Abstract
Background:
Olfactory dysfunction (OD) is a frequent symptom of Parkinson’s disease (PD) that appears years prior to diagnosis. Previous studies suggest that PD-related OD is different from non-parkinsonian forms of olfactory dysfunction (NPOD) as PD patients maintain trigeminal sensitivity as opposed to patients with NPOD who typically exhibit reduced trigeminal sensitivity. We hypothesize the presence of a specific alteration of functional connectivity between trigeminal and olfactory processing areas in PD.
Objective:
We aimed to assess potential differences in functional connectivity within the chemosensory network in 15 PD patients and compared them to 15 NPOD patients, and to 15 controls.
Methods:
Functional MRI scanning session included resting-state and task-related scans where participants carried out an olfactory and a trigeminal task. We compared functional connectivity, using a seed-based correlation approach, and brain network modularity of the chemosensory network.
Results:
PD patients had impaired functional connectivity within the chemosensory network while no such changes were observed for NPOD patients. No group differences we found in modularity of the identified networks. Both patient groups exhibited impaired connectivity when executing an olfactory task, while network modularity was significantly weaker for PD patients than both other groups. When performing a trigeminal task, no changes were found for PD patients, but NPOD patients exhibited impaired connectivity. Conversely, PD patients exhibited a significantly higher network modularity than both other groups.
Conclusion:
In summary, the specific pattern of functional connectivity and chemosensory network recruitment in PD-related OD may explain distinct behavioral chemosensory features in PD when compared to NPOD patients and healthy controls.
Keywords
INTRODUCTION
Olfactory dysfunction (OD) is a very common non-motor symptom of Parkinson’s disease (PD) that appears in preclinical stages, years prior to the typical motor symptoms characterizing the disease and thus diagnosis [1–3]. This highlights the interest to use olfactory loss as a potential prodromal biomarker of PD [4]. An essential first step, however, is to differentiate PD-related OD from other forms of acquired non-parkinsonian OD (NPOD: such as post-viral, sinonasal or post-traumatic OD [5]). In this context, it is particularly interesting that PD-related OD has a specific impact on the trigeminal system which is distinct from other forms of NPOD [6, 7].
The trigeminal system is a chemosensory system that allows for the nasal and oral perception of sensations, such as the cooling of menthol, the burning or pain of chili peppers as well as touch, pressure, and temperature that are mediated by stimulation of the trigeminal nerve [8, 9]. This system interacts closely with the olfactory system for the perception of odorants [10, 11]. Consequently, patients with NPOD typically exhibit reduced trigeminal sensitivity as well [12–15]. In contrast, PD patients do not show any such decrease in trigeminal sensitivity and perception when measured behaviorally [6, 16], but rather exhibit a distinct pattern of electrophysiological activation that is different from both patients with NPOD and healthy controls [7]. Even though the cause of this distinctiveness is not fully understood, it suggests that PD specifically impairs the neural interaction between the trigeminal and the olfactory systems.
The olfactory and the trigeminal system interact at central levels and mutually enhance and suppress each other [17–19]. As a consequence, patients with anosmia typically show smaller central activation in response to trigeminal stimulation [13]. A possible neurobiological explanation is the overlap in central activation patterns between both sensory systems, especially in piriform cortex, insula, orbitofrontal cortex and middle frontal gyrus [20–22]. The connections between trigeminal and olfactory centers are therefore of prime interest if one aims at understanding how loss of olfactory function, PD-related or not, affects trigeminal sensitivity. To do so, functional connectivity characterizes networks, i.e., neural interactions amongst brain regions, and therefore allows to elucidate the mechanisms underlying human brain function in health and their changes in disease [23]. PD impairs several resting-state networks that include motor, striatal, cortical, limbic and sensorimotor areas [24–28], which may provide reliable biomarkers to monitor functional changes of PD [25, 29]. Alterations in functional connectivity patterns are associated with the severity of motor deficits [26, 30] but also to non-motor symptoms frequently observed in PD, such as cognitive impairment and dementia [31, 32], depression [33], dysphagia [34], REM sleep behavior disorder [35], misperception and hallucinations [36], and olfactory loss [37–40]. Even though we do not yet completely understand how brain network changes and neurodegeneration are related, resting-state fMRI studies suggest a link between alterations of functional connectivity and the clinical phenotype [29, 41]. This is further supported by longitudinal studies that revealed a disruption of functional connectivity [42] and decline of resting-state network integrity [43] as PD progresses, similar to what is observed in Alzheimer’s disease [44]. Functional connectivity may therefore elucidate how PD affects trigeminal-olfactory brain networks differently compared to that of NPOD patients.
This study aimed to assess PD-specific alterations in functional connectivity within the network responsible for processing of chemosensory information, both during chemosensory tasks and at resting-state, by comparing PD patients with NPOD patients and healthy controls. More specifically, we aimed to assess functional connectivity between olfactory and trigeminal brain processing areas using a seed-based correlation approach and network organization using a community detection approach at (1) resting-state and (2) following chemosensory stimulation. Importantly, during chemosensory stimulation, participants carried out tasks that relied on (2a) olfactory or (2b) trigeminal features of mixed olfactory-trigeminal stimuli, i.e., stimuli that activate both the olfactory and the trigeminal system [11].
