Abstract
Background:
It has been speculated that stains are neuroprotective and are associated with a reduced risk of Parkinson’s disease (PD), but only a few studies have investigated the influence of statins on the progression of PD.
Objective:
To evaluate whether long-term statin use may affect motor progression in a large cohort of
Methods:
We conducted a 4-year retrospective observational cohort study to assess patients with PD. The patients were consecutively recruited from a single tertiary center between January 2015 and January 2017. Information on motor function was obtained using the MDS-Unified Parkinson Disease Rating Scale (UPDRS)-III and all subjects were extensively characterized, including information about lifestyle habits, cardiovascular risk factors and cholesterol blood levels.
Results:
Of the 181 participants included in the study, 104 patients were evaluated for eligibility (42 patients were exposed to statin therapies and 62 were not treated with statins). They presented similar scores in UPDRS III at baseline but the statin users had a lower motor impairment at 4 years compared to non-user PD patients. Additionally, statin treatment resulted in slower progression of the rigidity score of UPDRS over 4 years. No other significant differences were observed between PD patients with and without statins.
Conclusion:
Early PD patients with long-term statin usage showed lower motor deterioration after 4 years of disease duration compared with patients not taking statins at diagnosis, suggesting a possible influence of statins on disease progression in PD. Further investigation is warranted to understand the potential beneficial effects of statin treatment on clinical symptoms in PD.
INTRODUCTION
Parkinson’s disease (PD) is a common neurodegenerative disorder affecting 6.2 million people globally and with a prevalence projected to steadily increase in the coming years due to population ageing [1]. Clinically, PD is characterized by a tetrad of motor features—bradykinesia, rigidity, resting tremor and postural instability—attributed mostly to dopaminergic cell loss in the substantia nigra. Nigrostriatal degeneration is accompanied by the intraneuronal accumulation of alpha-synuclein containing Lewy bodies and Lewy neurites in several brain regions, which are the biological hallmark of PD [2]. However, the etiology of PD is poorly understood and the relationship between Lewy bodies and neuronal dysfunction is uncertain. The mechanisms responsible for PD neuronal degeneration are complex and common to age-related diseases, including oxidative stress, excitotoxicity, mitochondrial dysfunction, altered protein handling and neuroinflammation cascade [3].
Despite considerable advances in our understanding of the etiology and pathogenesis of PD over the last years, there are currently no disease-modifying agents or effective interventions that can slow or halt its progression. Beyond their cholesterol lowering action and clear cardiovascular benefits in at-risk populations, it has been theorized that statins have neuroprotective effects in neurodegenerative diseases, such as Alzheimer’s disease (AD) and PD, modulating many cholesterol-independent cellular signaling pathways, which are relevant to their pathogenesis [4]. Statins have attracted considerable attention in view of their ‘pleiotropic effects’, involving antioxidant, anti-inflammatory, immunomodulatory and anti-thrombotic/antiplatelet effects [5, 6]. Furthermore, it has been demonstrated that statins prevent the degeneration of dopaminergic neurons and suppress α-synuclein aggregation
However, individual studies exploring the association between statin use and the incidence and/or progression of PD have produced conflicting results [9, 10], and several authors have argued that the apparent protective effect of statins on risk of PD could be confounded by potential biases, including their effect on serum cholesterol [11]. Indeed, hypercholesteremia seems to be associated with a reduced risk of developing PD, supporting the results of studies suggesting that short-term exposure to statins can increase PD risk [12, 13].
While there is an abundance of literature examining association between statin use and the development of PD [14], how or whether the statin use may affect motor progression is scarcely studied in patients who already have PD. To our knowledge, only two studies so far have investigated the potential effects of statins on the course of PD, yielding conflicting results on motor symptoms [15, 16]. Moreover, contrasting results were obtained in two different, recently completed clinical trials on statins in PD [17, and Carroll CB, unpublished data]..
