Abstract
Background:
As mutations in glucocerebrosidase 1 (
Objective:
We investigated whether glucosylceramide accumulation and abnormal immune status in the brain are associated with PD.
Methods:
We measured glucosylceramide by liquid chromatography-electrospray ionization-tandem mass spectrometry (LC-ESI-MS/MS) as well as levels of the active fragment of complement C5, C5a, in the CSF of 33 PD, 15 amyotrophic lateral sclerosis (ALS) and 22 neurologically normal control (NNC) subjects. Serum C5a levels in all PD and ALS cases and in a limited number of NNC subjects (
Results:
C5a levels in CSF were significantly downregulated in PD compared with NNC. Moreover, CSF C5a/serum C5a ratio showed pronounced perturbations in PD and ALS patients. LC-ESI-MS/MS revealed a statistically significant accumulation of a specific subspecies of glucosylceramide (d18 : 1/C23 : 0 acyl chain fatty acid) in ALS, but not in PD. Interestingly, CSF glucosylceramide (d18 : 1/C23 : 0) exhibited a significant correlation with CSF C5a levels in PD, but not ALS. No correlation was observed between C5a levels or glucosylceramide subspecies content and disease duration, levodopa equivalent daily dose or Hoehn & Yahr staging in PD.
Conclusion:
Our findings demonstrate complement dysregulation without glucosylceramide accumulation in PD CSF. Furthermore, we found an association between a specific glucosylceramide subspecies and immune status in PD.
INTRODUCTION
Parkinson’s disease (PD) is the second most common neurodegenerative disorder, and is characterized by motor symptoms, including akinesia, tremor and rigidity, as well as non-motor symptoms [1]. Although the pathogenesis of PD remains unclear, heterozygous mutations in the gene for glucocerobrosidase 1 (
The complement system is an evolutionarily conserved component of the innate immune system [16]. It has three different activation pathways, which are each in turn activated by specific triggers. All three pathways converge on C3, and successive cleavage of C3 and C5 leads to the production of the membrane attack complex. Dysregulated complement cascades have been associated with neuro-infectious disease [17], as well as neurodegenerative diseases [18], although their involvement in PD remains to be elucidated. C1q, iC3b (an activated form of C3) and C9 are upregulated in PD patient brains [19, 20]. A recent study, however, has suggested that activated complement, such as C5 and C3, as well as their receptors exert neuroprotective actions on neuronal cells [21].
Here, we investigate whether GlcCer and complement activation in CSF are associated with PD.
MATERIALS AND METHODS
Participants
We recruited 33 consecutive patients with PD, 15 consecutive patients with amyotrophic lateral sclerosis (as a disease control [DC]) and 22 neurologically normal control (NNC) subjects between October, 2012 and September, 2017 at Fujita Health University Hospital.
Diagnoses of PD and ALS were made according to the clinical diagnostic criteria of the UK Parkinson’s Disease Society Brain Bank and the revised El Escorial criteria, respectively [22, 23]. PD disease stage in the “ON” condition was assessed using the modified Hoehn & Yahr staging system for PD cases [24]. All recruited patients did not have any positive family history or juvenile onset of the disease. The NNC subjects volunteered to participate in the study when they received surgical operation with lumbar anesthesia at the Department of Urology of Fujita Health University Hospital because of urological problems (e.g., renal, ureteral, or bladder stone). None of the NNC subjects had a previous history of any psychiatric or neurological disorder or positive family history. All samples from patients and NNC subjects were obtained in the morning. Age and sex did not differ significantly between groups.
All subjects provided written informed consent before participation according to the Declaration of Helsinki. This study was approved by the internal ethics review board committees of Fujita Health University Hospital and RIKEN Institute.
Materials
β-d-Glucopyranosyl-(1→1)-N-lauroyl-d-
Lipid extraction
Lipids were extracted from CSF according to the method of Bligh and Dyer [25]. Briefly, 180μl of CSF was mixed with 675μl of chloroform:methanol (1 : 2, v/v) containing 400 fmol of glucosylceramide (GlcCer) (d18 : 1-C12 : 0) as internal standards. After vortexing and sonicating the solution, 225μl of chloroform and 225μl of distilled water were added. After mixing this solution, the lower phase was collected and dried under a stream of nitrogen.
Quantitative HILIC-ESI-MS/MS
GlcCer, was analyzed as previously reported by hydrophilic interaction chromatography (HILIC)-ESI-MS/MS, with minor modifications [26]. The HILIC-ESI-MS/MS analysis was performed on an LC system Nexera X2 (SHIMADZU, Kyoto, Japan) attached to a triple-quadrupole linear ion trap mass spectrometer (QTRAP4500, SCIEX, Tokyo, Japan). The lipid extracts were dissolved in chloroform:methanol (2 : 1, v/v) and diluted 10-fold with mobile phase A (acetonitrile:methanol:formic acid, 97 : 2:1 [v/v/v], with 5 mM ammonium formate). Aliquots (10μl) were applied to an Atlantis silica HILIC column (2.1 mm i.d.×150 mm; particle size, 3μm; Waters, Milford, MA, USA) maintained at 40°C. Samples were eluted at a flow rate of 0.05 ml/min using the following gradient of mobile phase B (methanol:water:formic acid, 89 : 9:1 [v/v/v], with 20 mM ammonium formate): 3.3 min, 0%; 23.4 min, 0% –35% linear gradient; 1.3 min, 35% –70% linear gradient, 0.05–0.15 ml/min linear gradient flow rate; 8 min, 70% (washing step); 29 min, 0%, flow rate increased to 0.2 ml/min (equilibration); 2 min, 0%, flow rate decreased to 0.05thinsp;ml/min. The mass spectrometer was set to positive ion mode (ion spray voltage: 5,500 V; curtain gas pressure: 30 psi; nebulizer gas pressure: 90 psi; heating gas pressure: 30 psi; temperature: 100°C). Analysis of target lipids was conducted in the multiple reaction monitoring (MRM) mode using specific precursor-product ion pairs [25]. The resulting peak areas were integrated using MultiQuant (ver. 2.1) and Analyst software (SCIEX), and quantified based on the peak areas of the associated internal standard. Lipid contents were normalized against the internal standard recovery and expressed as fM in CSF.
