SCOPA motor scale (right-left) [1] have been used to define asymmetry in PD. One of the disadvantages of using arbitrarily defined asymmetry cut-off indices is that they do not correspond to the objective measures of asymmetry. For example, although the disease may be “clinically asymmetric”, a symmetrical distribution of image-based findings, such as DatSCAN is often the case [18], although in another single-centre study [19] of 634 drug-naive PD patients with median 4.28 years of follow-up, subjects had 18F-FP-CIT positron emission tomography (PET), and a higher PET-asymmetry index was associated with slower longitudinal changes in L-dopa equivalent daily doses (LEDD), suggesting a slower progression of the disease, as higher LEDD is associated with worse motor symptoms, a finding which is contrary to the findings of our study. Nevertheless, we have decided to consider asymmetry as a continuum, rather than dividing patients into asymmetric and symmetric groups. Indeed, this was suggested in previous studies [14], as the dichotomous approach to asymmetry is not compatible with the normal distribution of the difference score. In future studies, it might also be useful to define asymmetry not solely based on motor symptoms, but also based on lateralised non-motor symptoms, such as pain and possibly auditory dysfunction.
There are some limitations of our study. First, the results of our study are based on the data from the PPMI database, which is a multicentric study including data from 33 centres. The multi-centre nature of the PPMI dataset may have led to added variance in clinical and imaging data, which in the end might have resulted in a lack of correlation between the clinical/image-based asymmetry index and disease progression as explored in our study. However, the data collected in PPMI database, even though multicentric are gathered using rigorous study protocol. Second, PPMI database includes data of PD patients only. It would be useful to investigate whether early detection of asymmetry on clinical grounds, alone or together with other biomarkers, could successfully distinguish between PD and symmetric types of atypical parkinsonism, such as progressive supranuclear palsy (PSP) and multiple system atrophy in the early stages of the disease [2] on a heterogeneous population of subjects with parkinsonism, such as those from the everyday outpatient clinic for movement disorders. There is already evidence from neurophysiological studies that certain asymmetry characteristics, such as the asymmetry index of the blink reflex recovery cycle could successfully differentiate PD from progressive supranuclear palsy and multiple system atrophy [20]. This renders the importance of asymmetry unquestionable and requires further research on this phenomenon in different clinical situations.
Footnotes
ACKNOWLEDGMENTS
Data used in the preparation of this brief report were obtained from the Parkinson’s Progression Markers Initiative (PPMI) database (http://www.ppmi-info.org/data). For up-to-date information on the study, visit http://www.ppmi-info.org. PPMI, a public-private partnership, is funded by the Michael J. Fox Foundation for Parkinson’s Research and funding partners. The list with full names of all the PPMI funding partners could be found at ![]() .
.
The research was supported by the Slovenian Research Agency (ARRS) under the Artificial Intelligence and Intelligent Systems Programme (ARRS No. P2-0209).
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
