Abstract
Background/Objective:
To compare the longitudinal trajectories of cognition according to the presence of the apolipoprotein E (APOE) ɛ4 allele in male and female Parkinson’s disease (PD) patients.
Methods:
This study included a total of 361 patients with recently diagnosed de novo PD (mean age [standard deviation], 61.4 [9.8] years). The patients were classified into the following groups: APOE ɛ4 + /M (n = 65), APOE ɛ4-/M (n = 173), APOE ɛ4 + /F (n = 25), and APOE ɛ4-/F (n = 98). Cognitive decline was assessed annually over 5 years of follow-up using the Montreal Cognitive Assessment (MoCA). To assess the sex-specific impacts of the APOE ɛ4 status on cognitive decline, we used generalized linear mixed effects (GLME) models separately for men, women, and the two sexes combined.
Results:
In the sex-stratified GLME models adjusted for covariates, the interaction results showed that the males with APOE ɛ4 had a steeper rate of cognitive decline than those without APOE ɛ4. In contrast, there was no significant interaction between APOE ɛ4 and time on longitudinal MoCA performance in the females. The main effect of APOE ɛ4 on the change in the MoCA score was not significant for either men or women. When the data from both men and women were used, the APOE ɛ4 + /M group exhibited a steeper rate of cognitive decline than did the APOE ɛ4 + /F and APOE ɛ4-/F groups. These results were consistent with those of sensitivity analyses.
Conclusion:
Sex may be considered when APOE ɛ4-related vulnerability to early cognitive decline is evaluated in PD patients.
INTRODUCTION
Cognitive impairment is one of the major nonmotor complications that occurs in Parkinson’s disease (PD) patients [1]. The risk of this complication among PD patients increases as the disease progresses [2]. Longitudinal studies have shown that mild cognitive deficits affect approximately 20% of patients with early PD [3], and dementia occurs in up to 80% of PD patients over the long term [4, 5]. Cognitive function in PD patients is regarded as a key determinant of their quality of life, functional independence, and health care costs [6, 7]. Importantly, accumulating evidence shows that even mild cognitive deficits in PD patients may negatively impact functional status and quality of life [8, 9]. These findings indicate that interventions for cognitive decline in PD patients should be considered at an early stage in the disease process.
There are many types of drugs that are used to improve motor aspects of PD, but generally, they do not significantly improve cognition. Moreover, although rivastigmine, a cholinesterase inhibitor, can be effective for the treatment of dementia in PD patients, its efficacy in improving cognitive impairment in PD patients without dementia remains unclear [10]. These facts emphasize the need for clinical trials designed to slow or stop the progression of cognitive decline in the early stage of the disease. In this context, the ability to identify PD patients who are at risk of early cognitive decline is crucial so that patients can be stratified in future clinical trials.
Apolipoprotein E (APOE) is a major cholesterol transporter apolipoprotein in the central nervous system that supports neuronal development, plasticity and repair [11]. The APOE gene has three major alleles that are defined by the two single nucleotide polymorphisms (SNPs) at amino acid residues 112 and 158: ɛ2 (Cys112/Cys158), ɛ3 (Cys112/Arg158), and ɛ4 (Arg112/Arg158) [12]. Among these alleles, APOE ɛ4 is well known to increase the risk of Alzheimer’s disease (AD) [11, 12]. However, growing evidence has shown that the APOE ɛ4 allele also has a potential role in cognitive impairment in PD. A meta-analysis including 17 studies found that the dementia risk in PD patients with APOE ɛ4 was 1.72 times higher than that in those without APOE ɛ4 [13]. Similar results have been reported in a recent genome-wide association study using data from 12 longitudinal cohorts where the APOE ɛ4 allele was related to lower cognitive performance in PD patients [14]. A previous study using Parkinson’s Progression Markers Initiative (PPMI) data showed that APOE ɛ4 status was associated with cognitive decline in early PD over 2 years of follow-up [15].
