Abstract
Background:
Co-occurrence of β-amyloid (Aβ) pathology has been reported in Parkinson’s disease (PD), and Aβ deposition in the brain may contribute to cognitive decline in patients with PD. Whether striatal dopamine uptake and cognitive status differ with amyloid deposition has been reported in only a few studies.
Objective:
The purpose of this study was to investigate the association among striatal dopaminergic availability, Aβ-positivity, and motor and cognitive status in early and non-demented PD.
Methods:
A total of 98 newly-diagnosed, non-medicated, and non-demented patients with PD were included in this study. Cognitive status was assessed using neuropsychological testing. Patients with mild cognitive impairment (MCI) were stratified into two groups: amnestic MCI (aMCI) and non-amnestic MCI (naMCI). Patient motor status was examined using the Unified Parkinson’s Disease Rating Scale (UPDRS) and positron emission tomography (PET) with 18F-N-(3-fluoropropyl)-2beta-carbon ethoxy-3beta-(4-iodophenyl) nortropane (18F-FP-CIT). All patients also underwent 18F-florbetaben (18F-FBB) PET and were divided based on the results into Aβ-positive and Aβ-negative groups.
Results:
Eighteen patients had Aβ-positivity in 18F-FBB PET and 67 had MCI. Sixteen of 18 with Aβ-positive patients had MCI. The Aβ-positive group had higher frequency of MCI, especially amnestic-type, and lower dopaminergic activities in the left ventral striatum, but not with UPDRS motor score.
Conclusion:
Amyloid pathology was associated with MCI, especially amnestic-subtype, in early and non-demented PD patients and with low dopaminergic activities in the left ventral striatum. This finding suggests that PD patients with Aβ-positivity have AD-related cognitive pathophysiology in PD and associated impaired dopaminergic availability in the ventral striatum can affect the pathophysiology in various ways.
Keywords
INTRODUCTION
β-amyloid (Aβ) is regarded a hallmark of Alz-heimer’s disease (AD). Amyloid is also associated with preclinical (Aβ-positive cognitively normal) and prodromal AD (Aβ-positive mild cognitive impairment, MCI) [1]. Amyloid deposition is a risk factor in patients with amnestic-subtype of MCI (aMCI) and is an independent predictor for conversion to AD [2, 3].
Parkinson’s disease (PD) is the second most comm-on neurodegenerative disease. Neuronal degeneration in PD is accompanied by Lewy bodies, intraneuro-nal inclusions composed of aggregated α-synuclein [4]. The patients with PD dementia have higher levels of cortical and limbic α-synuclein pathology than PD patients without dementia as evidenced by post-mortem research [5]. However, the neuropatho-logical correlates of cognitive impairment in PD are heterogeneous and inconsistent. Co-occurrence of Aβ pathology has been reported in PD [5]. Amyloid in the brain was reported to contribute to cognitive decline in PD patients [6]. Aβ contributed to cognitive decline over time in PD subjects without dementia and Aβ deposition was associated with the severity of cognitive function [7, 8].
Aβ and parkinsonian motor symptoms have also been investigated. Decreased cerebrospinal fluid Aβ was associated with increased risk of freezing of gait and postural instability-gait disturbance motor phenotype [9, 10]. Freezing of gait was associated with decreased cerebrospinal fluid Aβ and caudate dopamine transporter uptake in PD patients [10]. Aβ deposition in dementia with Lewy bodies was associated with reduced dopamine transporter uptake in the anterior putamen and ventral striatum [11].
However, data regarding the association of Aβ with motor and cognitive status in the early, non-demen-ted PD patients are limited. We hypothesized that non-demented. Aβ-positive PD patients might have different motor deficits and cognitive status than Aβ-negative PD subjects. In addition, the striatal subregional dopamine activities in the early, non-demented patients with PD was investigated based on the Aβ-uptake status.
METHODS
Subjects
The study protocol was approved by the Institutional Review Board at Seoul St. Mary’s Hospital, and all subjects provided written informed consent to participate. All experiments were performed in accordance with relevant guidelines and regulations. This study was registered (Identification Number: KCT0005552) in the Clinical Research Information Service (CRIS; http://cris.nih.go.kr), which is an on-line clinical trial registration system established by the Korea Centers for Disease Control and Prevention (KCDC) with support from the Korea Ministry of Health and Welfare (KMOHW) and embodied as a part of the Primary Registries in the World Health Organization (WHO) Registry Network.
