Abstract
Background:
There is a lack of recommendations for selecting the most appropriate gait measures of Parkinson’s disease (PD)-specific dual-task costs to use in clinical practice and research.
Objective:
We aimed to identify measures of dual-task costs of gait and turning that best discriminate performance in people with PD from healthy individuals. We also investigated the relationship between the most discriminative measures of dual-task costs of gait and turning with disease severity and disease duration.
Methods:
People with mild-to-moderate PD (n = 144) and age-matched healthy individuals (n = 79) wore 8 inertial sensors while walking under single and dual-task (reciting every other letter of the alphabet) conditions. Outcome measures included 26 objective measures within four gait domains (upper/lower body, turning and variability). The area under the curve (AUC) from the receiver-operator characteristic plot was calculated to compare discriminative ability of dual-task costs on gait across outcome measures.
Results:
PD-specific, dual-task interference was identified for arm range of motion, foot strike angle, turn velocity and turn duration. Arm range of motion (AUC = 0.73) and foot strike angle (AUC = 0.68) had the largest AUCs across dual-task costs measures and they were associated with disease severity and/or disease duration. In contrast, the most commonly used dual-task gait measure, gait speed, showed an AUC of only 0.54.
Conclusion:
Findings suggest that people with PD rely more than healthy individuals on executive-attentional resources to control arm swing, foot strike, and turning, but not gait speed. The dual-task costs of arm range of motion best discriminated people with PD from healthy individuals.
INTRODUCTION
The observation that nursing home residents who stopped walking when talking had a higher risk of falls [1] motivated the exploration of dual-task protocols to investigate cognitive-motor interactions and fall risk in older adults and clinical populations [2, 3], such as people with Parkinson’s disease (PD). Difficulty walking while performing another task has been reported to be worse in people with PD than healthy older adults [4–6]. This might be explained, at least in part, by the increased prefrontal executive-attentional control of walking (due to reduced automaticity) [7] and cognitive impairments [8–11] that occur in PD. Walking in people with PD is slow; with reduced arm swing; short, variable and shuffling (lack of heel strike) steps [12, 13]. Turning amplitude and velocity is also reduced with excessive steps, even early in the disease [12, 13]. Cognitive impairments in PD are broad and include reduced executive functions and attention and slow processing speed that may affect dual-task performance [8–10].
Dual-tasking protocols simulate challenging situations of daily life, such as walking while talking or manipulating objects. The dual-task condition often yields a decrement in performance of one or both tasks compared to the single-task condition [4–6]. The magnitude of the decrement, called “dual-task costs”, arises from the two tasks interfering with each other and competing for the same brain resources [14]. It is often reported that people with PD present greater dual-task costs while walking than healthy individuals [4, 15–19], and a number of potential mechanisms have been proposed to explain this finding [6]. Notably, greater dual-task costs have been shown to predict the risk of falling [20] and to be associated with more severe symptoms [11, 21], freezing of gait [22, 23] and reduced executive functions [11, 24] in PD. Thus, measures of dual-task costs to walking provide meaningful information for clinical assessment in PD. However, there is a lack of standardization in carrying out the dual-task and a lack of recommendations for selecting the most appropriate measures of PD-specific, dual-task costs while walking to use in clinical practice and research.
The majority of studies have been focused on reporting the dual-task on gait speed [5, 25–27]. However, functional mobility includes other relevant and independent domains, such as the control of the trunk, upper limbs and turning while walking [28]. These domains of functional mobility are also impaired in PD [28–30] and may rely more on attention than the pace of gait. In fact, gait speed may not be sensitive enough to identify PD-specific, dual-task impairments. For example, previous studies have demonstrated similar reductions in gait speed, as well as stride length, from single to dual-task walking in people with PD and healthy individuals [15, 27]. Therefore, there is an unmet need to expand the scope of mobility measures assessed to include trunk and upper limb measures of walking as well as turning. In addition, there is an unmet need to determine which specific objective measures of dual-task costs of gait and turning best discriminate performance in people with PD from healthy individuals. Sensitive and specific measures of dual-task costs on gait could serve as potential biomarkers for future clinical trials investigating the effects of interventions on cognitive-gait interference in people with PD.