In line with previous reports of a profound alteration of functional connectivity due to PD, we hypothesized that PD alters the chemosensory network in a disease specific manner, in contrast to NPOD which causes no direct alterations to the brain [45]. Specifically, we expected changes in resting-state connectivity in PD patients, but not in NPOD. However, during stimulation, we expected both patient groups to exhibit reduced connectivity between olfactory processing areas when performing an olfactory task as the result of impaired olfactory function in both groups. In contrast, we expected no connectivity changes in PD patients when performing a trigeminal task, as opposed to NPOD patients, in line with the unaltered trigeminal sensitivity in PD. Moreover, we further expected effects on the modularity of these networks. Modularity is a property of brain networks and describes to what extent modules are distinguishable within a network, i.e., brain regions forming a module have a strong functional connection between themselves, but are sparsely connected with other modules [46]. Since behaviorally PD patients have a dissociation between olfactory and trigeminal sensitivity, we expect olfactory and trigeminal modules to be less connected, resulting in higher modularity in this group, in line with the notion of altered modular organization of several brain functional networks and consequently increased modularity in PD [31, 47].
METHODS
Participants
A total of 45 participants completed the study of the total of 48 enrolled; three participants couldn’t complete the study and were excluded, 2 patients were claustrophobic, and one had a severe tremor and could not complete the scanning session.
We included 15 PD patients recruited through the Quebec Parkinson Network and the local PD association. Patients were diagnosed with PD according to the United Kingdom PD Society Brain Bank diagnostic criteria [48]. All Parkinson patients were on stable anti-Parkinsonian medication. Patients with unclear diagnosis or symptoms compatible with an atypical Parkinsonian syndrome were excluded, as were patients with nasal pathology that might have caused concurrent OD non-related to the disease (such as sinonasal pathologies, head trauma, or viral infections) [5].
Furthermore, we included 15 patients with NPOD that were recruited from our lab database of participants. The probable cause of OD was subjectively evaluated in an interview with the patient and using a questionnaire based on the position paper on OD [5]. We only included patients with post-infectious olfactory loss (n = 10) and olfactory loss due to sinonasal disease (n = 5), as idiopathic olfactory loss may be associated with an elevated risk of developing PD [49], post-traumatic olfactory loss may be associated with neural damages unrelated to the OD [50], and congenital anosmia is associated with altered cerebral structure [51]. The mean duration of OD was 10±9 years.
Finally, we enrolled 15 control participants from the community. They were matched in terms of age and sex to PD patients and they were in good general health with a normal sense of smell.
There was no significant differences in age between the 3 groups [One way ANOVA: F(2, 42) = 1.18; p = 0.31]. We further ascertained olfactory status by means of the “Sniffin Sticks” test (Burghart, Wedel, Germany), including olfactory detection threshold, odor quality discrimination and cued identification tasks [52, 53]. Univariate ANOVA revealed an effect of group [F(2,42) = 44.51; p < 0.001, ηp 2 = 0.68] and post hoc comparisons confirmed lower olfactory function in both PD patients and NPOD patients when compared to controls (both p < 0.001), but no difference between PD patients and NPOD patients. Next, screening for cognitive impairment using the Montreal Cognitive assessment (MoCA) [54] did not show any group differences [One way ANOVA: F(2, 42) = 0.04; p = 0.96]. However, there were a significant group difference in respect of depressive symptoms, as assessed using the Beck Depression Inventory (BDI) questionnaire [55] [One way ANOVA, F(2, 42) = 11.32; p < 0.001, ηp 2 = 0.35], with PD patients being significantly more depressed than both controls (p < 0.001) and NPOD patients (p = 0.003). See Table 1 for participant’s characteristics and Table 2 for PD patients’ individual profile.
Participants’ characteristics
Data are shown as means and standard deviation. LEDD, Levodopa equivalent daily dose; MoCA, Montreal Cognitive Assessment; UPDRS, Unified Parkinson Disease Rating Scale; BDI, Beck Depression Inventory; TDI, summation of the 3 olfactory subtests; threshold, discrimination and identification.
Demographic, non-motor symptoms, and co-morbidities information for each PD patient
H & Y, Hohen & Yahr disease stage; LEDD, Levodopa equivalent daily dose; RBD, Rapid-eye movement sleep behavior disorder; No, symptom not present; Yes, symptom present; Pr, probable.
All aspects of the study were performed in accordance with the Declaration of Helsinki on biomedical research involving human subjects. The study protocol was approved by the local ethics committees (Research Center of the Institut Universitaire de Gériatrie de Montréal (IUGM) at University of Montréal; at University of Quebec at Trois-Rivières). After being thoroughly informed on the study protocol, participants provided written consent prior to their inclusion in the study. All participants took part in an hour and a half behavioral testing session and an hour scanning session including structural and functional MRI imaging. Both sessions either took place in the same day or in two separates days according to the participant’s choice.
MRI data acquisition
MRI data were acquired on a 3.0 Tesla Prisma Fit MRI scanner (Siemens Magnetom) using a 32 channels head coil, at the Functional Neuroimaging Unit (UNF) of the IUGM research center. A T1-Weighted 3D magnetization prepared rapid acquisition gradient echo (MPRAGE) sequence (echo time (TE): 2.17 ms, repetition time (TR): 2400.0 ms, flip angle: 8°, voxel size: 1.0 * 1.0 * 1.0 mm, 176 contiguous sagittal slices, Field of view (FOV): 224 mm) was acquired for anatomical reference. Resting-state and task-related functional scans were also acquired with a whole brain echo-planar imaging (EPI) sequence sensitive to blood oxygen level-dependent (BOLD) contrast (acceleration factor slice: 3, TR: 832 ms, TE: 20.0 ms, flip angle: 58, Voxel size: 3.0×3.0×3.0, FOV:192, 51 slices, no gap). Resting-state data were collected before task related scans as resting-state networks might be influenced by odors [56]. For the resting state, one single measurement of 570 scans was acquired while the participants were instructed to lie still for 8 min with their eyes open and to fixate their gaze at a white cross projected on a black screen. For the activation task, 2 sequences of approximately 7 min, each including a single measurement of 565 scans, were acquired. Headphones were used to attenuate scanner noise. Patients with PD were on normal dopaminergic medication state during MRI scanning to control for involuntary head motions.
