Abstract
Introduction:
Improved analytical tools for detailed characterization of synucleins in pre-clinical models of Parkinson’s disease (PD) and related synucleinopathies are needed.
Objective:
Develop a multiple reaction monitoring (MRM) liquid chromatography tandem mass spectrometry (LC-MS/MS) assay to quantify species-specific sequences and structural heterogeneity in soluble α- and β-synucleins in brain tissue.
Methods:
Using a proteolytic digestion workflow, the MRM LC-MS/MS method assayed six proteotypic peptides from the α-synuclein sequence; three unique to mouse or human α-synuclein and three conserved in α- and β-synuclein. For quantification, we used labeled α-synuclein as the internal standard and an external calibration curve. As proof of concept, the synuclein LC-MS/MS method was applied to brain tissue specimens from M83 transgenic PD mice, which overexpresses human α-synuclein, relative to wild-type littermate controls.
Results:
The synuclein MRM assay was linear over a wide concentration range (at least one order of magnitude). The assay had several advantages over ligand binding analytical methods, such as western blotting and enzyme-linked immunosorbent assays. These advantages included the ability to: quantify 1) total α-synuclein, 2) combined α- and β-synucleins, 3) species-specific contributions to total α-synuclein (e.g., in mice expressing both mouse and human α-synuclein), and 4) identify peptide-specific profile differences that may reflect post-translational modifications, all within a single analysis.
Conclusion:
With improved and expanded analytical characteristics coupled with a streamlined sample preparation workflow, the quantitative synuclein profiling LC-MS/MS assay provides a versatile and efficient platform to characterize synuclein biology in pre-clinical models and the potential for application to human tissues and fluids.
Keywords
INTRODUCTION
Parkinson’s disease (PD) is a slow, progressive neurodegenerative disease that results in cardinal motor symptoms of tremor, bradykinesia and rigidity. PD is defined postmortem by loss of nigral dopamine cells and the presence of proteinaceous aggregates termed Lewy bodies within remaining cells of the substantia nigra pars compacta [1]. Lewy bodies, and related Lewy neurites (together termed Lewy pathology), are primarily composed of α-synuclein. Lewy pathology is the hallmark of a wider family of neurodegenerative diseases, termed synucleinopathies, that includes PD dementia, dementia with Lewy bodies, and multiple system atrophy. Mutations in the α-synuclein gene (SNCA), were the first identified as causal to PD through familial linkage, and several other genes have since been similarly linked to autosomal-dominant clinically-typical forms of parkinsonism (e.g., LRRK2 and VPS35) [2]. Genetic models expressing PD point mutations, or α-synuclein multiplications, in addition to several neurotoxin-lesion based models of PD, recapitulate several features of the disease. For example, M83 mice overexpressing human α-synuclein are commonly used as PD models. M83 mice express A53T human α-synuclein, and develop severe motor impairment in parallel with the formation of intracytoplasmic neuronal α-synuclein inclusions, reminiscent of Lewy pathology [3, 4]. Pathology and phenotype onset can be induced in younger M83 mice by intracerebral inoculation of brain homogenate from older M83 mice [3, 4]. Such models are useful for studying disease etiology and pathophysiology [5], and essential for pre-clinical testing of potential therapeutics.
Quantifying α-synuclein in tissue from humans and experimental models by antibody-based methods (e.g., western blotting, sandwich immunoassays, and immunohistochemistry) has yielded a wealth of information about synucleinopathies [6]. These techniques have revealed that α-synuclein exists in post-translationally modified and multimeric forms, and that several of these forms are found in higher concentrations in biospecimens from PD patients and PD models, compared to relevant controls [7–9]. Although extremely useful, immunometric approaches can lack specificity and accuracy, due to vagaries of antigen recognition and indirect detection of the target analyte [10]. Further, antibody-based approaches generally lack the resolution desired for detailed characterization of primary structure, and are challenging to effectively multiplex [10, 11].