In the current study, we used body-worn inertial sensors to record a broad set of objective measures of walking and turning [28] in both single and dual-task conditions in people with PD and healthy control individuals. The primary aim of this study was to identify measures of dual-task costs of gait and turning that best discriminate dual-task performance of people with PD from healthy individuals. Since gait variability, turning and the control of upper limbs (e.g., arm swing) are markedly affected by PD and associated with executive control of walking [17, 31–36], we hypothesized that measures of dual-task costs in these domains would better discriminate people with PD from healthy individuals than does dual-task costs of gait speed. We also investigated the relationship between the most discriminative measures of dual-task costs with disease severity and disease duration. We expected that greater dual-task costs would associate with more severe motor symptoms and longer disease duration in people with PD.
MATERIALS and METHODS
Participants
One hundred and forty-four people with idiopathic PD and 79 age-matched healthy control subjects participated in this study. For both groups, participants were included if they were aged 50–90 years. Specifically for PD, inclusion criteria were: 1) diagnosis of idiopathic PD from movement disorders neurologist with the United Kingdom Parkinson’s disease Society Brain Bank criteria [37]; 2) no major musculoskeletal or peripheral disorders that could significantly affect their balance and gait; 3) ability to stand and walk unassisted; 4) no recent changes in medication (six weeks of stable medications). Exclusion criteria for all subjects included any other neurological disorders or musculoskeletal impairments that interfere with gait or balance, and inability to follow instructions. The experimental protocol was approved by the Oregon Health and Science University Institutional Review Board (#4131) and the joint OHSU and Veterans Affairs Portland Health Care System Institutional Review Board (#8979). All the participants provided written informed consent prior to participation.
Experimental procedures, equipment, and outcomes
Subjects with PD were tested in the practical Off levodopa state, after at least 12 h of anti-parkinsonian medication wash-out. Participant characteristics of age, sex, height and weight were recorded. The following functional and cognitive tests were administered in both groups: Mini Balance Evaluation System Test (Mini-BEST) [38]; the Activities-specific Balance Confidence scale (ABC-scale) [39] and; the Montreal Cognitive Assessment (MoCA) [40]. Specifically for subjects with PD, disease severity was measured using the motor section of the Movement Disorders Society (MDS-revised) Unified Parkinson’s disease Rating Scale (MDS-UPDRS part III) [41]; and PD stage was assessed by the Hoehn and Yahr Rating Scale [42].
At the beginning of the mobility assessment, participants performed a seated cognitive task of reciting every other letter of the alphabet for 1 min. The performance on the cognitive task was quantified by the accuracy in the given responses (i.e., percentage of correct responses out of the total responses). Then, participants were instrumented with eight inertial sensors (Opals, APDM, Portland, OR, USA) that included triaxial accelerometers, gyroscopes and magnetometers recording at 128 Hz. They were fixed, with Velcro straps, at the sternum, lumbar spine, bilaterally on the wrists, shanks and feet. Participants were instructed to walk at a comfortable pace back and forth continuously between two lines 7.62 m apart, with a 180° turn at each end. They received no specific instruction about the side to turn. Two conditions were tested: single-task and dual-task walking. In the single-task condition, participants were instructed to walk for 2 min with no additional task. In the dual-task condition, participants were instructed to walk for 1 min while reciting every other letter of the alphabet; no information about task priority was assigned to participants. The single-task condition was always completed before the dual-task condition.
A total of 26 objective measures (including mean and standard deviation) within four mobility domains (upper body, lower body, turning and variability [28, 43]) were extracted using Mobility Lab software (Mobility Lab v2, APDM, Portland, OR, USA) [44–46]. Briefly, the core of the algorithm used by Mobility Lab is the automatic identification of gait events, such as heel-strike and toe-off, and turns [45]. A candidate turn is defined as the trunk rotation around the vertical plane with a minimum of 40 °/s, and a start/end of each turn is defined with a threshold of 15 °/s [47]. Only turns with durations between 0.5 and 10 s, and turn angles of 40° or more are considered [47]. Before the actual calculation of gait measures, the algorithm excludes the very first steps during the gait initiation acceleration phase and also steps prior-to and right after each turn so that the gait measures are representative of straight ahead gait. The choice of the objective measures was based on prior knowledge about test-retest reliability, validity, and discriminative ability in separating people with PD from healthy controls [43–48]. The complete list of measures is presented in Fig. 1 (see Supplementary Table 1 and [43] for the definitions of objective measures). Definitions and illustrations of the objective measures can be found at APDM whitepaper (https://www.apdm.com/publications/).