Patients with newly diagnosed PD who visited the movement disorder clinic at Seoul St. Mary’s hospital between November 2018 and November 2019 were enrolled. PD was diagnosed based on the UK Parkinson’s Disease Society Brain Bank clinical diagnostic criteria [12] and Movement Disorder Society clinical diagnostic criteria for PD [13]. Patient demographics including age, sex, handedness, motor symptom duration, education years, and medical his-tory of hypertension and diabetes mellitus were collected. Motor symptoms have also been evaluated using the Unified Parkinson’s Disease Rating Scale (UPDRS) [14] and modified Hoehn and Yahr (H&Y) stage score [15]. Symptom laterality index was calculated using the absolute difference of the left- and right-sided motor score (left - right) in UPDRS part III [16].
All patients underwent brain magnetic resonance imaging (MRI), positron emission tomography (PET) with 18F-N-(3-fluoropropyl)-2beta-carbon ethoxy-3beta-(4-iodophenyl) nortropane (18F-FP-CIT), and 18F-florbetaben (18F-FBB) at the time of diagnosis. Apolipoprotein E (APOE) polymorphism was evaluated and the subjects were categorized based on the presence of the APOE ɛ4 allele, because this allele has pivotal role for developing dementia [17].
Patients were excluded based on the following criteria: (1) normal dopamine transporter scan based on the Movement Disorder Society clinical diagnostic criteria for PD [13], (2) dementia based on the clinical diagnostic criteria for probable PD dementia according to the Movement Disorder Society Task Force Level I criteria [18], (3) atypical or secondary parkinsonism, (4) previous stroke or structural lesions on the basal ganglia, (5) history of symptomatic cerebrovascular disease severe enough to cause cognitive decline or impairment in general performance, (6) taking anti-parkinsonian medications or medications that influence striatal dopamine uptake, and (7) taking medications to influence cognitive function.
Neuropsychological test
The neuropsychological evaluations were performed in drug-naïve state, and the tests were rated by experienced psychologists who were blinded to patient clinical and neuroimaging data. Information about memory problems and other subjective cognitive deficits was obtained from caregiver interviews. General cognitive status and dementia severity were evaluated using the Korean version of the Mini-Mental Status Examination (MMSE), the Clinical Dementia Rating (CDR) scale, and Global Deterioration Scale (GDS). All patients were evaluated using comprehensive neuropsychological tests, Seoul Neuropsychological Screening Battery-II, which include 31 subsets for cognition, assessment of activities of daily living (ADL), and cover five cognitive domains [19]. The attention and working memory domains were examined with the Digit Span Forward and Backward, Korean-Trail Making Test-Elderly version (K-TMT-E) Part A, and the Korean-Color Word Stroop Test (K-CWST). Executive function was ass-essed with the Controlled Oral Word Association Test (COWAT) appraising phonemic fluency, Digit Symbol Coding, and the K-TMT-E Part B. Language domain was evaluated with the Korean-Boston Naming Test (K-BNT), and COWAT appraising animal fluency, and visuospatial domain was investigated with the Rey Complex Figure Test (RCFT) and clock drawing test. In the domain of memory, the Seoul Verbal Learning Test (SVLT) and RCFT were utilized for assessing its subdomains: immediate recall, delayed recall and recognition. Each quantifiable neuropsychological test score was converted into a standardized score (z-score) based on age-, sex-, and education-specific norms, and the scores were classified as abnormal when they were below 1.5 standard deviation below the norms. The z-scores of each test were calculated for analyses. In domains with multiple z-scores, the average scores of each domain were defined as the representative values [20]. MCI cases had unimpaired functional activities of daily living, as verified by ADL assessment, and scored 1.5 SD or more below normative data on at least two measures within at least one of the five cognitive domains according to the Movement Disorder Society Task Force Level II criteria (comprehensive assessment) [21]. Depending on the impaired cognitive domain, MCIs were further stratified into MCI with memory impairment (aMCI) and MCI without memory impairment (non-amnestic MCI; naMCI). In addition, patients who had more than two abnormal domains on neuropsychological tests were classified as multi-domain MCI, otherwise, single-domain MCI [21, 22].