In light of the limitations associated with antibody binding methods, and the structural heterogeneity of the target analyte synuclein, it is desirable to develop a method with improved selectivity. Ideally, a multiplex quantification method would quantitate total synuclein, the various synuclein family members (e.g., α-, β-, γ-synuclein), differentiate source species (e.g., murine or human), and isoform-specific contributions to total-synuclein, while using small quantities of the biospecimen. Since a wide range of post-translational modifications (PTMs) is possible, a method that enables high-throughput screening of PTMs would also be desirable. For example, the widely utilized M83 PD mouse model expresses both endogenous mouse α-synuclein and human A53T α-synuclein, and is often assayed using antibodies that recognize both murine and human proteins; discrimination of the different species of α-synucleins requires that separate sandwich immunoassays are used, complicating analyses [4, 7]. Furthermore, there is growing interest in selective characterization and quantitation of β-synuclein, in the context of synucleinopathy, Alzheimer’s disease, and encephalopathy [12, 13]. The current requirements of multiple antibody pairs to achieve the desired resolution for synucleins is cumbersome and costly. With additional antibodies needed for each target of interest, the number of PTMs that can be quantified in a single study is also limited.
Multiple reaction monitoring (MRM), an approach used in liquid chromatography triple quadrupole tandem mass spectrometry (LC-MS/MS), is a powerful tool for detecting and quantifying protein biomarkers with high selectivity in a multiplexed fashion [14]. For MRM analysis, proteins are enzymatically digested into peptides using a protease such as trypsin, and generated peptides are separated, typically by hydrophobicity, by LC before introduction into the MS/MS. Ionized peptides are selected in the first quadrupole of the mass spectrometer based on their mass-to-charge (m/z) ratio, fragmented in the second quadrupole, and then relevant fragment ions are detected in the third quadrupole, again by their m/z. The high selectivity of this method arises from the direct detection of the molecule of interest, and the fragmentation of the molecule – akin to a sequencing experiment – to confirm its identity. It is recommended that three transitions (precursor/fragment ion pairs) are monitored per peptide in a quantitative assay. In addition, there are several safety nets in place in an MRM quantitative workflow to confirm what is detected is in fact the target analyte, including monitoring the relative ratios of the fragments generated and comparison to known standards.
In this study, we developed a quantitative MRM method for soluble α, β-synuclein proteins to enable reliable and precise measurement of total α-synuclein, dissect the roles of both α-synuclein and α, β-synuclein, selectively identify human and murine constructs and also regionally map synuclein modifications in brain tissue. Existing analytical methods monitor synuclein from one or two 5–7 residue regions (e.g., as would occur in a western blot targeting a single epitope and sandwich immunoassay targeting two epitopes), but with the multiplexing capacity of LC-MS/MS, the sequence coverage has been extended dramatically. MRM analysis of unmodified peptides from across a protein’s sequence can be used to indirectly detect structural alterations in the target protein, including those occurring as a result of PTM. For example, in the presence of non-uniform and differential PTM of the protein sequence in disease versus controls, the relative ratios of unmodified peptides from the target protein would be altered [15]. As proof-of-concept, the method was applied to a mouse model of PD, specifically, human α-synuclein overexpressing M83 mice. The development of this new analytical tool was undertaken to improve our understanding of how α-synuclein alterations (i.e., mutations, increased expression and PTMs) relate to insoluble aggregate formation.
MATERIALS AND METHODS
This study was conducted with research ethics board approval from Providence Health Care Research Institute (H18-03050) and McGill (#7544 and #7888). A complete description of reagents and their sources, LC-MS/MS conditions and assay validation procedures can be found in the Supplementary Material.
Mouse models
WT littermate (herein WT) and M83 mice, maintained on a C56.BL6/C3 H background, were bred and housed in the McGill University Centre for Neurological Disease Modeling. M83 mice express human A53T α-synuclein under the direction of the mouse prion promoter [4]. Brain homogenate from heterozygous (M83+/–, 6 mo, n = 3), homozygous (M83+/+, 6 mo, n = 3), and non-transgenic wild-type (WT, 6 mo, n = 3) mice were subject to MRM MS analysis. LC-MS/MS validation and method optimization experiments were initially performed using C57.BL/6 WT mice (3 mo).