In this context, the aim of the present observational study was to examine whether long-term statin use influences PD motor progression in a cohort of
MATERIALS AND METHODS
Patient enrolment
We evaluated the 4-year disease course of 181 participants enrolled in a cohort of newly diagnosed, treatment-naïve, early-stage PD patients referred consecutively to the Movement Disorders Clinic of the Neurology Unit at Santa Chiara Hospital, Pisa University (Pisa, Italy) from January 2015 to January 2017. All patients fulfilled the Movement Disorder Society (MDS) clinical diagnostic criteria for PD [18]. Inclusion criteria at baseline were: 1) PD diagnosis within 24 months of first symptom onset; 3) available cholesterol blood measurement taken on the day of their first evaluation; 3) detailed information on lifestyle and cardiovascular risk factors; 4) pre-diagnosis statin use (≥3 years). Serum cholesterol levels were determined in a clinical laboratory using standard clinical methods. Patients were excluded if they had a history of cerebrovascular and psychiatric disorders, or other neurological diseases, including atypical or secondary parkinsonisms. We also excluded patients with significant vascular changes in the subcortical gray and white matter on magnetic resonance imaging (MRI) brain scans. Patients were enrolled in the study if they had undergone a 123ioflupane-fluoropropyl-carbomethoxy-3-beta-4-iodophenyltropane SPECT ([123I]FP-CIT-SPECT) at baseline, confirming the degeneration of the nigrostriatal projections. In particular, we retrospectively identified 42 patients (24 M/18 F) who were taking statins to lower cardiovascular risk or to treat dyslipidemia when diagnosed with PD, and 62 (38 M/24 F) who were not taking any lipid-lowering drugs (Fig. 1).
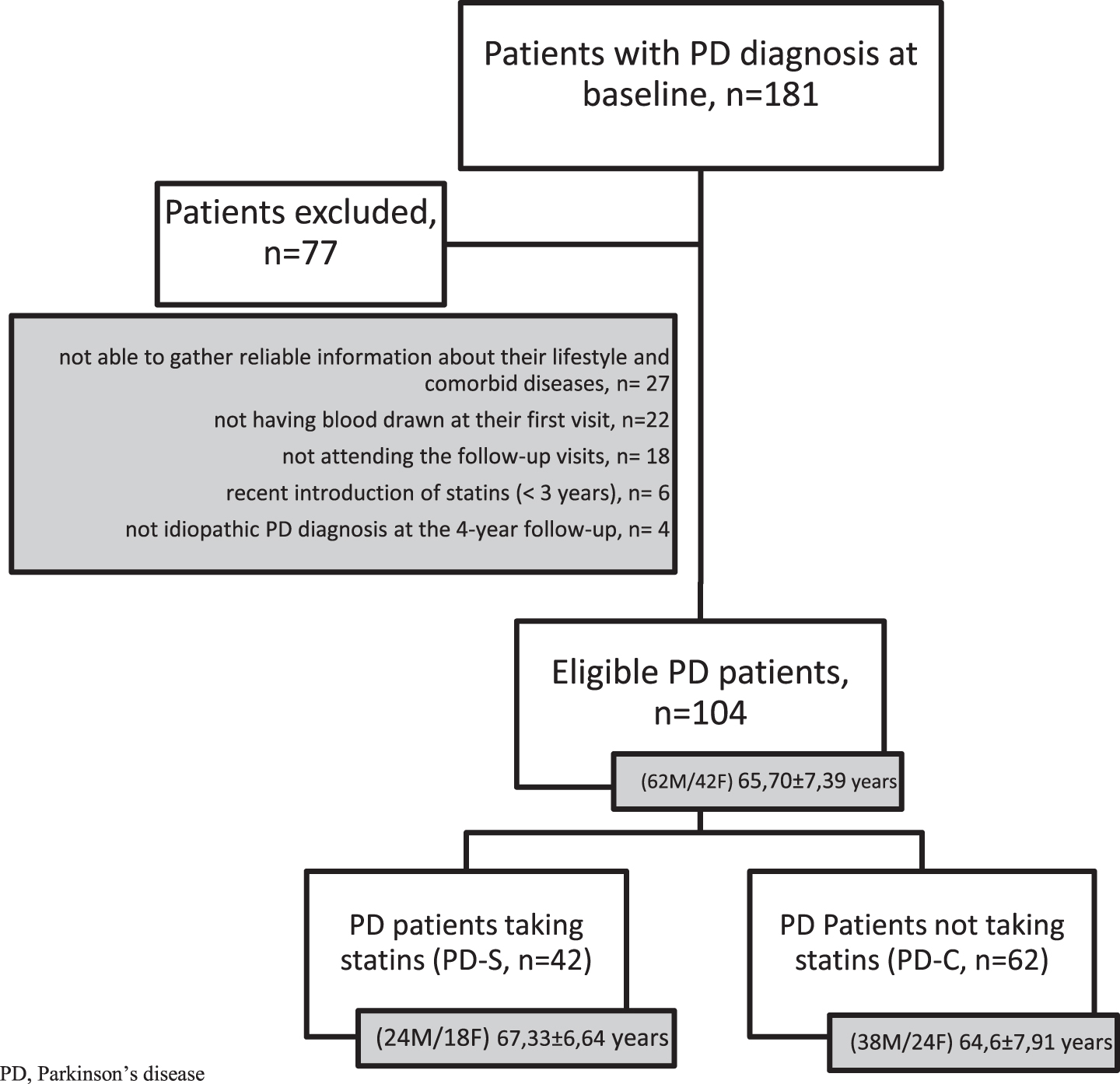
Flowchart of patient inclusion. PD, Parkinson’s disease.
