Abstract
Background:
There is evidence that alterations in functional connectivity (FC) of the striatocortical circuits may appear before the onset of clinical symptoms of Parkinson’s disease (PD).
Objective:
The aim of this study was to investigate FC of the striatocortical circuitry in asymptomatic carriers of heterozygous glucocerebrosidase (GBA) mutations, which pose a significant risk for developing PD.
Methods:
Twenty-one parents of confirmed Gaucher disease patients who were carrying heterozygous GBA mutations and 18 healthy individuals matched for age and gender were included. GBA mutation analysis was performed in all participants. Clinical evaluation included neurological examination, Mini Mental State Examination, and UPDRS Part III. Structural and functional MRI data of 18 asymptomatic GBA mutation carriers (asGBAmc) and 17 healthy controls (HC) were available. FC was analyzed with seed-based approach.
Results:
Eleven asymptomatic mutation carriers had heterozygous p.L483P mutation, 6 subjects heterozygous p.N409S mutation and 1 subject heterozygous p.R392G mutation in GBA gene. Mini-Mental State Examination mean score was 28.77 (±1.16) and 29.64 (±0.70) in asGBAmc and HC groups, respectively (p = 0.012). Significant increased connectivity
Conclusion:
Our results suggest that alterations in striatocortical FC can be detected in asymptomatic heterozygous GBA mutation carriers who are at risk of developing PD. These findings may provide insight into network changes during the asymptomatic phase of PD.
INTRODUCTION
Genetic studies in Parkinson’s disease (PD) have led to identification of various monogenic forms as well as genetic risk factors [1]. Heterozygous mut-ations in glucocerebrosidase (GBA) gene are associated with a significantly increased risk of developing PD [2], as well as dementia with Lewy bodies [3, 4]. GBA mutations are the strongest known genetic risk factors in PD and the risk for PD in carriers is estimated at 15%by age 75. [5]. GBA gene encodes a lysosomal enzyme, glucocerebrosidase, genetic mut-ations in GBA gene alter glucocerebrosidase activity. Decreased glucocerebrosidase activity was found both in sporadic as well as GBA gene related PD (GBA-PD) patients [6–10]. Glucocerebrosidase deficiency increases the aggregation of alpha synuclein, the main component of Lewy bodies; aggregated alpha synuclein, in turn, leads to further impairment of glucocerebrosidase enzyme activity [11].
There is accumulating evidence that in neurodegenerative diseases, alterations in brain functional connectivity (FC) may be detected even before the onset of clinical symptoms [12–14]. Resting-state functional magnetic resonance imaging (rs-fMRI) is an advanced neuroimaging technique that investigates synchronous activity between spatially distinct regions of the brain and thus has the potential to detect changes in FC. Studies using rs-fMRI showed that alterations in striatocortical FC, akin to those seen in patients with PD, occur in non-presenting (asymptomatic) leucine-rich repeat kinase 2 (LRRK2) mutation carriers [15, 16], which are at risk to develop an autosomal dominant form of PD. Likewise, an altered pattern of cortical responses to cognitive tasks was observed in both GBA and LRRK2 mutation carriers without any clinical symptoms [17]. In this study, we aimed to assess whether alterations in striatocortical FC can be detected in asymptomatic heterozygous GBA mutation carriers (asGBAmc) which have an increased risk to develop PD.
METHODS
Subjects
We identified heterozygous GBA mutation carriers by contacting parents of patients with Gaucher disease, caused by homozygous GBA mutations. We reasoned that since the Gaucher disease is inherited as an autosomal recessive pattern, parents of children with this disease ought to carry at least one mutated GBA gene. Patient database of Istanbul Faculty of Medicine, Department of Pediatrics, Division of Ped-iatric Nutrition and Metabolism was searched for patients with a diagnosis of Gaucher Disease. Thirty-three parents were identified; they were contacted by phone and asked if they were willing to attend a ses-sion at which the purpose and the procedures of the study would be explained. Twenty-one of them acc-epted to participate in the study. Exclusion criteria were clinical diagnosis of any past or present neuro-logical disorder, history of major psychiatric disease, severe head trauma, alcohol or drug abuse, being pre-gnant, history of claustrophobia or metallic implant of any sort that prevents MRI examination and pathological MRI findings. Eighteen healthy volunteers with no history of any health issues and having no relatives with a known diagnosis of Gaucher’s disease were included as a control group (HC).