Sample preparation
Mice were euthanized and whole brain removed, rinsed with saline, snap-frozen in liquid nitrogen, and stored at –80°C. Brain tissue homogenate (1 : 2 w/v) was prepared by mechanical homogenization in the presence of TBS with protease inhibitor cocktail (1 tablet in 10 mL TBS), and the supernatant separated from the pellet by centrifugation (20,000×g, 10 min). A 50μL aliquot of supernatant was then diluted with TBS to a concentration of 6.4 g/L of total protein, incubated at 90°C for 15 min (referred to herein as thermal enrichment) and centrifuged to pellet any debris (20,000×g, 10 min). The supernatant (referred to herein as the soluble fraction) was collected and stored at –80°C for further analysis.
LC-MS/MS
For LC-MS/MS analysis, the soluble fraction (10μL for WT and 2μL for M83, with final concentrations corrected for differences in the initial addition of soluble fraction volumes) was combined with 50 mM AMBIC (40μL) and the 15N-labelled α-synuclein internal standard stock solution (50μL) in a 1.5 mL tube. Protein precipitation was performed with the addition of 100% trichloroacetic acid in water (25μL), incubation with gentle mixing for 10 min, then centrifugation (20,000×g for 10 min). The supernatant was discarded and the pellet washed with cold acetone (20μL at 4°C). The wash was centrifuged (20,000×g, 10 min), the supernatant was discarded and the pellet was air dried in the fume hood for 5 min. The pellet was then dissolved in 1% sodium deoxycholate in 50 mM AMBIC (100μL), incubated at 95°C for 15 min, 4°C for 10 min, then 4.5μL L-1-tosylamido-2-phenylethyl chloromethyl ketone (TPCK) treated trypsin solution (5 mg/mL in 50 mM AMBIC) was added. Proteolytic digestion proceeded via incubation at 37°C for 18 h and was arrested with the addition of 1% formic acid in water (50μL). Samples were centrifuged (20,000×g, 10 min); the supernatant transferred to a 1.5 mL tube; and 5μL of this supernatant was injected for LC-MS/MS analysis. All experiments were repeated to obtain duplicate measurements for each sample. For synuclein quantitation, peptide concentrations were interpolated from external calibration curves of human recombinant α-synuclein (Supplementary Figure 1) and reported in ng/mL normalized to the total protein concentration of the sample.
RESULTS AND DISCUSSION
MRM development and method validation
A thermal enrichment, acid precipitation sample preparation workflow was developed for quantificaiton of synucleins from the solube fraction of mouse brain homogenates (Fig. 1). Synucleins, including monomeric, fibril and PTM forms, are heat stable and remain in the supernatant following heat-enrichment [16, 17]. As part of method development, the use of heat treatment was not found to alter synuclein peptide concentrations nor internal peptide ratios. Peptide concentrations were found to be similar in TBS-soluble fractions analyzed before and after heat enrichment (data not shown), which was consistent with previous work showing no detectable loss of synucleins post-heat enrichment to the insoluble pellet fraction [16].
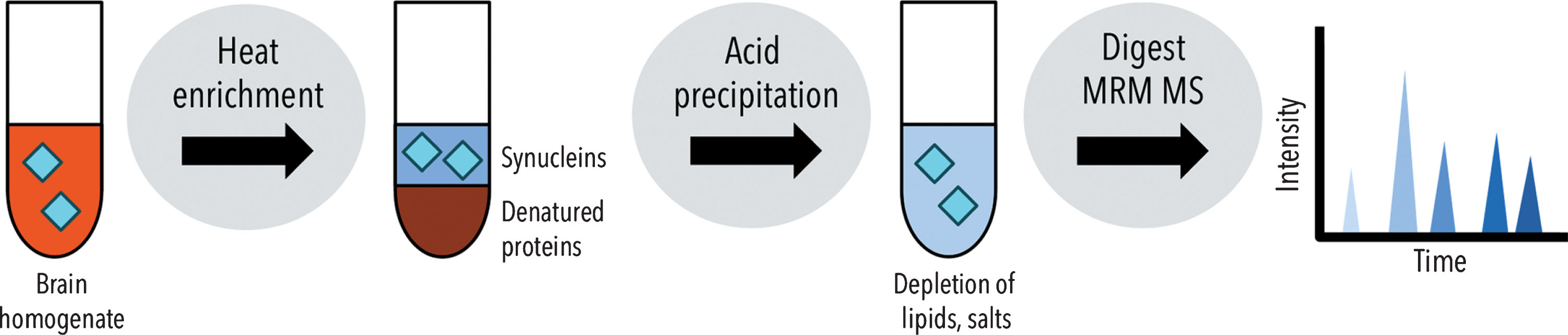
Schematic of the sample preparation workflow for quantification of synucleins in brain tissue by LC-MS/MS.