ELISA for C5a in serum and CSF
All samples were centrifuged and stored at –80°C until assay. C5a levels were quantified by ELISA (Abcam, Cambridge, UK) from all subjects in duplicate according to the manufacturer’s instructions.
Statistical analysis
R, an open-source software environment for statistical computing and graphics, was used for statistical analyses. Fisher’s exact test was used for comparison of sex between groups. The Kruskal–Wallis test was used for comparisons of age, CSF C5a, and CSF GlcCer levels. We added the Steel–Dwass test if there was a significant difference after false discovery rate controlling procedures [27]. Pearson tests were used to evaluate the relationship between age, disease duration, C5a levels and levodopa equivalent daily dose (LEDD) in groups. Spearman tests were used to evaluate the relationship between the Hoehn & Yahr scale and other factors in the PD group. Differences were considered significant at
RESULTS
Recruited subjects
All demographic data for recruited patients with PD, ALS, and NNC are shown in Table 1. There was no significant difference in age or sex between subjects (Table 1). Disease duration, Hoehn & Yahr stage and LEDD for PD patients are also shown in Table 1. The numbers of PD patients at the various Hoehn & Yahr stages were as follows (stages indicated in Roman numerals): I: 3; II: 8; III: 12; IV: 7; V: 3.
Demographic characteristics of the recruited subjects
†There is no significant difference in age between groups. ††There is no significant difference in sex between groups. †††There is a significant difference between PD and ALS (
ELISA for C5a in serum and CSF
CSF C5a levels were significantly decreased in PD patients compared with NNC subjects (Fig. 1a). There was no significant difference between the NNC and ALS groups. Furthermore, neither disease duration, Hoehn & Yahr staging nor LEDD was correlated with CSF C5a levels (Fig. 2a–c).
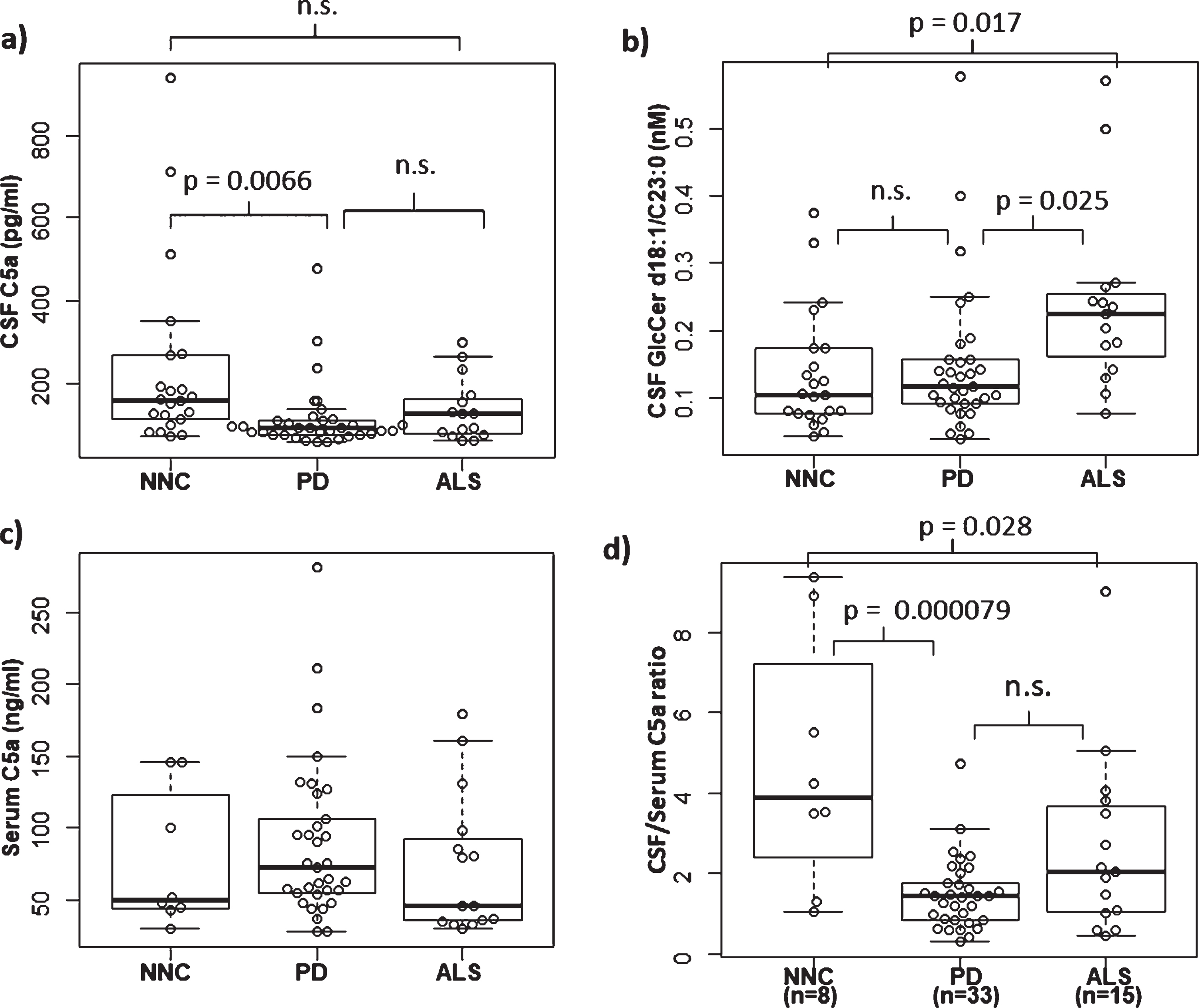
Comparison of serum C5a, CSF C5a and CSF GlcCer d18 : 1/C23 : 0. a) Comparison of CSF C5a among groups. There was a significant difference between PD (
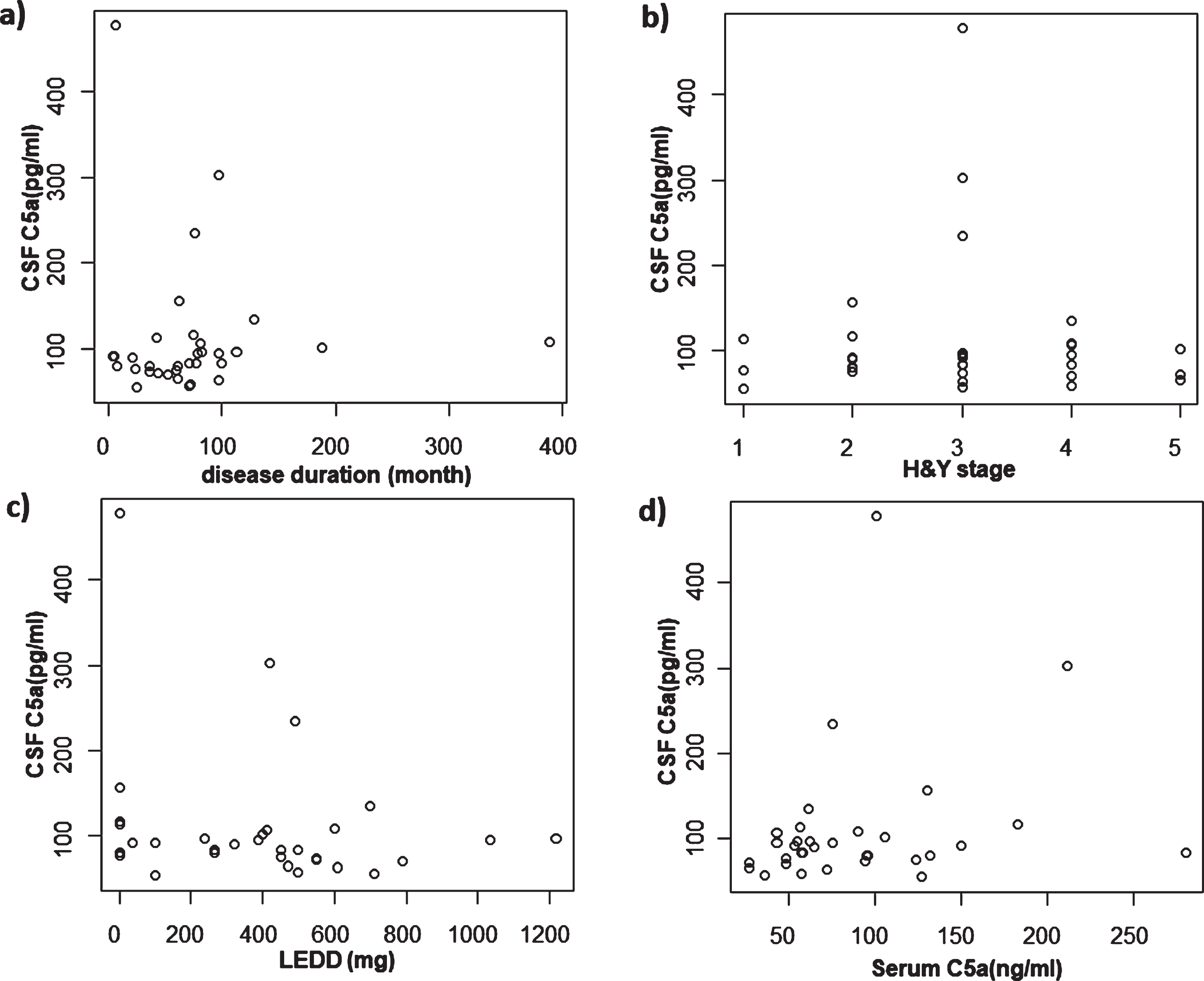
Correlation between CSF C5a and other factors in PD. a) Correlation between CSF C5a and disease duration. There was no significant correlation. b) Correlation between CSF C5a and H & Y stage. There was no significant correlation. c) Correlation between CSF C5a and LEDD. There was no significant correlation. d) Correlation between CSF C5a and serum C5a. There was no significant correlation.PD, Parkinson’s disease; CSF, cerebrospinal fluid; H&Y, Hoehn & Yahr; LEDD, levodopa equivalent daily dose.