Area under the curve (in descending order) for each gait measure discriminating people with PD from healthy individuals. The color-coding scheme is based on domains of mobility as shown in the legend. ROM, range of motion; SD, standard deviation.
To quantify the interference between the two tasks when performed simultaneously, we calculated the dual-task costs for both mobility and cognitive tasks using the following equation: Dual-task costs [% ] = 100 * (dual-task metric –single-task metric) / single-task metric. In order to make the interpretation of dual-task costs consistent across all the objective measures, some measures of dual-task costs were multiplied by -1 so that positive values correspond to dual-task costs (and negative values to eventual improvements with the dual-task).
Statistical analysis
The normality of data was determined with Shapiro-Wilk tests and parametric analysis was used, unless otherwise stated. Independent t-tests or Mann-Whitney U tests (if not normally distributed) were used to compare demographic, clinical and cognitive measures between groups. To investigate which specific measures best discriminate walking and turning characteristics in people with PD from healthy individuals for single-task, dual-task and dual-task costs, we performed Receiver Operating Characteristic (ROC) curves [49] analysis and computed the area under the curve (AUC) [50], and then ordered them based on the highest to lowest AUC value. To investigate dual-task interference on the top six measures discriminating people with PD from healthy individuals in the single-task condition, we used a two-way (2 groups×2 conditions) repeated measures analysis of variance (ANOVA). Pearson’s correlation was used to assess the relationship between mobility measures that showed significant group×condition interaction and severity of PD (MDS-UPDRS part III and disease duration). In addition, to check if the inclusion of participants with cognitive impairment could somehow influence findings, we performed ROC curves analysis considering only those with intact global cognition (MoCA ≥26). To investigate which measure of dual-task costs best discriminate early and late PD from healthy individuals, we performed additional subanalysis splitting the PD group into two subgroups according to disease duration (early PD:≤5 years; late PD: > 5 years). AUCs were calculated separately for early and late PD for measures of dual-task costs. All statistical analysis was performed using R Version 1.1.456 software. The statistical significance was set to p < 0.05.
RESULTS
Group characteristics
Group characteristics are summarized in Table 1. There were no differences between groups for age, height, weight or global cognition (MoCA). As expected, people with PD had poorer dynamic balance (Mini-BEST) and worse self-reported balance confidence (ABC-scale) than healthy individuals. People with PD had mild-to-moderate disease severity (MDS-UPDRS part III: 40.5±12.5 (mean±SD); disease duration: 6.2±4.9 years; Levodopa equivalent daily dosage: 617.5±408.6 mg/day; Hoehn and Yahr stage: I (n = 1), II (115), III (15), and IV (13)).
Demographic data
Mini-BEST, Mini Balance Evaluation System Test; ABC, Activities-specific Balance Confidence; MoCA, Montreal Cognitive Assessment; aMann-Whitney U test; bChi-squared test.
Arm range of motion and foot-strike angle were the most discriminative measures of dual-task costs between people with PD and healthy controls
Single and dual-task conditions
Similar gait characteristics discriminated walking performance in people with PD from healthy individuals for both single and dual-task conditions. The gait measures with the largest AUC during single-task/dual-task walking included: turn velocity (AUC = 0.89/0.90; specificity = 0.82/0.85; sensitivity = 0.85/0.82), turn duration (AUC = 0.87/0.89; specificity = 0.76/0.90; sensitivity = 0.84/0.74), arm range of motion (AUC = 0.84/0.87; specificity = 0.81/0.87; sensitivity = 0.79/0.74), foot strike angle (AUC = 0.84/0.85; specificity = 0.81/0.72; sensitivity = 0.78/0.84), stride length (AUC = 0.83/0.83; specificity = 0.80/0.66; sensitivity = 0.73/0.85) and gait speed (AUC = 0.80/0.74; specificity = 0.80/0.86; sensitivity = 0.67/0.50) (see Fig. 1). With the exception of gait speed, which did not discriminate between PD and healthy individuals as well during the dual-task as during the single-task, the same measures best discriminated people with PD from healthy individuals in the dual-task condition (Fig. 1).