FMRI activation task
Stimulus were delivered during the functional scan using a MR compatible and computer-controlled air-dilution olfactometer [57]. Mixed olfactory-trigeminal stimuli embedded in a constant air flow (0.5 L/min) were presented intranasally to the participant through a cannula, that had been modified so that odors were presented independently to the right or left nostril. Two mixed olfactory-trigeminal stimuli were used, eucalyptus odor (eucalyptol 99% (470–82–6): Sigma-Aldrich, St. Louis, MO, USA) and almond odor (benzaldehyde 99% (100–52–7), Sigma-Aldrich). Participant were familiarized with the odorants and the task prior to the scanning session.
Participants performed two tasks in the scanner: an olfactory identification task and a trigeminal localization task; tasks were performed in a pseudorandomized order. The same odorants and the same stimulation paradigm were used for both tasks. Odors were presented in 8 stimulation blocks of 16 s separated by blocks of odorless air of 30 s to avoid habituation. For the olfactory identification task, participants identified the presented odor by button pressing. For the trigeminal localization task, participants identified the stimulated nostril (right or left nostril), no matter the presented odor using similar button presses. This trigeminal localization task is specific to the trigeminal system and based on the fact that localization of an odorous stimulus presented to only one nostril is not possible if the stimulus activates the trigeminal system [58, 59] (Fig. 1).
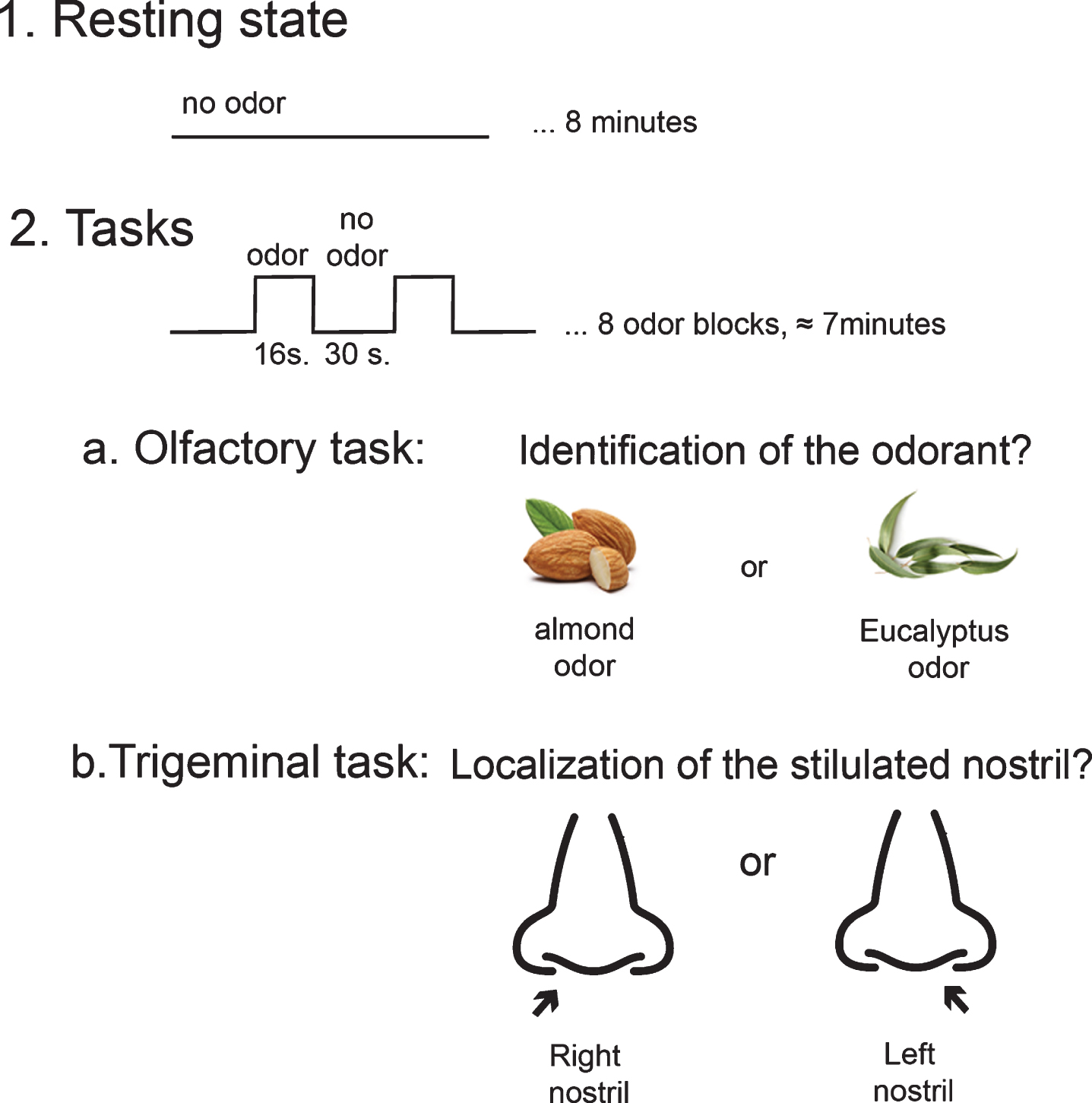
fMRI design. Participants were invited to first perform a resting-state of 8 minutes and then two tasks, in a pseudorandomized order, that rely on a) olfactory or b) trigeminal features of mixed olfactory-trigeminal stimuli (almond or eucalyptol odor).