We additionally measured serum C5a activity in a limited number of samples for comparison with values in CSF. Unfortunately, we were able to obtain serum samples from only eight NNC subjects who were also tested for CSF C5a levels. Although PD subjects showed a tendency towards a higher concentration of serum C5a compared with both the NNC and ALS groups, there were no significant differences among the groups (Table 1; Fig.1c). Furthermore, there was no significant correlation between serum C5a and CSF C5a in PD (Fig. 2d). We also calculated the CSF/serum C5a ratio, and found a significant reduction of this ratio in PD cases. Moreover, the ratios from ALS cases showed a significant reduction compared with NNC subjects (Fig. 1d). Notably, the CSF/serum C5a ratios showed a tendency towards a correlation with not disease duration, but with Hoehn & Yahr staging, in PD patients (
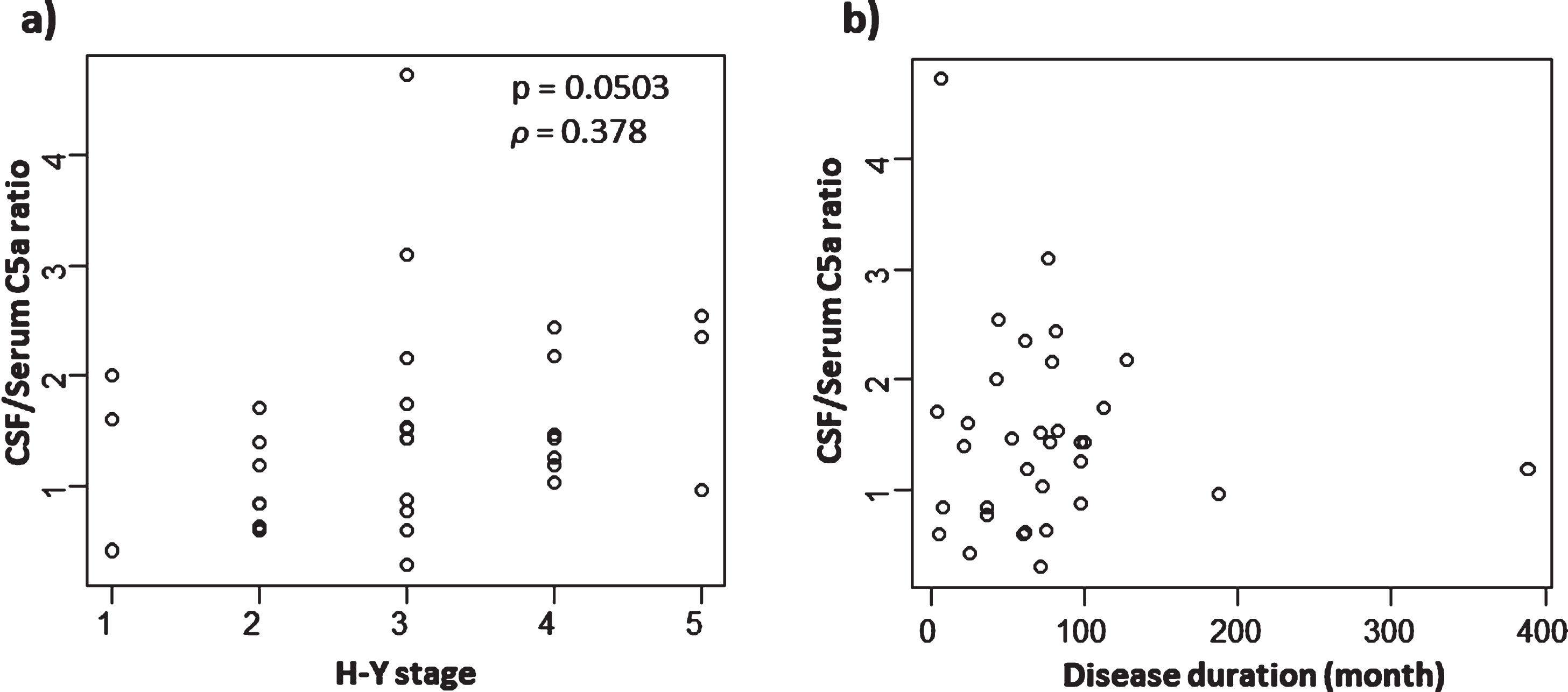
Correlation between CSF/serum C5a ratios and disease severities and disease duration in PD. a) Correlation between CSF/serum C5a ratio and H&Y stage. Although it was not statistically significant, there was a tendency towards a correlation (
Neutral GSL determination with HILIC-ESI-MS/MS
CSF samples from PD, ALS and NNC subjects were subjected to lipid extraction and analyzed with HILIC-ESI-MS/MS. HILIC columns are widely used for separating isomeric glycolipids such as GlcCer and galactosylceramide (GalCer) [26]. We were able to measure GlcCer and GalCer separately by use of HILIC in combination with ESI-MS/MS. Among the GlcCer subspecies, those with d18 : 1/C23 : 0 acyl chain fatty acids showed a significant difference between the groups (Table 2). GlcCer with the d18 : 1/C23 : 0 acyl chain length fatty acid was more abundant in ALS compared with NNC and PD (Fig. 1b). Interestingly, although there was no significant correlation between GlcCer (d18 : 1/C23 : 0) content and CSF C5a levels (Fig. 4a) or disease duration (Fig. 4b) in ALS cases, we found a significant positive correlation between the GlcCer (d18 : 1/C23 : 0) subspecies and CSF but not serum C5a in PD cases (Fig. 4c, d). We did not detect any correlation between the levels of GlcCer (d18 : 1/C23 : 0) and disease duration, H & Y staging or LEDD in PD patients (Fig. 5a–c). Furthermore, there was no correlation between age and levels of CSF C5a or CSF GlcCer (d18 : 1/C23 : 0) in NNC subjects (Fig. 5d, e).