Dual-task costs
The measures of dual-task costs on gait showed smaller AUCs to discriminate PD from healthy individuals compared to the gait measures themselves in single and dual-task conditions (Fig. 1). Specifically, the following measures of dual-task costs showed the largest AUCs: arm range of motion (AUC = 0.73; specificity = 0.81; sensitivity = 0.60), foot strike angle (AUC = 0.68; specificity = 0.94; sensitivity = 0.41), trunk coronal range of motion (AUC = 0.67; specificity = 0.70; sensitivity = 0.60), turn velocity (AUC = 0.66; specificity = 0.73; sensitivity = 0.56) and trunk transverse range of motion (AUC = 0.65; specificity = 0.71; sensitivity = 0.57). These dual-task costs measures had larger AUC than measures of dual-task costs on gait speed (AUC = 0.54; rank = 20), stride length (AUC = 0.62; rank = 7) or accuracy in the cognitive task (AUC = 0.59; rank = 9).
Dual-task costs (MoCA≥26)
Findings were consistent even when only a subset of participants with preserved global cognition (MoCA ≥26) was considered in the analysis (i.e., 87 people with PD and 54 healthy individuals). Top six measures were the same identified in the full sample, with similar AUCs as well. Example: Dual-task costs of arm range of motion was ranked #1 in both analysis (AUCMoCA > 26 = 0.716) and the only measure of dual-task costs with AUC > 0.7.
Descriptive statistics (means and standard deviations) for single-task, dual-task and dual-task costs are reported in Supplementary Table 2.
People with PD showed specific dual-task impairments on arm swing, foot strike angle and turning compared to healthy controls
Only two measures, arm range of motion and foot strike angle, showed a significant group×condition interaction, indicating that the dual-task affected these measures more in PD than healthy subjects (Table 2). Post-hoc comparisons showed that although both groups reduced arm range of motion and foot strike angle from single to dual-task condition, the reduction was greater in people with PD (Fig. 2). Of note, arm range of motion and foot strike angle were ranked #1 and #2, respectively, among measures of dual-task costs in discriminating people with PD from healthy individuals (Fig. 1).