MRI data preprocessing
Preprocessing was performed using Statistical Parametric Mapping (SPM12) (https://www.fil.ion.ucl.ac.uk/spm/software/spm12/) implemented in MATLAB (R2017b, MathWorks Inc., Natick, Massachusetts, USA). DICOM files were first converted to Nifti format. Standard preprocessing pipeline was applied to fMRI data including slice time correction, realignment, co registration to the participant’s T1 structural images, spatial normalization to Montreal Neurological Institute (MNI) standard brain template and spatial smoothing with a 6 mm full width half maximum (FWHM) isotopic Gaussian kernel filter.
Seed-based functional connectivity analysis
We aimed to specifically investigate the functional connectivity between trigeminal and olfactory central processing areas to assess potential differences between groups. According to this a-priory hypothesis, we chose a seed-based correlation approach that investigate the relationship between a specific region of interest in the brain with other region of interest. More specifically, this analysis indicates the functional coupling between the average BOLD time courses of a particular seed to the signal of other selected seeds [24].
We therefore selected specific regions of interest (ROI) contributing mainly to trigeminal and olfactory processing to assess potential alterations in functional connectivity between those regions. These regions were specifically selected from a meta-analysis employing a quantitative voxel-based activation likelihood estimation (ALE) approach on functional imaging data to identify human brain areas activated following chemosensory, i.e., olfactory, trigeminal, and mixed olfactory-trigeminal, stimulation [20]. The following regions of interest were selected: (1) piriform cortex, (2) medial insula, (3) amygdala, and (4) postcentral gyrus and orbitofrontal cortex (OFC). The original publication presented regions in Talairach coordinates; we converted them to MNI coordinates in Matlab [60]. By doing so, we noticed that the region that had been labelled OFC was in fact located in the anterior part of the insula; we thus labelled it (5) anterior insula (Fig. 2). In the original publication, postcentral gyrus, anterior insula, medial insula and piriform cortex had been characterized as activated following trigeminal stimulation, whereas amygdala and anterior cingulate cortex had been characterized as activated following olfactory stimulation [20]. See Table 3 for brain coordinates.
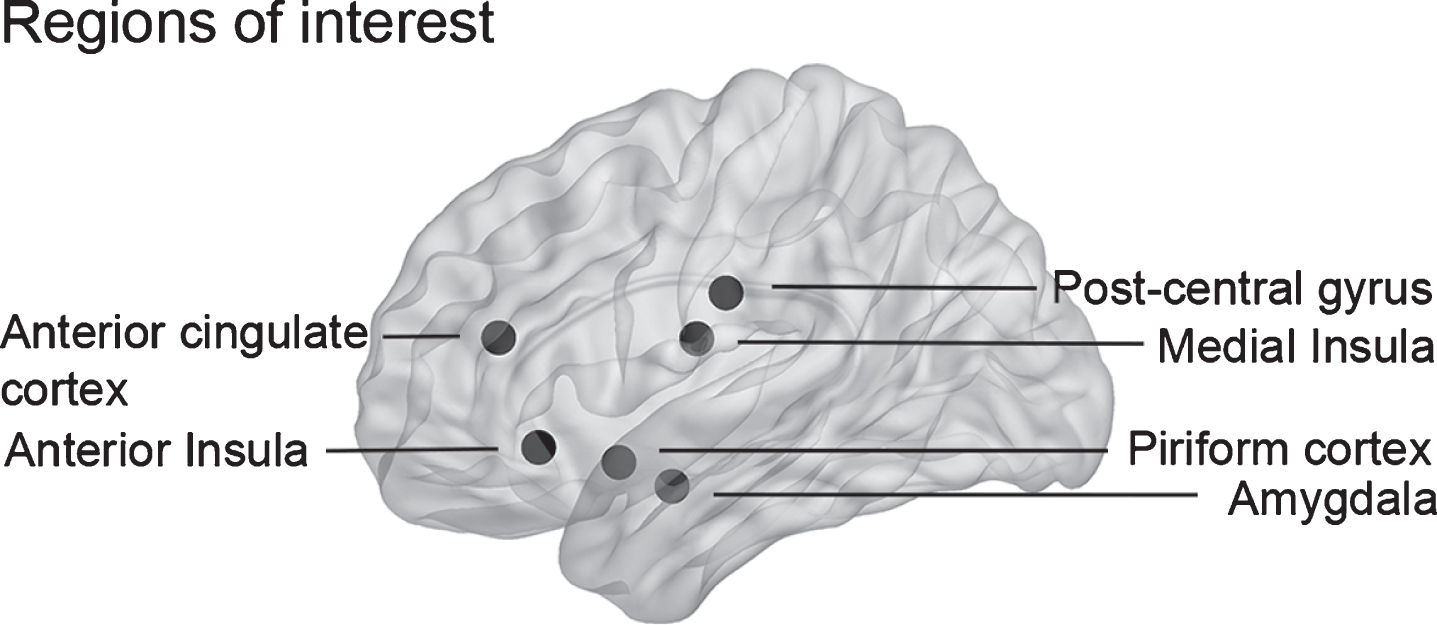
Schematic localization of the selected regions of interest. The following brain regions implicated in the processing of olfactory and trigeminal information were selected for seed-based correlations analysis: anterior cingulate cortex, anterior insula, postcentral gyrus, medial insula, piriform cortex, and amygdala.
