Abstract
Background:
Several dietary components have been shown to be neuroprotective against risk of neurodegeneration. However, limited observational studies have examined the role of overall diet quality on risk of Parkinson’s disease.
Objectives:
We examined the associations between diet quality and risk of Parkinson’s disease in a prospective cohort study and meta-analysis.
Methods:
Included in the cohort study were 3,653 participants (1,519 men and 2,134 women; mean age: 81.5 years) in the Geisinger Rural Aging Study longitudinal cohort in Pennsylvania. Diet quality was assessed using a validated dietary screening tool containing 25 food- and behavior-specific questions in 2009. Potential Parkinson’s cases were identified using electronic health records based on ICD9 (332.*), ICD10 (G20), and Parkinson-related treatments. Hazard ratios (HRs) and 95% confidence intervals (CIs) across diet quality tertiles were calculated using Cox proportional hazards models after adjusting for potential confounders. We further performed a meta-analysis by pooling our study with four published papers on this topic. Random-effects model was utilized to calculate the pooled risk ratios and 95% CIs.
Results:
During a mean of 6.94 years of follow-up, 47 incident Parkinson’s cases were documented. Having high diet quality at baseline was associated with lower Parkinson’s disease risk (adjusted HR for the highest vs the lowest diet quality tertile = 0.39; 95% CI: 0.17, 0.89; p-trend = 0.02). The meta-analysis including 140,617 individuals also showed that adherence to high diet quality or a healthy dietary pattern was associated with lower risk of Parkinson’s disease (pooled risk ratio = 0.64; 95% CI: 0.49, 0.83).
Conclusion:
Having high diet quality or a healthy dietary pattern was associated with lower future risk of Parkinson’s disease.
INTRODUCTION
Parkinson’s disease is the second most common neurodegenerative disease. With a rising trend of prevalence, more than one million individuals in the United States may suffer from Parkinson’s disease by 2030 [1]. Motor symptoms, such as tremor, rigidity, bradykinesia, are commonly observed in individuals with Parkinson’s disease [2]. Growing evidence also suggests that a wide range of non-motor symptoms, including cognitive impairment, dementia, depression, constipation, and sleep disorders, can also occur in individuals with Parkinson’s disease [3].
A number of observational studies have examined the association between individual dietary component and risk of Parkinson’s disease [4, 5]; however, observational studies investigating the role of overall diet quality and/or dietary pattern on the pathogenesis of Parkinson’s disease are limited. Understanding the impact of overall diet quality may provide better insights in relation to diet and Parkinson’s disease because synergistic effects of food components and potential nutrient interactions in diets can also be considered [6, 7].
In this study, we aimed to prospectively examine the association between overall diet quality and risk of incident Parkinson’s disease in a longitudinal cohort of 3,653 community-dwelling older adults who were not diagnosed with Parkinson’s disease at baseline. Diet quality was assessed by a validated Dietary Screening Tool (DST). In our previous validation analysis including 122 oldest old aged 80 years and over, significant correlation between the DST score and the Healthy Eating Index (HEI)-2015 score was observed [8]. Participants categorized as having high diet quality by the DST also had significantly higher HEI-2015 score which represents better adherence to the Dietary Guidelines for Americans [8, 9]. In addition, our previous study including 204 older adults aged 65 years and older showed that participants categorized as having high diet quality by the DST had better overall biochemical indicators of nutritional status compared with those being categorized as having low diet quality [10]. The evidence suggests that the DST is a valid measurement of diet quality among older adults and the oldest old [8, 10]. We also performed a meta-analysis of observational studies to summarize current evidence on diet quality and the development of Parkinson’s disease. We hypothesized that having high diet quality or adherence to a healthy dietary pattern would be associated with lower risk of developing Parkinson’s disease.
METHODS
Prospective study
Study population
The Geisinger Rural Aging Study (GRAS) originated in 1994 as a longitudinal cohort including 21,645 community-dwelling older persons aged 65 years and over in rural Pennsylvania with limited diversity in race and lifestyle factors. Participants who enrolled in a Medicare-managed health organization through the Geisinger Health System at the time of entry were eligible. Detailed information regarding the GRAS cohort has been reported previously [11].
