Abstract
Background:
Scientific research increasingly focuses on visual symptoms of people with Parkinson’s disease (PD). However, this mostly involves functional measures, whereas self-reported data are equally important for guiding clinical care.
Objective:
This review provides an overview of the nature and prevalence of self-reported visual complaints by people with PD, compared to healthy controls.
Methods:
A systematic literature search was performed. Studies from three databases (PubMed, PsycInfo, and Web of Science) were screened for eligibility. Only studies that reported results of visual self-reports in people with idiopathic PD were included.
Results:
One hundred and thirty-nine eligible articles were analyzed. Visual complaints ranged from function-related complaints (e.g., blurred vision, double vision, increased sensitivity to light or changes in contrast sensitivity) to activity-related complaints (e.g., difficulty reading, reaching, or driving). Visual complaints were more prevalent in people with PD compared to healthy controls. The presence of visual complaints leads to a reduced quality of life (QoL). Increased prevalence and severity of visual complaints in people with PD are related to longer disease duration, higher disease severity, and off-state.
Conclusion:
A large proportion of people with PD have visual complaints, which negatively affect QoL. Complaints are diverse in nature, and specific and active questioning by clinicians is advised to foster timely recognition, acknowledgement, and management of these complaints.
Keywords
INTRODUCTION
Parkinson’s disease (PD) is a neurodegenerative disorder characterized by a number of well-known motor and non-motor symptoms, including visual problems [1–5]. During recent years, visual symptoms in PD have received increasing attention in the literature. In clinical practice however, these symptoms are still underrecognized. Visual symptoms are less conspicuous than motor symptoms, and may therefore be overlooked [6, 7]. However, visual symptoms have a substantial impact on the daily lives of people with PD and may already occur in the early and prodromal stages of the disease [1, 8]. Consequently, several studies state the importance of tools to improve the recognition, diagnostic accuracy, and management of non-motor symptoms, including visual symptoms, in clinical practice [2, 9–11].
To date, most tools used to assess visual symptoms are quantitative tests of visual functions. These measurements, in most cases part of an ophthalmological assessment, objectify functions and impairments, such as visual acuity and visual field loss. In contrast, few studies include self-report data, asking people to reflect on their own visual functioning. It is acknowledged that both functional and self-reported data are required to obtain a clear picture of the impact of PD on individuals [9], which is necessary to guide care and rehabilitation [12, 13].
Despite the importance of self-reported data, no reviews have been published on this topic so far. Therefore, this paper presents a systematic review on self-reported visual complaints in PD, compared to healthy controls.
METHODS
Literature search
A literature search was performed according to the guidelines of Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) [14]. The search encompassed the scientific databases PsycInfo, PubMed, and Web of Science (Core Collection) and was performed on literature up to the date of February 5, 2021. The search terms are presented in Table 1. Two complementary searches were performed. The first one focused on subjective dysfunction in general, since the current review was part of a project that also examined self-reported cognitive complaints of people with PD. The second search addressed self-reported visual complaints specifically.
Search terms for literature search
aTerm should be present in title or abstract, bOne of the terms should be present in the content of the paper.
Eligibility criteria
Only papers fulfilling the following criteria were included: written in English, published in peer-reviewed academic journals, including human participants with idiopathic PD, including a self-report measure for visual functioning, and data (i.e., prevalence) of this measure for the group of people with PD or a comparison (i.e., prevalence or score) with healthy controls. Visual self-report measures were defined as items, subscales or total scores representing an individual’s reflection on their own visual functioning, performance of visual activities or vision-related quality of life (QoL). We made the assumption that a self-report measure was filled out by the patient, unless otherwise stated. Longitudinal or intervention studies were included if relevant baseline measures were presented. Review articles and case reports were excluded, as well as meeting abstracts and letters to editors. Since visual hallucinations in PD are widely recognized and extensively studied [15], these were not incorporated in this review.
Literature extraction
Figure 1 shows the PRISMA outline of papers identified through the literature search. Articles were first screened by title and abstract, and when they seemed relevant, reviewed by the full text. In addition to the database search, reference lists of included papers were screened according to the predefined inclusion criteria. Two authors (IvdL and FH) and a research assistant (EV) systematically judged the eligibility of articles and discussed this in case of doubt. In case uncertainty remained, a third author (GdH) was involved until consensus was reached.