Brain coordinates of the selected regions of interest
To define the ROI, we centered a spherical volume of interest around the activation maxima in each predefined coordinate, on both right and left hemispheres separately (radius = 6 mm) [61]. We then created a binary mask for each seed and used it to extract time courses of the BOLD response for each ROI of both the resting-state and the task related scans for each subsequent analysis. To correct for head movements, 6 parameters motions were regressed out from the times series (x, y, z translations and rotations) [62]. Then a band pass Butterworth filter (cutoff frequency 0.01–0.1 Hz, order 4th) was applied to the times series. As head motion might affect functional connectivity [63], we compared frame wise displacement metric between the 3 groups to exclude systematic differences in motion and we confirmed no group differences for resting-state [One way ANOVA: F(2, 42) = 0.76; p = 0.48], olfactory task [One way ANOVA: F(2, 42) = 0.77; p = 0.47] and trigeminal task [One way ANOVA: F(2, 42) = 0.41; p = 0.67]. Then, static functional connectivity network was estimated over the duration of the entire scan session of the resting-state session and for each task session. Functional connectivity values were obtained for each participant based on the Pearson’s correlation matrices from the ROI. The correlated values were then Fisher r to z- transformed to improve normality. For group comparisons, 3 separates pair wise t-tests were applied, between controls and PD patients, controls and NPOD and between PD patients and NPOD patients. To ensure intra patient reproductibility, we assess the biases of each individual on connectivity results using Jacknife resampling method (Supplementary Fig. 1).
Community detection and modularity of brain networks
To further assess how these regions of interest are connected and to identify brain networks, a community detection analysis was performed using the brain connectivity toolbox in Matlab (http://www.brain-connectivity-toolbox.net) [64]. To do so, the Louvain modularity optimization algorithm was applied on the data (resolution parameter: 0.95) [65, 66]. This is a well-suited method for brain analysis questions that allows for detecting communities in networks by optimizing the modularity. A community, or a module, is a group of nodes (in this context: regions of interest) from the network, where nodes in the same community are strongly connected with each other but sparsely connected to nodes from different communities. The modularity globally evaluates the difference between the actual probability of the connection of two nodes in a same community and the estimated randomly connection probability between these two nodes [67]. The modularity index (Q, between – 1 to 1) is a measure of the strength of the network division into sub-networks which are characterized by strong interconnections, with positive values indicating higher levels of connectivity and greater functional specialization of a network, negative values indicating reduced connectivity and a Q value of 0 reflecting a random network [65, 68]. The Louvain algorithm optimizes the modularity index for each community (or here grouping of regions of interest) until convergence to a modularity maximum. In other words, this is a hierarchical analysis that is repeated as many times as necessary so that the modularity index doesn’t change anymore, which result in the identification of the best possible modular partitions and ensures the validity of the results. We compared the modularity distributions of our data with the modularity of a 1000 randomly generated networks derived from topological rewiring procedure [69], group comparison was performed using the non-parametric Monte Carlo resampling test.
