Abstract
Background:
Sleep disturbances and neuropsychiatric symptoms are some of the most common nonmotor symptoms in Parkinson’s disease (PD). The effect of subthalamic stimulation (STN-DBS) on these symptoms beyond a short-term follow-up is unclear.
Objective:
To examine 36-month effects of bilateral STN-DBS on quality of sleep, depression, anxiety, and quality of life (QoL) compared to standard-of-care medical therapy (MED) in PD.
Methods:
In this prospective, controlled, observational, propensity score matched, international multicenter study, we assessed sleep disturbances using the PDSleep Scale-1 (PDSS), QoL employing the PDQuestionnaire-8 (PDQ-8), motor disorder with the Scales for Outcomes in PD (SCOPA), anxiety and depression with the Hospital Anxiety and Depression Scale (HADS), and dopaminergic medication requirements (LEDD). Within-group longitudinal outcome changes were tested using Wilcoxon signed-rank and between-group longitudinal differences of change scores with Mann-Whitney U tests. Spearman correlations analyzed the relationships of outcome parameter changes at follow-up.
Results:
Propensity score matching applied on 159 patients (STN-DBS n = 75, MED n = 84) resulted in 40 patients in each treatment group. At 36-month follow-up, STN-DBS led to significantly better PDSS and PDQ-8 change scores, which were significantly correlated. We observed no significant effects for HADS and no significant correlations between change scores in PDSS, HADS, and LEDD.
Conclusions:
We report Class IIb evidence of beneficial effects of STN-DBS on quality of sleep at 36-month follow-up, which were associated with QoL improvement independent of depression and dopaminergic medication. Our study highlights the importance of sleep for assessments of DBS outcomes.
GLOSSARY
deep brain stimulation Hospital Anxiety and Depression Scale levodopa equivalent daily dose standard-of-care medical therapy nonmotor symptoms Parkinson’s Disease PD Questionnaire-8 PD Sleep Scale quality of life subthalamic nucleus
INTRODUCTION
Sleep disturbances and depressive symptoms are amongst the most common nonmotor symptoms (NMS) in patients with Parkinson’s disease (PD) and are major predictors of negative health-related quality of life (QoL) [1]. Deep brain stimulation (DBS) of the subthalamic nucleus (STN) is effective in improving medication-refractory motor impairment [2], QoL [3, 4], and a wide range of NMS [5, 6] in patients with PD. Long-term effects of STN-DBS on NMS have only been studied to a limited extent. Previous studies have documented beneficial effects of STN-DBS on sleep up to three years postoperatively [7–9]. However, there is a lack of controlled studies comparing effects of STN-DBS on sleep symptoms to standard-of-care medical treatment (MED).
It has been described that in PD patients mood disturbances such as depression and anxiety are associated with sleep disturbances [10] and worse quality of life. Furthermore, sleep problems and depressive symptoms in PD are partly responsive to dopaminergic medication [11, 12]. However, little is known about the long-term effects of STN-DBS on sleep, mood, and dopaminergic medication and their relationship with quality of life.
Therefore, in this study, we aimed to investigate 36-month effects of STN-DBS on these parameters in comparison to patients treated with MED. We hypothesized that STN-DBS improves subjective sleep symptoms at 36-month follow-up independent of mood symptoms and dopaminergic medication and that these sleep improvements are correlated with improvements of quality of life.
MATERIALS AND METHODS
Study design
We prospectively recruited patients between 03/2011 and 10/2015 in an ongoing, observational, controlled, multicenter international study as part of the DBS and medication arms of the NILS study that investigates NMS in patients with PD [6]. Here, we report the results of the 36-month follow-up. The study was carried out in accordance with the declaration of Helsinki and authorized by the medical ethics committees of the participating centers (master votes for Germany: Cologne, study number: 12–145, German Clinical Trials Register: DRKS00006735, and for the United Kingdom: National Research Ethics Service South East London REC 3, 10/H0808/141; NIHR portfolio, number: 10084). All patients gave written informed consent prior to any study procedures. Enrollment criteria and study procedures have been reported previously [13] and will only be described briefly.