A semi-structured clinical interview was conduc-ted with all participants to evaluate the presence of non-motor features of PD such as hyposmia, paraso-mnia, autonomic symptoms or subjective cognitive impairment. Mini-Mental State Examination (MMSE) test and clinical neurological examination including part III of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) were performed to all subjects.
Genomic DNA was isolated from 2 ml venous blood samples in all participants. Primers were desig-ned for coding region and exon-intron boundaries of GBA gene region (NM_001005741; NP_001005741), samples were amplified and sequenced on ABI 3500 (Thermo Fisher). Sequences were blasted against Human Reference Genome (GRCh38/hg38), and se-quence alterations were expressed with the recom-mended nomenclature (Human Genome Variation Society).
The study was approved by the Ethics Committee of Istanbul Faculty of Medicine (2015/1862), and written informed consent was provided by all participants.
Image acquisition
Structural and functional imaging data were acqu-ired on a 3 T MRI scanner, using a 32 channel head coil (Philips Achieva, Best, The Netherlands) High resolution 3D T1-weighted TFE images were acq-uired with the following parameters; 250×250 mm field of view (FOV), 180 axial slices, voxel size 1 mm3 isotropic, flip angle 8°. Axial FLAIR images were also obtained and visually inspected (by MS and BB) to exclude participants with pathological MRI findings except for mild white matter hyperintensities.
We collected rs-fMRI data using EPI sequence (TR = 2 s, TE = 30 ms, flip angle = 77°, FOV = 224 x 240 mm, 36 axial slices, voxel size = 2×2× 4 mm). Following 10 dummy volumes, 214 volumes were collected with a total scan duration of 7.5 min. Subjects were instructed not to sleep and to keep their eyes closed during functional scanning.
MRI data analysis
Voxel based morphometry
Voxel-based morphometry (VBM) analysis was conducted in order to asses possible structural cha-nges between asGBAmc and control groups. Local distribution of gray matter was investigated using the VBM8 Toolbox (r445, http://dbm.neuro.uni-jena.de/vbm8/) runs within SPM8 software package (r6313, https://www.fil.ion.ucl.ac.uk/spm/). VBM8 processing pipeline incorporates Maximum A Posterior (AMAP) based segmentation [18] of T1 weigh-ted structural images into grey matter, white matter and cerebrospinal fluid compartments and high-di-mensional DARTEL normalization [19]. Normaliz-ation was implemented for the spatial registration of the structural images to the DARTEL template derived from 550 IXI healthy subjects (https://brain-development.org/ixi-dataset/) in MNI space with iso-tropic voxel size of 1.5 mm. Normalized grey matter images were smoothed with a Gaussian kernel (FWHM = 8 mm) before the statistical analysis.
Statistical inference about the VBM outputs was performed by permutation test via Statistical non-Parametric Mapping toolbox (SnPM13.1.0.8, http://nisox.org/Software/SnPM13/). Gray matter images were masked so that only the voxels higher than the absolute value of 0.1 were submitted to permutation based group level analysis for the comparison of asGBAmc and healthy controls with 10000 permutations. Resulting clusters (cluster forming threshold p < 0.001) were assessed for significance at Familywise α<0.05, as determined by permutation distribution [20].
MRI functional data pre-processing
Pre-processing was performed using the SPM8 toolbox (Wellcome Center for Human Neuroimaging). In order to correct motion artifacts functional data were realigned to the first functional volume. Afterwards structural and functional images were coregistered and normalized to MNI template with the spatial resolution of 2 mm3. Smoothing was ap-plied to the functional images with an isotropic Gaussian kernel (FWHM = 8 mm). Outlier volumes were labeled by ART toolbox using unsmoothed functional data and realignment parameters (http://www.nitrc.org/projects/conn) [21]. Difference in gl-obal signal (GS) between two successive volumes and framewise displacement (FD) were calculated and used to mark outlier volumes. FD more than 0.9 mm or GS more than five standard deviation from the mean were the criteria for outlier detection. One healthy subject who had outliers more than 20%of total functional volume (44 outliers) was excluded from further analysis. Effect of outlier volumes were also removed via regression (scrubbing) in the de-noising step of the connectivity analysis.