In October 2009 (baseline of this current study), mailed surveys were sent to 5,939 surviving GRAS participants to obtain demographic and descriptive information and diet quality. After excluding participants who did not return completed surveys and diet quality assessment or had prevalent Parkinson’s disease at baseline or lost to follow up, 3,653 participants were included in the current analysis (Fig. 1).
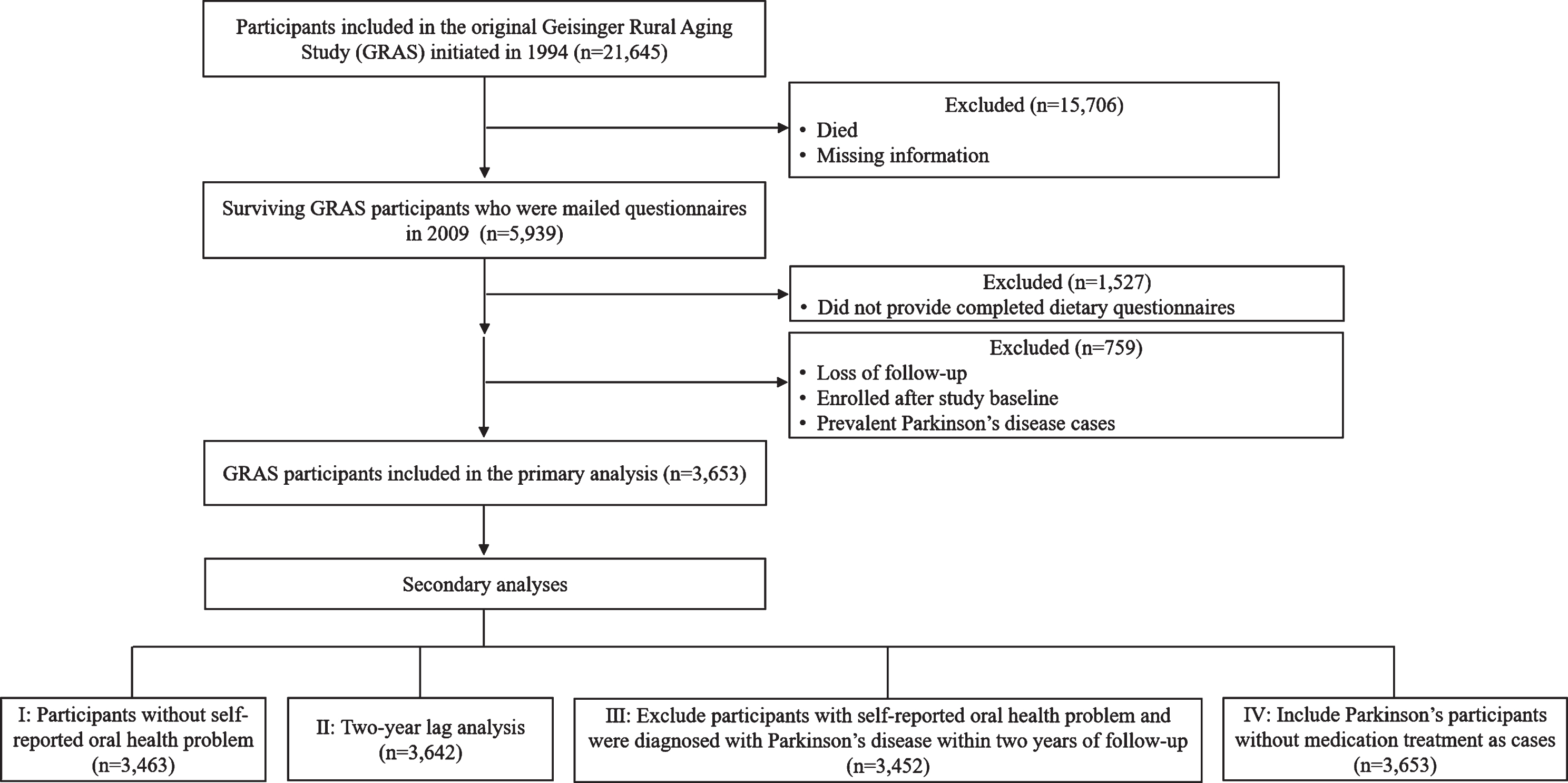
Flow chart of Geisinger Rural Aging Study (GRAS) participants. A total of 21,645 older adults were enrolled in GRAS longitudinal cohort starting from 1994. Mailed surveys were sent to 5,939 surviving GRAS participants in 2009. Among participants who received surveys, 3,653 of them returned completed survey and did not have prevalent Parkinson’s disease at baseline and were therefore followed through September 2019.