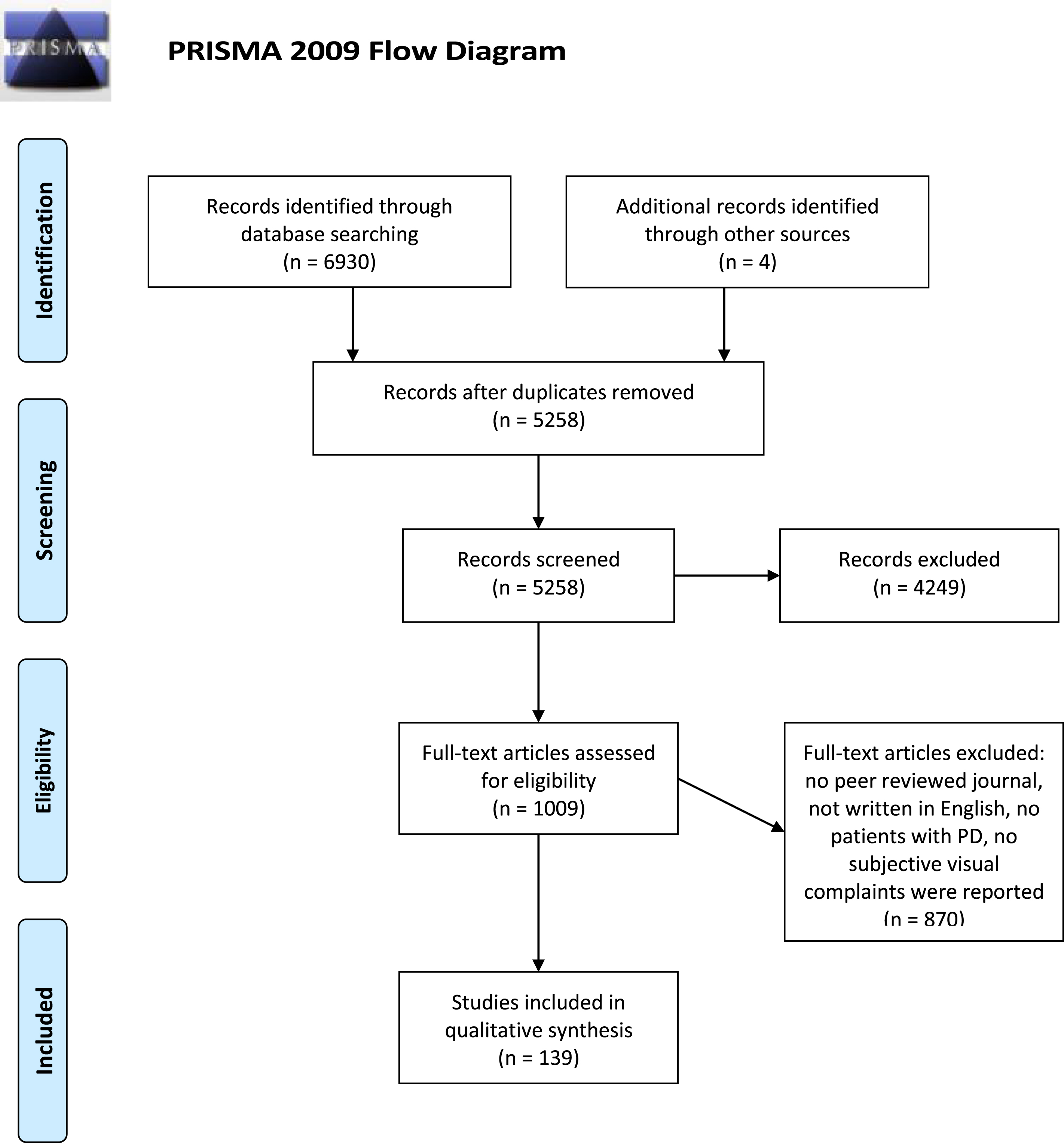
PRISMA flow diagram of systematic literature search [14].