Results of repeated measures ANOVA for the top six measures in discriminating walking and turning characteristics in people with PD from healthy individuals during single-task walking
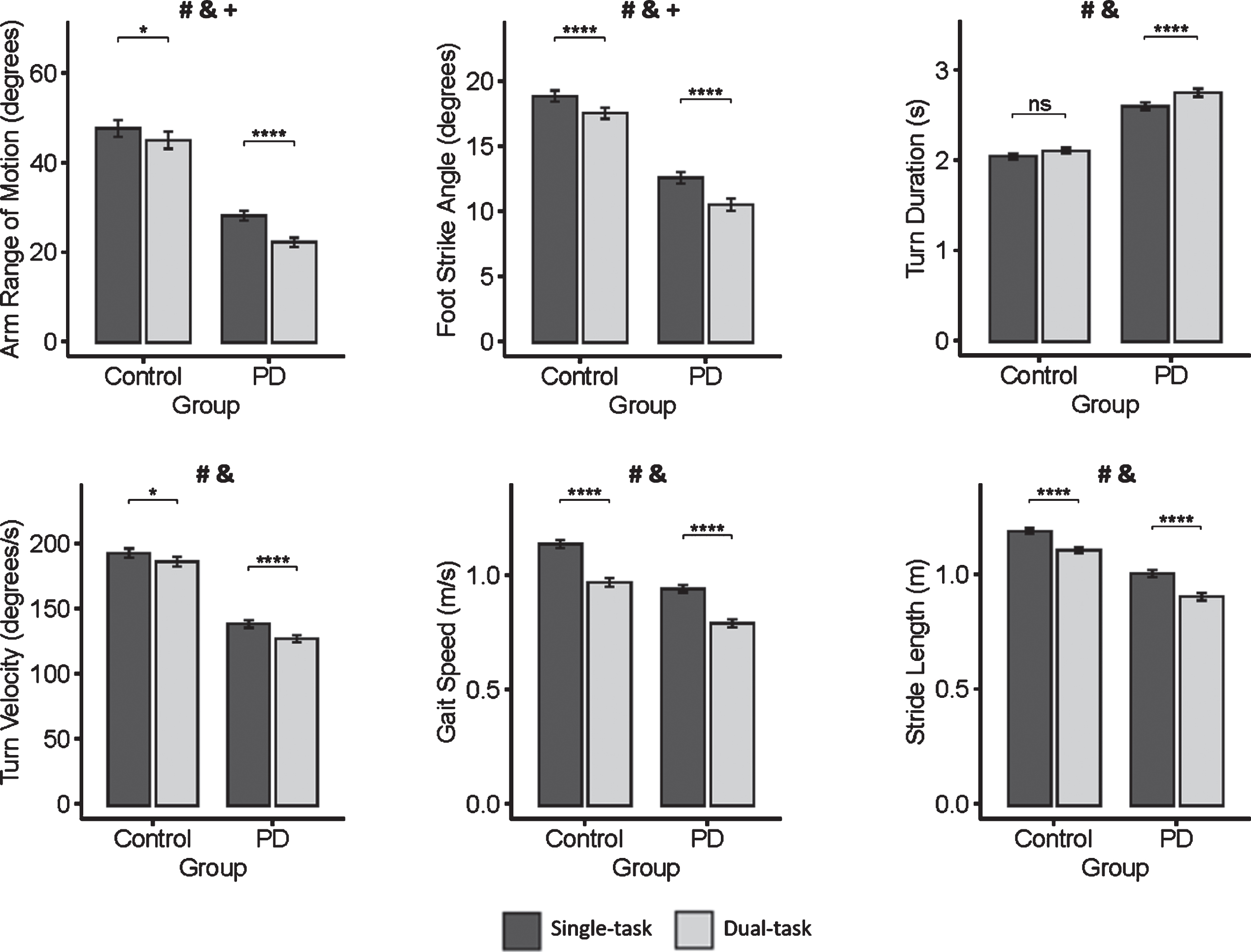
Means and standard error of the mean of gait measures for people with Parkinson’s disease (PD) and healthy control individuals during single-task (black) and dual-task (gray) walking. # indicates a significant group effect (p < 0.05); & indicates a significant condition effect (p < 0.05); and+indicates a significant interaction effect (p < 0.05); asterisks indicate significant differences in post hoc paired comparisons (*p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001).
Turn velocity and turn duration may also capture PD-specific dual-task interference. Group×condition interaction approached statistical significance for turn velocity (p = 0.06) and turn duration (p = 0.07) (Table 2) and, therefore, we checked the corresponding pairwise comparisons. They showed that people with PD increased turn duration from single to dual-task condition while healthy individuals did not change turn duration across conditions (Fig. 2). In addition, although both groups reduced turn velocity from single to dual-task condition, the reduction was greater in people with PD (Fig. 2).
Stride length and gait speed had a general (non-PD-specific) dual-task interference effect, as revealed by significant condition main effect combined with a non-significant group×condition interaction (Table 2). The two groups similarly decreased stride length and gait speed from single to dual-task condition (Fig. 2).
Dual-task costs of arm range of motion and foot strike angle are associated with PD severity and duration
Greater dual-task costs of arm range of motion while walking were associated with longer disease duration (r = 0.21, p = 0.012) and more severe motor signs (r = 0.18; p = 0.03) (Fig. 3). Similarly, greater dual-task costs of foot strike angle were associated with longer disease duration (r = 0.24; p = 0.004) (Fig. 3). Further, the results were similar even when partial correlations controlled for age and global cognition (MoCA; see Supplementary Table 3). The dual-task costs of gait speed were not significantly associated with disease severity (r = 0.1; p = 0.19) or disease duration (r = 0.04; p = 0.58).