Seed-based functional connectivity analyses
CONN toolbox version 17 was used for seed-to-voxel connectivity analysis of the rs-fMRI data [21]. On the grounds that alterations in striatocortical connectivity by placing seeds at striatum were shown in a study including asymptomatic familial PD patients, using FSL Harvard-Oxford Atlas, we chose bilateral anterior and posterior putamen and caudate nucleus as seed regions (Fig. 1) [16]. Due to functional differences between anterior and posterior parts of the putamen, each of the left and right putamen seeds of AAL atlas were divided into two parts. The border between anterior and posterior parts of the putamen was defined as the line passing through the anterior commissure [22], a gap of 2 mm was left between these subdivisions.
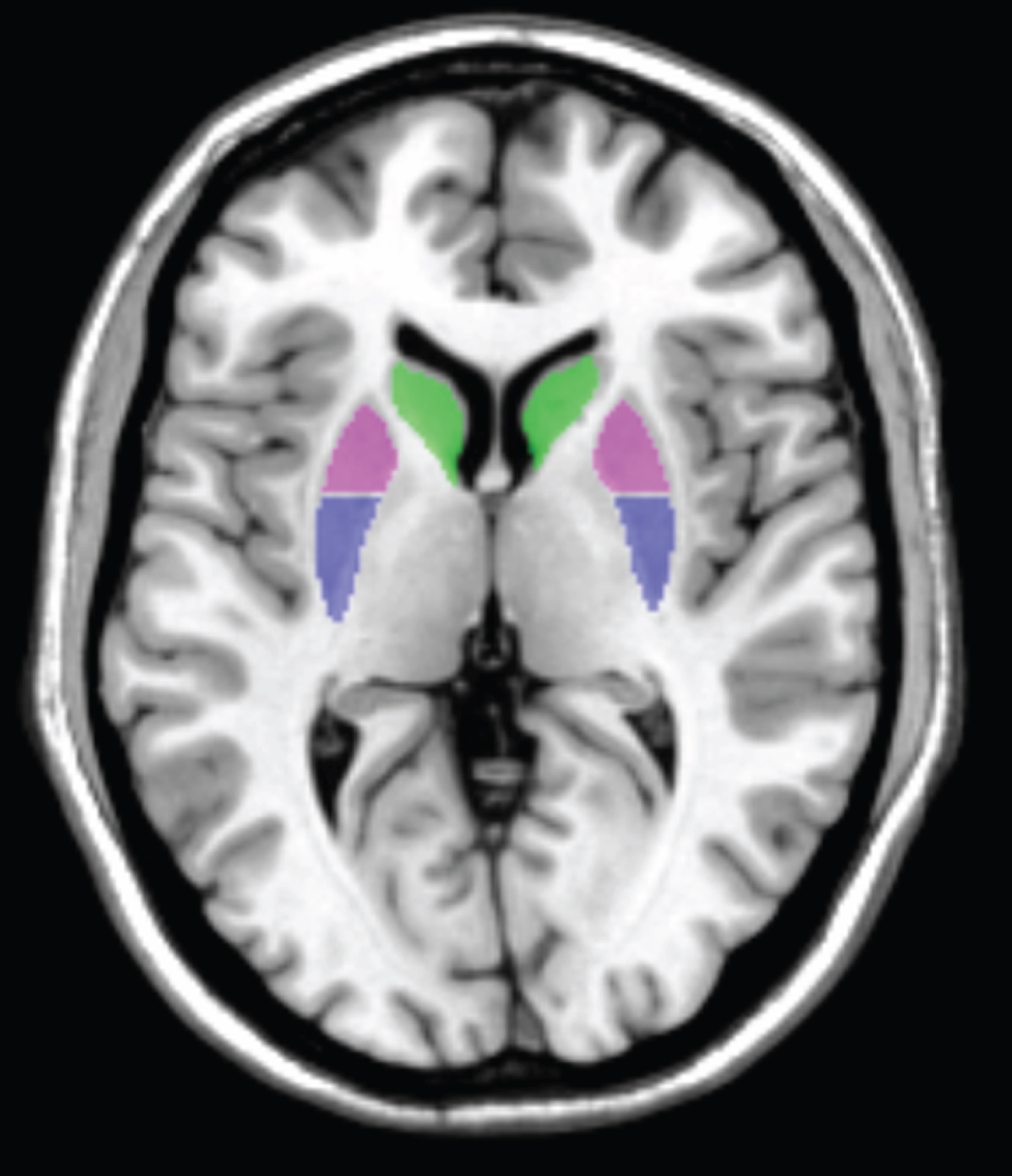
Subcortical seeds shown on axial slice: caudate nuclei (green), anterior putamen (violet), posterior putamen (blue).