Glucosylceramide subspecies profiles in the CSF (nM)
mean ± SD (nM). *Bold numbers indicate significant differences among the three groups.
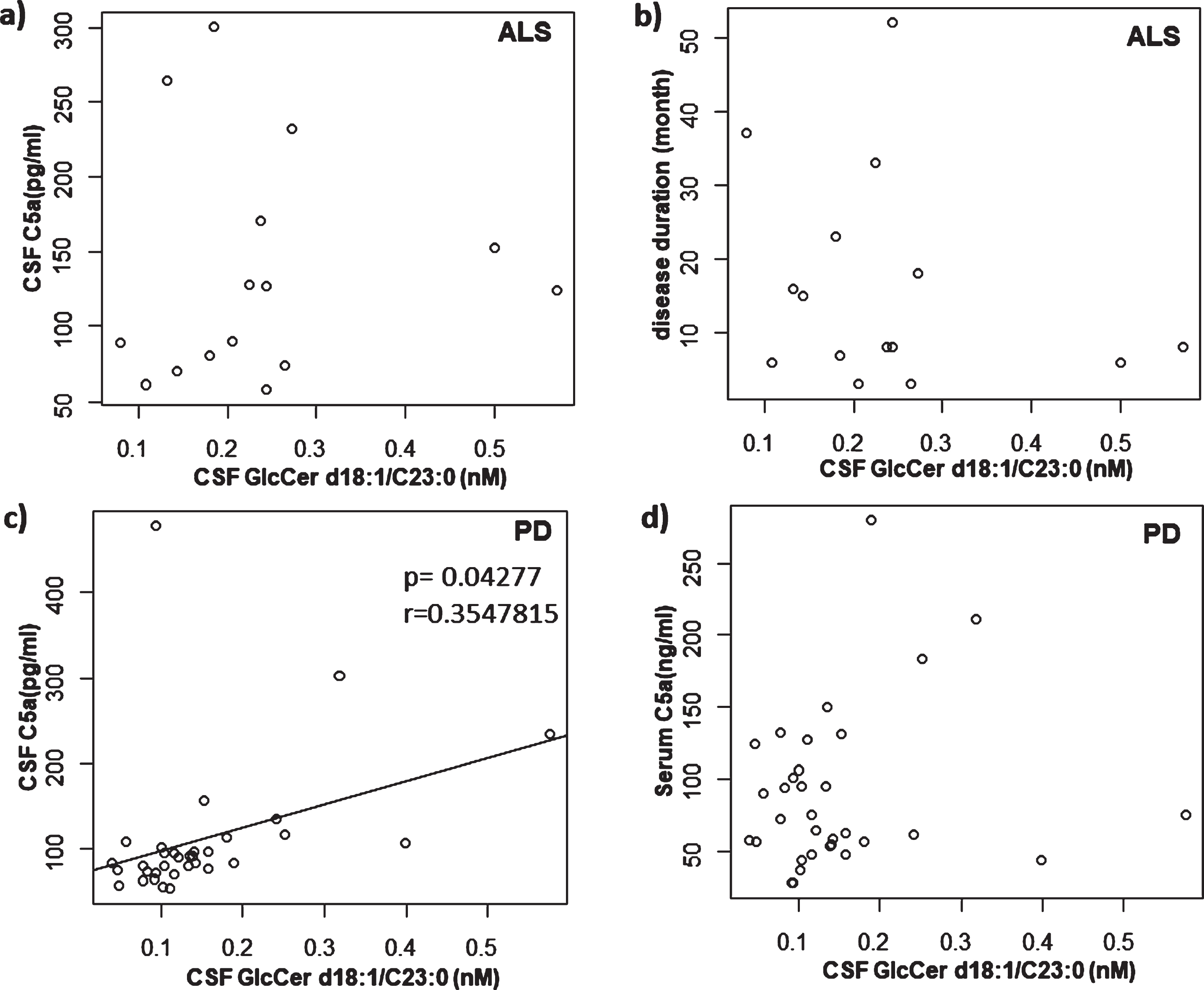
Correlation between CSF GlcCer d18 : 1/C23 : 0 and other factors in PD and ALS. a) Correlation between CSF GlcCer d18 : 1/C23 : 0 and CSF C5a in ALS. There was no significant correlation. b) Correlation between CSF GlcCer d18 : 1/C23 : 0 and disease duration in ALS. There was no significant correlation. c) Correlation between CSF GlcCer d18 : 1/C23 : 0 and CSF C5a in PD. There was a significant correlation (
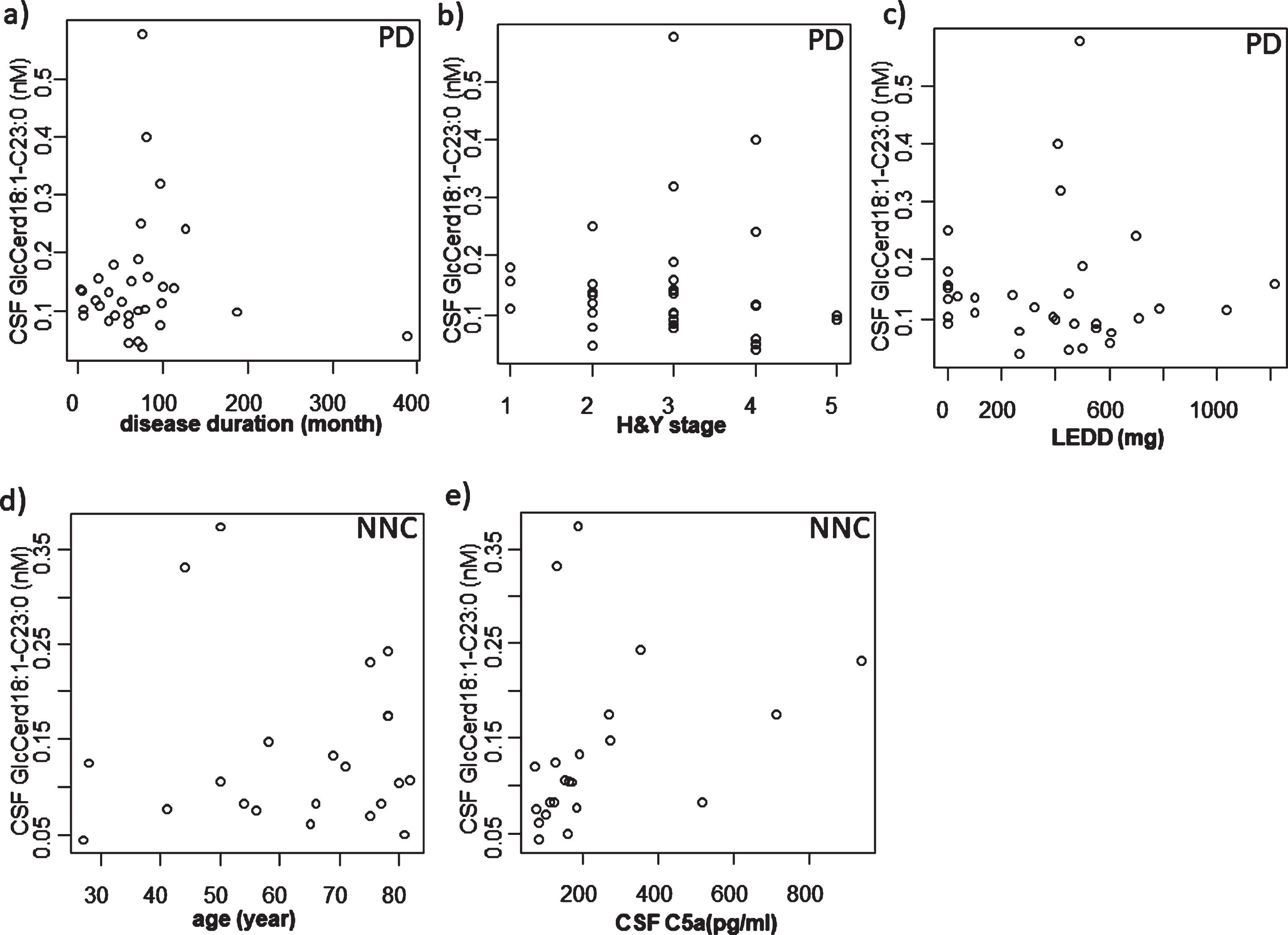
Correlation between CSF GlcCer d18 : 1/C23 : 0 and other factors in PD and NNC. a) Correlation between CSF GlcCer d18 : 1/C23 : 0 and disease duration in PD. There was no significant correlation. b) Correlation between CSF GlcCer d18 : 1/C23 : 0 and H&Y stage in PD. There was no significant correlation. c) Correlation between CSF GlcCer d18 : 1/C23 : 0 and LEDD in PD. There was no significant correlation. d) Correlation between CSF GlcCer d18 : 1/C23 : 0 and age in NNC. There was no significant correlation. e) Correlation between CSF GlcCer d18 : 1/C23 : 0 and CSF C5a in NNC. There was no significant correlation. PD, Parkinson’s disease; CSF, cerebrospinal fluid; H&Y, Hoehn & Yahr; LEDD, levodopa equivalent daily dose; NNC, neurologically normal control.