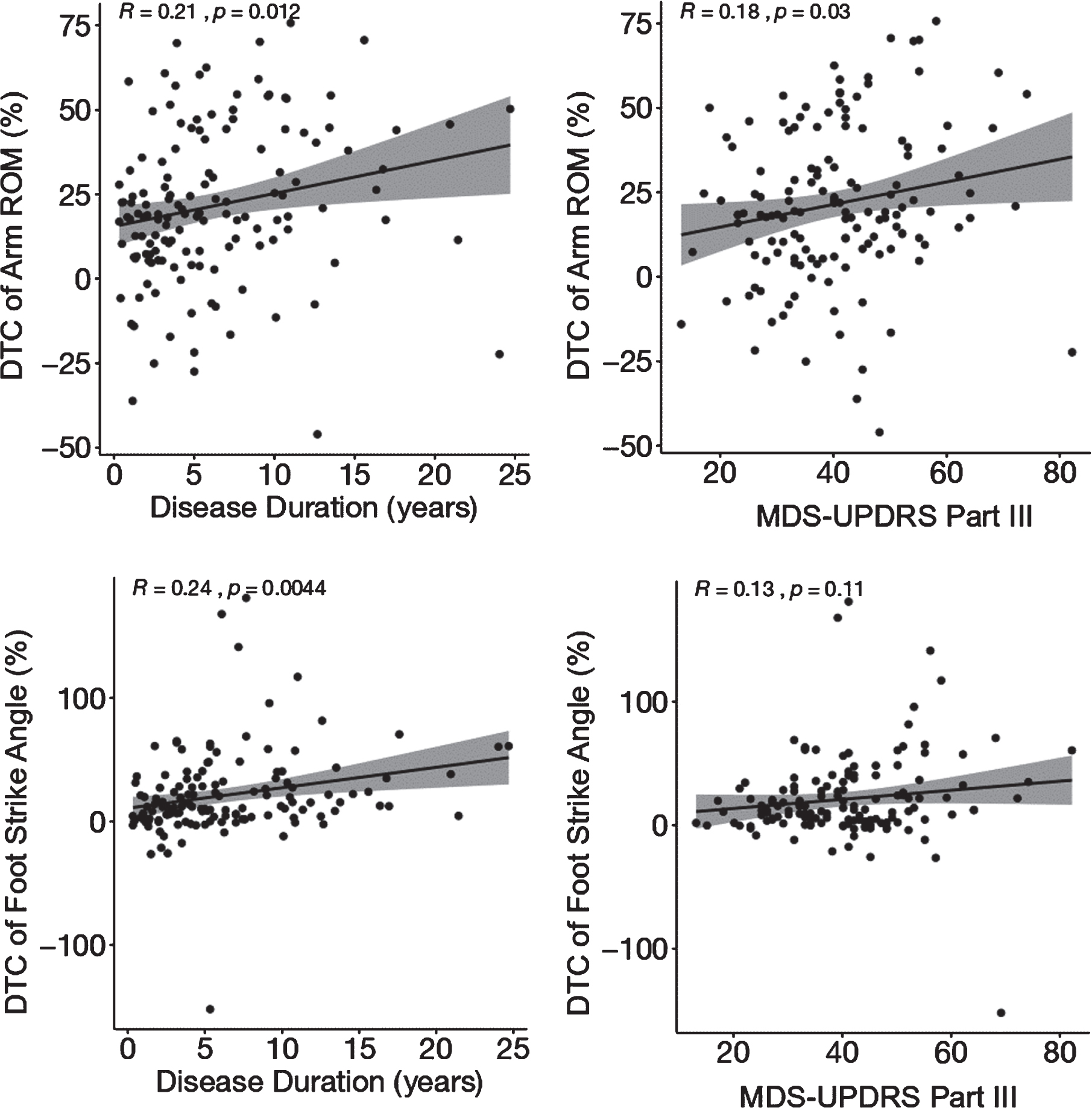
Pearson’s correlation between top two measures of dual-task costs to gait and disease severity and disease duration. ROM, range of motion; MDS-UPDRS, Movement Disorders Society - Unified Parkinson’s disease Rating Scale.
Dual-task costs of arm range of motion and foot strike angle better discriminate people with PD from healthy controls later in the course of PD
Correlations between dual-task costs and disease duration, above, are consistent with a subanalysis in which AUCs were calculated separately for early versus late PD, i.e., individuals with PD were split into two subgroups according to disease duration (early PD:≤5 years, n = 73; late PD: > 5 years, n = 71). The measures of dual-task costs of arm range of motion and foot strike angle showed larger AUCs to discriminate PD from healthy individuals later in the course of PD compared to early PD: Dual-task costs of arm range of motion (AUCearlyPD = 0.67 / AUClatePD = 0.79) and dual-task costs of foot strike angle (AUCearlyPD = 0.61 AUClatePD= 0.75).