PET imaging acquisition and processing
18F-FBB PET images were acquired 90 minutes after intravenous injection of 300 MBq±20% 18F-FBB according to a standard acquisition and imaging processing protocol using a Discovery STE PET/computed tomography (CT) scanner (GE Healthcare, Milwaukee, WI, USA). PET imaging was interpreted by nuclear medicine physicians as either positive or negative based on regional 18F-FBB uptake assessment of the lateral temporal cortex, frontal cortex, posterior cingulate cortex/precuneus, and parietal cortex, as previously described [23].
18F-FP-CIT-PET images were acquired using the same PET/CT sca-nner. Patients underwent intravenous injection (average 3.7 MBq/kg) of 18F-FP-CIT over three hours and brain CT scans were acquired for attenuation correction, followed by a 10-minute 18F-FP-CIT emi-ssion PET scan. PET images were reconstructed in a 512×512×110 matrix with an ordered-subsets expectation maximization algorithm. The voxel size was 0.668×0.668×2 mm. Axial T1-weighted brain MR images with 3D-spoiled gradient-recalled seq-uences (512×512 matrix, voxel spacing 0.469×0.469×1 mm) were also acquired with a 3.0-T sca-nner (Magnetom Verio, Siemens, Erlangen, Germa-ny). Statistical Parametric Mapping 8 software (SPM8; Wellcome Trust Centre for Neuroimaging, London, UK) implemented in MATLAB 2015a (MathWorks, Natick, MA, USA) was used for co-registration and spatial normalization of images and voxel-based comparisons. To spatially normalize 18F-FP-CIT PET images, an MR-guided conventional spatial normalization method was used [24]. PET images were co-registered to individual MR images and spatially normalized to the Montreal Neurolo-gical Institute space with the parameter normal-izing, skull-stripped MR [24]. Volume of interest (VOI) templates of striatal subregions were obtained after subcortical parcellation and partial volume correction using FreeSurfer 5.1 (Massachusetts General Hospital, Harvard Medical School; http://surfer.nmr.mgh.harvard.edu). VOI templates of five striatal subregions and cerebellum were spatially normalized to the MR template and then subregional uptake values of each side of the caudate, putamen, globus pallidus, thalamus, ventral striatum, and cerebellum were calculated using in-house MATLAB 2015a programs (MathWorks, Natick, MA, USA) [24–26]. Mean standardized uptake value ratio (SUVR) was calculated as target SUV divided by cerebellar SUV.
Statistical analysis
Pearson’s χ2 test was used to compare frequencies for categorical variables and the independent sample t-test was used to compare mean differences between the two groups. Individual neurocognitive tests and SUVRs of the two groups were analyzed using analysis of covariance (ANCOVA) after controlling for age, sex, education years, and APOE ɛ4 allele status. All statistical analyses were performed with SPSS software version 24.0 for Mac (IBM Corporation, New York, NY, USA). Statistical significance was set at p < 0.05.
RESULTS
A total of 98 patients with PD were enrolled in this study. Eighteen (18.4%) patients had positive Aβ on 18F-FBB PET based on visual analysis and 67 (68.4%) were MCI. Sixteen of 18 with Aβ-positive patients had MCI. Age at examination, disease duration, education years, sex distribution, and frequency of right handedness, hypertension, and diabetes mellitus were similar between Aβ-positive and Aβ-negative groups. UPDRS and modified H&Y stage scores were not different between the two groups and motor symptoms were not different between left and right sides.
The proportion of normal cognition in the Aβ-positive group was significantly lower than in the Aβ-negative group (Aβ-positive; 2/18, 11.1% vs. Aβ-negative; 29/80, 36.3%). Among MCI patients, the proportion of aMCI in the Aβ-positive group was significantly higher than in the Aβ-negative group (Aβ-positive; 81.3% vs. Aβ-negative; 51.0%). The Aβ-positive group had higher scores on CDR, sum of box of CDR, and GDS than the Aβ-negative group, and the MMSE score tended to be lower in the Aβ-positive group than in the Aβ-negative group, although without statistical significance. The APOE ɛ4 carrier was more frequent in the Aβ-positive group (Table 1).