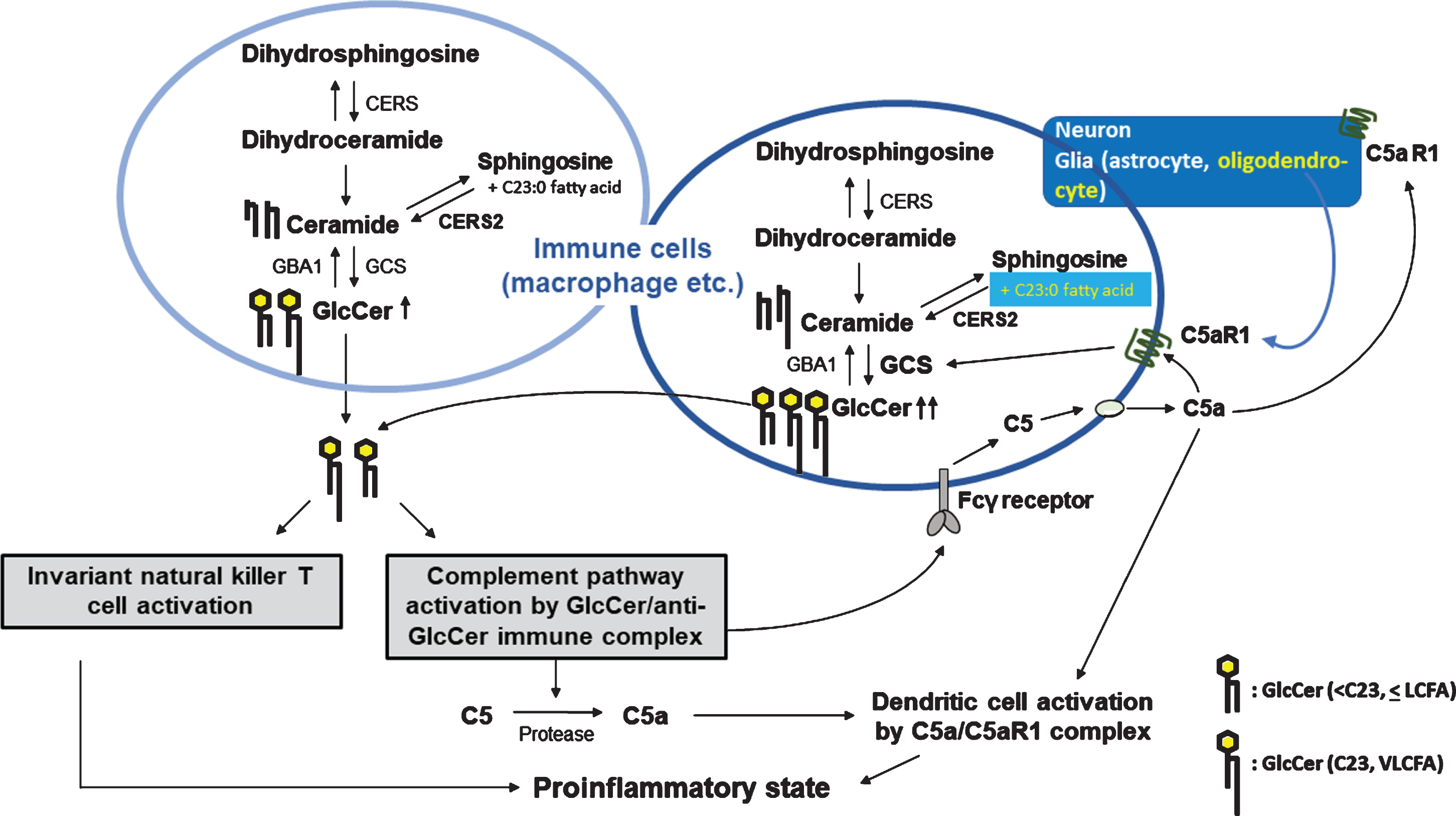
Hypothetical correlation between odd-numbered very-long-chain fatty acids containing GlcCer (glucosylceramide) and abnormally downregulated complement C5 activation status: comparison with normal and abnormal GlcCer-mediated inflammatory reactions in the induction of C5a-mediated inflammation. Accumulation of GlcCer in macrophages induces activation of the classical complement pathway by forming of GlcCer/anti-GlcCer antibody-immune complexes. The immune complexes induce protease-mediated C5a production from C5. The immune complexes also bind to Fcγ receptors on macrophages and induce local C5a production. C5a/C5a receptor 1 complexes induce dendritic cell activation and lead to a proinflammatory state. Odd-numbered fatty acids containing GlcCer are considered to be derived from oligodendrocytes and neurons as well as oligodendrocytes, which are known to express C5aR1. Therefore, these different cell types might be sensitive to the active fragment, C5a. The very-long-chain fatty acid (VLCFA) GlcCer can activate and modulate invariant natural killer T cells (ivNKT cells), provoking inflammatory reactions.
DISCUSSION
The present findings demonstrate dysregulation of the central innate immune system as revealed by a significant reduction in CSF C5a levels in PD patients. Moreover, the central/peripheral C5a ratios measured in a limited number of NNC subjects (
Our present study is the first to perform GlcCer analysis in CSF from PD patients using HILIC-ESI-MS/MS. With this method, we could measure levels of GlcCer of various acyl chain fatty acid lengths in the CSF. Although there was no significant change in total GSLs in the PD and ALS group, we found a significant increase in GlcCer with the d18 : 1/C23 : 0 acyl chain length fatty acids in ALS compared with the NNC and PD groups. It should be noted that odd-numbered acyl chain fatty acids (C23 : 0 in this case) are produced from phytosphingosine as well as 2-hydroxy fatty acids by the action of HACL2 (2-hydroxyacyl-CoA lyase 2) in the endoplasmic reticulum (ER) of cells [30, 31]. Thus, it is reasonable to speculate that increased levels of C23 : 0 containing GlcCer are possibly derived from oligodendrocytes, which contain a large amount of hydroxylated C24 acyl chain fatty acids in GalCer in myelin. Interestingly, very-long-chain fatty acids have been reported to protect against α-synuclein neurotoxicity in a yeast model of PD [32]. As discussed before, GlcCer (d18 : 1/C23 : 0) levels are associated with CSF C5a levels, and its levels were slightly higher compared with NNC subjects. However, this increase in the GlcCer subspecies could not prevent the severe decline in CSF C5a levels, which might be caused by an unidentified immunological abnormality in PD.