In the denoising step, BOLD signal was band pass filtered at 0.01 - 0.1 Hz. In order to remove the potential sources of noise, confounding effects of the white matter and cerebrospinal fluid signal, scrubbing parameters, the rest condition and the six head motion parameters were regressed out.
In the first level analysis, Fisher transformed biv-ariate correlation coefficients (Z-scores) between the average BOLD signal of each seed region and all other voxels (seed-to-voxel) were calculated for each subject. Multiple comparisons correction was perfor-med with Analysis of Functional NeuroImages (AFNI)’s 3dClustSim (version = AFNI_20.1.13) to determine a cluster size threshold for statistical signific-ance (significance level: voxel p = 0.001, α=0.05 with 10000 Monte Carlo simulations) [23]. Areas showing significant connectivity changes was defined based on the intrinsic connectivity networks using the cortical parcellation atlas developed by Yeo et al. [24].
Statistics for clinical variables
We used SPSS 21 software (SPSS Inc, Chicago, IL, USA) for the statistical analysis of demographic and clinical data. Significance level was set at p < 0.05. Descriptive statistics were applied and Chi-square test and Fisher’s exact test were used for the group comparison. Non-normally distributed variables were analyzed with Mann-Whitney U test. Cor-relation analysis was performed with Spearman test.
RESULTS
Demographics
Twenty-one asGBAmc and 18 healthy control subjects participated in this study. Even though there was no prior history of claustrophobia, three subjects from the aGBAmc group experienced severe anxiety during MRI scanning and were excluded from study. One subject from the HC group was not also included in the neuroimaging analysis due to poor quality of MRI data. Finally, 18 aGBAmc subjects and 17 healthy subjects were included in the structural and functional analysis of MRI data. A detailed flow diagram of the study is shown in Fig. 2. Mean age of the asGBAmc and control subjects included in the neuroimaging analysis were 43.77 (±7.85) and 44 (±9.17) years respectively. Demographic data of the subjects are shown in Table 1. Family history of essential tremor and family history of PD were reported in 5 and 3 asGBAmc subjects, respectively. Neither essential tremor nor PD was reported in the first or second-degree relatives of the control subjects.