Emerging data indicate that sex modifies the APOE ɛ4 effect on the development and progression of AD [16–24]. Accordingly, we hypothesize that sex also plays a critical role in modulating the association between APOE ɛ4 and cognition in PD patients. However, no studies have been conducted to assess the sex-dependent effects of APOE ɛ4 on cognitive function in PD patients. The aim of this study was therefore to investigate longitudinal trajectories of cognition according to the presence of the APOE ɛ4 allele in male and female PD patients over 5 years of follow-up.
METHODS
Study design and participants
For this study, we used data from the PPMI database. PPMI is a multicenter, international, longitudinal cohort study designed to identify biomarkers of PD progression. The methodology and details of the study assessments have been published elsewhere [25, 26] and are available on the PPMI website (http://www.ppmi-info.org/study-design). PD patients who were recently diagnosed with idiopathic PD, were not treated for PD, and had a dopamine transporter deficit, as assessed by single photon emission computed tomography, were included in the PPMI study cohort. The patients underwent baseline and scheduled follow-up evaluations at 3-month intervals for the first year and 6-month intervals in the subsequent years. We downloaded all the information required for this analysis from the PPMI database in July 2019.
Four hundred and twenty-three patients with early de novo PD were enrolled in the PPMI between 2010 and 2013. We excluded patients who had no data for the APOE genotype (n = 39), were lost to follow-up within the first year after enrollment (n = 16), or had no yearly follow-up data (n = 1). We also excluded patients who had APOE ɛ2/ɛ4 (n = 6), given that the presence of the APOE ɛ2 allele, which has potential neuroprotective effects [12], may attenuate the harmful impact of the APOE ɛ4 allele. Finally, 361 patients were included in this study.
Standard protocol approvals, registrations, and patient consents
The PPMI study was approved by the institutional review boards of all participating institutions, and written informed consent was obtained from all participants at each site. The study was registered at clinicaltrials.gov as NCT01141023.
Assessments
We examined cognitive decline by using the Montreal Cognitive Assessment (MoCA) [27]. The MoCA is widely used to evaluate global cognitive function in PD patients [28–30], and it has been shown to be more sensitive than the Mini-Mental State Examination in detecting early baseline and longitudinal cognitive impairment in PD patients [31]. We obtained data on the MoCA score at baseline and at the 1-, 2-, 3-, 4-, and 5-year follow-up visits.
The APOE genotype was identified from DNA extracted from the whole blood of the patients. Two SNPs (rs429358 and rs7412) were genotyped to distinguish between the APOE ɛ2, ɛ3, and ɛ4 alleles. TaqMan assays or the NeuroX genotyping platform were used to genotype these SNPs [32].
We also obtained data on age, sex, disease duration, age at onset, educational level, the Movement Disorders Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) score, and the use of medications for PD at the follow-up visits. The use of dopaminergic drugs was expressed as the levodopa equivalent daily dose (LEDD) [33].
Statistical analysis
Analyses were performed using R version 3.6.0 (R Foundation for Statistical Computing, Vienna, Austria). The data are presented as the means, standard deviations, and frequencies. The normality of the data was tested with the Shapiro-Wilk test. The baseline characteristics of the APOE ɛ4 carriers and noncarriers in men and women were summarized, and differences were tested by Student’s t-tests, Mann-Whitney U tests, or chi-square tests, as appropriate. We calculated the annual decline in MoCA scores using the following formula: (MoCA score at baseline –MoCA score at the last follow-up) / duration of follow-up. To investigate the sex-specific effects of the APOE ɛ4 status on cognitive decline, we used separate generalized linear mixed effects (GLME) models with a random effect for participants for men and women using the R package “lme4”. In these models, the covariates were age, disease duration, years of education, the baseline MoCA score, and their interactions with time. Power calculations were performed using the R package “simR”. To further explore the interactions between the APOE ɛ4 status and sex, we analyzed the data from both men and women, and pairwise comparisons were performed for the following groups: APOE ɛ4 + /M, APOE ɛ4-/M, APOE ɛ4 + /F, and APOE ɛ4-/F. We conducted two different sensitivity analyses. First, we excluded patients who had cognitive impairment at baseline. For this analysis, cognitive impairment was defined as a total score of less than 26 on the MoCA [34]. Second, considering that dopaminergic treatment may affect cognitive function, particularly in the early stage of PD [35], we additionally included LEDD and the interaction between LEDD and time as covariates in the models.