Participants
PD diagnosis was based on the UK Brain Bank criteria and DBS screening was carried out according to the international guidelines. Patients were eligible for DBS treatment if they were responsive to levodopa (>30% improvement of motor examination assessed by the Unified PD Rating Scale–III). We excluded patients with clinically relevant neuropsychiatric disorders or neuropsychological impairment [14] as assessed in a multidisciplinary team including specialized neuropsychiatrists and neuropsychologists. For the final analysis, we included only patients with advanced PD with dyskinesia, ON/OFF fluctuations or medication-refractory tremor in the MED group, to achieve better comparability between the STN-DBS and MED group. Patients in the MED group selected for the final analyses were regarded as potential candidates for STN-DBS in the multi-disciplinary assessments, but preferred non-surgical treatment at this point in time according to published standard-of-care recommendations [15]. Various parameters may have influenced patients’ informed decisions including patients’ age, disease duration, dopaminergic medication requirements, and severity of motor and NMS [15]. Patients in both groups received oral medical therapy, which could include levodopa, dopamine agonists, monoamine oxidase type B inhibitors, catechol-O-methyltransferase inhibitors, amantadine, or other drugs for treatment of PD symptoms. We have published results regarding other non-motor aspects of PD in the same cohort elsewhere [13].
Clinical assessment
Baseline and follow-up assessments took place in the ON-medication state (MedON). Patients in the STN-DBS group were also in the ON-stimulation state at follow-up (medication and stimulation ON, MedON/StimON). Clinical assessments were carried out using the following scales:
The
The
The
We calculated
Motor impairment, activities of daily living and motor complications were assessed with the
Statistical analysis
We used SPSS 26.0 (IBM Corporation) for the statistical analysis. Propensity score matching was applied to identify sub-cohorts with balanced baseline characteristics, which is a method of estimating treatment effects in observational studies, when random treatment allocation is difficult to implement [26]. Propensity score matching aims to reduce bias caused by preexisting differences between treatment groups [27]. The technique includes separate logistic regressions to compute propensity scores of the different subjects. The most common implementation of propensity score matching is 1:1, in which pairs of patients with overlapping propensity scores within a predefined calliper are formed [28]. The matching was computed with Propensity Score Matching for SPSS (version 3.04) [29]. For matching, we used the following variables: age at baseline, disease duration since diagnosis, dopamine agonist LEDD and preoperative SCOPA total score. We implemented a 1:1 ratio nearest-neighbor matching algorithm with a 0.25 caliper without replacement. Afterwards, balance diagnostics of the entered covariates based on Cohen’s effect size | d|<0.25 were calculated as a threshold for an accurate balance of the covariates [28].
The assumption of normality distribution was assessed with the Shapiro-Wilk test. Differences of baseline characteristics between the two groups were analyzed using Chi2-test for dichotomous variables and for continuous variables with Mann–Whitney U or unpaired t-tests, when parametric criteria were fulfilled. To determine outcome changes from baseline to follow-up within each group, Wilcoxon signed-rank or paired sample t-tests were calculated. Mann-Whitney U tests of change scores between STN-DBS and MED groups (mean testbaseline – mean testfollow - up) were conducted to analyze differences of outcome parameter changes. As we assessed a variety of tests, we employed Benjamini-Hochberg’s procedure for multiple comparisons and report corrected P-values (threshold: P < 0.05). Information on the clinical relevance of the responses can be found in the supplementary table e-3 (relative changes and respective effect sizes Cohen’s d). In addition, we explored the relationship between changes of PDQ-8, PDSS, HADS, and LEDD from baseline to 36-month follow-up using Pearson correlations and partial correlations, respectively Spearman correlations and partial correlations if the assumption of normality distribution was violated.
RESULTS
A total of 323 patients were screened, of whom 147 patients were excluded as they did not suffer from dyskinesia, ON/OFF fluctuations or medication-refractory tremor and, therefore, were no DBS candidates (see Fig. 1). Five patients were excluded because they underwent DBS in the globus pallidus interna or ventral intermediate nucleus of the thalamus, and ten patients because of neuropsychological impairment (Mini-Mental State Examination < 25). A total of 159 patients with PD (103 male) undergoing STN-DBS (n = 75) or MED (n = 84) were assessed eligible for this study. The 159 patients in the final sample had a mean age of 64.0 years±9.6 with mean disease duration of 8.8 years±5.0. The mean time to follow-up was 3.0 years±0.5.