MRMs were developed for eight peptide sequences in total (Table 1, Supplementary Table 1, Fig. 2): six tryptic peptides from human α-synuclein and mouse α-synuclein (both 52% sequence coverage), with three of the tryptic peptides sharing 100% sequence identify between human [hu] and mouse [mu], as well as between α-synuclein and β-synuclein (residues 1–6, 13–21 and 35–43, denoted as peptides α/β1–6, α/β13–21, α/β35–43, where α/β denotes the 100 % sequence identity). If additional coverage of specific synuclein sequence regions is desired, alternate proteases — in place of or in addition to trypsin — can be explored to attempt to produce alternate proteotypic peptides (i.e., peptides that uniquely identify synuclein and are consistently observed via LC-MS/MS).
Synuclein peptide sequences monitored by MRM MS, and their species and synuclein family specificities
aIncludes the human A53T variant.
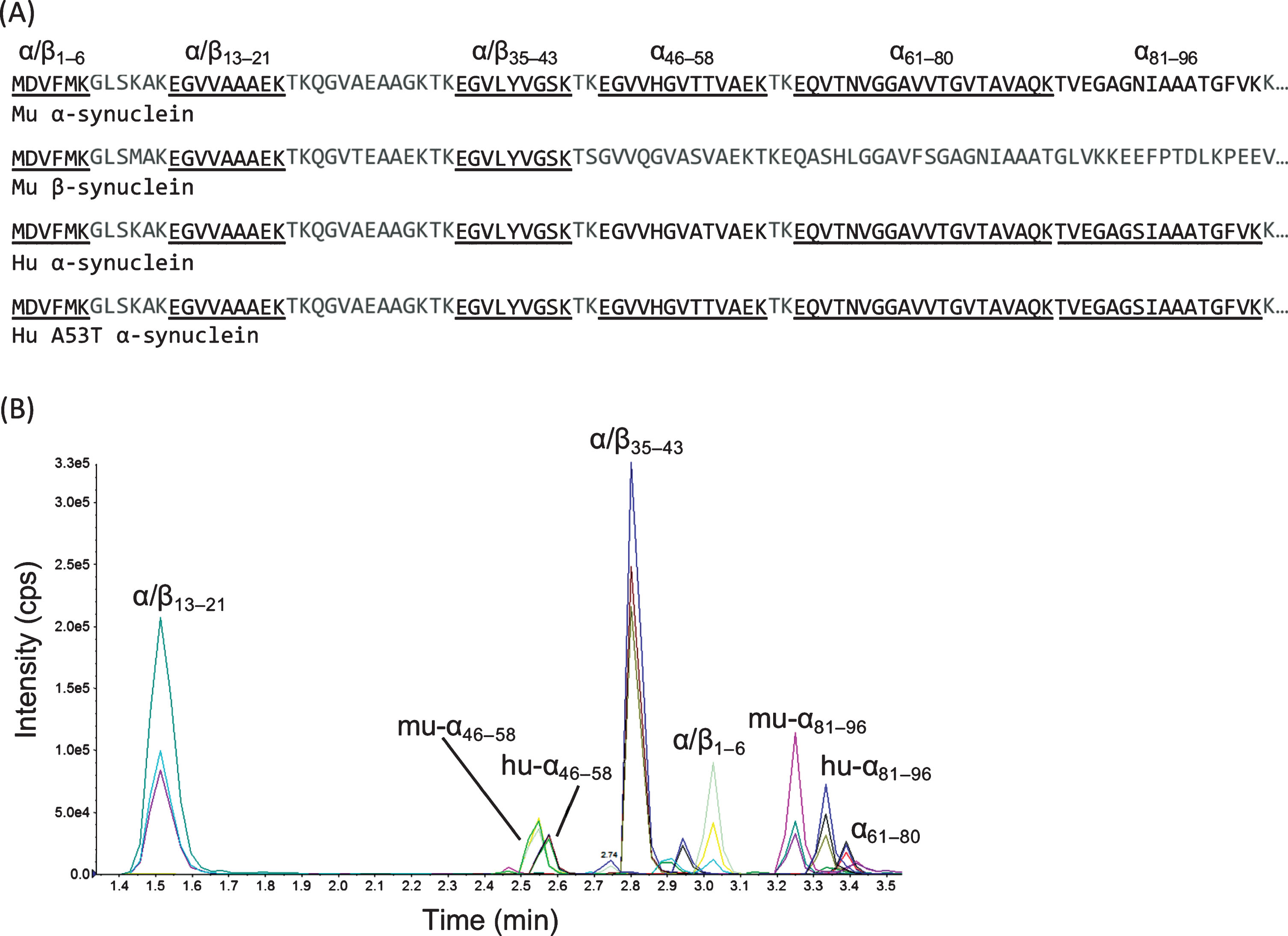
A) Sequence alignments of human and mouse synuclein variants with MRM peptides noted (black), including those shared across species or variants (underlined). B) Representative chromatogram from mouse brain (WT C57.BL/6, with five tryptic synuclein peptides monitored) and recombinant human α-synuclein (three peptides monitored: α/β1–6, hu-α46–58, and hu-α81–96) demonstrating chromatographic resolution and the three transitions monitored per peptide.
A parallelism experiment was performed to select a suitable surrogate matrix for our calibrators (Supplementary Table 2, Supplementary Figure 2). By statistical comparison, the slopes of the surrogate and biological curves were comparable in all peptides up to 3000 ng/mL of spiked recombinant α-synuclein, except for peptide hu-α81–96 (p-value = 0.0006). The