No adjustments were made to control for type II error. Thus, all analyses should be considered exploratory. All tests were 2-sided, and p values less than 0.05 were regarded as statistically significant.
RESULTS
Study population characteristics
The baseline characteristics of the whole study population are shown in Table 1. The mean age and age at PD onset of the patients were 61.4±9.8 years and 59.6±9.8 years, respectively, and 238 (66%) patients were men. The annual decline in MoCA scores was 0.24±0.58. Ninety patients were carriers of APOE ɛ4, including the genotypes ɛ3/ɛ4 (n = 81) and ɛ4/ɛ4 (n = 9), and 271 were noncarriers, including ɛ2/ɛ2 (n = 2), ɛ2/ɛ3 (n = 46), and ɛ3/ɛ3 (n = 223).
Baseline characteristics
Data are n (%) and the mean (standard deviation). APOE, apolipoprotein E; MDS-UPDRS, Movement Disorder Society Unified Parkinson’s Disease Rating Scale; MoCA, Montreal Cognitive Assessment; PD, Parkinson’s disease.
In the male group, 65 patients (27%) were APOE ɛ4-positive and 173 (73%) were APOE ɛ4-negative. In the female group, 25 patients (20%) were APOE ɛ4-positive, and 98 (80%) were APOE ɛ4-negative. The proportion of patients with the APOE ɛ4 allele did not differ between men and women (p = 0.146). In both the male and female groups, there were no significant differences between the carriers and noncarriers in age, disease duration, age at PD onset, or the scores for parts 1, 2, and 3 of the MDS-UPDRS. The MoCA scores were also not different between the APOE ɛ4 carriers and noncarriers in both the men (p = 0.168) and women (p = 0.621). The LEDD values of the patients at each follow-up visit are presented in Supplementary Table 1.
The sex-specific effects of APOE ɛ4 on cognitive trajectories
The results of the sex-stratified GLME models are shown in Table 2. The main effect of APOE ɛ4 on the change in the MoCA score was not significant in either the men (p = 0.242) or women (p = 0.995). Regarding the interaction between APOE ɛ4 and time, the males with APOE ɛ4 showed a steeper rate of cognitive decline than did those without APOE ɛ4 (estimate –0.009, standard error [SE] 0.004, t = –2.50, p = 0.012). In contrast, the longitudinal changes in the MoCA scores did not differ according to the presence of the APOE ɛ4 allele in females (p = 0.469). The power to detect the interaction effect between APOE ɛ4 and time was 78.0% (95% confidence interval [CI], 68.6 to 85.7) for men and 14.0% (95% CI, 7.9 to 22.4) for women. A diagrammatic depiction of the model slopes for APOE ɛ4 in men and women can be found in Figs. 1, 2, respectively.
Sex-stratified generalized linear mixed effects models
The APOE ɛ4- was used as the reference group. The significant values are in bold. AIC, Akaike Information Criterion; APOE, apolipoprotein E; BIC, Bayesian Information Criterion; MoCA, Montreal Cognitive Assessment; SE, standard error.