Availability of data
Data will be made available upon request.
RESULTS
Seed-based functional connectivity
At resting-state, we observed reduced functional connectivity in PD patients compared to controls between the right anterior insula and the right medial insula (t(27) = –2.44, p = 0.022). No differences were found between NPOD patients and controls. PD patients further showed increased connectivity compared to NPOD between the right piriform and the right postcentral gyrus (t(27) = –2.20, p = 0.036).
When performing the olfactory identification task within the scanner, PD patients showed increased functional connectivity compared to controls between the right amygdala and the bilateral anterior cingulate gyrus (left: t(28) = 2.23, p = 0.034; right: t(28)=2.15, p = 0.040). Similarly, NPOD patients also showed increased functional connectivity compared to controls, between the right piriform and the left anterior cingulate gyrus (t(28) = 2.09, p = 0.045). No differences were found between PD patients and patients with NPOD.
When performing the trigeminal localization task, no differences were found between PD patients and controls. In contrast, compared to controls, NPOD patients showed reduced connectivity between the right medial insula and the bilateral postcentral gyrus (left: t(28) = –2.06, p = 0.049; right t(28) = –3.20, p = 0.003). When we compared both patient groups, PD patients showed reduced connectivity between the left piriform and the left amygdala (t(28) = –2.75, p = 0.010) compared to NPOD (Fig. 3).
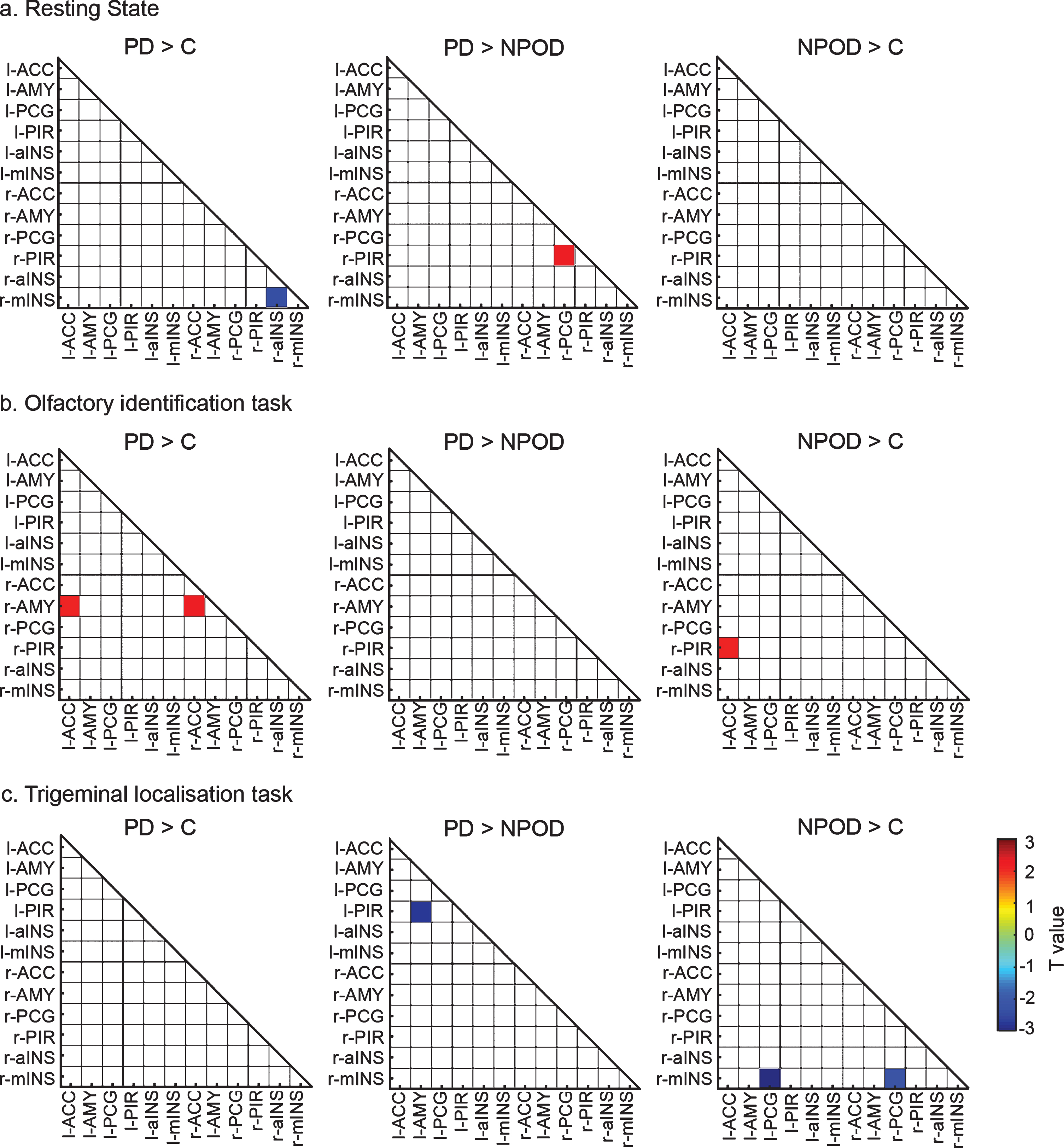
Functional connectivity matrices. Differences in functional connectivity between groups at a) resting state, b) during the olfactory identification task and c) during the trigeminal localization task. Contrast between PD and controls, PD and NPOD and NPOD and Controls are presented for the left and right selected region of interest. Only significant results are shown. Colors represent t-values, red correspond to increase connectivity and blue correspond to reduced connectivity. ACC, anterior cingulate cortex; aINS, anterior insula; PCG, postcentral gyrus; mINS, medial insula; PIR, piriform cortex; AMY, amygdala.
Community detection and modularity of brain networks
The community detection analysis revealed different modules amongst the ROIs with an optimal modularity during resting-state and task-related scans (Fig. 4). Next, we compared network modularity between groups for each task.
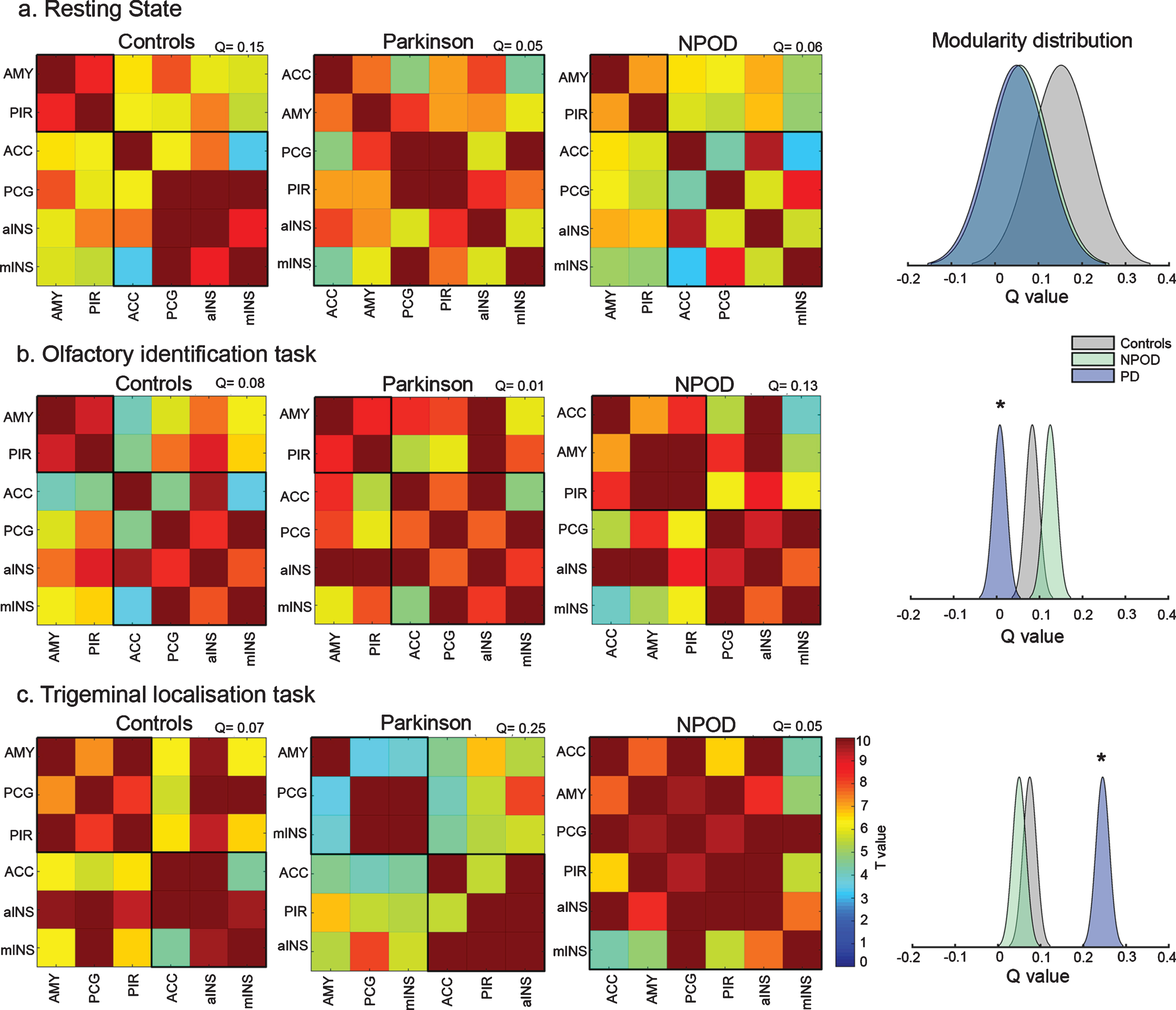
Community detection of brain networks and modularity distribution. Detection of community amongst the selected regions of interest for the left and right brain side, for each group at a) resting state and while performing b) an olfactory task and c) a trigeminal task. Black squares regroup the identified networks. Colors represent T-values. Modularity (Q) distribution of the identified networks for the 3 groups is presented for each condition. ACC, anterior cingulate cortex; aINS, anterior insula; PCG, postcentral gyrus; mINS, medial insula; PIR, piriform cortex; AMY, amygdala.