Standard protocol approvals and patient consent
Approval for implied consent through completion of the mailed surveys was obtained from the Office of Research Protections at The Pennsylvania State University and the Human Research Protection Program of the Geisinger Health Systems Institutional Review Board.
Assessment of diet quality
Diet quality was assessed at baseline in 2009 using the mailed Dietary Screening Tool (DST) survey questionnaire that included twenty-five food- and behavior-based dietary questions. Some sample questions in the DST are “How often do you usually eat whole grain breads?” and “How often do you usually eat candy or chocolate?”. The total DST score could range from 0 to 100 and 5 bonus points would be scored for dietary supplement usage [10]. Participants’ diet quality categories were determined based on tertiles of the DST score. More detailed information regarding DST development and validation in older persons has been described previously [8, 12].
Assessment of incident Parkinson’s disease cases
Parkinson’s disease cases were identified using Electronic Health Record (EHR) based on ICD9 (332.*) and later verified by ICD-10-CM code G20 through September 9, 2019. Incident Parkinson’s disease cases were defined as being diagnosed at least 1 year after completing diet quality assessment and receiving Parkinson-related medication treatment(s), including medication containing carbidopa/levodopa (Sinemet, Parcopa, Stalevo, Duodopa), ropinirole (Requip), pramipexole (Mirapex), rasagiline (Azilect), pergolide (Permax), rotigotine (Neupro), entacapone (Comtan), selegiline (Eldepryl, Zelapar), carbidopa (Lydosyn), or amantadine (Symmetrel).
Assessment of covariates
Self-reported demographic and descriptive variables, including age, sex, race, weight, height, educational level, self- or proxy-reporting, smoking status, oral health status, living status (live alone or with family members) and living arrangement (at home, skilled nursing, or other facility), were collected using the mailed survey questionnaire at baseline. Body mass index (BMI) was calculated as weight (kg)/height (m)2.
Statistical analyses
Statistical analyses were performed using SAS version 9.4 (SAS Institute, Cary, NC). Characteristic and demographic variables were presented as means along with standard errors for continuous variables and percentages for categorical variables. Hazards ratios (HRs) and 95% confidence intervals (CIs) across diet quality tertiles were calculated with the lowest tertile as a reference in Cox proportional hazards models after adjusting for potential confounders, including age, sex, race, BMI, educational level, self- or proxy-reporting, smoking status, oral health status, living status, and living arrangement at baseline. The proportional assumption was tested by including covariate by time interaction effects into the model (p > 0.05 for all) [13]. We examined trends in risk of incident Parkinson’s disease between diet quality tertile in Cox proportional hazards models by assigning each tertile’s median diet quality score to each participant based on their corresponding diet quality tertile. Possible interactions between diet quality and related covariates, including age, sex, BMI, and educational level, were tested by adding a multiplicative term in Cox proportional hazards model after adjusting for the aforementioned covariates.
Four sensitivity analyses were conducted to test the robustness of our results: 1) restricting the statistical analyses to 3,463 participants without any self-reported oral health problem to avoid potential impact of self-reported oral health problem(s) on diet quality as our previous study found poor oral health to be strongly associated with poor diet quality [14]; 2) excluding participants who were diagnosed with Parkinson’s disease within two years of follow-up (n = 11) to address potential reverse causality; 3) excluding participants who were diagnosed with Parkinson’s disease within two years of follow-up and with self-reported oral health problem(s) (n = 201); and 4) further including 31 participants who were diagnosed with incident Parkinson’s disease while not receiving Parkinson-related treatment as cases in the model to determine whether incident Parkinson’s cases were underestimated by defining cases as being diagnosed and receiving medication treatment(s).