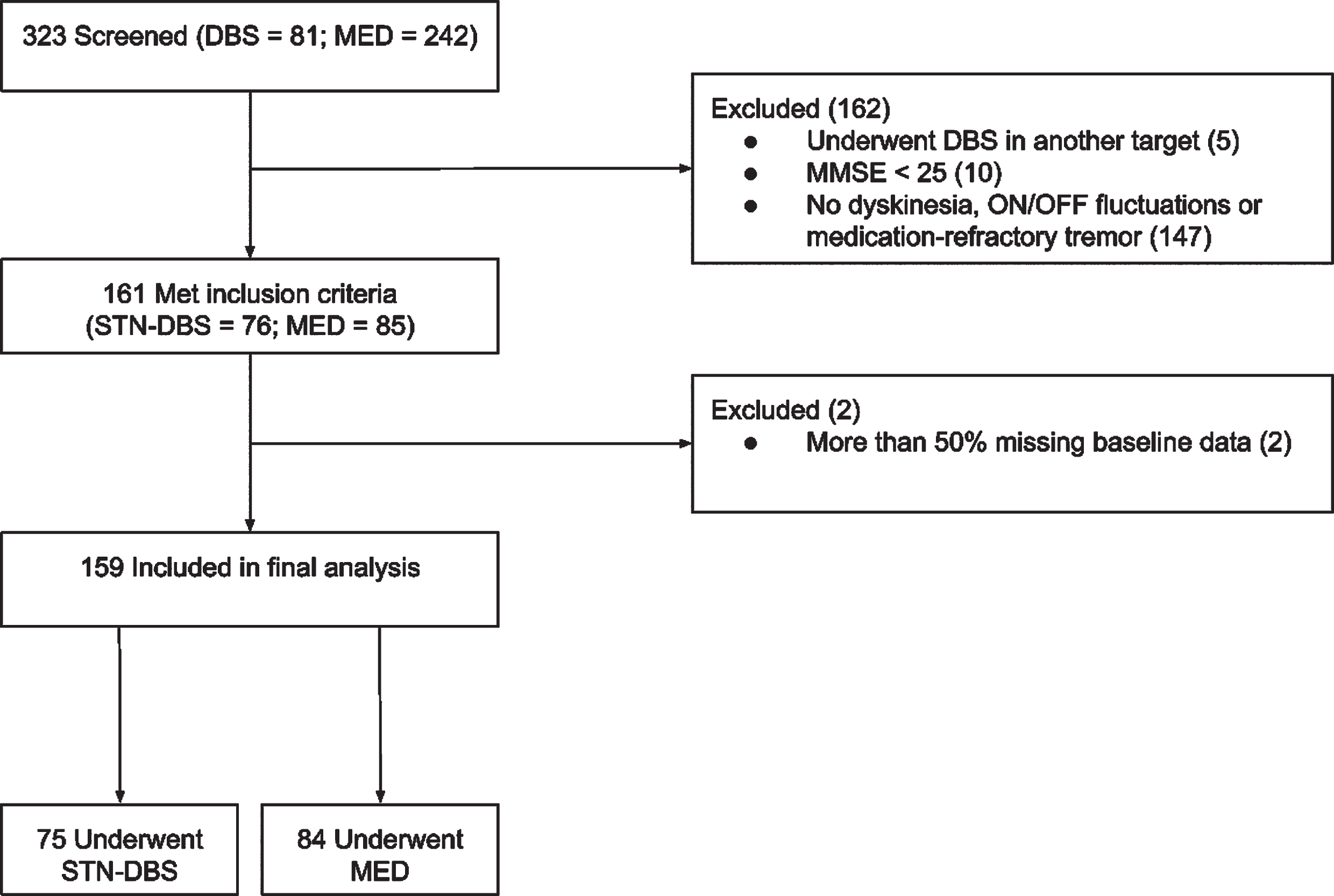
Patient selection. Abbreviations: MMSE = Mini-Mental State Examination; MED = standard-of-care medical treatment group; STN-DBS=subthalamic nucleus deep brain stimulation.