slopes related to peptide hu-α81–96 were comparable up to 1000 ng/mL of spiked recombinant α-synuclein. Thus, the upper limits of the measuring interval were set accordingly. Parallelism was also assessed by comparing the synuclein concentration in the biological matrix obtained from standard addition versus that interpolated from the calibration curve, revealing relative errors of less than 12% (Supplementary Table 3).
The assay was linear over one order of magnitude (Supplementary Table 4, Supplementary Figure 3), and this wide linear range was desirable for measuring both α,β-synuclein (as measured by peptides α/β13–21 and α/β35–43) and α-synuclein specifically, as the former was five-fold higher in concentration in the WT mouse (see next section below). The large dynamic range was also advantageous for measuring unknown concentrations in a variety of mouse models. Reproducibility was assessed by measuring intra- and inter-assay precision at three concentrations, including at the LLMI; the CV for all peptides was < 16 % (Supplementary Table 5).
To assess the required proteolytic digestion time, digestion time course experiments were performed as previously described by our laboratory (Supplementary Figure 4) [18]. From the digestion time-course profiles, tryptic peptide intensities reached a digestion asymptote within ∼1 h and remained stable out to 24 h. As such only a brief 1 h digestion was required, which facilitates high-throughput analyses, and decreases overall assay turn-around time.
Via post-column infusion experiments, there was no evidence of ion suppression or enhancement observed for hu-α81–96 or any other peptide originating from human α-synuclein during their expected retention times (Supplementary Figure 5).