Dodge et al. reported that the levels of GlcCer are elevated in spinal cords of ALS patients [33]. Intriguingly, GlcCer upregulation by inhibition of GBA1 activity has neuroprotective effects in a mouse model of ALS [34]. Furthermore, the GlcCer synthesizing enzyme, GlcCer synthase, has been reported to be upregulated in ALS patients and SOD1 model mice [35]. Unfortunately, although these previous studies reported an increase in total GlcCer content, they did not report the levels of the GlcCer d18 : 1/C23 : 0 subspecies.
Mielke et al. recently reported that ceramides are significantly elevated in serum from PD cases. However, these investigators only measured serum levels and did not measure GlcCer and GalCer separately (only total hexosylceramide) [36]. The levels of GlcCer in CSF are dependent on the balance of the synthesis from ceramide by GlcCer synthase and degradation by glucocerebrosidase, GBA1. They are also dependent on the specific fatty acid elongase activities, which elongate acyl chain fatty acids by even-numbered addition. Currently, it is unknown why total and individual GlcCer subspecies are not elevated in CSF from PD cases, although decreased GBA1 activities have been previously reported in the brain and CSF. However, possible reasons may include differences in disease duration or age, because GlcCer expression has been reported to diminish with age [37]. In this study, the disease duration in PD was much longer than that in ALS. Therefore, elevated GSLs in CSF might be present at the initial phase of the disease, as in ALS, and decrease with time. Of course, this hypothesis should be fully investigated in a future study.
Our present results reveal no obvious accumulation of GlcCer in CSF, although the levels of GlcCer (C23 : 0) (very-long-chain GlcCer) were slightly higher than those in NNC subjects. GlcCer (C23 : 0) showed a significant correlation with CSF C5a levels only in the PD group, and their C5a levels were significantly downregulated. These positive correlations between GlcCer subspecies containing very-long-chain fatty acids and complement activation status suggest a mechanistic link that has not yet been clarified. Indeed, a previous study indicated that very-long-chain sphingolipids strongly influence invariant natural killer T cell development and homeostasis, and therefore activate immune responses [38]. Moreover, Pandey and colleagues detected autoantibodies against β-GlcCer in GD patients as well as in GD model mice, and these autoantibodies initiate neuroinflammatory reactions through complement activation, C5a production, and cognate receptor activation [14]. They also suggested that neuronal death in GD is caused by anti-GlcCer antibodies generated against accumulated GlcCer, which produce neuroinflammatory responses. Furthermore, GSLs have been recently reported to play an important role in inflammation by being presented to immunocytes, such as natural killer T cells, via CD1d. α-GlcCer, from fungi and bacteria, had already been shown to be loaded onto CD1d for presentation to immune cells [39].
GSLs in CSF may cause a significant change in the lipid composition of the plasma membrane and membranous organelles in susceptible neurons, resulting in impaired neuronal function in GD, as demonstrated by a previous study [40]. To the best of our knowledge, our present results reveal, for the first time, no significant accumulation of total or any subspecies of GlcCer in the CSF in PD patients, although levels were slightly higher than in the NNC group.
This study has some limitations. First, the sample size was rather small. Therefore, the present findings should be confirmed with a larger cohort of patients. Second, the molecular mechanisms underlying the accumulation of the specific GlcCer subspecies (C23 : 0) in CSF in ALS and its correlation with CSF C5a levels in PD, but not in ALS, remain to be elucidated. It should be mentioned that ceramide synthesis is dependent on six different ceramide synthases (CERS1–6). Each enzyme produces a distinct ceramide subspecies differing in acyl chain-lengths in the fatty acid moieties [41]. Among them, CERS2 is responsible for the synthesis of ceramide having very-long-chain fatty acids (C22–C24) and is highly expressed in oligodendrocytes in the brain [42]. Moreover, another important enzyme family that elongates acyl chains in fatty acids, FA elongases, were not examined in the current study. Interestingly, in the present study, we found a significant accumulation of specific GlcCer with very-long-chain fatty acids (C23 : 0) in CSF from ALS patients. In a future study, we will investigate in greater detail the changes in the metabolism of other glycosphingolipids as well as gene expression of different ceramide synthases and their activities in PD and ALS.
In conclusion, our findings suggest an association between a specific subspecies of GlcCer with an odd-numbered fatty acid acyl chain and complement system perturbation in patients with PD. Our results also provide potential therapeutic targets for regulating GSLs metabolism and complement system activation. Intriguingly, Sardi and colleagues recently showed that prolonged pharmacological antagonism of GlcCer synthase reduces α-synuclein accumulation and GlcCer levels in the brain, and thereby improves behavioral outcomes in PD model mice [43].
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
We would like to thank Toshiki Maeda, MD, Eri Muto, PhD, and Mitsuko Ide, MAgr, for their excellent technical assistance. We also thank Drs. Shinji Ito, Akihiro Ueda, Koichi Kikuchi, Ryunosuke Nagao, and Seiko Hirota for recruiting patients involved in the present work.
This work was partly supported by a JSPS KAKENHI Grant Number JP 16k09685 [Grant-in-Aid for Scientific Research (C)] from the Ministry of Education, Culture, Sports, Science, and Technology (MEXT) of Japan, and Research Grants for Intractable Diseases from the Ministry of Health, Labor, and Welfare of Japan to TM. The present work was also partly supported by a JSPS KAKENHI Grant Number JP17K15608 [Grant-in-Aid for Young Scientists (B)] from MEXT to HA.