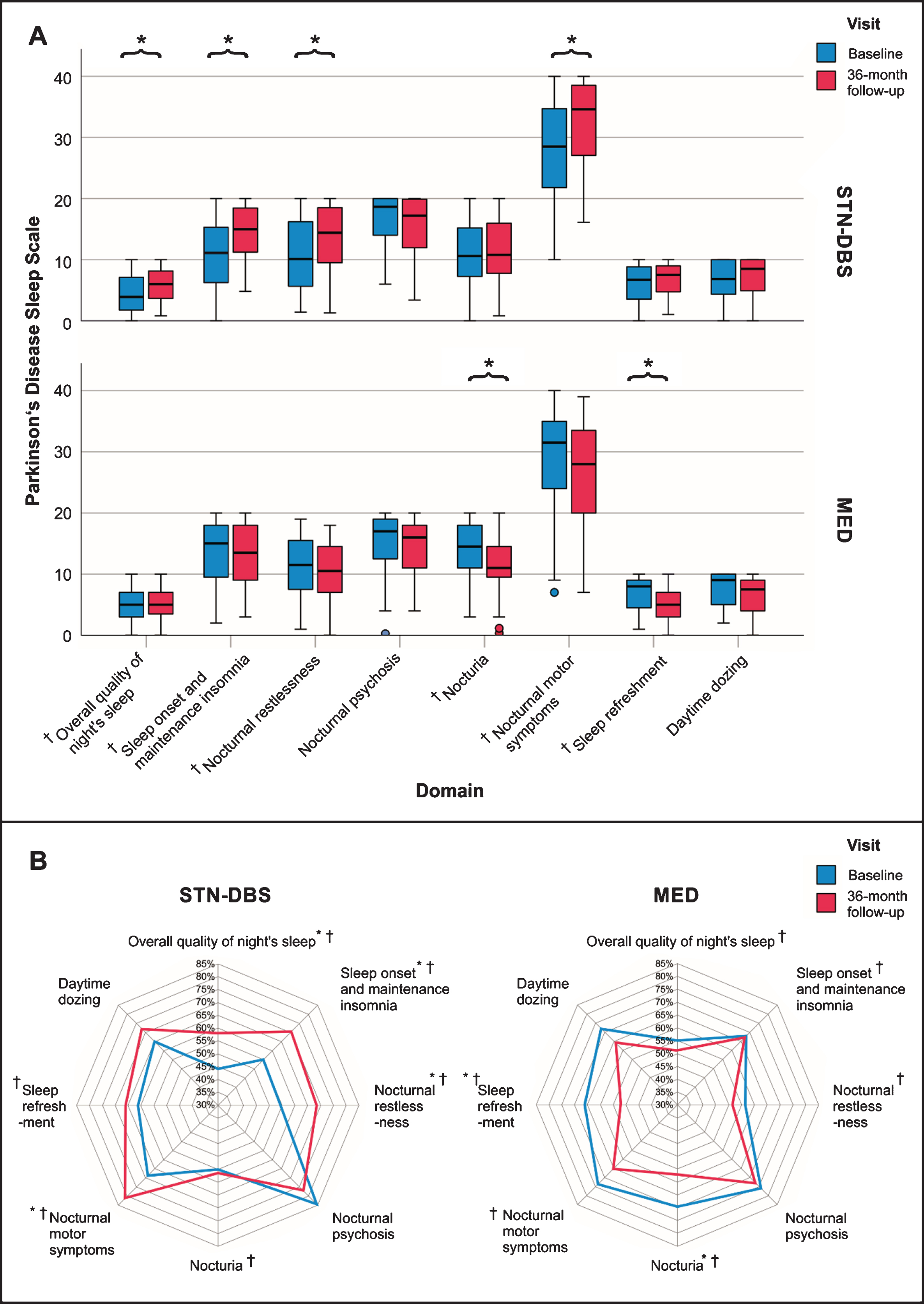
Parkinson’s Disease Sleep Scale domain scores at baseline and 36-month follow-up for the STN-DBS and MED groups in clustered boxplots and radar charts. PDSS domain scores at baseline (blue) and 36-MFU (red) for the STN-DBS and MED groups as clustered box plots (A) and radar charts (B). Significant within-group changes of PDSS domains from baseline to 36-MFU are highlighted with a black star and significant between-group differences (STN-DBS vs MED) with a cross. (A) In the clustered box plot, small circles represent outliers (2–3 SD). (B) PDSS domain scores are presented as percentage of maximum domain scores. Smaller areas represent more severe sleep impairment. In the STN-DBS group, PDSS domains overall quality of night’s sleep, sleep onset and maintenance insomnia, nocturnal restlessness, and nocturnal motor symptoms significantly improved from baseline to 36-MFU. In the MED group, nocturia and sleep refreshment significantly worsened from baseline to 36-MFU. Between-group longitudinal differences in PDSS domain scores were significant for overall quality of night’s sleep, sleep onset and maintenance insomnia, nocturnal restlessness, nocturia, nocturnal motor symptoms, and sleep refreshment. Abbreviations: 36-MFU = 36-month follow-up; MED = standard-of-care medical treatment; PDSS = Parkinson’s Disease Sleep Scale-1; STN-DBS = subthalamic nucleus deep brain stimulation.