Assessment methods
The enrolled subjects were followed up for 4 years after baseline and all clinical assessments were performed by a movement disorder specialist during the scheduled annual assessments as part of the usual standard of care in our department. Several demographical and clinical data were collected at baseline. In particular, at each visit, motor symptoms were investigated in the off-state using the Unified Parkinson Disease Rating Scale (MDS-UPDRS) part III motor score and cognition was rated using the Mini-Mental State Examination (MMSE) questionnaire. Patients were initially phenotypically classified as Tremor Dominant (TD), Akinetic-rigid (AR) or Mixed according to the clinical judgment of experienced movement disorder specialists. Then, we examined the subscores of the UPDRS motor section, calculating a tremor score from the sum of UPDRS III items 15 (postural tremor of the hands), 16 (kinetic tremor of the hands), 17 (rest tremor amplitude) and 18 (constancy of rest tremor); bradykinesia score from the sum of UPDRS III items 4 (finger tapping), 5 (hand movements), 6 (pronation-supination movements of hands), 7 (toe tapping), 8 (leg agility) and 14 (body bradykinesia); rigidity score from the UPDRS III item 3; postural and gait score from the sum of UPDRS III items 10 (gait), 11 (freezing of gait), 12 (postural stability) and 13 (posture). For dopaminergic therapy, the total levodopa equivalent daily dose (LEDD) was calculated according to a validated conversion table [19].
The local ethical committee approved the study, and all subjects gave their written informed consent.
Statistical analysis
Descriptive statistics were used to describe baseline characteristics. Continuous variables were reported as the mean±standard deviation (SD) while categorical variables were described by the absolute and relative frequencies. Mann-Whitney U and Chi square test, as appropriate, were used to test for between-group differences both at baseline and the 4-year follow-up. Longitudinal changes of the MDS-UPDRS were calculated in both PD-statins and PD-without statins using Wilcoxon signed-rank. A
RESULTS
Results at baseline
A total of 77 patients were excluded if they did not attend the follow-up visits, were re-diagnosed as not having idiopathic PD during the 4-year follow-up period, did not have blood drawn at their first visit or because we could not gather reliable information about their lifestyle and comorbid diseases. Thus, a total of 104 participants contributed to this study and were divided into a group of patients taking statins (PD-S,
The PD-S group had a similar age at onset, gender, motor phenotype at onset and disease severity according to the MDS-UPDRS III compared with the PD-C group (Tab.1). Moreover, there were no statistically significant differences at baseline (T0) between the two groups regarding demographic, other clinical features, SPECT measures, lifestyle habits (including smoking status), cardiovascular risk factors (including homocysteine and glucose blood levels), and concomitant comorbidities (Table 1). Interestingly, both PD-S and PD-C had comparable levels of circulating cholesterol at baseline and after 4 years of follow-up (T1) and neither was there a correlation between serum cholesterol levels and clinical variables measured at baseline and follow-up assessments.
Demographic and clinical features of PD patient at baseline
Data are expressed as mean±SD for continuous variables and as absolute and relative frequency for categorical variables. MAO-B, monoamine oxidase-B; MDS-UPDRS, Movement Disorder Society Unified Parkinson’s Disease Rating Scale; MMSE, Mini-Mental State Examination; PD, Parkinson’s disease; PD-S, PD patients taking statins; PD-C, PD patients not taking statins; SPECT, single-photon emission computed tomography. †Reference range for adults: serum cholesterol < 200 mg/dL; blood homocysteine 4.3–11.1μmol/L; blood glucose 74–109 mg/dL *
18 patients were lost to follow-up, but study completers were not different from dropout patients in terms of baseline characteristics.