Analysis
Relevant results were extracted from included papers. Results were categorized per complaint. Complaints were divided amongst three categories: function-related complaints, activity-related complaints, and vision-related QoL. Results were ordered based on the number of studies investigating each complaint. Based on this systematic categorization, a descriptive table was included (Table 2). If data on subgroups of PD were reported (e.g., tremor dominant versus akinetic-rigidity subtypes, or young versus late onset PD), but no total prevalence, it was calculated by the authors. The weighted prevalence of a complaint was calculated for studies using the same method to assess the complaint. Relationships of self-reported visual complaints with demographics and disease-related variables were gathered in an additional table (Table 3).
Overview of the prevalence of visual complaints in people with PD and the comparison to healthy controls
Per complaint, each self-report measure used to describe this complaint is presented individually, along with the number of visual items it includes and the criterion that must be fulfilled for people to belong to the group with visual complaints. Weighted averages (prevalence of visual complaints) are presented for each complaint. References to the studies are placed in brackets []. ADLs, activities of daily living; CIRS-G, Cumulative Illness Rating Scale-Geriatric; CISS, Convergence Insufficiency Symptom Survey; COMPASS, Composite Autonomic Symptom Score; ICF, International Classification of Functioning; N, number/population size; N/A, not available; NMSQ, Non-Motor Symptoms Questionnaire, NMSS = Non-Motor Symptom Scale, OSDI = Ocular Surface Disease Index, PD = Parkinson’s disease, PRO-PD = Patient Reported outcomes in Parkinson’s Disease; QoL, quality of life; SCOPA-AUT, SCales for Outcomes in PArkinson’s disease– autonomic dysfunction; VAQ, Visual Activities Questionnaire; VFQ-25, Visual Function Questionnaire (25-item); VIPD-Q, Visual Impairment in Parkinson’s Disease Questionnaire. a Difference in score or prevalence between the groups, b Weighted prevalence of complaints on the ICF functions ‘seeing functions’ (72.9%) and ‘difficulty with watching’ (78.2%), c Items that may to some extent have arisen from motor symptoms (e.g., bumping into objects and freezing in narrow spaces) were included in the analysis. Prevalence was 57% after removing these items, dKhedr et al. (2012) [90] & Khedr et al. (2013) [55], and Giorelli et al. (2014) [65] & Giorelli et al. (2014) [66] used the same group of people with PD and reported the same results. For this reason, results of only one of these studies was reported, e Results only account for the non-tremor group (N = 12), f Results only account for the tremor group (N = 13), g Weighted prevalence of complaints on size (17%) and shape perception (21%), h Weighted prevalence of reading related complaints: ‘letters disappear while reading’ (20% in patients and 5% in controls) and ‘read better with one eye closed’ (17% in patients and 6% in controls), iWeighted prevalence of the complaints freezing (60% in patients and 3% in controls) and difficulty at doorways (56% in patients and 3% in controls) [119].
Relationship of demographic and disease-related variables with visual complaints
Supplementary Table 2 provides an overview of investigated relationships per complaint. >, first group reported more visual complaints than second group; ADL, activities of daily living; DBS, Deep Brain Stimulation; LEDD, Levodopa Equivalent Daily Dose; N, number of studies; PIGD, postural instability and gait disorder; QoL, quality of life. aSide of onset influenced the relationship of gender with difficulty estimating spatial relations; the difference between males and females was bigger (and significant) for the left onset group than right onset group (not significant), bDisease severity measured by Hoehn & Yahr classification and Clinical Impression of Severity Index for Parkinson’s Disease (CISI-PD) [151], cDisease severity measured by Cumulative Illness Rating Scale – Geriatrics (CIRS-G) [151], dOnly significant when adjusted for disease duration and dopaminergic treatment duration and dose [70], eResult accounts for a group of patients with late onset PD [79], fResult accounts for a group of patients with young onset PD [79], gNot significant when adjusted for age, gender, disease duration, disease severity, dopamine agonist, and levodopa [33], hMore colorful dreams were reported after using Pergolide, and relaxation of the eye muscles and blurry vision after using Benzhexol [119].