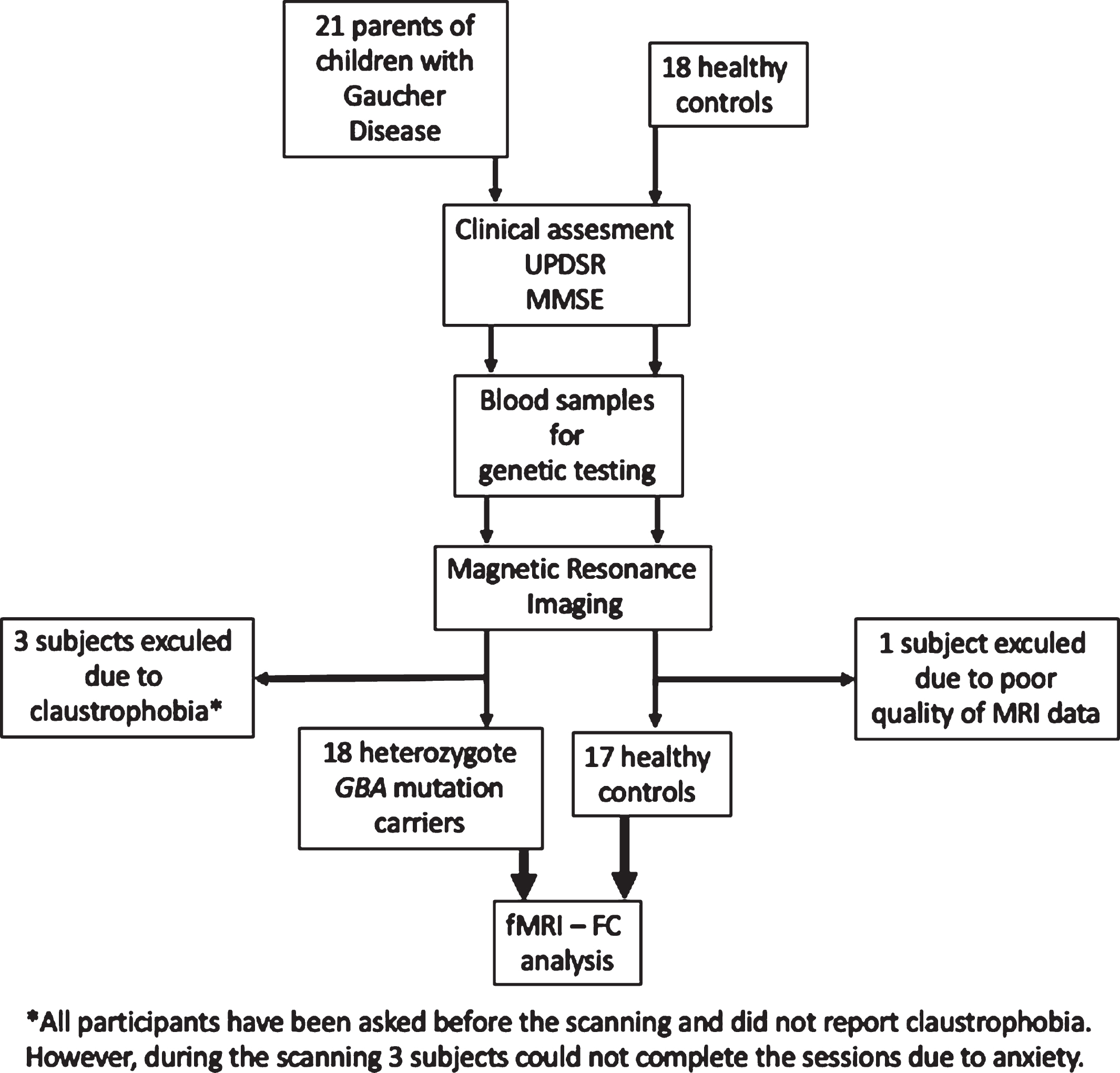
Flow diagram of the study. UPDRS, Unified Parkinson’s Disease Rating Scale; MMSE, Mini-Mental State Examination; GBA, Glucocerebrosidase; fMRI-FC, functional magnetic resonance imaging-functional connectivity.
Demographical and clinical features of subjects included in the neuroimaging analysis
asGBAmc, asymptomatic GBA mutation carriers; HC, healthy control; M, male; F, female; MMSE, Mini-Mental State Examination scores; UPDRS III, Unified Parkinson’s Disease Rating Scale Data are expressed as the mean±SD (range).
Molecular genetic analysis
Out of the 18 asGBAmc who participated in the fMRI study heterozygous c.1448T>C (p.L483P) mutation in exon 11 was identified in 11 subjects, heterozygous c.1226A>G (p.N409S) mutation in exon 10 in 6 subjects and heterozygous c.1174C>G (p.R392G) mutation in exon 9 in 1 subject (Table 2). Homozygous form of all these mutations have been reported to be associated with Gaucher disease [8, 25–27]. No pathogenic GBA gene mutations were identified in the HC subjects.
The type of mutations in GBA gene in asymptomatic carriers participating in the neuroimaging part of the study
#, number; M, male; F, female; het, heterozygous.
Clinical features
All subjects were right-handed. There was mild postural tremor in two subjects in the asGBAmc group without any other pathological findings, in particular no signs of parkinsonism. None of the participants had signs or history of autonomic dysfunction or REM sleep behavior disorder. Neurological examination was normal in the HC group. Mean UPDRS III score was 0.17 in the asGBAmc group (due to asymmetric mild postural tremor in two subjects) and 0 in the HC group. Six subjects in the asGBAmc group reported subjective mild cognitive problems. Mean MMSE score was 28.77 (±1.16) and 29.64 (±0.70) in asGBAmc and HC groups, respectively (p = 0.012).