As additional analysis, PD patients taking brain penetrant statins (simvastatin and lovastatin users) were compared with patients taking not brain penetrant statins (rosuvastatin and pravastatin users) (Table 2), and each subgroup was then compared with controls (Tables 34), to evaluate whether the ability of statins to cross the blood-brain barrier is associated with better outcomes, both at baseline and follow-up. We separately conducted a subgroup analysis for atorvastatin users because not all studies are concordant about its lipophilicity (Table 5) [20]. Thus, statin arms were not significantly different for any baseline measure, except UPDRS II when compared separately with controls (Tables 34), but PD patients taking simvastatin and lovastatin exhibited lower motor scores than patients taking rosuvastatin and pravastatin (Table 2).
Characteristics of PD patients taking brain penetrant statins (simvastatin/lovastatin) and PD patients taking not brain penetrant statins (rosuvastatin/pravastatin)
Data are expressed as mean±SD. FU, follow up; H&Y, Hoehn & Yahr; LEDD, Levodopa Equivalent Daily Dose; MDS-UPDRS, Movement Disorder Society Unified Parkinson’s Disease Rating Scale; MMSE, Mini-Mental State Examination. †Reference range for adults: serum cholesterol < 200 mg/dL. *Results in bold letters indicate significant difference between groups (
Characteristics of PD patients taking brain penetrant statins and PD-C
Data are expressed as mean±SD. H&Y, Hoehn & Yahr; FU, follow up; LEDD, Levodopa Equivalent Daily Dose; MDS-UPDRS, Movement Disorder Society Unified Parkinson’s Disease Rating Scale; MMSE, Mini-Mental State Examination; PD-C, PD Patients not taking statins. †Reference range for adults: serum cholesterol < 200 mg/dL. *Results in bold letters indicate significant difference between groups (
Characteristics of PD patients taking not brain penetrant statins and PD-C
Data are expressed as mean±SD. H&Y, Hoehn & Yahr; FU, follow up; LEDD, Levodopa Equivalent Daily Dose; MDS-UPDRS, Movement Disorder Society Unified Parkinson’s Disease Rating Scale; MMSE, Mini-Mental State Examination; PD-C, PD Patients not taking statins. †Reference range for adults: serum cholesterol < 200 mg/dL. *Results in bold letters indicate significant difference between groups (
Characteristics of patients taking atorvastatin and PD-C