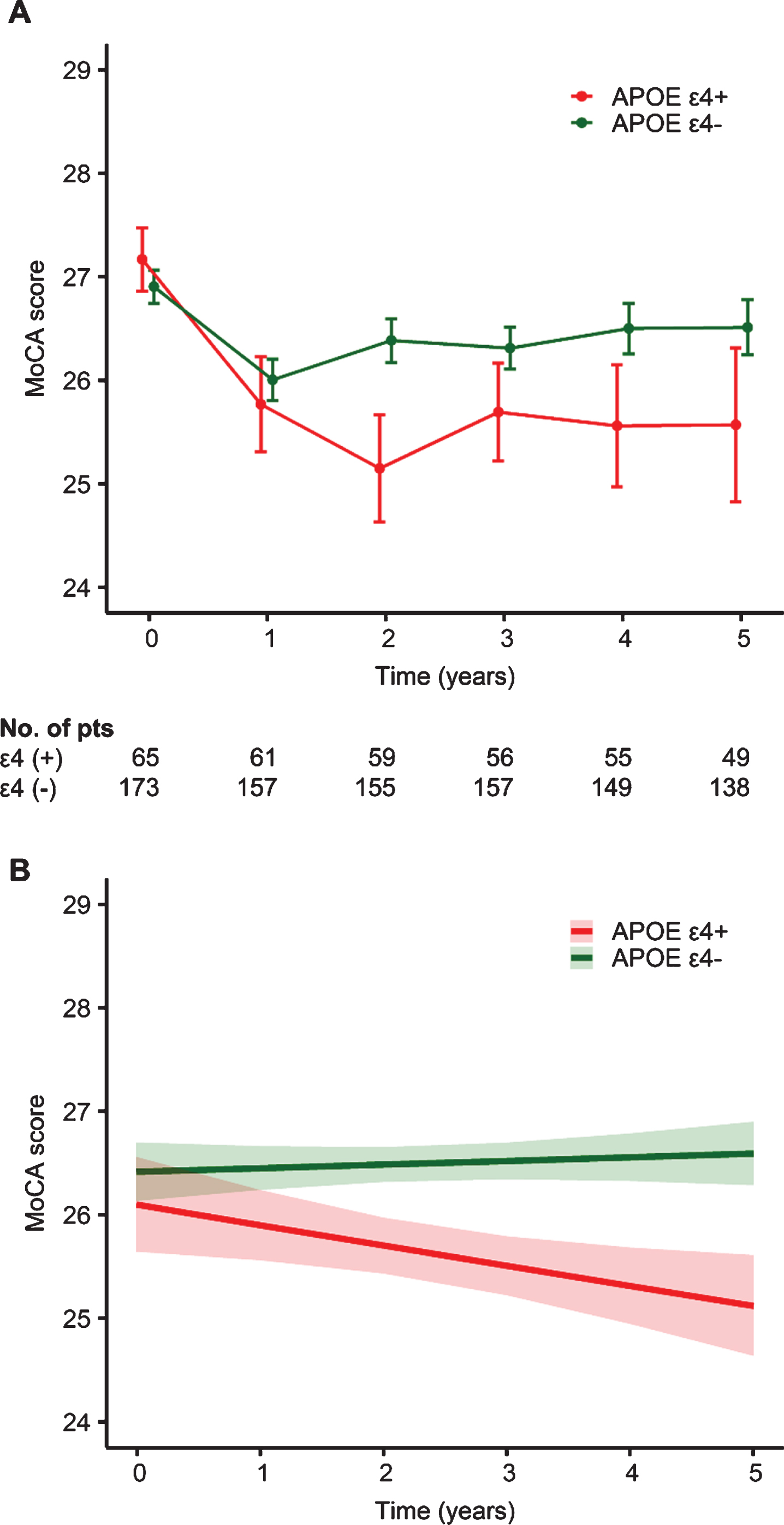
Unadjusted (A) and adjusted (B) longitudinal changes in the Montreal Cognitive Assessment (MoCA) score according to the presence of apolipoprotein E (APOE) ɛ4 in the male group. I bars indicate standard errors, and shaded regions indicate 95% confidence intervals.