Clinical characteristics
Values represent mean with standard deviation (SD) or numbers of patients (percentage). Analyses were performed using the independent t-test and *χ2 test. UPDRS, Unified Parkinson’s Disease Rating Scale; H&Y, Hoehn and Yahr; MCI, mild cognitive impairment; aMCI, amnestic mild cognitive impairment; naMCI, non-amnestic mild cognitive impairment; MMSE, Mini Mental Status Examination; CDR, Clinical Dementia Rating; GDS, Global Deterioration Scale; APOE, apolipoprotein E.
In comparison of neuropsychological test results, the Aβ-positive group had lower composite z-scores for language (p = 0.002), memory (p = 0.001), and frontal/executive function domains (p = 0.014) than the Aβ-negative group, however, differences were not found in attention and visuospatial composites (Table 2). In individual cognitive tests, the Aβ-pos-itive group exhibited lower z-scores in the Korean Boston naming test, verbal and visuospatial memory tests, Controlled Oral Word Association Test, Trail Making Test part B, and Digit Symbol Coding Test than the Aβ-negative group (Supplementary Table 1).
Neuropsychological domain composite z-scores of the two groups
Values represent mean with standard deviation. Analyses were performed using the independent sample t-test.
The SUVRs of 18F-FP-CIT PET imaging were also analyzed. The Aβ-positive group showed a lower SUVR in the left ventral striatum than the Aβ-negative group (4.57±0.93 vs. 5.34±1.11; p = 0.031). In other subregions including caudate nucleus, putamen, globus pallidus, right ventral striatum, and thalamus, the SUVRs did not differ between the two groups (Table 3, Fig. 1).
Comparison of SUVRs in the two groups
Values represent mean with standard deviation. Analyses were performed using analysis of covariance (ANCOVA) test after controlling for age, sex, education, and apolipoprotein E (APOE) ɛ4 allele; SUVRs, standardized uptake value ratios; *< 0.05.
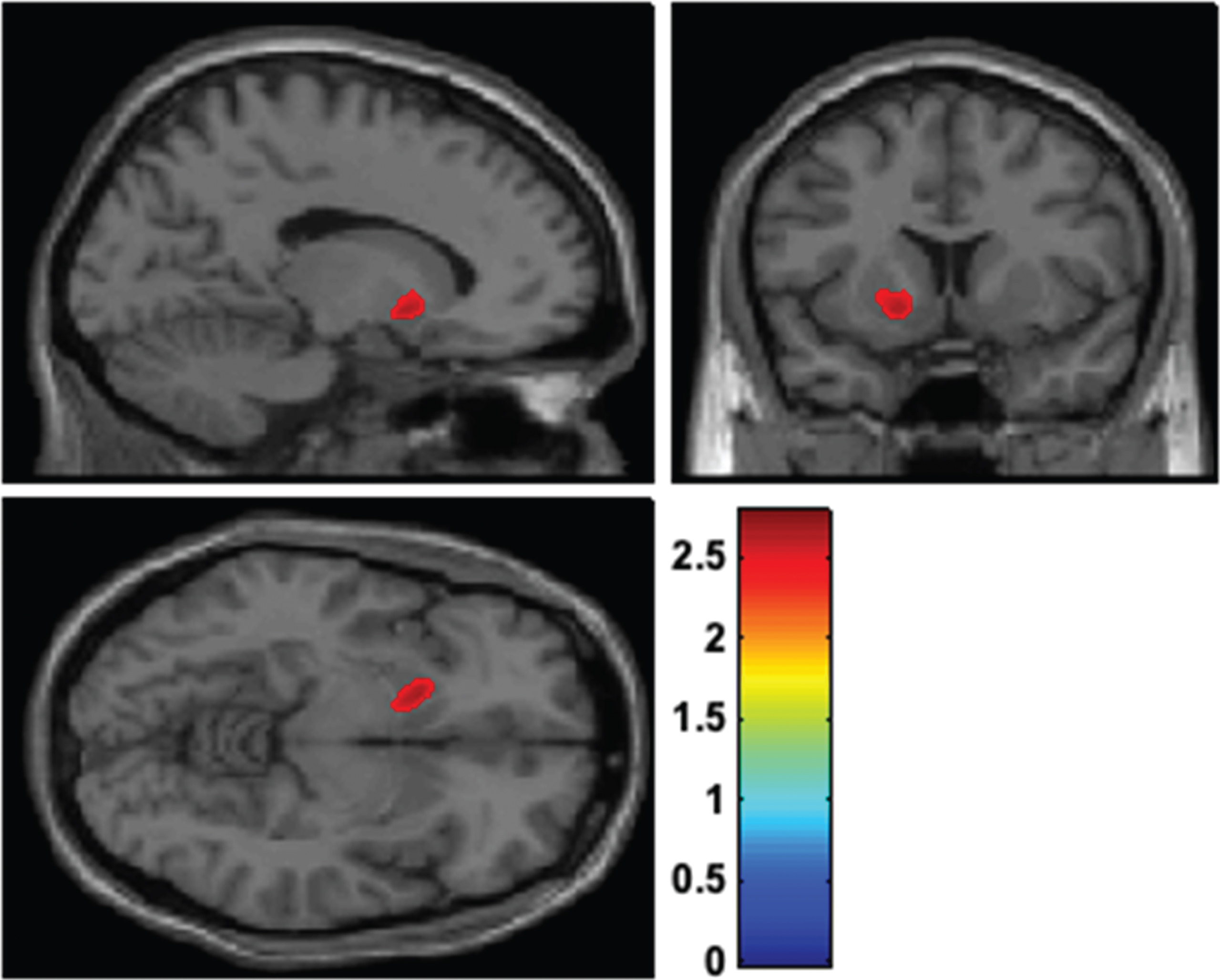
Voxel-based comparison of 18F-N-(3-fluoropropyl)-2beta-carbon ethoxy-3beta-(4-iodophenyl) nortropane positron emission tomography (18F-FP-CIT PET) after controlling for age, sex, education, and apolipoprotein E (APOE) ɛ4 allele status between Aβ-positive and Aβ-negative groups. The differences in the standardized uptake value ratios (SUVRs) between the two groups are shown in the colored region. The SUVR in the left ventral striatum of Aβ-positive patients was more decreased than in Aβ-negative patients. T values are indicated by the color bar.