Structural MRI findings
In the VBM analysis, no significant clusters of grey-matter showing volumetric changes were found in the asGBAmc group compared to healthy subjects.
Functional connectivity findings
Left posterior putamen and left caudate nuclei showed significant increased functional connectivity changes in the seed-to-voxel analysis (Table 3). For the left posterior putamen seed, between-group comparisons revealed significant hyper-connectivity in the asGBAmc group compared to HC with a cluster of 148 voxels corresponding to the left postcentral gyrus (Familywise α<0.05, adjusted for cluster size) (Fig. 3A). For the left caudate seed, between-group comparisons revealed significant hyper-connectivity in asGBAmc group compared to HC with a cluster of 201 voxels corresponding to parts of the right parietal operculum and right planum temporale (Familywise α<0.05, adjusted for cluster size) (Fig. 3B).
Seed-to-voxel results with threshold-cluster method (significance level: voxel p = 0.001, α= 0.05 with 10000 simulations)
asGBAmc, asymptomatic GBA mutation carrier; HC, healthy control; FC, functional connectivity.
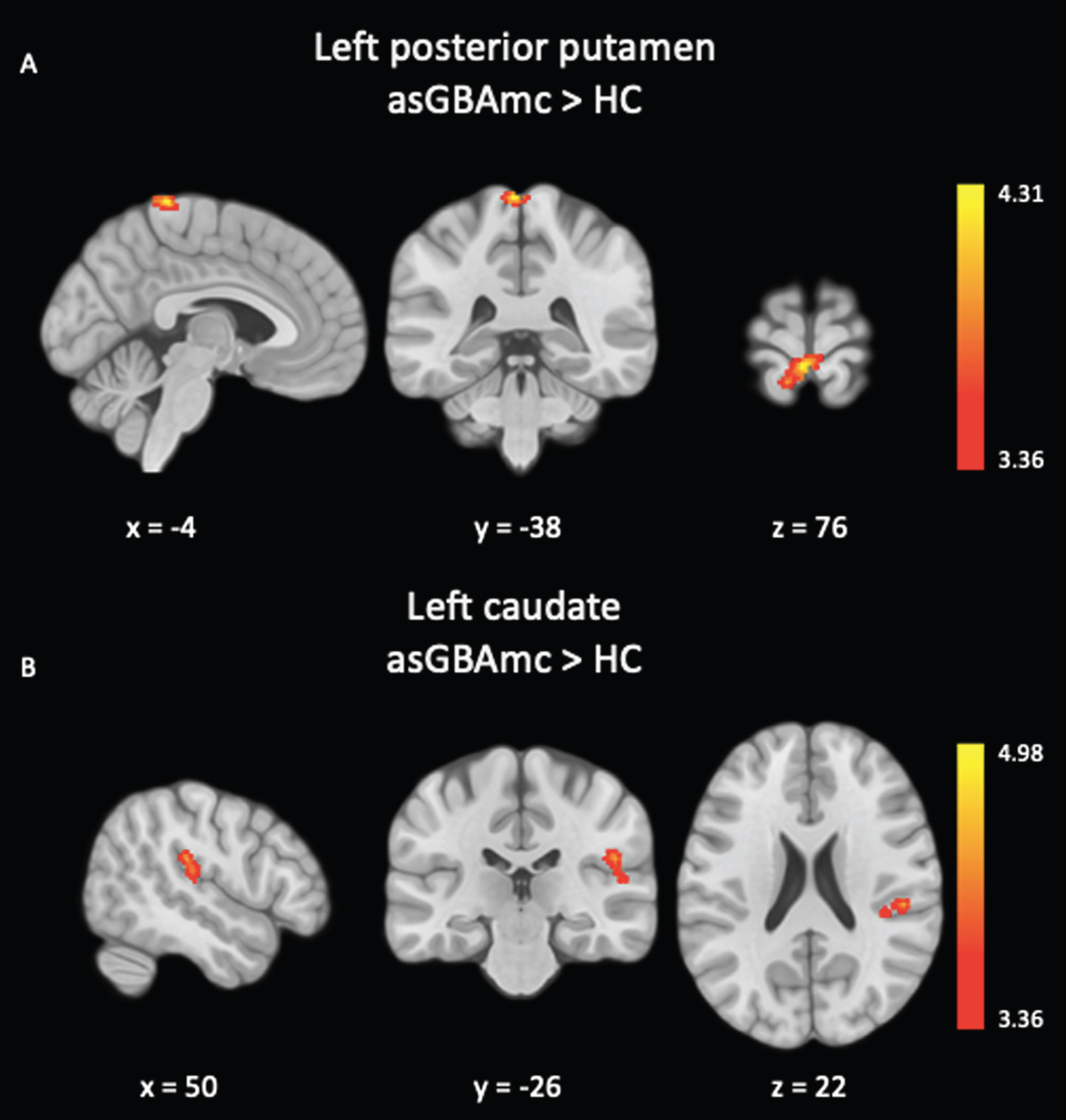
A) Functional connectivity differences for left posterior putamen seed. asGBAmc >HC: hyper-connectivity with left postcentral gyrus (cluster size: 148 voxels). B) Functional connectivity differences for left caudate seed. asGBAmc >HC: hyper-connectivity with right parietal operculum and right planum temporale (cluster size: 201voxels). Multiple comparisons correction was performed with Analysis of Functional NeuroImages (AFNI)’s 3dClustSim to determine a cluster size threshold for statistical significance (significance level: voxel p = 0.001, α= 0.05 with 10000 Monte Carlo simulations). asGBAmc, asymptomatic GBA mutation carriers; HC, healthy control.
Correlation analysis between the Z-scores (Fisher transformed bivariate correlation coefficients) of the FC values (left caudate and left putamen) and the MMSE and UPDRS III scores were performed in the asGBAmc group with Spearman test (Bonferroni-adjusted alpha level of 0.0125) and there was no significant association between FC values and clinical parameters in the asGBAmc group (Table 4).
Relationship between clinical parameters and connectivity of the left caudate and putamen in asGBAmc group