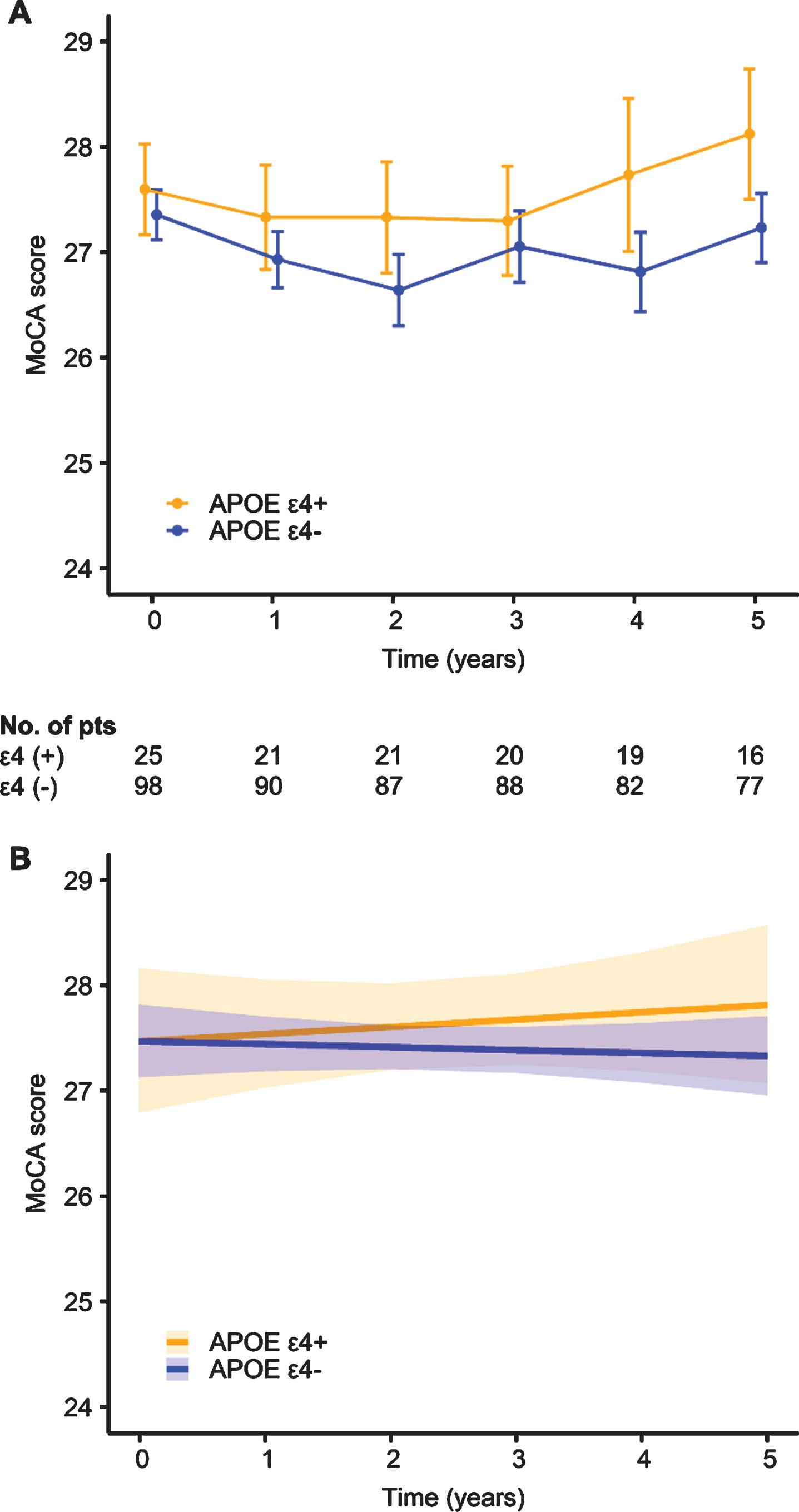
Unadjusted (A) and adjusted (B) longitudinal changes in the Montreal Cognitive Assessment (MoCA) score according to the presence of apolipoprotein E (APOE) ɛ4 in the female group. I bars indicate standard errors, and shaded regions indicate 95% confidence intervals.