Meta-analysis
Search strategy
We systematically searched in the PubMed, Web of Science, and Cumulative Index for Nursing and Allied Health (CINAHL) databases starting from January 1, 1981 up to June 8, 2020. Search terms related to diet quality, dietary pattern, and Parkinson’s disease in controlled vocabulary (in PubMed and CINAHL) as well as text were used. Studies assessing either diet quality or dietary pattern were included in order to provide a comprehensive picture of diet assessed by a priori and a posteriori methods in relation to Parkinson’s disease. Detailed search words for the three databases with results can be found in Supplementary Table 1.
Eligibility criteria
Study selection was completed using the inclusion and exclusion criteria listed in Supplementary Table 2. Observational studies, including prospective, retrospective, and case-control studies, written in English as a primary research article in a peer-reviewed journal were included. We aimed to include longitudinal studies, however, case-control studies were also included due to outcome studied being relatively rare. Studies that assessed overall dietary patterns or diet quality using either an a priori or a posteriori approach were eligible. Eligible studies must have outcome(s) of clinically diagnosed Parkinson’s disease and reported associations in the form of relative risks or odds ratios. Ineligible studies included those that focused on a single nutrient or selected dietary components. We did not include studies which reported only the mean difference in diet quality score between Parkinson’s disease cases and controls [15].
Study selection process, data extraction, and quality assessment
Study selection and coding process were conducted and managed using Rayyan online software by one reviewer (YL) and verified by a second reviewer (MN). Data extraction for each included study was conducted by one reviewer (YL) and verified the other reviewer (MN). Extracted data included study characteristics, cohort, characteristics of participants, covariates, dietary assessment, outcome assessment, statistical methods, and primary results. Quality of all included studies was assessed using Newcastle-Ottawa Scale stars [16]. Possible stars for quality assessment ranged from 0 to 9 stars with 7 stars or higher representing good study quality [16]. The results of study quality assessment are shown in Table 3.
Statistical analyses
Statistical analyses were carried out using Stata/SE software version 15.1 (StataCorp, College Station, TX) to pool results from all included studies. Pooled risk ratios and 95% CIs between two dietary pattern scores or diet quality categories (the highest versus the lowest) were calculated with the lowest dietary pattern score or diet quality group as reference using random effects models in generic inverse variance method. For a study that reported relative risks from both diet quality index and dietary pattern score, we extracted relative risks from diet quality index in order to be consistent with the current cohort study [17]. Two results were pooled as one risk ratio and included in the models for a study with separate results for men and women [18]. When risk ratios from diet quality index were not available, we extracted results from dietary pattern score [19]. Meta-regression was conducted to examine whether there was potential bias resulted from prospective cohort and case-control designs. Heterogeneity among studies was examined by Q statistics and I2 index with 25%, 50%, and 75% representing low, moderate, and high heterogeneity [20]. Publication bias was tested by the Egger’s test.
Registration
The search strategy and protocol of this meta-analysis have been registered on the PROSPERO (https://www.crd.york.ac.uk/PROSPERO) as CRD42020169169.
RESULTS
Cohort analysis
After a mean of 6.94 years of follow-up, 47 incident Parkinson’s cases were documented. The mean age at onset was 81.3 years for women and 80.4 years for men (p-value = 0.44). Participants with better diet quality were more likely to be women and never smoker, have higher educational level, and live with family members (Table 1). We did not observe other significant differences across diet quality tertiles in other demographic and descriptive variables (Table 1).
Demographic and characteristic information of participants at baseline in the Geisinger Rural Aging Study (GRAS) by diet quality category (n = 3,653)
1Shown as mean±standard error. 2Adjusted for sex. 3Adjusted for age and sex.
Having high diet quality was associated with lower risk of incident Parkinson’s disease during a mean of 6.94 years of follow-up (adjusted HR = 0.39 comparing two extreme tertiles; 95% CI: 0.17, 0.89; p-trend = 0.02) after adjusting for potential confounders (Table 2). The sensitivity analysis excluding participants who had self-reported oral health problem(s) generated similar results (adjusted HR = 0.39; 95% CI: 0.17, 0.90; p-trend = 0.02) (Table 2). Similar trends between diet quality and risk of Parkinson’s disease were also observed in the sensitivity analysis excluding participants who were diagnosed within two years of follow up plus with self-reported oral health problem(s) (p-trend=0.05) and the sensitivity analysis further including Parkinson’s cases without medication treatment, however, the association lost significance (p-trend = 0.16) (Table 2).