Peptide α/β1–6 was detected in human recombinant α-synuclein preparations but not mouse brain homogenates, which may be attributed to in vivo N-terminal acetylation of α,β-synuclein [19], which would alter the mass of the of the peptide. The remaining three peptides monitored were unique to α-synuclein (i.e., residues 46–58, 61–80 and 81–96; denoted as α46–58, α61–80, α81–96). The α61–80 sequence is identical in murine and human α-synuclein, whereas the other two peptides (i.e., α46–58 and α81–96) are not. Notably, in the M83 model the A53T mutation from the human α-synuclein transgene, results in conversion of the human sequence into the mouse sequence within the α46–58 peptide, and thus huA53T-α46–58 shares 100% sequence identity with mu-α46–58 . Species-specific contributions were, in turn, determined from the concentrations of mu-α81–96 and hu-α81–96 peptides. The retention times of the murine-specific peptides (i.e., mu-α46–58 and mu-α81–96) were comparable to the analogous human peptides from the 15N-labeled internal standard, likely due to high sequence similarity.
Several features of the synuclein LC-MS/MS method illustrate sample preparation and analytical advances over previously described approaches. Previous mass spectrometric analyses of synucleins (e.g., for analysis of cerebrospinal fluid [13, 20], PD brain tissue [21] and melanoma tumor tissue [22]) have relied on micro/nanoflow LC for separation of tryptic peptides. The method described herein uses a standard HPLC column, which is a more robust setup (e.g., less prone to column clogging, relatively short chromatographic run times) for high throughput analyses of complex biosamples [23]. The method herein also utilized a streamlined sample preparation scheme involving heat enrichment for rapid purification of synuclein.
Quantification of α-synuclein
M83+/+ and M83+/– mice begin to develop α-synuclein inclusions and motor symptoms, features reminiscent of PD, at ∼8 and ∼22 months of age, respectively [4]. Lesions primarily accumulate in the spinal cord, brainstem, cerebellum and thalamus [4]. To assist in the characterization of these PD mouse models, peptides from across the α- and β-synuclein sequences were quantified via the synuclein LC-MS/MS assay (Fig. 3A).
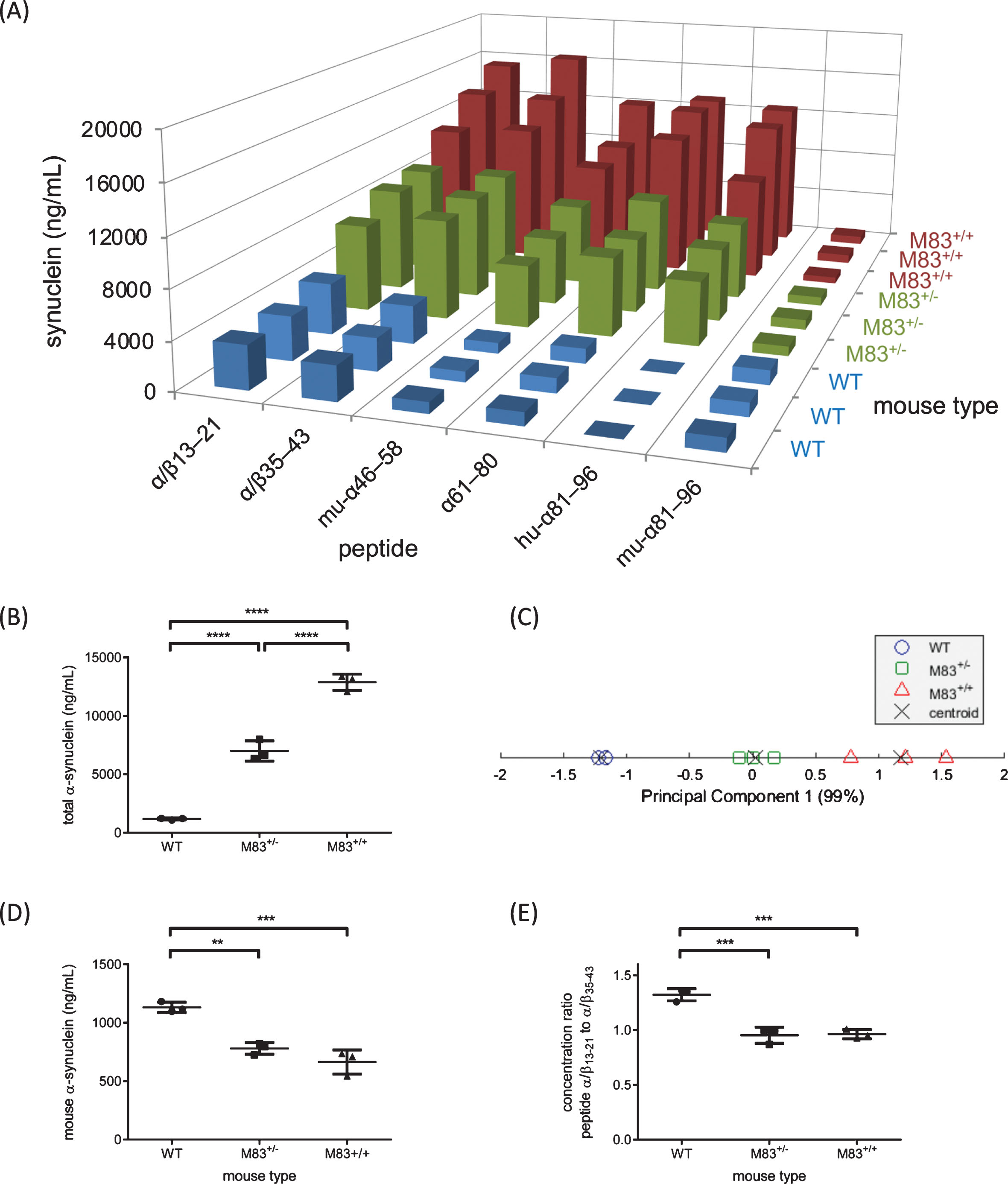