When the data from both men and women were used, there were no significant main effects of APOE ɛ4/sex groups on the change in the MoCA score (Supplementary Table 2). However, the interaction results showed that the rate of cognitive decline was steeper in the APOE ɛ4 + /M group than in the APOE ɛ4-/M group (estimate –0.009, SE 0.003, t = –2.59, p = 0.010), APOE ɛ4 + /F group (estimate –0.012, SE 0.006, t = –2.11, p = 0.035), and APOE ɛ4-/F group (estimate –0.009, SE 0.004, t = –2.33, p = 0.020) (Fig. 3). The rate of change in cognitive performance over time was not significantly different between the APOE ɛ4-/M group and the APOE ɛ4 + /F (p = 0.583) and APOE ɛ4-/F groups (p = 0.953). Similarly, there was no significant interaction between APOE ɛ4 and time on longitudinal cognitive performance in females (p = 0.577).
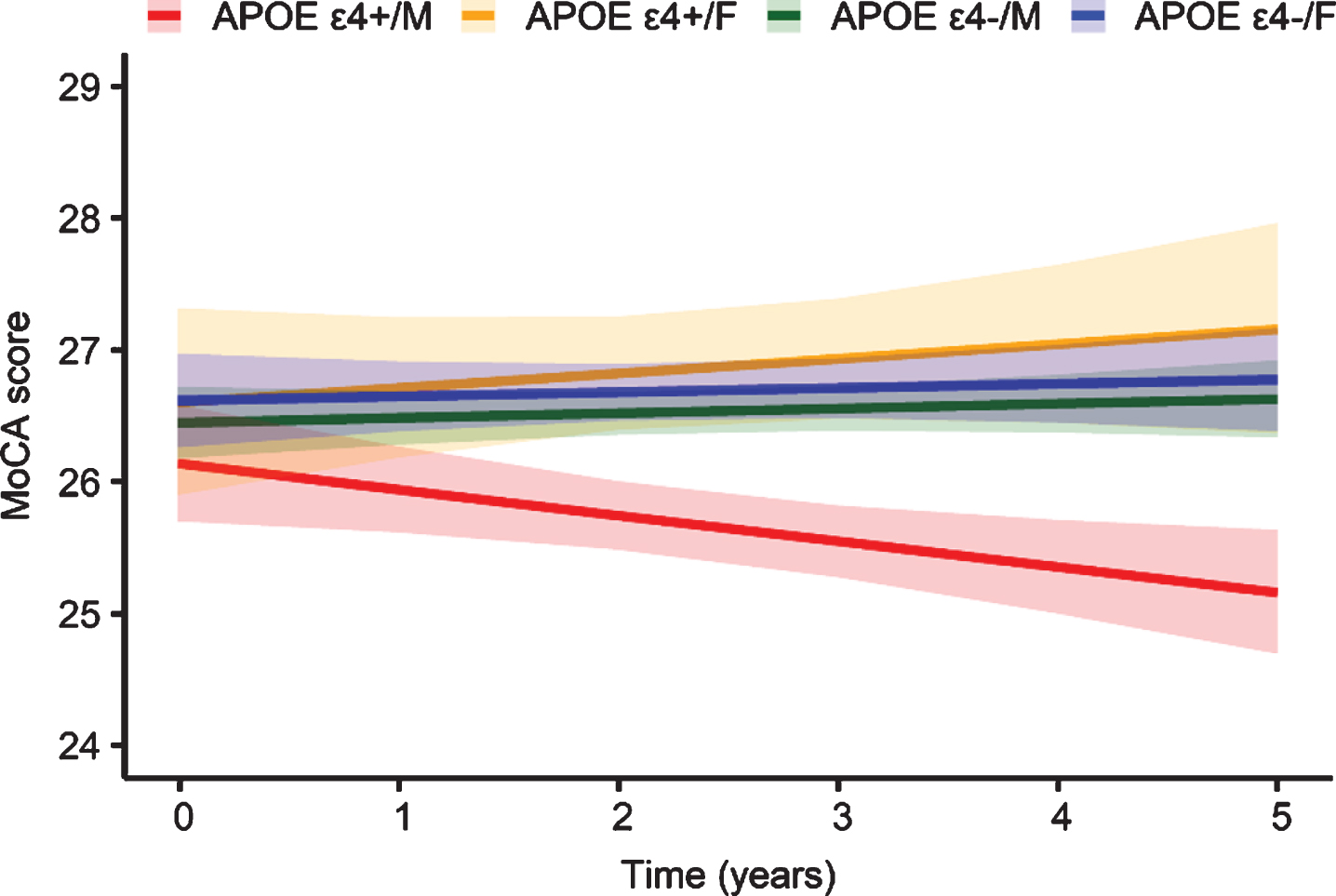
Adjusted longitudinal changes in the Montreal Cognitive Assessment (MoCA) score by apolipoprotein E (APOE) ɛ4 status and sex. Shaded regions indicate 95% confidence intervals.
Sensitivity analyses
The association of the APOE ɛ4 allele with cognitive decline remained significant in men when those who had normal cognitive performance at baseline were included in the study population (n = 281; APOE ɛ4 + /M [n = 54], APOE ɛ4-/M [n = 131], APOE ɛ4 + /F [n = 20], and APOE ɛ4-/F [n = 76]; Supplementary Table 3) or LEDD variables were included as covariates in the models (Supplementary Table 4). The comparisons of the APOE ɛ4 + /M group with the other groups also showed consistent results.
DISCUSSION
Using reliable data recorded with strict participant inclusion criteria from the PPMI study cohort, we investigated the impact of the APOE ɛ4 allele on changes in cognitive performance over time in male and female patients with early PD. The analyses presented here showed that a significant association of the APOE ɛ4 allele with cognitive decline existed in men but not in women. Furthermore, male PD patients with APOE ɛ4 showed a steeper rate of cognitive decline than females. These findings were shown to be robust in the sensitivity analyses. To the best of our knowledge, this is the first study to demonstrate sex-specific effects of the APOE ɛ4 allele on cognitive decline in patients with PD.