DISCUSSION
To the best of our knowledge, this is the first study comparing a broad set of objective measures of gait and turning to investigate which measures of dual-task costs best discriminate performance of people with PD from healthy individuals. As expected, measures of dual-task costs on traditional gait parameters, such as gait speed and stride length, were not the best measures to differentiate people with PD from healthy individuals. PD-specific, dual-task interference was identified for arm range of motion, foot strike angle, turn velocity and turn duration. However, all measures of dual-task costs on gait had lower discriminative ability than gait measures themselves in the single and dual-task conditions. The dual-task costs of arm range of motion had the largest AUC among measures of dual-task costs. Furthermore, measures of dual-task costs to reducing arm range of motion and reducing foot strike angle were associated with disease duration and/or severity. Overall, our results suggest that dual-task costs of arm range of motion best discriminated dual-task performance of people with PD from healthy individuals (while walking and reciting every other letter of the alphabet).
Gait speed and stride length were among the top measures in discriminating people with PD from healthy individuals during single-task walking. These findings suggest that gait speed and stride length are able to capture the bradykinetic characteristic of parkinsonian gait [12]. However, the dual-task costs on gait speed and stride length are similar between people with PD and healthy controls. In fact, gait slowed and stride length shortened with a dual task in both the PD and the control groups, as reported previously [15, 27]. Not surprisingly, the discriminative ability of the corresponding dual-task costs measures was drastically reduced compared to the gait measures themselves in the single-task condition. These findings suggest that gait speed and stride length are unable to capture PD-specific dual-task impairments. Instead, dual-task related changes in gait speed and stride length may represent a general conservative/protective strategy [18] and/or age-related increase in attention needed for the pace of gait [25].
PD-specific dual-task impairments were observed for arm range of motion, foot strike angle, turn velocity and turn duration, indicating that these measures may be good markers of reduced automaticity in PD. Thus, our findings suggest that people with PD may rely on additional executive-attentional resources to control foot strike, arm swing and turning, consistent with previous studies [15, 51]. A recent paper from our laboratory showed that PD-specific dual-task costs occur for local dynamic stability immediately before and during foot strike (i.e., phase corresponding to weight transfer): people with PD, but not healthy controls, became less dynamically stable in the dual-task condition [15]. Baron and colleagues recently showed that several different measures of arm swing (e.g., arm swing path length and jerkiness) are affected by dual-tasking in PD [36]. The increased executive load imposed by the dual-task may have led people with PD to decrease/freeze some degrees of freedom, as manifested by reduced arm range of motion and foot strike angle in the dual-task condition. Further, evidence for increased executive control of turning come from functional infrared spectroscopy studies showing increased prefrontal cortical activity (i.e., executive-attentional processing) during turning in people with PD compared to healthy individuals [33, 51].
Compared to gait measures themselves, measures of dual-task costs while walking seem to have limited ability to discriminate the performance in people with PD from healthy individuals. Notably, measures of dual-task costs showed lower discriminative ability compared to gait measures in either the single- or dual-task conditions. This can be explained by the large variability and overlapping values in measures of dual-task costs between groups, which is driven by the fact that some individuals in both groups had their gait affected by the dual-task whereas others did not. For measures of dual-task costs (considering the full sample), only dual-task costs of arm range of motion achieved an acceptable (0.7 < AUC<0.8 [52]) performance in discriminating people with PD from healthy individuals. Other measures of dual-task costs achieved, at most, a fair discrimination level. It is possible that reduced arm swing in people with PD, which is observed early in the disease [12] and exacerbated under dual-task conditions, is one of the best markers of reduced automaticity and increased executive control.
Dual-task costs of arm range of motion, as well as dual-task costs of foot strike angle (but not gait speed), better discriminated between healthy controls and people with PD performance later in the course of PD (duration > 5 years). This could be explained by gait control becoming less automatic as the disease progresses [35], so arm swing and foot placement are more affected by distraction. In addition, cognitive impairments progress with duration of PD [53], leaving patients with less executive-attentional resources to compensate for less automatic gait. Dual-task costs of arm range of motion and of foot strike angle were also associated with disease severity and disease duration, consistent with worse performance later in the course of PD.
Surprisingly, measures of gait variability were not top ranked among measures of dual-task costs in discriminating people with PD from healthy individuals. This finding suggests that our measures of gait variability may be unable to capture PD-specific dual-task impairments, which differs from previous studies [18, 54]. Although the gait response to a cognitive dual-task is task-specific [19, 55], measures of gait variability were not very discriminative for PD, even for single-task walking. Thus, discrepancy may be due to differences in methodology to collect gait variability. Another explanation is that measures of gait variability recorded in the laboratory may not be as sensitive in discriminating people with PD and healthy individuals as measures of turning or control of the upper body while walking [43].
