Abstract
Still little is known about the nature of the gastrointestinal pathological alterations occurring in Parkinson’s disease (PD). Here, we used multiplexed mRNA profiling to measure the expression of a panel of 770 genes related to neuropathological processes in deep submucosal rectal biopsies of PD patients and healthy controls. Altered enteric neuropathological traits based on the expression of 22 genes related to neuroglial and mitochondrial functions, vesicle trafficking and inflammation was observed in 9 out of 12 PD patients in comparison to healthy controls. These results provide new evidences that intestinal neuropathological alterations may occur in a large proportion of PD patients.
Keywords
INTRODUCTION
Parkinson’s disease (PD) is a major neurodegenerative disease worldwide. It has become clear over the last decades that neuroinflammation plays a central role in PD pathology. Neuroinflammatory mechanisms appear to be linked to multiple biological pro-cesses in PD, encompassing altered trafficking of α-synuclein or generation of oxidative mediators, which may ultimately contribute to the spreading of the pathology and to the degeneration of dopamine neurons in the substantia nigra of PD patients [1].
Gastrointestinal (GI) disorders constitute one of the main early, non-motor, symptoms in PD with up to 60–90% of PD patients presenting with GI symptoms. Accordingly, the study of the GI tract in PD has gained a particular focus in the recent years and the GI tract has even been proposed to serve as a potential entry route for the disease, although the initiation and spreading of the pathology may occur through multiple ways [2, 3].
Recent works have revealed that neuroinflammatory manifestations in PD are not limited to the central nervous system but are also observed in GI tissues and may contribute to the initiation and progression of the disease [4, 5]. For instance, altered α-synuclein expression patterns [5, 6], increased expression of inflammatory cytokines, defects of the intestinal barrier (“leaky gut”), enteric gliosis and altered enteric neurochemical coding have been observed in the gut of PD patients [5, 8]. However, it remains un-clear whether such alterations may constitute a specific hallmark of PD. Here, we aimed to further characterize and determine the specificity of enteric neuropathological traits in PD using digital multiplex expression profiling.
MATERIAL AND METHODS
Biopsy acquisition was performed as previously described [6]. Patients collective includes 12 healthy controls (age range: 45–82 y; mean age: 65.2 y; male: 3 / female: 9) undergoing colonoscopy for col-orectal cancer screening and 12 patients with PD diagnosed according to the official criteria [9] (age range: 43–77 y; mean age: 64.9 y; male: 9 / female: 3). The study has received approval from the Local Ethics Committee of the Faculty of Medicine, Chr-istian-Albrechts-University of Kiel, Germany (D455/10; B299/07). For each patient or control, deep sub-mucosal biopsies from the upper dorsal rectal wall were retrieved. Constipation (determined by Wex-ner’s score [10]), motor symptoms (determined by UPDRS-III score [11]), L-Dopa equivalent daily dose and disease duration were determined for each patient (Supplementary Table 1).
For each biopsy, total RNA was extracted and expression of 770 genes related to neuropathological diseases and neuroinflammatory pathways, as well as internal control genes, was quantified using digital multiplex expression profiling. Correlation between gene expression and clinical parameters was further evaluated. Fluorescence immunohistochemistry for characterization of candidate genes was performed on human colonic specimens from patients who underwent partial colectomy for non-obstructive colorectal carcinoma as previously described [12]. Detailed procedures are described in supplementary methods.
RESULTS
Expression of 184 genes related to neuropathological pathways was altered in PD patients compared to controls (p < 0.05, Supplementary Table 3), with 22 candidate genes remaining significant after correction for multiple testing (FDR p < 0.05, fold change ≥ ±1.4, Table 1 and Supplementary Table 3). Particularly, pathways involved in neuro-glial functions, mitochondrial functions, vesicle trafficking, and regulation of gene expression/splicing showed significant differences in PD patients compared to controls as determined by analysis of similarities (ANOSIM, data not shown).
Candidate genes showing altered gene expression in rectal biopsies of Parkinson’s disease (PD) patients in comparison to controls as determined using nCounter analysis
Discrimination between PD patients and controls was achieved based on the gene expression profile of the 22 genes that remained significant after correction for multiple testing (FDR-adjusted p < 0.05, Table 1 and Supplementary Table 3) as shown by non-metric dimensional scaling analysis (NMDS, ANOSIM, p < 0.002) (Fig. 1A). An altered expression pattern for these 22 genes was observed in 9 out of 12 PD patients, as visualized using hierarchical clustering (Fig. 1B).