RESULTS
The search procedure resulted in 139 relevant articles. Table 2 presents the prevalence of visual complaints reported by people with PD and the comparison with healthy controls. One hundred and thirty-one studies investigated the prevalence of visual complaints in people with PD. Some studies reported on more than one complaint, resulting in 185 prevalence numbers. Forty-four studies compared the presence of visual complaints between people with PD and healthy controls. Some studies reported group differences for multiple complaints, resulting in 122 numbers on group comparisons. Of these, 102 (83.6%) indicated that people with PD reported more complaints than healthy controls, whilst 20 (16.4%) found either no difference between these groups or reported healthy controls to experience more complaints than people with PD.
Function-related visual complaints
Visual complaints in general
Seventeen studies [16–32] reported on visual complaints in general. Six of these studies compared people with PD with a control group. In all cases, the prevalence of visual complaints was higher among patients than among controls [16, 26]. One study examined the number of complaints experienced. Three out of four patients (77.3%) experienced at least one complaint, and 43.2% experienced multiple visual complaints [25]. Only 31.6% of healthy controls reported a visual complaint [25].
Double vision
Sixty-one studies reported relevant results on double vision based on the Non-Motor Symptoms Questionnaire (NMSQ) [33–93] and 24 studies based on the Non-Motor Symptom Scale (NMSS) [9, 94–115] (see Supplementary Figure 1). The prevalence ranged from 2.9% [72] to 23.5% [59] in studies with a sample size≥150. Studies with a smaller sample size (N < 150) showed a range from 0% [35, 94] to 60.9% [45]. One study found that for most people with PD experiencing double vision (9.6% of total sample) complaints started after motor symptoms had manifested. Only a small proportion (1.8% of total sample) already experienced double vision before the onset of motor symptoms [71].
Most studies (15/18) on double vision showed a higher prevalence in people with PD compared to controls [16, 116–119]. One study reported prevalence numbers in more detail and showed that 16% of people with PD experienced double vision ‘often’, and 36% ‘sometimes’ [118].
Increased sensitivity to light
Nineteen studies included the SCales for Outcomes in PArkinson’s disease–autonomic dysfunction (SCOPA-AUT) [120] and reported relevant results for the item on oversensitivity to light in people with PD [120–138]. The prevalence ranged from 1.9% [130] to 61.0% [120] (see Supplementary Figure 2). Most studies (13/15) showed that the prevalence was higher in people with PD compared to controls [25, 138–140].
Other light-related complaints were more prevalent in people with PD as well. People with PD had significantly more difficulty adapting to changing light conditions than controls (patients: 27% vs. controls: 11%) [117], and experienced reduced night vision more often (patients: 16%–47.6% vs. controls: 3%–18.5% [117, 140]).
Altered color vision
Prevalence of altered color vision did not significantly differ between people with PD and healthy controls in the majority of studies [18, 141–143]. Three studies indicated a trend toward more complaints among people with PD [18, 118]. Only one study found that significantly more people with PD (11%) reported the perception of colors to be more pale than controls did (3%) [117]. Another study found that 10% of people with PD perceived less color than before, while 5% perceived more color [118].
Painful, dry or watery eyes
Complaints related to painful, dry or watery eyes were more frequently reported by people with PD than by controls (5/6 studies [18, 145]). Also, severity seems to differ. People with PD had ‘moderate dry eye disease’ and controls ‘mild dry eye disease’ [145]. It was reported that 28% of people with PD had a burning sensation or gritty feeling in their eyes, and only 8% of controls [117]. While some studies could not confirm significance [25, 26], painful eyes were present in 27.3% of people with PD compared to 22.4% of controls [25]. Watery eyes were more frequently reported by people with PD (26.8%–31%) than controls (15%) [24, 117].
Blurred vision
The prevalence of blurry vision was higher in people with PD compared to healthy controls (4/4 studies [25, 146]). Patients experiencing blurry vision indicated that their vision was worse in the dark and they needed more light to see clearly [119].