Association between diet quality and incident Parkinson’s disease in Geisinger Rural Aging Study (GRAS) over 6.94 years of follow-up by diet quality (n = 3,653)
DST, Dietary Screening Tool. 1Adjusted for age and sex at baseline. 2Adjusted for covariates listed in model 1 plus race, baseline body mass index (BMI), educational level, self- or proxy-reporting, smoking status, living status, living arrangement, and oral health status. 3Adjusted for covariates listed in model 1 plus race, baseline body mass index (BMI), educational level, self- or proxy-reporting, smoking status, living status, and living arrangement.
Higher intake frequency of fruit, whole grain cereals, cakes or pies, hot or cold breakfast cereals, and juice at breakfast was associated with lower risk of Parkinson’s disease (p-value <0.05 for all), however, we did not observe significant association between other DST components and risk of Parkinson’s disease (data not shown). No significant interactions between diet quality and potential confounders, including age, sex, BMI, and educational level, were observed (p-interaction >0.05 for all) (data not shown).
Meta-analysis
A total of 175 studies were identified based on our search strategy and 4 studies (5 study populations) met our inclusion criteria (Supplementary Figure 1). Among the 4 studies included, 2 studies utilized hypothesis-driven a priori diet assessments [18, 21], 1 study used data-driven a posteriori approaches [19], and 1 study utilized both a priori and a posteriori assessments [17]. In addition, 2 were prospective studies with follow-up period ranging from 16 to 41 years [17, 18] and 2 were case-control studies [19, 21]. Within the 4 studies identified, 3 of them showed a statistically significant inverse association between diet quality or dietary pattern and risk of Parkinson’s disease (Table 3).
Characteristics of studies included in the meta-analysis examining the associations between dietary pattern, diet quality, and Parkinson’s disease
PD, Parkinson’s disease. 1Assessed using Newcastle-Ottawa Scale star.
A significant association between having high diet quality or high dietary pattern scores and lower risk of Parkinson’s disease compared with those with low diet quality or low dietary pattern scores was observed in our meta-analysis (pooled risk ratio = 0.64; 95% CI: 0.49, 0.83) (Fig. 2). The pooled risk ratios were 0.72 (95% CI: 0.50, 1.02) for the four prospective cohorts and 0.51 (95% CI: 0.35, 0.74) for the two case-control studies (p for heterogeneity = 0.27). There is no strong evidence of publication bias (p-value = 0.87).