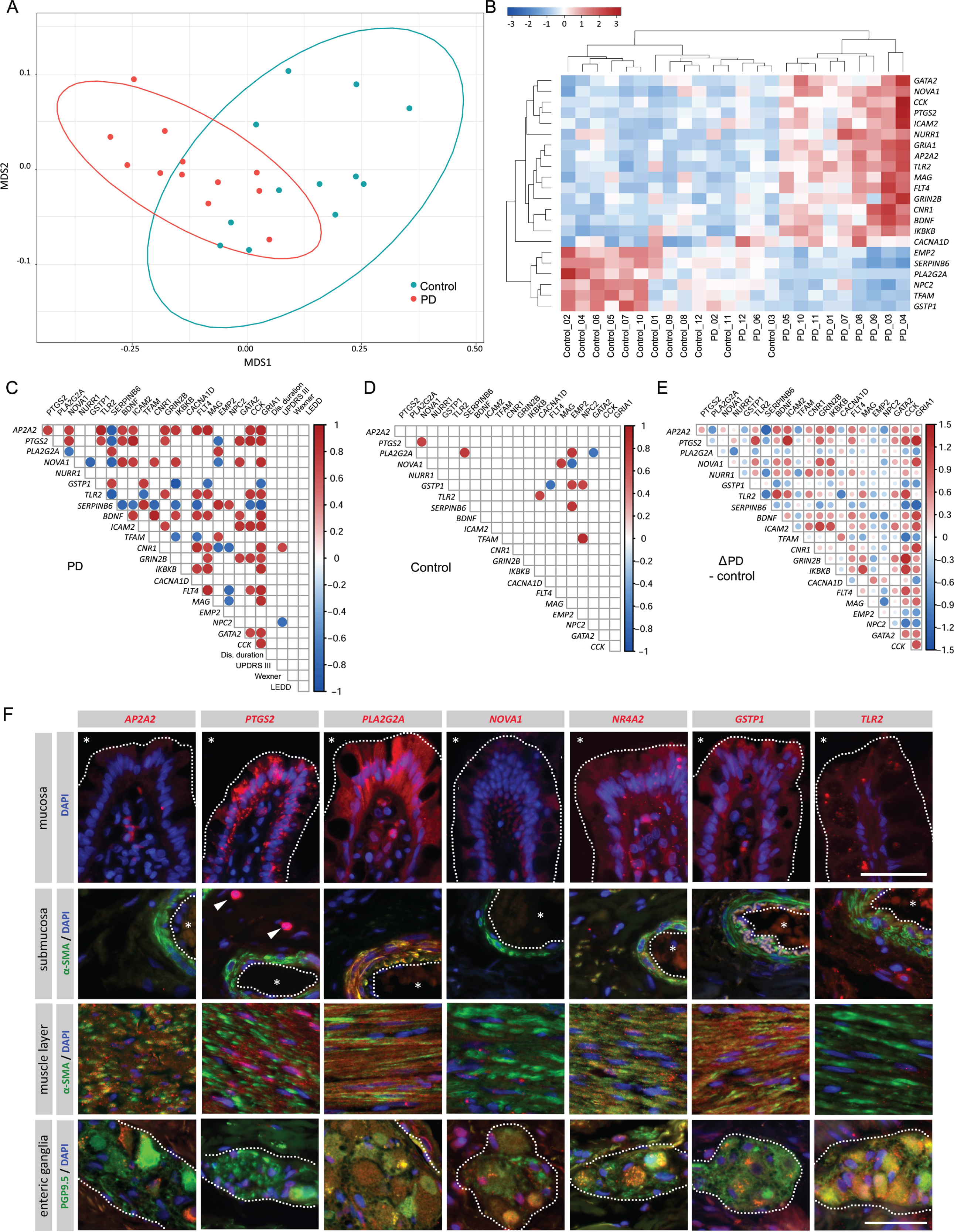
A) Non-metric dimensional scaling profiles based on the expression of the 22 most significantly altered genes (FDR p < 0.05) were determined for Parkinson’s disease (PD) patients (red spots) and controls (blue spots). The 90% confidence ellipses circumscribe the centroids of PD (red line) and control group (blue line). B) Hierarchical clustering for these 22 genes showed a global altered expression in 9 out of 12 PD patients. The scale indicates relative fold change variations: red, increase; blue, decrease. C–E) Correlation matrix of gene expression levels for the top 22 genes assessed in colonoscopic biopsy samples in PD patients (C, additionally showing correlation to clinical scores) and controls (D). Pearson’s correlation coefficients are represented as blue (negative correlation) and red circles (positive correlation) when significant (p < 0.01). Contrast matrix (E) indicating differences in correlation between controls and PD patients (C minus D) highlights the differential expression pattern in PD (blue, decreased correlation; red, increased correlation). Dis. duration: disease duration; UPDRS III: MDS-UPDRS III score; Wexner: Wexner’s score; LEDD: L-Dopa equivalent daily dose. F) Fluorescence immunohistochemistry for AP2A2, PTGS2, PLA2G2A, NOVA1, NR4A2, GSTP1, TLR2 and co-staining with the neuronal marker PGP9.5 or the smooth muscle marker α-SMA in the mucosal (epithelial border delimited by dashed line, *: intestinal lumen), submucosal (blood vessels delimited by dashed line, *:vessels lumen, arrowheads: interstitial immune cells), muscular layer and in enteric ganglia (delimited by dashed line) of human control colonic tissue, as indicated. Cell nuclei were counterstained with DAPI (blue). Scale bar = 50μm.