Changes in contrast sensitivity
Changes in contrast sensitivity were more often reported by people with PD compared to controls (2/2 studies [117, 118]). Roughly a quarter of people with PD (27%) reported changes in the perception of contrast (23% perceived less contrast, 4% perceived more contrast) [118]. People with PD (26%) reported more difficulty reading plain text on a colored or gray background compared to controls (10%) [117].
Changes in size, shape, or depth perception
People with PD reported more complaints regarding size, shape, or depth perception than controls [117, 119]. Seventeen percent of people with PD experienced difficulty with size, shape, and depth perception, compared to 2% (depth) – 5% (size/shape) of controls [117].
Peripheral vision
People with PD experienced significantly more limitations in peripheral vision (i.e., ‘noticing objects off to the side’) than controls [18, 26]. People with PD (43%) indicated to need to turn their head to the side to see peripheral objects more often than controls did (14%) [119].
Difficulty estimating spatial relations
Half of the patients (50%) reported misjudging spaces (5% ‘often’ and 45% ‘sometimes’), compared to 28% of controls [118]. Most people with PD (27%) indicated that spaces appear smaller, while 17% noted that spaces seem larger.
Visual field impairment
People with PD experienced visual field impairments more often (6.1% [24] – 9% [117]) than healthy controls (2% [117]).
Changes in motion perception
Changes in motion perception were reported by a significantly higher number of people with PD (19%–25%) than controls (3%) [118, 119]. Eight percent of people with PD had difficulties ‘often’, and 17% ‘sometimes’. Most people with PD perceived motion as faster (13%), while few perceived motion to be slower (4%). Others perceived motion to be less smooth (3%) [118]. In addition, people with PD had more difficulty following quick movements with their eyes (patients: 25% vs. controls: 4%) [117].
Activity-related visual complaints
Reading difficulties
People with PD reported more difficulty reading than controls (4/4 studies [16, 117]). While one study could not confirm significance, a higher prevalence was found in people with PD (26.7%) compared to controls (9.7%) [16]. Compared to controls, people with PD reported significantly more often that letters disappeared while reading (patients: 20% vs. controls: 5%) and that they had fewer reading difficulties when having one eye closed while reading (patients: 17% vs. controls: 6%) [117].
Near and distant visual activities
People with PD reported more problems with near (patients: 44.3% vs. controls: 25.5%) and distant visual activities (patients: 38.6% vs. controls: 15.0%) than controls [21]. All five studies confirmed this result [18, 148]. Most individuals who had difficulty working nearby reported that their eyes felt tired or uncomfortable [147, 148]. In contrast, people with PD ‘often’ do not have trouble judging close (95%) or distant objects (87%), comparable to healthy controls, of which 96% and 99% did not have problems judging close or distant objects, respectively [118].
Difficulty/freezing in narrow spaces
People with PD had more difficulty in narrow spaces than controls (4/4 studies [25, 119]). Walking through narrow spaces was difficult for 70% of people with PD, compared to 1% of controls [118]. Eighteen percent of people with PD indicated to have difficulties ‘often’, and 52% ‘sometimes’. People with PD indicated to freeze (26%), bump (29%), feel uneasy (10%), or have other problems (3%) while walking through narrow spaces.
Freezing (patients: 60% vs. controls: 3%) and difficulty at doorways (patients: 56% vs. controls: 3%) was also significantly more common in people with PD compared to controls [119]. Half of the people with PD reported to bump into objects [17].
Driving
People with PD experienced more driving difficulties compared to controls (3/3 studies [18, 149]). This could be attributed to poor vision, especially at night. Along with that, people with PD reported difficulties with reading maps or using navigational devices [17]. Besides finding driving more difficult in low visibility conditions (patients: 21.6% vs. controls: 2.7%), people with PD avoid these situations more frequently compared to controls (patients: 35.1% vs. controls: 5.4%) [149].
Difficulty reaching for objects
People with PD reported significantly more difficulty reaching for objects (46%) than controls (3%). Nine percent of people with PD had difficulties ‘often’, and 37% ‘sometimes’. Most patients indicated to not reach far enough (27%), while others reached too far (7%) or reached off to one side of the object (8%) [118].