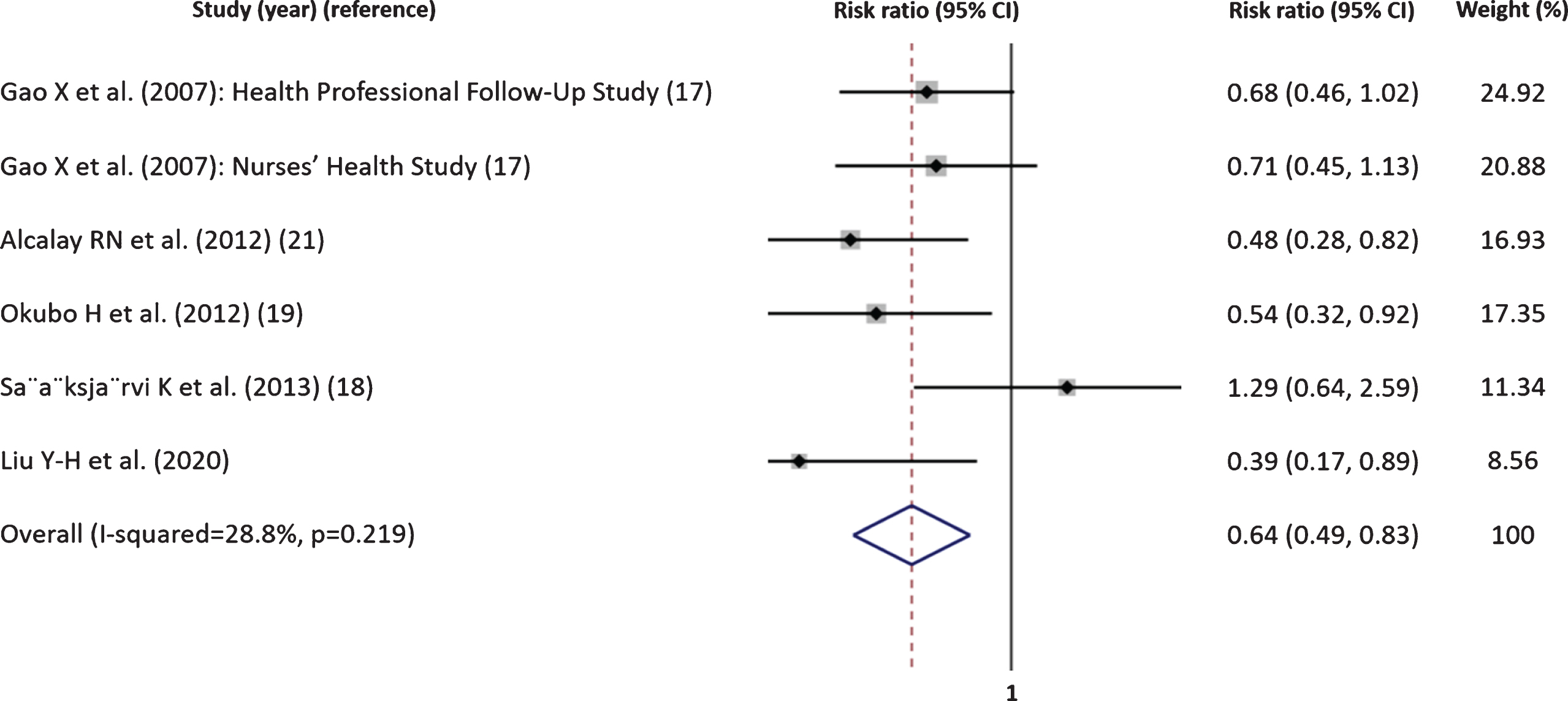
The association between diet quality 1 and risk of Parkinson’s disease (risk ratio for the highest versus the lowest diet quality or dietary pattern) (95% CI: confidence interval). 1Studies using diet quality: Gao X et al. (2007), Alcalay RN et al. (2012), S
DISCUSSION
In our prospective study conducted in over 3600 older adults living in a rural region, we observed that having high diet quality, as assessed by a validated diet quality screening tool, was associated with lower future risk of Parkinson’s disease. This association was independent of several potential confounders, including age, sex, BMI, and educational level. Similarly, our meta-analysis of 6 study populations revealed that adherence to a healthy dietary pattern or having high diet quality was associated with lower odds of Parkinson’s disease. Our results suggest that a healthy diet may be a potential modifiable lifestyle factor that may delay or prevent the onset of Parkinson’s disease. To the best of our knowledge, our study is the first meta-analysis investigating the relationship between overall diet quality and risk of Parkinson’s disease.
Parkinson’s disease is a complex and multifactorial disease which could be related to genetic, environmental, and lifestyle factors. In particular, a recent study examining the heritability of Parkinson’s disease showed the overall heritability is low (∼0.27), strengthening the potential importance of environmental and lifestyle factors in the development of Parkinson’s disease [22]. Diet has gained increased attention as dietary components and dietary patterns are modifiable lifestyle factors which could positively or negatively impact Parkinson’s disease pathogenesis [23]. Dietary factors have emerged as one of the main determining factors in the metabolic health of gut microbiota, which may in turn regulate the progression of Parkinson’s disease [23]. Some vitamins and antioxidants, for example, vitamin B6, vitamin E, flavonoids, and magnesium were shown to be inversely associated with risk of Parkinson’s disease in some [4, 24–26] but not all published studies [27, 28]. In contrast, dietary components commonly observed in the Western diet, such as high saturated fats and low dietary fiber were related to higher risk of Parkinson’s disease in a recent review [23]. Adherence to a healthy dietary pattern, characterized as high consumption of fruits and vegetables, polyunsaturated fatty acids, plant-based proteins and low intakes of red meat and saturated fats may therefore be associated with lower risk of Parkinson’s disease [23].