There is ample evidence showing that the APOE ɛ4 allele affects cognitive function mainly via early and abundant amyloid accumulation in the brain [11, 12]. Cerebral amyloid deposition is regarded as one of the important contributors to cognitive impairment in patients with PD [36]. However, it has been reported that the APOE ɛ4 allele also influences tau-mediated neurodegeneration, abnormal glucose metabolism, neuroinflammation, and impaired cerebrovascular function [12]. In addition, two recent studies found that the allele directly impacts the development of α-synuclein pathology [37, 38]. These amyloid-unrelated effects may also contribute to cognitive decline in PD patients. Unfortunately, we do not understand the mechanism underlying sex differences in the effect of the APOE ɛ4 allele on cognition in PD patients from the current study design, and this topic requires further investigation.
Previous data have shown that the incidence rates of cognitive impairment in PD patients are higher in men than in women [39–44], indicating that male sex is an important risk factor for cognitive decline in PD patients. However, these observations are partly consistent with our results because the change in cognitive performance over time in male patients without the APOE ɛ4 allele was not significantly different from that in female patients. In this regard, a direct effect of APOE ɛ4 by sex interaction would, at least in part, explain why there is a sex difference in the cognitive function of PD patients. Although other potential mechanisms, including exposure to estrogens and a relatively higher exposure of men to environmental risk factors, have been suggested [43], these hypotheses are not supported by our findings.