During resting-state, we identified modules in both, the control participants and NPOD patients, namely Module 1, including amygdala and piriform cortex, and Module 2, including the anterior cingulate gyrus, medial and anterior insula as well as the postcentral gyrus. We did not observe any modules in PD patients. Nevertheless, modularity was not significantly different between groups (z = 1.06; p = 0.2).
While performing the olfactory identification task, we identified the same modules as at resting-state for controls. These same modules were also weakly coupled in PD patients. In NPOD patients, however, we identified a different network, with Module 1, including amygdala, piriform cortex, and anterior cingulate gyrus and Module 2, including postcentral gyrus, anterior insula and medial insula. The modularity was significantly lower in PD patients when compared to both controls (z = 3.43; p < 0.001) and NPOD (z = 5.32; p < 0.001), but not different between controls and NPOD.
While performing the trigeminal localization task, controls exhibited a different network as Module 1 regrouped amygdala, piriform cortex and postcentral gyrus while Module 2 comprised anterior cingulate gyrus, anterior insula and medial insula. In contrast, PD patients’ network consisted of Module 1 including amygdala, postcentral gyrus and medial insula and Module 2 including anterior cingulate gyrus, piriform cortex and anterior insula. No modular network was identified for patients with NPOD. The modularity distribution of the networks was significantly higher in PD patients when compared both to controls (z = 7.59; p < 0.001) and NPOD patients (z = 8.68; p < 0.001), but not different between controls and NPOD(Fig. 4).
DISCUSSION
In this study, we compared functional connectivity of the chemosensory network between PD patients, patients with NPOD, and controls. We found impaired functional connectivity in PD patients at resting-state while no such changes were observed for NPOD patients, when compared to healthy controls. However, when assessing the chemosensory network organization, we found no group difference in modularity of the networks. When participants carried out a chemosensory task, a different picture emerged. Both patient groups exhibited impaired connectivity when they executed an olfactory task. Furthermore, the network modularity was significantly lower for PD patients than in controls and NPOD patients. However, when performing a trigeminal task, no changes were found for PD patients, but NPOD patients exhibited impaired connectivity. In contrast, PD patients’ network exhibited a significantly higher modularity than both other groups.
With regards to the resting-state results, PD patients exhibited reduced connectivity between the anterior and medial part of the insula. This is in line with the notion of PD affecting functional connectivity in several regions and networks, including the default mode network, limbic, motor and sensorimotor networks, with a large heterogeneity [25, 28]. For instance, PD leads to both reduced, e.g., in mesostriatal and corticostriatal networks, and increased, e.g., in fronto-parietal regions and cerebellum, functional connectivity [70]. Further, the olfactory deficit in PD is associated with a changed pattern of functional connectivity between primary sensory area and the posterior cingulate cortex [37], a reduced connectivity between the orbitofrontal cortex and the insula [40], a reduced connectivity of the amygdala [39], and the limbic/paralimbic cortex [38]. We found similar connectivity changes in the insula, which is a known center of chemosensory and multisensory integration as well as pain processing [71]. Specifically, the anterior insula receives projections from the piriform cortex and is consistently activated by olfactory stimulation [72], whereas the medial insula receives somatosensory, pain, temperature and trigeminal related information [73]. Our result therefore suggest that PD impairs connectivity between olfactory (anterior insula) and trigeminal (medial insula) processing areas, in line with the notion of an altered interaction between these two systems. In contrast to the finding in PD, there are no connectivity changes in NPOD at rest, when compared to healthy controls. This is in line with the literature on patients with anosmia [74] and result of the fact that NPOD is not a disease of the brain.
In contrast to the resting-state data, both groups of patients showed increased connectivity compared to controls while they were carrying out an olfactory task. Increased connectivity may be found when a system is recruited for a specific task, especially if participants struggle to perform the task [75]. Therefore, this result is in concordance with impaired olfactory performance in both groups of patients. Similar networks were identified in PD patients and controls, but the modularity distribution was significantly weaker in PD patients than in controls and NPOD patients. The alteration in network modularity is in line with previous reports showing that PD alters modular organization of different brain networks [31, 47]. In PD patients connectivity between the right amygdala and the bilateral anterior cingulate gyrus is changed; both areas are impaired in PD [76, 77] and associated to PD-related OD [38, 79].