Known factors that are associated with altered risk of Parkinson’s disease include cigarette smoking, socioeconomic status, and sex [29]. Several studies suggested that cigarette smokers have lower risk of developing Parkinson’s disease [30, 31]. In addition, some studies suggested that people with low socioeconomic status or low work complexity had lower risk of Parkinson’s disease compared with individuals with high socioeconomic status or high work complexity [32, 33]. Studies also suggested that sex is one of the key factors in Parkinson’s disease as women have lower risk of disease development compared with men [34, 35]. In our prospective study, we also observed that being a smoker, having lower educational level, or being female may be associated with lower risk of developing Parkinson’s disease in our multivariate model (data not shown). However, the observed inverse association between overall diet quality and low Parkinson’s disease risk was not significantly modified by these factors.
To date, the pathogenesis of developing Parkinson’s disease and associated metabolic changes before disease onset remain unclear. However, some studies suggested that non-motor symptoms (i.e., prodromal Parkinson’s symptoms), such as anxiety or constipation, could have more than 20 years of preclinical phase while neuropathology might have 5 to 6 years of preclinical period [36]. Reverse causality could thus be another potential interpretation for our findings. Because of long pre-clinical stage prior to being diagnosed, individuals with underlying prodromal Parkinson’s symptoms may have already changed their dietary habits. Although our 2-year lag analysis generated a similar association between high diet quality and lower risk of Parkinson’s disease, we still cannot totally exclude the possibility of reverse causality. Alternately, these results could suggest that healthy diet quality might be associated with lower risk of conversion from prodromal to clinical Parkinson’s disease.
There are some additional limitations that need to be carefully considered when interpreting our study results. First, in our prospective study, we were unable to estimate participants’ energy consumption because of the design and limited food items included in the DST. Although the DST contains 25 individual food- and behavior-specific questions, it was designed for evaluating overall diet quality which should thus be interpreted as a whole instead of individually by single DST component. Moreover, only 3.4% of participants reported a past- or current smoking habit. Smoking may well be under-reported and so result in potential residual confounding. Interestingly, smoking behavior has also been associated with poor diet quality [37]. We may thus underestimate the true diet-Parkinson relationship. Generalizability of findings from our cohort needs to be further studied due to limited diversity in the GRAS cohort with most of participants being non-Hispanic white. However, similar association between diet quality and risk of Parkinson’s disease was observed in the meta-analysis, in which studies with different study designs and populations were included. The diagnosis of Parkinson’s disease in our study was based on electronic medical record review. Diagnosis was not confirmed by a neurologist so we cannot rule out the possibility that participants diagnosed with Parkinson’s disease may have had other neurological diseases with similar symptoms. We tried to minimize this potential misclassification, accepting as incident Parkinson’s disease cases only those who had both ICD-10-CM code G20 and the use of relevant treatments. This was supported by the observations that the association between diet quality and risk of Parkinson’s disease was attenuated after we further included participants with incident Parkinson’s disease diagnosis, but were not receiving Parkinson-related medication as cases in the model. Although we conducted a very comprehensive systematic literature search for our meta-analysis, only 6 cohorts were selected for inclusion. The small number of studies included have limited our ability to further perform subgroup analyses to examine the potential impact of heterogeneity across cohorts. Various dietary assessments (i.e. a priori and a posteriori) were utilized in the selected individual studies which may in turn impact interpretation of results in our meta-analysis [38]. However, similar characteristics, for example, increased consumption of fruits and vegetables and decreased consumption of red meat and saturated fats, are likely shared in most of the identified healthy dietary patterns or high-quality diets.
In summary, results from our prospective study and meta-analysis suggest that having high diet quality or adherence to a healthy dietary pattern is associated with lower risk of Parkinson’s disease. More observational studies with larger sample size and longer follow-up are needed to better understand the temporal relationship between dietary pattern and the development of Parkinson’s disease.
CONFLICT OF INTEREST
All authors declare no conflict of interest.
*A copy of the Dietary Screening Tool (DST) is available upon request.
Footnotes
ACKNOWLEDGMENTS
The authors gratefully acknowledge the coordinating, recruiting, and screening staff from the Geisinger Obesity Research Institute.
This study is funded by the United States Department of Agriculture, Agricultural Research Service agreement 8050-51530-012-01A and National Institute of Health 1R01NS102735-01A1.