A) Abundance of synuclein peptides as measured by MRM MS in WT, M83+/– and M83+/+ mice whole brains. B) Quantification of peptide α61–80 reflective of total α-synuclein. C) Principal component plot of mouse types grouped by k-means clustering of the synuclein LC-MS/MS peptide data. D) Quantification of peptide mu-α81–96 reflective of total mouse α-synuclein. E) Relative amounts of α/β13–21 and α/β35–43 differentiate M83 mice from WT mice.
To create a quantitative MRM assay representative of the “total” amount of a protein, akin to an enzyme-linked immunosorbent assay that non-specifically detects multiple isoforms, a peptide from a region that is present across all desired isoforms to be included in the measurement is used as the surrogate peptide. The α61–80 peptide was selected as the surrogate peptide for a total α-synuclein on the basis of relatively few empirical observations of PTMs within this region [9]. The total α-synuclein concentrations can be found in Supplementary Table 6. Based on the total α-synuclein concentration, WT and M83 mice were readily distinguished by one-way ANOVA (F2,6 = 248; p < 0.0001, Fig. 3B) as were M83+/– and M83+/+ mice. Notably, the three mouse groups studied formed distinct clusters via principal component analysis followed by k-means clustering using all six peptide concentrations measured (Fig. 3C).
By comparing mean total α-synuclein in WT, M83+/– and M83+/+ mice, we observed that M83+/– and M83+/+ mice expressed 6- and 11-fold higher total α-synuclein than WT mice, respectively. These findings were consistent with western blot analysis demonstrating 6- and 9-fold greater total α-synuclein in M83+/– and M83+/+ mice, respectively, versus WT (Supplementary Figures 6 and 7). These relative increases are consistent with previous western blot studies demonstrating ∼3.3- (M83+/–) and 4.6- (M83+/+) fold higher α-synuclein (human and mouse) in the cortex of 3 mo old M83 mice compared to non-transgenic mice [4]. While comparing to this orthogonal technique provides evidence in support of the utility of the synuclein LC-MS/MS method; comparisons are limited by the fact that western blotting is a semi-quantitative technique. The quantitative LC-MS/MS results obtained here thus provide a more precise description of total soluble α-synuclein that is useful for interpreting the role of soluble proteins in the formation of pathological aggregates.