Vision-related QoL
People with PD scored significantly worse than controls on vision-related QoL (Visual Function Questionnaire; VFQ [150]) [18, 26]. Social functioning and mental health are influenced, role difficulties exist, and people with PD are more dependent on others due to their visual problems. Except for role difficulties, vision-related QoL was influenced to a greater extent in people with PD than in controls.
Relationship of demographic or disease-related variables with visual complaints
Table 3 reports on the relationship of demographic and disease-related characteristics with self-reported visual complaints. Visual complaints did not relate to age (3/4 studies [66, 122]) and gender (12/13 studies [34, 122]). Most studies did find a relationship with disease duration (6/9 studies [32, 137]) and severity (12/18 studies [18, 152]), indicating that visual complaints were more prevalent in patients with a longer disease duration and a more severe disease. Dominant symptoms of the disease (7/9 studies [50, 142]), as well as side of onset (1/1 study [81]) did not relate to visual complaints.
A higher Levodopa Equivalent Daily Dose (LEDD) was not related to visual complaints (5/7 studies [54, 132]), while patients did report more complaints during off-state (3/3 studies [30, 119]). Some medicaments (i.e., Pergolide and Benzhexol) led to visual adverse effect (1/1 study [119]). Deep Brain Stimulation (DBS) was found to relieve double vision (2/2 studies [22, 61]).
The relationship of visual complaints with objectified visual disorders and non-motor symptoms was also investigated (see Supplementary Table 1). Double vision was related to abnormal ocular alignment and aberrant movements of the eyes [116]. Regarding painful or dry eyes, decreased tear production, abnormalities in Meibomian glands, and ocular surface problems could confirm the complaint [16, 145], but it was not related to decreased tear film break-up time or tear osmolarity [145]. Visual complaints were not related to cognitive functioning (3/4 studies [102, 151]), but it did co-occur with psychotic symptoms (e.g., hallucinations; 3/3 studies [24, 151]).
Visual complaints were found to negatively relate to the performance of activities of daily living (ADL) and QoL (5/6 studies [117, 151]).
Supplementary Table 2 presents an overview of investigated relationships with demographic and disease-related variables per visual complaint (an aid for understanding Table 3 and Supplementary Table 1).
DISCUSSION
This systematic review aimed to provide an overview of the literature on self-reported visual complaints in people with PD. Visual complaints occur in a wide variety in people with PD. Moreover, there is a fundamental difference between people with PD and healthy controls. More than 80% of comparisons showed that visual complaints were more common in people with PD than in controls.
According to the literature found, the prevalence of visual complaints varies greatly. Nevertheless, we can conclude that visual complaints were more often present in people with PD than in controls. Visual complaints in general appear to occur in at least a quarter of people with PD, with prevalence ranging from 26.8%–98%. Other complaints with a relatively high prevalence are blurred vision (19.7%–46%), watery eyes (26.8%–31%), light-related complaints (reduced night vision (16%–47.6%) and light/dark adaptation (27%)), ocular fatigue (43.6%), and all activity related complaints (> 21.6%; i.e., reading, driving, reaching, near or distant activities, and difficulty/freezing in narrow spaces). Complaints that appear less prevalent are altered color vision (4.1%–12.7%), and visual field deficits (6.1%–9%).
Vision-related QoL in people with PD appears to be affected in a variety of ways. This is not surprising given that vision is essential in performing a wide variety of activities. Moreover, people with PD need their vision to compensate for their motor loss [6]. Therefore, vision loss may lead to even more difficulties in daily life.
Strengths and limitations
We conducted a systematic literature search and selected all articles that met the inclusion criteria. By involving at least three researchers in the process of data extraction, we sought to ensure reliable and standardized data extraction.