Baseline characteristics in the original and matched cohort
In the original cohort, patients in the STN-DBS group were significantly younger, had longer duration of PD, higher LEDD, and greater dopamine agonist requirements than the MED group (see Table 1). Also, the STN-DBS group had more severe impairment in all of the clinical scales except for HADS (all P < 0.05).
Baseline characteristics in the original cohort and matched sub-cohort
Demographic characteristics and outcome parameters at baseline in the original cohort and matched sub-cohort. Significant results are highlighted in bold font. aMann-Whitney U test or t test when parametric test criteria were fulfilled. Abbreviations: HADS = Hospital Anxiety and Depression Scale; LEDD = levodopa equivalent daily dose; MED = standard-of-care medical treatment; PDSS = Parkinson’s Disease Sleep Scale-1; PDQ-8 = 8-item Parkinson’s Disease Questionnaire; SCOPA = Scales for Outcomes in Parkinson’s Disease; STN-DBS=subthalamic nucleus deep brain stimulation.
Propensity score matching resulted in a sub-cohort of 80 patients (40 patients in each group). Balance diagnostics revealed no significant differences for the demographic and clinical outcome parameters. Baseline motor subscores and Mini-Mental State Examination scores of the matched cohort are reported in the Supplementary table e-1. The following results refer to the matched cohort. Additionally, clinical outcomes of the original cohort are presented in the Supplementary table e-2.
Clinical outcome changes at 36-month follow-up
Between-group longitudinal differences (STN-DBS vs. MED) and longitudinal within-group changes are reported in Table 2. The time of follow-up assessment did not differ significantly between the groups (STN-DBS = 3.0±0.2 years, MED = 3.1±0.4 years; P = 0.167).
Outcomes at baseline and 36-month follow-up in the matched sub-cohort
Outcome parameters at baseline and follow-up for STN-DBS and MED groups. Multiple comparisons due to multiple outcome parameters were corrected with Benjamini-Hochberg’s method. Post-hoc exploratory analyses were conducted for PDSS domains and HADS subscales. Significant results are highlighted in bold font. aWilcoxon signed rank test between baseline and 36-month follow-up to analyze within-group changes of outcome parameters. bMann-Whitney U test to analyze between-group longitudinal differences of change scores between STN-DBS and MED group. Significant results are highlighted in bold font. Abbreviations: 36-MFU = 36-month follow-up; HADS = Hospital Anxiety and Depression Scale, LEDD = Levodopa equivalent daily dose; MED = standard-of-care medical treatment group; PDSS = Parkinson’s Disease Sleep Scale-1; PDQ-8 = 8-item Parkinson’s Disease Questionnaire; SCOPA = Scales for Outcomes in Parkinson’s Disease; STN-DBS=subthalamic nucleus deep brain stimulation.
Between-group longitudinal differences of change scores were significant for the PDSS total, PDQ-8 SI, SCOPA-total, LEDD total, and LEDD of dopamine agonists, all favoring STN-DBS. In the STN-DBS group, the PDSS total score significantly improved from baseline to 36-month follow-up. We further observed beneficial effects of STN-DBS on the SCOPA-total score. In contrast, in the MED group, we found a significant worsening of the PDSS total score, whereas the SCOPA total score remained stable. As expected, the LEDD total and LEDD of dopamine agonists were reduced significantly in the STN-DBS group and remained stable in the MED group. In both groups, the HADS total score, as well as its depression and anxiety subscales did not change significantly at 36-month follow-up.
Post-hoc analyses of the different PDSS domains revealed significant beneficial effects of STN-DBS on ‘Overall quality of night’s sleep’, ‘Sleep onset and maintenance insomnia’, ‘Nocturnal restlessness’, and ‘Nocturnal motor symptoms’. In contrast, in the MED group, we observed a significant worsening of ‘Nocturia’ and ‘Nocturnal motor symptoms’. Significant between-group longitudinal differences were found for ‘Overall quality of night’s sleep’, ‘Sleep onset and maintenance insomnia’, ‘Nocturnal restlessness’, ‘Nocturia’, ‘Nocturnal motor symptoms’, and ‘Sleep refreshment’.