The picture was different for NPOD patients, in whom connectivity between the right piriform and the left anterior cingulate gyrus was altered. However, in contrast to PD patients, when looking at the network organization, we found no group differences in modularity despite different identified networks. In fact, previous studies showed that olfactory stimulation in NPOD typically leads to reduced activation in olfactory brain processing areas, and the severity of the olfactory deficit is related to the extent of the chemosensory network recruitment [74]. Further, the piriform cortex was found to be more activated in response to olfactory stimulation in NPOD patients in contrast to pure sniffing [74, 80], which is a task capable of activating the olfactory network even in anosmia [81]. In line with this, NPOD patients show reduced connectivity in response to sniffing of odorless air in a broad variety of brain areas including chemosensory areas such as the entorhinal cortex, piriform cortex, and primary somatosensory cortex [74]. In summary, both patients’ groups show impaired connectivity while performing an olfactory task, but different regions are involved, and distinct mechanism seems to be implicated as PD-related OD specifically affect modularity.
When performing a trigeminal task, connectivity was reduced in NPOD patients but not in PD patients. Similar to the results in the olfactory task, this reflects behavioral findings as trigeminal sensitivity is not impaired in PD patients, but typically reduced in NPOD patients [6]. Interestingly, our data demonstrate a specific chemosensory network organization with significantly higher modularity in PD while they perform the task. In the PD network, the two modules consisted of (1) amygdala, postcentral gyrus and medial insula, and (2) anterior cingulate gyrus, piriform cortex and anterior insula. This suggests that although showing low modularity at rest and during the olfactory task, PD patients recruit the chemosensory network in a distinct fashion while executing a trigeminal task. Densely intra-connected regions, as reflected by a higher modularity, in the nervous system is an indication of effective segregate processing for specialized function [82]. This result is concordant with our previously published results suggesting that PD patients process central trigeminal information differently, when compared to controls and patients with NPOD [7]. More precisely, network organization showed that the piriform cortex and the amygdala were grouped together in the same module in all conditions and groups, with the only exception being PD patients while performing the trigeminal task. Rather, in PD patients the piriform cortex is replaced, in the module, by the medial insula. We hypothesize that reduced functional connectivity between piriform cortex and insula, or between piriform cortex and amygdala, or both, might explain this network reorganization. In fact, alterations of the insula were found to be implicated in several non-motor symptoms of PD, by its interaction with several brain networks, and are particularly contributing to somatosensory alterations in PD [83]. Thus, reduced connectivity of this region might explain different trigeminal processing in PD patients. Interestingly, when focusing on specific differences observed between PD and NPOD patients, reduced connectivity between the piriform cortex and the amygdala is observed while performing the trigeminal task. Both regions are known to be implicated in PD pathology and PD-related OD [84, 85]. Therefore, investigating the link between these two brain processing areas might be the key towards the development of discriminating tool between both forms of OD. Other changes between both groups of patients include reduced connectivity of the right piriform cortex and postcentral gyrus at resting state. Again, the piriform cortex seems to be specifically implicated in PD-related OD. Atrophy and altered brain metabolism of the piriform cortex was indeed previously associated to OD in PD [84, 86]. Futures studies should focus on the piriform cortex in both forms of OD, using seed-based functional connectivity to characterize its connections with the rest of the brain.
As for patients with NPOD, no modularity changes were observed but they showed reduced connectivity between the medial insula and the postcentral gyrus while performing the trigeminal task. Both regions are important for processing of trigeminal information and this result is in line with the notion of impaired trigeminal sensitivity in NPOD. Trigeminal stimulation in these patients is associated with reduced activation in somatosensory brain processing areas (e.g., S1, S2, insula), but increased activity in regions such as parahippocampal, cingulate gyrus; which was suggested to be the consequence of potential compensatory mechanism [13, 88]. In line with our results, NPOD patients have unchanged network recruitment but reduced connectivity of these network [89].
In summary, while performing a trigeminal task, functional connectivity changes are observed only in NPOD patients and not in PD patients, as opposed to control. However, modular organization of the chemosensory network is specifically altered in PD patients as opposed to controls and NPOD. As modularity was affected during both tasks, reduced during olfactory and increased during trigeminal tasks in PD patients, futures studies should further focus on modular organization of the chemosensory network as a potential biomarker of olfactory-trigeminal alterations in PD.
We acknowledge that the sample size used in this study is relatively small, the results should therefore be interpreted carefully. Moreover, only a limited number of nodes were selected for connectivity analysis while more nodes are typically selected in network neuroscience; therefore, the results should only be interpreted within the network of interest and not be interpreted as a general effect. However, these regions of interest were specifically selected to assess the olfactory and trigeminal systems and to precisely answer our hypothesis. Nevertheless, this study is well controlled, the inclusion of both a healthy control group and a control group of patients with NPOD is crucial towards a better understanding of PD-related OD. A rigorous screening procedure was performed to include only patients with post-viral and sinonasal OD that are difficult to recruit. A larger study including both healthy controls and controls with NPOD should be carried out to better define a specific pattern of functional connectivity in PD and network modularity.
In conclusion, we here report preliminary findings showing a potentially specific pattern of functional connectivity and chemosensory network recruitment in PD-related OD, when compared to patients with NPOD and healthy controls, that has to be further investigated.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
This work was supported by grants from the Quebec Bio-imaging Network, by the Fonds de la recherche du Québec – Santé and by the Université du Québec in Trois-Rivières. We would like to thank the Quebec Parkinson Network for the help with the recruitment of PD patients, the IUGM staff, Lison Guibert-Bérengier for the help with the testing and all the participants implicated in this study.