Quantification of α,β-synuclein
The synuclein LC-MS/MS method enabled quantification of not only α-synuclein, but also total α,β-synuclein based on α/β13–21 (Supplementary Table 6). Peptide α/β35–43 was also representative of both synuclein family members. In WT mice, the lower observed concentration of α/β35–43 compared to α/β13–21 may reflect PTMs within this region that change the mass of α/β35–43 (e.g., Y39 nitration/phosphorylation [9]), highlighting an area for further investigation. In general, α, β-synuclein peptides were both found in significantly higher concentrations in the overexpressing mice, consistent with increased total α-synuclein.
Species-specific contributions
Concentrations of mouse and human α-synuclein in M83 mice were monitored from species-specific peptides hu-α81–96 and mu-α81–96. As expected, with significantly greater concentrations of hu-α81–96 in M83+/– and M83+/+ mice compared to mu-α81–96, soluble α-synuclein was predominantly human in these mice. Although the difference was subtle, mu-α81–96 was found in higher concentration in WT compared to M83 mice (p-values < 0.0023, Fig. 3D and Supplementary Table 6). This trend is not observed in human brain tissue when comparing concentrations of TBS-soluble α-synuclein in PD compared to controls by western blot analysis [24]. Genetic differences and the consequences of massively increased synuclein expression in M83 mice could account for varying amounts of peptide mu-α81–96, while sequestration of soluble mouse α-synuclein by aggregates in M83 mice is another plausible explanation.
Synuclein-ome analysis
An examination of within-subject relative concentrations of synuclein peptides revealed discriminating patterns in WT vs. M83 mice (Fig. 3E). The ratio of α/β13–21:α/β35–43 in WT compared to M83 mice was significantly different (p-values = 0.0006), with M83+/– and M83+/+ mice being indistinguishable. Differences in internal peptide ratios may reflect structural modification in the synucleins, including changes in three-dimensional structure and/or PTMs, within the soluble fraction of synuclein, which in turn may alter tryptic digestion and/or peptide abundance observed by LC-MS/MS. Ubiquitination at Lys-21 of α-synuclein has been identified in Lewy bodies [9], and would in theory decrease the observed abundance of the unmodified peptide α/β13–21, as would N-terminal truncation and other PTMs that either modify tryptic cleavage or the mass of the peptide [25, 26]. The identification of such disease-related synuclein peptide signatures in M83 mice, identifies areas for future investigations. It is promising that quantitatively monitoring synuclein with an expansive coverage of the protein’s sequence, all within a single analysis, may be beneficial for screening sites of pathological modifications and identifying the most meaningful peptides (synuclein regions).
Conclusions
A multiplex MRM MS method was developed to analyze α- and β-synuclein, and validated in a transgenic mouse model of PD. The method enabled multiple simultaneous analyses, including i) quantification of total α-synuclein, ii) assessing contributions from different forms (α, β) and iii) species of synuclein (human vs. mouse) and iv) identifying differences within α- and β-synucleins that may be reflective of structural changes. Selectivity, sequence coverage, and multiplexing capacity were three key advantages of this approach over ligand binding methods such as ELISAs. The synuclein LC-MS/MS method also employs a streamlined sample preparation workflow relying on thermal enrichment of the target analytes instead of immuno-enrichment. This approach results in a simple and cost-effective workflow that is amenable to automation and free from antibody-related confounds. This is the first time, to our knowledge, that this highly efficient purification strategy has been utilized in tandem with MRM MS for characterization of synucleins. Analyses were further streamlined by using relatively short chromatographic run times (less than 10 min), enabling routine and high-throughput analyses. The synuclein LC-MS/MS method thus provides a versatile and efficient platform to understand how synuclein abundance and its different forms contribute to phenotypic outcomes, and will be a valuable resource to researchers investigating disease etiology, progression of disease, response to therapeutics and disease subtypes.
CONFLICTS OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
The authors acknowledge the support of the Michael Smith Foundation for Health Research (MLD and SS) and the Pacific Parkinson’s Research Institute (SS). We also thank Dr. Edward Fon of the Montreal Neurological Institute for supplying mouse tissue.