Explorative correlation analyses
Better PDSS total score outcomes were significantly correlated with better SCOPA total score (r s = –0.35, P = 0.002) and PDQ-8 SI (r s = –0.40, P < 0.001) outcomes. A partial correlation between changes in PDSS total score and PDQ-8 SI was still significant after controlling for the SCOPA outcome (r s = –0.34, P = 0.003). No significant correlations were found between changes of PDSS total score and other parameters. PDQ-8 SI changes were also significantly correlated with changes in HADS total scores (r s = 0.48, P < 0.001), its anxiety and depression subscales (both (r s = 0.42, P < 0.001), and with SCOPA total score (r s = 0.28, P = 0.016). There were no significant correlations between changes in overall LEDD or LEDD dopamine agonists with any other outcome changes including the different PDSS domains.
DISCUSSION
In this prospective, controlled, observational, propensity score matched, international multicenter study with a 36-month follow-up in 80 patients with PD, we report beneficial effects of STN-DBS on subjective sleep symptoms which were significantly correlated with improvements of QoL and motor symptoms. Depressive and anxiety symptoms remained unchanged from baseline to 36-month follow-up. Beneficial effects of STN-DBS on subjective sleep disturbances were not associated with changes in depression, anxiety, or dopaminergic medication requirements. The relationship between quality of life and subjective sleep disturbances was still significant when controlling for motor symptoms.
Motor symptoms, quality of life, and LEDD
In line with previous studies with up to 36-month follow-up, we observed long-term improvements in motor symptoms and QoL [2, 4]. In the MED group, standard-of-care resulted in stabilized motor symptoms and QoL at 36-month follow-up. As expected, STN-DBS led to a significant LEDD reduction, while medication requirements remained stable in the MED group.
Specific aspects of sleep
To our knowledge, this is the first controlled study demonstrating sustained improvements in various aspects of sleep dysfunction in PD patients following STN-DBS at 36-month follow-up. Compared to a control group receiving MED, STN-DBS significantly improved the following specific sleep aspects:
The present work is the first observation, to our knowledge, of a significantly better outcome of symptoms of
We observed a significant improvement of symptoms of
In our study, we observed significant between-group longitudinal differences of symptoms of
The present study shows beneficial effects on
To our knowledge, this is the first study to report significantly favorable effects on self-reported
Anxiety and depression
In the present study, we did not observe significant changes of anxiety and depression at 36-month follow-up. This result is in line with previous studies, on depressive and anxiety symptoms by Weaver et al. and Berney et al. with a follow-up period up to one year [38, 39]. In contrast, Funkiewiez et al. found improvements in depressive symptoms after bilateral STN-DBS one year and three years postoperatively [40]. Recent studies have reported that depression and anxiety depend on the location of active DBS contacts, the volume of tissue activated by DBS or the structural connectivity profiles between the STN and prefrontal cortex [41].
Mechanisms of improved sleep following STN-DBS
Heterogeneous causes may account for improvements of sleep disturbances following STN-DBS. Firstly, enhanced motor function may be an important contributor to improvements in sleep. This is in line with our observation of significantly better nocturnal motor symptoms and the observed significant correlation between changes in motor examination and sleep disturbances. Secondly, a pathophysiologic correlate for a direct DBS effect on sleep could be the modulation of regions in proximity of the STN. In particular, the pedunculopontine nucleus, which is reciprocally connected to the STN, regulates the sleep-wake cycle and serves as a critical ‘mesencephalic locomotor region’ [42]. As a reticular structure, the precise topographic boundaries of the pedunculopontine nucleus are indistinct. However, a location 5 mm ventral of the STN with even closer projections has been discussed [43]. Thirdly, reduced LEDD following STN-DBS may enhance sleep symptoms like daytime sleepiness although we observed no linear relationship with changes in LEDD. As a fourth possible explanation, the interplay between anxiety, depression, and sleep symptoms could play an important role. In the present study, improvements of PDSS total score and HADS anxiety and depression subscale scores were not significantly correlated, indicating that sleep and mood disorders could be independently modulated by STN-DBS. This is in line with studies in which we observed distinct ‘sweet spots’ for sleep and mood symptoms connected to specialized functional brain networks [41, 44].
Relationship between dopaminergic medication, sleep dysfunction and depression
Dopamine has a complex role for sleep disturbances in PD. Dopamine receptor agonists can both alleviate and aggravate specific aspects of sleep dysfunction in PD. We observed no association between sleep disturbances and overall LEDD or dopamine agonists LEDD. Ricciardi et al. found a significant relationship between improvement of sleep and dopamine agonists dose reduction [45]. On the other hand, there are also sleep-related adverse events caused by dopamine receptor agonists, such as excessive daytime sleepiness and sleep attacks [46].