Data are expressed as mean±SD. H&Y, Hoehn & Yahr; FU, follow up; LEDD, Levodopa Equivalent Daily Dose; MDS-UPDRS, Movement Disorder Society Unified Parkinson’s Disease Rating Scale; MMSE, Mini-Mental State Examination; PD-C, PD Patients not taking statins. †Reference range for adults: serum cholesterol < 200 mg/dL. *Results in bold letters indicate significant difference between groups (
Results at 4 years
Four years after PD diagnosis, all patients were treated with dopaminergic drugs and both groups experienced motor decline as measured by the MDS-UPDRS part III, even though this was more modest in the PD-S group compared with controls. As a consequence, the UPDRS motor score was higher in the PD-C group than in the PD-S (
Characteristics of PD patients with and without statin use after 4-year follow-up
Data are expressed as mean±SD for continuous variables and as absolute and relative frequency for categorical variables. LEDD, Levodopa Equivalent Daily Dose; MDS-UPDRS, Movement Disorder Society Unified Parkinson’s Disease Rating Scale; MMSE, Mini-Mental State Examination; PD, Parkinson’s disease; PD-S, PD patients taking statins; PD-C, PD Patients not taking statins. †Reference range for adults: serum cholesterol < 200 mg/dL. *Results in bold letters indicate significant difference between groups (
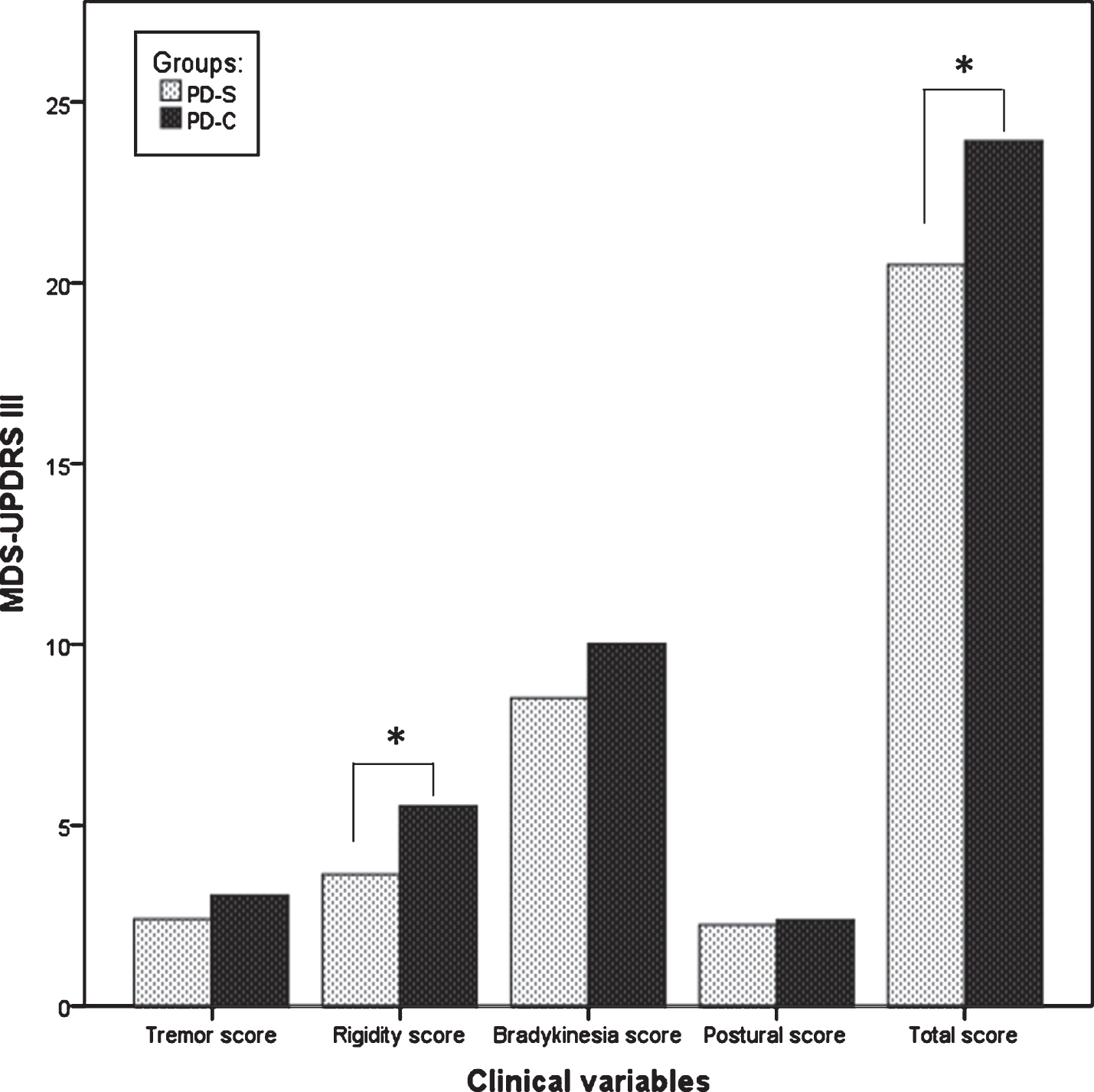
Comparison of the motor symptoms between the two groups of PD after 4-year follow-up. LEDD, Levodopa Equivalent Daily Dose; MDS-UPDRS, Movement Disorder Society Unified Parkinson’s Disease Rating Scale; MMSE, Mini-Mental State Examination; PD, Parkinson’s disease; PD-S, PD patients taking statins; PD-C, PD Patients not taking statins. *
Changes in clinical variables between baseline (T0) and year 4 (T1)
Data are expressed as mean±SD. LEDD, Levodopa Equivalent Daily Dose; MDS-UPDRS, Movement Disorder Society Unified Parkinson’s Disease Rating Scale; MMSE, Mini-Mental State Examination; PD, Parkinson’s disease; PD-S, PD patients taking statins; PD-C, PD Patients not taking statins. †Reference range for adults: serum cholesterol < 200 mg/dL. *Results in bold letters indicate significant difference between groups (
Interestingly, although numbers were small, both the total UPDRS III score and rigidity subscore significantly differed between PD patients taking brain penetrant statins and control group (Table 3), but not between hydrophilic statin users and controls (Table 4). Additionally, while no significant difference was observed in clinical measures among statin subgroups at follow-up, except UPDRS part II, the increases in UPDRS motor scores were lower with simvastatin and lovastatin than with the hydrophilic statins (Table 2).