DISCUSSION
In the present study, the differences of cognitive status and striatal monoamine transporter availability in Aβ-positive and Aβ-negative groups were investigated. Aβ-positivity was associated with cognitive impairment in early and non-demented PD patients; approximately 89.9% of Aβ-positive PD patients had MCI and mainly had amnestic-subtype. The Aβ-positive group had poorer cognitive performances in language, memory, and frontal/executive domains, and the difference was significant especially in verbal and visuospatial memory among the neurocognitive tests. Aβ-positive patients showed more reduced dopaminergic activities in the left ventral striatum although they had similar motor deficits compared with patients in the Aβ-negative group.
Recent in-vivo imaging techniques showed in the subgroup of MCI subjects with memory impairment, that aMCI is a likely prodrome of AD and carries very high risk of progression to AD compared with naMCI [2, 3]. aMCI represents a transitional state between non-pathological brain aging and the severe cognitive pathology of AD [27]. In this study, Aβ-positivity was more frequently found in patients with aMCI (13/39; 33.3%) than in patients with naMCI (3/28; 10.7%) and normal cognition (2/31; 6.5%). This finding suggested an association that Aβ pathology, in early, non-demented PD, can lead to cognitive impairments through AD-associated pathophysiology. However, this correlation does not imply causation. The possibility should be considered that these patients also have a higher cortical (or subcortical) alpha-synuclein burden which might predominantly account for their greater cognitive impairment [28, 29]. Recent study found contrasting result that Aβ deposition was not associated with cognitive impairment and dementia in patients with PD [30].
In the present study, decreased dopamine transporter uptake was observed in the left ventral striatum in Aβ-positive patients, and more severe cognitive impairment was associated with Aβ positive PD patients. Because decreased dopamine transporter in the striatum expresses nigrostriatal dopamine cell loss, striatal dopamine can also be a surrogate marker of the pathological aggregation of α-synuclein in the basal ganglia [31]. Lewy bodies and amyloid pathologies were associated with earlier onset of dementia in PD [32].