r, correlation coefficient; MMSE, Mini-Mental State Examination scores; UPDRS III, Unified Parkinson’s Disease Rating Scale part III scores.
DISCUSSION
The main findings in this study were, as compared to HC asGBAmc group showed: (i) increased FC between left posterior putamen and left postcentral gyrus, involving the striatocortical network, (ii) increased FC between left caudate and right parietal operculum and right planum temporale.
There have been several fMRI studies in patients with clinically manifest PD, with somewhat varying results. Both increased and decreased FC between basal ganglia and sensorimotor areas were reported in PD patients [22, 28–30]. These FC alterations were also correlated with the severity of motor symptoms [29]. A meta-analyis of the studies showed that the most consistent resting-state functional alteration in PD patients was the decreased connectivity of the posterior putamen [31], a primary basal ganglia nucleus receiving dense dopaminergic projections from substantia nigra which shows initial neurodegeneration in PD. In line with these findings, subjects with an increased risk of developing PD in our study exhibited alterations of the connectivity in striatocortical circuits. Unlike the pattern observed in symptomatic PD patients, at-risk subjects in the current study demonstrated hyper-connectivity of the putamen. This may be the initial pattern of connec-tivity in at-risk subjects which then evolve to hypo-connectivity of the striatocortical circuits in the course of the disease. In our study, we have found FC alterations between striatum and sensorial cortex but not between striatum and motor cortices. It was proposed that FC alterations between striatal regions and primary motor cortex, primary somatosensory cortex and inferior parietal cortex could be an implication of impaired sensorimotor integration in PD patients [22]. Our finding may reflect the early impairment of sensorial system before the involvement of motor system in the sensorimotor disintegration in at-risk individuals. Additionally, sensorial symptoms like paresthesia and pain are frequent complaints in PD and they may emerge as initial symptoms [32] before the manifestation of motor systems, suggesting the early impairment of sensorial system in PD. However, our study is limited by the lack of elaborative evaluation of pain and sensorial disturbances in asGBAmc group.