For participants with atorvastatin there was a lower increase in the tremor and postural score after 4 years of follow-up when compared with controls (Table 5).
DISCUSSION
In this retrospective study comparing disease progression between statin users and non-statin users in a cohort of
The benefit of statins in PD may instead be related to a possible neuroprotective effect [12, 13]. Notably, statins seem to activate multiple biological pathways, including anti-inflammatory responses, reduced glial activation, antioxidant effects and augmentation of endothelial nitric oxide synthase with enhanced cerebral perfusion, which are all events involved in disease mechanisms of relevance to PD [3, 23]. Additionally, statins may have beneficial effects by elevating apolipoproteinA1 levels, which lower plasma concentrations have been associated both with earlier age at onset and greater motor severity in PD patients [24]. Moreover, statins demonstrated a protective effect against the loss of dopaminergic neurons in animal models of PD as well as a reduction of α-synuclein aggregation in
In this scenario, it is not surprising that epidemiologic studies have generally found that statin use is associated with a lower incidence of PD, particularly among younger participants, thus also supporting a strong dose-response relationship with the risk of PD [13, 31–33].
In contrast, only a few studies have investigated the influence of statins on disease progression when PD is already an established disease. A previous interview-based study on 173 patients with PD who used statins, using the Hoehn & Yahr scale, concluded that statins did not seem to affect motor symptoms of PD [15]. Conversely, in their retrospective study, Mutez and colleagues demonstrated that statin therapy was associated with a smaller increase of the LEDD over a period of 2 years in 42 PD patients when compared to controls not on statin therapy prior to diagnosis [16]. No differences in the UPDRS motor score were reported, suggesting good motor control conferred by the higher doses of dopaminergic drugs. However, they also found that statin exposure was associated to delayed onset of PD [16]. Although our findings suggest that statin use may be associated with slower motor progression of PD, we did not observe either a different age of disease onset or a different severity of disease between PD-C and PD-S at baseline. This could argue against a protective effect of statin use, but we cannot exclude the possibility that statins could have prolonged the premotor phase of disease in our patients taking statins. It would have been useful if all patients had clearly started statin therapy at baseline, and the first clinical data regarding ‘statin treatment’ in PD were available over the past few months.
The efficacy of lovastatin 80 mg/day (another lipophilic statin) in slowing PD progression was the focus of a very recent double-blind, randomized, placebo-controlled study, the results of which have suggested a favorable effect of statins in mitigating motor symptom deterioration in early-stage PD patients [17]. Consistent with this, the authors also found that PD patients receiving lovastatin had a slower decline in 18F-DOPA uptake compared with placebo [17]. Interestingly, their postwashout data (week 48 to week 52) showed a convergence between the placebo and lovastatin groups at 52 weeks, implying that lovastatin had a symptomatic rather than a disease-modifying effect on PD [17]. Our study confirms a beneficial effect of statins on motor symptoms of PD with slow progression in terms of motor scores in early-stage PD patients with long-term statin use. Experimental evidence suggests that statins enter the substantia nigra and improve motor function in the MPTP mouse model of PD, supporting the influence of statins on the dopaminergic system [8]. Nevertheless, it should be considered that few cellular and animal studies compared statin concentrations used in these experiments with the mean concentration of statins in human serum at therapeutic doses. Conversely, several studies used a high concentration of statins, which can be deleterious for the nigrostriatum and incompatible with their long-term administration in man [8, 34].