Surprisingly, cognitive decline over 5 years of follow-up was not clearly observed in females or even males without the APOE ɛ4 allele, suggesting that the follow-up duration in these groups was too short to characterize longitudinal cognitive trajectories. Alternatively, the small rate of cognitive change in those groups may be related to the effect of dopaminergic treatment on cognitive function. Although the PPMI cohort included patients with drug-naïve PD, all patients started dopaminergic treatment during the follow-up period. It is known that cognitive deficits in the early phase of PD are mainly mediated by frontostriatal dopaminergic dysfunction [35, 45], and several studies on PD have shown that dopaminergic drugs are beneficial for some cognitive functions involving frontal-lobe related tasks [46–49]. Although we cannot exclude the possibility that dopaminergic treatment may affect some of the main findings of the present study, the results remained the same after we adjusted for the dopaminergic effect on cognition; thus, this potential bias in our findings is likely to be minimal.
With respect to AD, previous studies have shown that the APOE ɛ4 allele leads to a higher risk of the disease in women than in men [50], which is supported by the results of a recent meta-analysis in which women with the APOE ɛ4 allele had an increased risk of incident AD between the ages 65–75 years [19]. Similarly, among individuals with normal cognitive performance and mild cognitive impairment, the negative impact of APOE ɛ4 on AD biomarkers was stronger in women than in men [21–23], although not consistently [18, 24]. Thus, our findings suggest that the sex-related effects of the APOE ɛ4 allele on cognitive impairment in PD patients differ from the effects in AD patients. This hypothesis may be supported by the fact that, in contrast to the prevalence of cognitive impairment in PD, the prevalence of AD is higher in women than in men [51].
However, this study should be considered with the following limitations in mind. First, the number of female PD patients who were APOE ɛ4 carriers was relatively small, which may hinder the detection of significant differences. Furthermore, lack of power attributable to the current sample size might undermine the reliability of our results. Second, because the MoCA reflects overall cognitive function, additional research should be conducted to determine which specific cognitive domains are affected by the interaction between sex and APOE ɛ4 in early PD patients. Third, there is a growing body of evidence showing that the APOE ɛ2 allele may have protective effects against AD [12]. Additionally, dose-dependent effects of the APOE ɛ4 and ɛ2 alleles were found on AD development [52]. Unfortunately, such effects on cognitive decline could not be investigated in our analysis because the sample size was small. Additional large-scale studies can be conducted to clarify these issues. Fourth, all analyses were considered exploratory; thus, we did not adjust for multiple comparisons. Last, because we assessed cognitive function as only a continuous variable, the clinical importance of the group differences in cognitive decline may be unclear.
In this cohort study, we found potential sex-related differences in the impact of the APOE ɛ4 allele on cognition in the early phase of PD, which may highlight the importance of considering sex when APOE ɛ4-related vulnerability to early cognitive decline in PD patients is evaluated. Additional research is needed to replicate our findings in other datasets and to reveal the exact mechanism of the sex-dependent effect of APOE ɛ4 on cognitive decline in PD patients.
CONFLICT OF INTEREST
No conflicting relationship exists for the authors.
Footnotes
ACKNOWLEDGMENTS
Data used in the preparation of this article were obtained from the Parkinson’s Progression Markers Initiative (PPMI) database (www.ppmi-info.org/data). For up-to-date information on the study, visit www.ppmi-info.org. PPMI (a public-private partnership) is funded by the Michael J. Fox Foundation for Parkinson’s Research and multiple funding partners, including AbbVie, Avid, Biogen, Bristol-Myers Squibb, Covance, GE Healthcare, Genentech, GlaxoSmithKline, Lilly, Lundbeck, Merck, Meso Scale Discovery, Pfizer, Piramal, Roche, Servier, Teva, and UCB.
This study was supported by a research grant of National Research Foundation (NRF) funded by the Ministry of Science and ICT (MSIT) in Korea (NRF-2020R1C1C1013382).