We aimed at providing a comprehensive overview of the literature on self-reported visual complaints in PD. The inclusion of lower quality studies may have biased the results. The prevalence of a complaint was sometimes determined by only few studies and small sample sizes (e.g., asthenopia prevalence was determined in 39 patients [152]). Moreover, the studies used different methods, response options and criteria to assess the complaints in patient groups with different disease characteristics. Therefore, the results presented should be interpreted with caution and results cannot be directly compared. For example, the two lowest rates of double vision are found in de novo PD patients [72, 94], which is in line with the reported increase of visual complaints with disease duration and severity (see Table 3). Also, it was not always clear in the phrasing of questions that problems were attributed to vision [17, 119]. People might have involved other symptoms when answering the questions (i.e., reading difficulties were attributed to both visual problems and difficulties with concentration [17]). For the sake of transparency, we listed the (number of) references, the number of participants in whom a complaint was investigated, and the criterion on which the prevalence was based.
Some visual complaints are reported relatively often, while there are also complaints that are hardly ever reported. An important explanation for this is that former complaints are addressed in frequently used self-reports or questionnaires, while the latter are not. This does not mean that visual complaints not included in those instruments are less common or should be taken less seriously. The majority of 139 included studies (N = 85, 61.2%) reported double vision, an item in the NMSS and NMSQ. Similarly, oversensitivity to light constitutes an item in the SCOPA-AUT and was therefore reported in multiple studies (N = 19, 13.7%). Only 35 articles (25.2%) investigated visual complaints using other measures. For example, only few studies asked for experienced visual field loss or reduced contrast sensitivity, which might nevertheless be relevant in the light of known visual field and contrast sensitivity deficits in PD [4, 153]. To obtain a more complete picture of the prevalence, nature, and severity of visual complaints in people with PD and the characteristics of people who experience these complaints, future research should systematically examine a broad range of visual complaints in a large group of people with PD.
Clinical implications
Detection of visual complaints is necessary to prevent unnecessary limitations in QoL. With knowledge of the visual complaints a patient experiences, clinicians are able to select treatments based on the patient’s priorities [12, 154]. In addition, the use of self-report measures within rehabilitation may provide insight into the changes a patient is experiencing and can indicate when individual goals have been achieved [13].
Visual complaints may be an indication of a treatable underlying disorder, which requires assessment and treatment by an ophthalmologist. Complaints may also relate to underlying disorders that cannot be treated, in which case vision-related rehabilitation may be recommended. As an example, complaints of double vision may be caused by cataract, ptosis, or corneal surface abnormalities. These conditions may be treated with cataract extraction, surgery, or artificial tears, respectively. Another possible cause of double vision is convergence insufficiency, which may require prism glasses or advice on how to compensate while reading.
In some cases, objectified functional disorders may not fully reflect self-reported complaints. For example, even though color vision deficits are more common in people with PD compared to controls [155, 156], people with PD do not tend to report more complaints with regard to color vision. In this example, treatment seems irrelevant because patients do not suffer from the underlying disorder. Therefore, care should always be based on self-reported complaints as well.
Although the literature is scarce, visual complaints seem to be related to disease duration and severity. In addition, visual complaints might be influenced by certain types of treatment. Therefore, we suggest that people with PD should be questioned more frequently and more thoroughly about these complaints in clinical practice, especially in advanced disease stages or with medication changes.
The importance of active questioning is also evident from the fact that visual problems might not always be spontaneously reported to healthcare professionals [48, 49], given that people with PD and their family members are often unaware of the relationship between visual complaints and PD [50]. In addition, Martinez-Martin et al. (2007) argued that these complaints may not be recognized unless detected by a questionnaire [60]. Recently developed visual self-report questionnaires may be used to improve the visual care for people with PD [157, 158].
Conclusion
This overview illustrates that a substantial number of people with PD report complaints in a variety of visual domains. These complaints are related to disease duration and severity and may be influenced by medication use. Since visual complaints have a clear negative impact on QoL, attention to and management of these complaints is essential. Active questioning is advised for timely recognition and treatment of visual complaints.
Footnotes
ACKNOWLEDGMENTS
This work was supported by Stichting NOVUM, Amsterdam, The Netherlands, ZonMw grant 637005001 (Expertisefunctie Zintuiglijk Gehandicapten, Meerjarig deelsectorplan 2020-2022 Visueel).
Research support: E. Vooijs (BSc), Psychology student at the University of Groningen; K.C. Klaassen (MSc) and J.E.M. Meijerink (MSc), Patient-Res-earchers for the Dutch Parkinson’s Disease Association.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