Asymptomatic subjects carrying a LRRK2 mutation, a monogenic form of PD, also comprise a risk group to develop PD. In rs-fMRI studies including asymptomatic LRRK2 mutation carriers, alterations in striatocortical and nigrostriatal connectivity have been reported: Vilas et al. found a decreased connectivity between the caudal motor part of the left striatum and the ipsilateral precuneus and superior parietal lobe and increased connectivity between the right substantia nigra and bilateral occipital cortical regions. In another rs-fMRI study in asymptomatic LRRK2 mutation carriers, inferior parietal region showed decreased connectivity with dorsoposterior putamen, but increased connectivity with the ventroanterior putamen [15, 16]. First, both results of these studies and our study suggest that alterations in distinct brain networks may appear before the onset of motor symptoms in individuals at high genetic risk for PD. Second, there may be differences in early pathophysiological changes in the asymptomatic carriers of LRRK2 and GBA mutations which may be the underlying factor of emergence of different pattern of connectivity seen in posterior putamen. In accordance with this assumption, in a recent study, asymptomatic GBA mutation carriers showed an increased striatal dopaminergic activity whereas asymptomatic LRRK2 mutation carriers showed a decreased activity [33] suggesting that the level of dopaminergic activity is different between these two distinct mutation carrier groups.
Functional neuroimaging studies in asymptomatic heterozygous GBA mutation carriers as well as in PD patients with GBA mutations are limited. In a task-based fMRI study using Stroop test, increased activity in right medial frontal gyrus and decreased activity in left lingual gyrus were described in asymptomatic GBA carriers as compared to both asymptomatic LRRK2 mutation carriers and healthy controls. In addition, increased activity was found in the left medial frontal gyrus and bilateral precentral gyrus in an incongruent task in the GBA group. Since there was no difference in the test performance between the two groups, authors interpreted increased fMRI activity during the task as a compensatory mechanism [17]. Altogether with these findings, our study provides additional evidence of functional changes in asymptomatic GBA mutation carriers. There are limited data on structural changes in GBA-PD patients. In a study using Freesurfer, no difference in cortical thickness and subcortical volumes was found between the manifesting GBA carriers and idiopathic PD patients, as well as between non-manifesting GBA carriers and healthy controls [34]. Taken together with our VBM results, there seem no structural changes in asGBAmc subjects in their midlife years, however, further longitudinal studies on this topic will be needed to show any structural changes that may occur in the following time period.
In the current study, even the asGBAmc subjects had no overt parkinsonian features, they had lower MMSE scores compared to healthy subjects. One of the feature of the GBA gene related PD is the more frequent presence and faster decline of the cognitive impairment. PD patients carrying a heterozygous GBA mutations have a significant increased risk for developing cognitive impairment compared to patients without a mutation [35]. A possible explanation for this might be that cognitive impairment may appear before the motor symptoms due to the altered connectivity of the caudate nucleus in GBA gene related PD; however, longitudinal studies following the asymptomatic carriers of GBA mutations are needed to confirm this assumption.
Our GBA group consisted of 11 subjects with L483P, 6 with N409S and 1 with R392G mutations. Different type of GBA mutations confer varying degrees of risk to develop PD and may be associated with different phenotypes. Compared to common N409S mutation, relatively rare L483P mutation is associated with an earlier onset of PD, more rapid cognitive decline [26, 30], and a higher risk of developing PD with an odds ratio of 8.17. Majority of our GBA subjects carried this particular mutation, hence they may be at greater risk of developing the disease and also demonstrating earlier changes in functional connectivity.
There are some limitations to our study. Despite the fact that this study included the highest number of asymptomatic GBA carriers in any fMRI study, the number of subjects is still relatively small. The small sample size did not allow to perform subgroup analysis such as comparison of fMRI findings in subjects with different mutations conferring varying degrees of risk to develop PD. Long-term follow-up and re-imaging of our asGBAmc subjects may reveal clinical relevance of network alterations found in this study. Since dopaminergic deficit is a very early feature in PD, and dopaminergic activity in the nigrostriatal pathway progressively decreases before the appearance of motor symptoms, alterations found in our study may presumably be linked to dopaminergic deficits. A future study with a combination of dopamine transporter imaging and rs-fMRI imaging may help to unravel such an association.
CONCLUSION
In conclusion, our results revealed that GBA mutations, which constitute the highest population attributable risk factor for PD, are associated with alterations in functional connectivity of the brain before the appearance of any clinical symptoms. Our results suggest that alterations in functional networks may be detectable not only in pre-symptomatic stages of monogenic forms of PD such as LRRK2 mutation carriers, but also in individuals who carry a genetic risk to develop PD.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
This work was supported by Scientific Research Projects Coordination Unit of Istanbul University (Project numbers 25144 and 42362).