The effect on mood of dopamine agonists is still controversial. We found no significant correlations between depressive symptoms and dosages of the other dopamine agonists. Future studies with larger cohorts are needed to investigate the relative effects of DBS and dopaminergic medication contributing to the observed changes.
Limitations
Our study has several limitations. The multicenter design of the study reduces bias caused by single center studies, increases external validity, and enables to capture data of large samples. Although the cohort size of the overall study population (n = 159) is one of the largest in studies of its kind, the matched treatment groups (STN-DBS n = 40; MED n = 40), however, were relatively small. This might be explained by the fact that we chose a conservative caliper for the matching procedure (0.25), as we wanted to implement a precise matching of the two groups. A narrower caliper would have resulted in smaller matched cohort sizes. Furthermore, although the controlled design of this study enabled us to separate between effects of PD progression and DBS, the treatment assignment was not randomized. In observational “real-life” studies, groups may differ systematically so that direct group comparisons may be misleading. By using propensity score matching, we aimed to increase causal inference by controlling preexisting differences in demographic and clinical parameters between the STN-DBS and MED group [27]. This method was used because a safe and effective treatment option cannot be withheld for three years from patients in advanced PD stages. Propensity score matching provides a precise method to match patients from two groups in these situations and diagnostic statistics in the present study indicated a well-balanced matching for all baseline parameters. However, inter-group comparisons results might have varied depending on the selection of matching variables. The choice of matching parameters was based on a previous publication of our group, [15] as these variables have led to balanced demographic and clinical baseline parameters between the treatment groups. However, we used dopamine agonists medication requirements instead of the overall LEDD for matching, as dopamine agonists dosage may be specifically important when investigating sleep dysfunction, because dopamine agonists have been observed to influence specific aspects of sleep dysfunction in previous publications [45, 46]. Propensity score matching cannot replace randomized trials, as there may be potentially relevant parameters, which were not assessed in the study, such as apathy or impulse control disorders. To account for this limitation, comparisons between the matched groups were carried out using independent sample tests. In the broader context of treatment choices, in the MED group, patients may have decided against surgical treatment options during the three year course of the study, in part, based on their short disease duration (mean 7.5 years in the original cohort) which was even shorter than in the EARLYSTIM study (7.7 years) [4]. Additionally, in the STN-DBS group, the matching led to the selection of less severely affected patients, as there were too few matching partners within the defined caliper in the MED group. Consequently, it cannot be ruled out that the observed effects of STN-DBS may be different in patients with very severe PD. Future studies should examine the dependence of DBS outcomes on the different levels of baseline impairment. Another limitation concerns the fact that medication changes were not resolved by an external panel in the present study, as e.g. in the EARLYSTIM study. However, standard-of-care medical therapy in each participating center was based on the same criteria [15] and motor scores and quality of life did not worsen significantly in the MED group over the 36-month period, indicating an effective medical treatment of these aspects. Moreover, as the minimal clinically important difference has not been reported for the PDSS-1 yet, we calculated Cohen’s effect size to quantify clinical relevance of the responses. Furthermore, we were interested in complex subjective sleep symptoms, such as nocturnal psychosis, sleep refreshment, nocturia, and motor state-related sleep symptoms, which cannot be captured by objective measurements of polysomnography or multiple sleep latency test, which measure parameters concerning sleep architecture. Further long-term studies are needed to investigate the relationship between sleep, depression, and anxiety with other neuropsychiatric symptoms, such as apathy, alexithymia, impulse control disorders, and mania [47] as they can develop late under treatment, have partly overlapping neuroanatomical pathways and may therefore be confounding factors in analyses of long-term outcomes.
CONCLUSION
This study provides Class IIb evidence for beneficial effects of STN-DBS compared to standard-of-care medical therapy on overall quality of nights’ sleep and different specific sleep disturbances, such as sleep onset and maintenance insomnia, nocturia, nocturnal motor symptoms, and sleep refreshment at 36-month follow-up. Sleep improvements were partly mediated by improved motor function. Furthermore, we observed no long-term effect of STN-DBS on depression and anxiety symptoms and no correlation between sleep and mood outcomes, indicating that sleep and mood disorders are separately influenced by STN-DBS. Beyond that, sleep and mood outcomes were significantly correlated with QoL improvement, highlighting their clinical relevance and the importance of holistic assessments of non-motor aspects in PD. Studies comparing different treatment options of PD pave the way to personalized medicine for individual patient profiles to better identify patients with the greatest benefits of each treatment option.