Potential pathophysiological concepts underlying PD-specific dual-task interference
The greater dual-task interference observed in people with PD (compared to healthy individuals) is commonly interpreted on the basis of PD-related reduced movement automaticity and cognitive impairments. In PD, reduced movement automaticity is likely due to impairment of basal ganglia function [14, 35]; and cognitive impairments have a broad spectrum, including deficits in cholinergic and norepinephrine pathways associated with cortical function [8–11]. To compensate for reduced automaticity, people with PD may rely on prefrontal executive-attentional resources for the control of single walking [7]. However, since the pool of executive-attentional resources is reduced in PD, less resources are available to deal with the increased executive-attentional demand imposed by the concurrent cognitive task. In fact, it has been recently demonstrated that appropriate levels of executive function are required for the efficiency of the prefrontal compensatory mechanism in controlling gait [56]. As a consequence, people with PD are more vulnerable to dual-task interference compared to healthy individuals.
Since the most discriminative measures of dual-task costs included arm range of motion and foot strike angle, we suggest that the increased executive load imposed by the dual-task may have led people with PD to decrease/freeze some degrees of freedom, which is a common strategy to facilitate the control of multi-joint movements [57, 58]. Indeed, conscious control of movement has been shown to be associated with freezing of degrees of freedom [58]. It has been also proposed that arm movements during walking are controlled by rhythmic activation of cervical central pattern generator networks, which are inhibited/suppressed when goal-directed upper limb control is desired [59]. The increased executive load under dual-task condition may thus have led to suppression of cervical central pattern generator networks.
Key study strength, limitations and clinical implications
A key strength of this study is the large sample size. We also compared a wide variety of walking and turning measures for dual-task costs. On the other hand, the following limitations have to be considered while interpreting the current results. Most of people with PD enrolled in this study were classified in Hoehn and Yahr stage II (∼80% of those with PD) and only one secondary cognitive task was tested. In addition, participants with PD were assessed Off medication. Hence, current results cannot be generalized to mild or more advanced PD stages or other secondary tasks. Since dopaminergic medications increase speed and prefrontal cortical activity and decrease variability during dual-task walking in PD [27, 54], our findings cannot be generalized to On medication state. Future studies implementing similar analysis in other secondary tasks and a more diverse sample of people with PD are encouraged to test if these findings can be generalized. Despite the existing limitations, our findings suggest that clinical trials investigating the effects of interventions on PD-specific dual-task interference, which is relevant for safe and independent mobility during daily activities, should assess the dual-task costs of arm range of motion (and perhaps foot strike angle), not on gait speed. This can be easily and quickly done with wearable inertial sensors. Further, our findings highlight aspects that should be targeted in the development of rehabilitation interventions to treat PD-specific dual-task interference. For example, interventions involving external cues, cognitive/attentional strategies, and dual-task gait training should focus on arm range of motion and foot strike angle.
CONCLUSION
Our findings suggest that people with PD may rely on additional executive-attentional resources to control foot strike, arm swing and turning, even more than speed of gait. Traditional spatiotemporal gait parameters related to footfalls during straight walking, such as stride length and gait speed, showed less discriminative ability to identify PD-specific dual-task impairments. Finally, the dual-task costs of arm range of motion best discriminated dual-task performance of people with PD from healthy individuals.
CONFLICT OF INTEREST
Dr. Horak has a significant financial interest in APDM, a company that may have a commercial interest in the results of this research and technology. Dr. Horak also consultants with Biogen, Neuropore, Sanofi, Pfizer and Takeda. This potential individual conflict has been reviewed and managed by OHSU.
Footnotes
ACKNOWLEDGMENTS
The authors thank all participants for generously donating their time to participate, Peter Fino, Carolin Curtze, Mike Fleming, Heather Schlueter, Peter Martin and Graham Harker for helping with data collection, Daniel Peterson and Katrijn Smulders for data collection and help with study procedures, and Edward King for helping with data collection and management. This research was funded by the National Institutes of Health under award number R01AG006457 (PI: Horak), and Department of Veterans Affairs Merit Award number 5I01RX001075 (PI: Horak).