In this study we found that moderate-intensity statin therapy was associated with a lower rigidity score at the follow-up, but not with tremor, posture, balance, and gait scores, which are instead largely secondary to degeneration of nondopaminergic pathways. However, we also observed a slower, yet still linear, progression over 4 years of all tremor, rigidity and bradykinesia subscores in PD patients taking statins compared to those not taking statins. Interestingly, although the incidence of motor complications was not significantly different between groups, we found a lower frequency of motor complications in patients taking statins. It is plausible that neuroprotective effects of statins could also account for protection against fluctuations and dyskinesia but there are currently no data available suggesting that statins may reduce OFF time and dyskinesia associated with standard ‘dopamine replacement’ treatments. It may be that, with proportionally few patients who develop motor complications, there was insufficient power to detect an effect of statin use on wearing-off and dyskinesias. A recent observational study suggested beneficial effects of statins on long term complications of PD showing lower rates of cognitive decline on global scales, semantic fluency and phonemic fluency tests for statin users in a PD cohort followed prospectively from 2 to 6 years [35]. Similarly, statins have also been associated with a reduced risk of dementia and slowed progression of AD [36]. We did not find a significant effect of statin therapy on MMSE in our cohort, but more detailed neuropsychological tests and a longer follow-up could have unveiled differences between statin users and non-users over time.
Based on encouraging data from both cell and animal studies, there were great expectations from the PD-STAT clinical study (NCT02787590) evaluating high-dose Simvastatin in patients with moderate-stage PD patients with a modified Hoehn and Yahr stage≤3.0 (ON state), on dopaminergic treatment and experiencing wearing-off [37]. Unfortunately, the trial has recently been concluded and preliminary results indicated that it was highly unlikely that simvastatin could slow the progression of PD [Carroll CB, unpublished data]. The study population was large (> 200 subjects) but included patients with different disease duration and who were already on dopaminergic medication. In contrast, Lin and colleagues investigated the neuroprotective effects of lovastatin in 77
Of course, our data should not be interpreted as evidence of symptomatic efficacy or neuroprotection but they should further stress the complex relationship between statin use and PD [38]. While waiting for the full published data of the PD-STAT study, we can say that the differential outcome could be attributed to several factors, such as age and gender, PD stage and etiology, vascular risk factors, the type and duration of statin treatment, specific DNA polymorphisms, as well as apolipoproteins and plasma cholesterol levels, which may impede our ability to identify consistent results in this field. Dysregulation of cholesterol homeostasis seems to drive some of the neuropathological features of PD and it is clear that a strong correlation exists between PD and abnormalities in the lipid metabolism [39]. Hence, more research is needed to reveal the exact function of cholesterol in the brain and to understand the full spectrum of statin effects in PD.
In particular, it should be noted that among the individual type of statins, the lipophilic ones, such as simvastatin, could have stronger efficacy than the hydrophilic statins in view of their greater ability to cross the blood-brain barrier [40]. Thus, despite small numbers of patients, we also sought to evaluate if different classes of statins could have a differential effects in terms of efficacy on clinical progression of PD. Subgroup analyses according to blood–brain barrier penetrating statins failed to show a greater protective effect against motor progression of simvastatin and lovastatin than hydrophilic statins (including rosuvastatin and pravastatin), even though it could be attributed to the small number of PD patients in each subgroup. Indeed, use of brain-penetrant statins was associated with a lower increase in UPDRS total and subdomain scores over time than patients taking hydrophilic statins. In addition, the longitudinal differences in UPDRS part III and rigidity subscore between statin users and non-users remained statistically significant only when comparing the lipophilic statin group and controls, suggesting that brain penetrant statins may be the most important determinant of our findings. Actually, due to the small number of patients, we are unable to draw concrete conclusions about possible link between some statins and PD. Nevertheless, it is worth mentioning that our patients were followed for twice as long as those in the PD-STAT [Carroll CB, unpublished data], and four times as long as those in the recent prospective trial of Lin et al. [17]. Another main strength of our analysis is that we took into account baseline cholesterol levels, together with several other potential confounders, such as comorbidities and lifestyle-related factors, all of which can modulate the risk of PD. Additionally, although this is a retrospective study, controls were not matched
To conclude, statins were associated with a potentially slower decline in the progression of motor dysfunction in early-stage PD patients, suggesting that statins might act favorably in modifying the course of PD. Further investigation of the potential role for statins and cholesterol in PD is warranted to confirm our findings. Deciphering the biologic effect responsible for the therapeutic activity of statins in PD may increase our understanding of the pathogenesis of the disease and open up new efficient therapeutic avenues in PD.
Footnotes
ACKNOWLEDGMENTS
We are grateful to the study participants and study team of the Movement Disorders Clinic, Neurology Unit, University of Pisa, who made this research possible.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