AUTHORS’ ROLES
Stefanie T. Jost – data acquisition, data analysis, drafting of the manuscript, tables and figures
K. Ray Chaudhuri – study concept and design, data acquisition, critical revision of the manuscript
Keyoumars Ashkan – surgical intervention, critical revision of the manuscript
Philipp A. Loehrer – data acquisition, critical revision of the manuscript
Monty Silverdale – data acquisition, critical revision of the manuscript
Alexandra Rizos – data acquisition, critical revision of the manuscript
Julian Evans – surgical intervention, critical revision of the manuscript
Jan Niklas Petry-Schmelzer – data acquisition, critical revision of the manuscript
Michael T. Barbe – data acquisition, critical revision of the manuscript
Anna Sauerbier – data acquisition, critical revision of the manuscript
Gereon R. Fink – data acquisition, critical revision of the manuscript
Veerle Visser-Vandewalle – surgical intervention, critical revision of the manuscript
Angelo Antonini – data acquisition, critical revision of the manuscript
Pablo Martinez-Martin – study concept and design, critical revision of the manuscript
Lars Timmermann – data acquisition, critical revision of the manuscript
Haidar S. Dafsari – study concept and design, data acquisition, critical revision of the manuscript
FINANCIAL DISCLOSURE/CONFLICT OF INTEREST
Stefanie T. Jost reports no financial disclosures.
K. Ray Chaudhuri has received funding from Parkinson’s UK, NIHR, UCB, and the European Union; he received honoraria from UCB, Abbott, Britannia, US Worldmeds, and Otsuka Pharmaceuticals; and acted as a consultant for AbbVie, UCB, and Britannia.
Keyoumars Ashkan has received honoraria for educational meetings, travel and consultancy from Medtronic, St Jude Medical and Boston Scientific.
Philipp A. Loehrer was funded by the SUCCESS-Program of the University of Marburg, the Parkinson’s Foundation, and the Stiftung zur Förderung junger Neurowissenschaftler.
Monty Silverdale has received honoraria from Bial, Britannia and Medtronic.
Alexandra Rizos has received honorarium from UCB and was supported by a grant from Medtronic.
Julian Evans reports no financial disclosures.
Jan Niklas Petry-Schmelzer has received travel grants from Boston Scientific.
Michael T. Barbe. received speaker’s honoraria from Medtronic, Boston Scientific, Abbott (formerly St. Jude), GE Medical, UCB, Apothekerverband Köln e.V. and Bial as well as research funding from the Felgenhauer-Stiftung, Forschungspool Klinische Studien (University of Cologne), Horizon 2020 (Gondola), Medtronic (ODIS), and Boston Scientific and advisory honoraria for the IQWIG.
Anna Sauerbier reports no financial disclosures.
Gereon R. Fink reports no financial disclosures.
Veerle Visser-Vandewalle is a member of the advisory boards and reports consultancies for Medtronic, Boston Scientific and St. Jude Medical. She received a grant from SAPIENS Steering Brain Stimulation.
Angelo Antonini reports personal consultancy fees from Zambon, AbbVie, Boehringer Ingelheim, GE, Neuroderm, Biogen, Bial, EVER Neuro Pharma, Therevance, Vectura grants from Chiesi Pharmaceuticals, Lundbeck, Horizon 2020 - PD_Pal Grant 825785, Ministry of Education University and Research (MIUR) Grant ARS01_01081, Cariparo Foundation, owns Patent WO2015110261-A1, owns shares from PD Neurotechnology Limited.
Pablo Martinez-Martin has received honoraria from Editorial Viguera and Movement Disorder Society for lecturing in courses; from AbbVie for speaking in experts’ meetings and from AbbVie and Zambon for participating in the Advisory Board of epidemiological studies. License fee payments for the King’s Parkinson’s Disease Pain Scale, and grants from the International Parkinson and Movement Disorder Society for development and validation of the MDS-Non-Motor Symptoms Scale.
Lars Timmermann reports grants, personal fees and non-financial support from SAPIENS Steering Brain Stimulation, Medtronic, Boston Scientific and St. Jude Medical.
Haidar S. Dafsari’s work was funded by the Prof. Klaus Thiemann Foundation and the Felgenhauer Foundation and has received honoraria by Boston Scientific and Medtronic.
This paper is independent research funded by the German Research Foundation (Grant KFO 219).