The dopamine of ventral striatum is considered to play an important role in cognition. The verbal and visual working memory input to the basal ganglia arises from the posterior parietal cortex and inferior temporal cortex, and is located in the nucleus accumbens, which is the main part of the ventral striatum, then sends a feedback signal to the anterior cingulate cortex if the response is incorrect [33]. The ventral striatum also receives inputs from the prefrontal cortex and hippocampus, thus, reduced dopaminergic signal in the ventral striatum is associated with processing impairment of visual and verbal memory in normal aging [34]. In the Tg2576 mouse model of AD, dopamine neuronal loss in the ventral tegmental area resulted in lower dopamine level in the nucleus accumbens via the mesocorticolimbic dopaminergic pathway and caused memory impairment [35]. In addition, because the ventral striatum receives a dopamine input originating from the frontal cortex, it is connected to the frontal/executive function including attention, action selection, decision making, conflict resolution, perception, behavioral flexibility, and reward processing [36]. The striatum also may affect language processing by maintaining morphemes and grammars [37]. In a functional MRI study, the ventral striatum was activated when the meaning of new words was successfully learned [38]. During word learning, the ventral striatum showed enhanced functional and structural connectivity associated with neocortical language areas [38]. In another study, the ventral striatum was suggested to function in language processing and computation [39]. In PD-MCI patients, dopamine transporter activity was decreased in the caudate, anterior putamen, and ventral striatum compared with PD patients with normal cognition [40]. In patients with dementia with Lewy bodies, the Aβ-positive group had decreased dopamine transporter uptake in the anterior putamen and ventral striatum [11]. Therefore, potentially, the cortical or limbic burden of Lewy bodies reflected on the left ventral striatum dopamine transporter activity could play an important role in memory dysfunction and occurrence of aMCI in PD patients with Aβ pathology. However, in this study of patients, we could not determine why the dopamine transporter activity differed, especially in the left striatum. There were no differences in the handedness and symptom laterality index between groups.
In the present study, APOE ɛ4 carriers were more frequent in the Aβ-positive group than in the Aβ-negative group. APOE ɛ4 is the main genetic det-erminant of AD risk [41]. In previous studies, the APOE ɛ4 allele was associated with faster progression of MMSE score and a risk for susceptibility to dementia in PD [42, 43]. In the present study, the APOE ɛ4 allele was associated with memory domain deficits and occurrence of aMCI and Aβ-positivity.
The present study had several limitations. The number of patients in the Aβ-positive group was relatively small, and the frequency of MCI in the study population was relatively high. In this study, we only enrolled patients at an early disease stage with short disease duration. Early cognitive decline in the subjects of this study might reflect a particular population within a spectrum of disorders between PD and dementia with Lewy bodies [44]. We tried to reduce selection bias by applying strict clinical diagnostic criteria for PD, however, it can be difficult to differentiate PD from dementia with Lewy bodies at early stage. In addition, there is considerable debate in distinguishing PD-MCI, PD-dementia, and dementia with Lewy bodies. This study was conducted based on the point prevalence at a specific study period, and therefore selection bias may be present. Second, the PD patients in this study had many systemic diseases, such as hypertension and diabetes mellitus, which are also associated with amyloid deposition and cognitive impairment. Third, the subregional analysis of 18F-FBB PET was not conducted. The quantitative analysis with multiple comparisons between amyloid uptake in the brain and dopaminergic activity in the striatum would provide more information on these associations.
To the best of our knowledge, there has been few studies in which the cognitive status and dopamine transporter activity were investigated based on Aβ uptake. In this study of early, non-demented patients with PD, Aβ pathology was associated with occurrence of aMCI and more memory domain impairment and associated with reduced dopaminergic activity in the ventral striatum irrespective of motor function. This finding suggests that PD patients with Aβ-positivity have AD-related cognitive pathophysiology in PD. The effects of Aβ on the progression and conversion to dementia in patients with PD may be determined in future long-term follow up studies.
CONFLICT OF INTEREST
The authors have no conflicts of interest.
Footnotes
ACKNOWLEDGMENTS
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT (NRF-2017R1D1A1B06028086 and NRF-2019R1G1A1099554). This study was also supported by the Catholic Medical Center Research Foundation made in the program year of 2019 and by Research Fund of Seoul St. Mary’s Hospital, The Catholic University of Korea made in the program year of 2020.