We then analyzed the differential co-expression of these 22 candidate genes. In controls, gene expression levels showed only few significant correlations (Fig. 1D), whereas in PD patients correlations were far more abundant (Fig. 1C) and showed several substantial directional differences compared to controls (Fig. 1E). A positive correlation was observed between CNR1 expression and motor symptoms (determined by MDS-UPDRS-III score), whereas expression of NPC2 was inversely correlated to UPDRS-III score (Fig. 1C). No further significant correlations between gene expression and constipation scores, UPDRS-III score or disease duration were observed (Fig. 1C). Immunohistochemistry was performed to determine the site-specific expression of selected significantly altered candidate genes in the human colon (AP2A2, PTGS2, PLA2G2A, NR4A2, NOVA1, GSTP1, and TLR2;Fig. 1F). The observed expression patterns support a potential functional involvement of these genes in the intestinal mucosa (PLA2G2A, PTGS2, NR4A2, TLR2), the intestinal smooth musculature (PLA2G2A, AP2A2, NR4A2, GSTP1), as well as in the enteric nervous system (PLA2G2A, NOVA1, AP2A2, PTGS2, NR4A2, GSTP1, TLR2;Fig. 1F, Supplementary Table 2).
DISCUSSION
The identified candidate genes mostly reflect and map to those pathologic pathways which have been previously associated with PD pathology. In-deed, increased expression of the cyclooxygenase 2 encoded by PTGS2 has been previously reported in colonic biopsies of PD patients and regulates the inflammatory state in PD [13, 14]. NR4A2 (also called NURR1) and TFAM both contribute to mitochondria-mediated neuroinflammation in PD [15, 16], although mitochondria functionality is not altered in enteric neurons of PD patients [17]. BDNF is an important neurotrophic factor which supports the development and maintenance of dopaminergic neurons [4, 18]. TLR2 expression is increased in the brain of PD patients where it contributes to the maintenance of a neuroinflammatory state [19] and is an important mediator of host-microbiota interactions [4]. Noteworthy, expression of TLR4 was slightly in-creased in biopsies of PD patients in this cohort, although this up-regulation did not reach statistical significance (Supplementary Table 3). Interestingly, further dysregulation of genes involved in host-int-estinal microenvironment interactions such as GSTP1 was also observed in our cohort, supporting the hypothesis that these pathways may play an important role in PD. Interestingly, CNR1 and NPC2 expression were respectively positively and negatively correlated to the UPDRS-III score in our cohort. AAT repeat polymorphism in the CNR1 locus is correlated to the occurrence of depression in PD patients [21]. NPC2 may contribute to intracellular α-synuclein trafficking [22]. However, how both genes may relate to motor symptoms and UPDRS III score in PD remains largely unknown.
While to our knowledge no association between PLA2G2A, NOVA1 and AP2A2 and PD has been identified so far, these genes have been previously linked to neurodegenerative disorders [23, 24] and to the regulation of brain functions [25] (Supplementary Table 3). Most particularly, AP2A2 interacts with and regulates α-synuclein trafficking in neurons [26] and may thus be involved in the tight regulation linking inflammation and α-synuclein life-cycle in PD [27, 28]. Immunohistological characterization confirms the potential involvement of these different candidates in the ENS. However, due to the broad expression pattern of these proteins (Supplementary Table 2) it remains to be determined which cellular components are primarily affected in PD.
Due to the rather small sample size, the present findings may be biased by heterogeneous patients’ characteristics, e.g., medication. In particular, a potential effect of unbalanced gender distribution between PD and control groups cannot be excluded in the present study. Other factors, such as body mass index or rapid-eye-movement sleep behavior disorders also have an influence on intestinal motility disorders and PD pathology [29, 30]. Whether altered enteric neuropathological traits are correlated to these parameters could not be assessed yet but should be addressed in future studies. Although data were normalized to five different housekeeping genes, cellular population heterogeneity between biopsies may similarly impacts the present results and further research is required to refine and confirm the specificity of the observed alterations in larger cohorts. Despite these limitations, these results support recent studies suggesting that common pathological pathways, including alterations of vesicle trafficking, mitochondrial and oxidative stress, as well as neuroinflammation, are affected along the gut-brain axis in a large subset of PD patients [4, 5]. These results are also in line with the increased expression levels of pro-inflammatory mediators observed in feces of PD patients [31, 32]. Moreover, this data further suggests that enteric neuropathological alterations may not represent a strict hallmark of PD, but may rather be indicative of individual PD patients’ clinical history and/or status. Whereas enteric neuronal cell numbers [6, 33] and functionality [17] appear normal in PD patients, our study supports recent hypotheses on the potential multifocal and systemic inflammatory progression of PD, in which a small proportion of PD patients may present without GI symptoms [2, 3]. To conclude, our observations suggest that characterization of enteric neuropathological traits using multiplexed gene expression analyses may help to better understand PD-related GI-disorders and their relevance in PD pathophysiology. Such approaches may facilitate the development of novel diagnostic tools aiming at stratifying subgroups amongst PD patients and ultimately define innovative therapeutic targets for the disease.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
The authors thank Gaby Steinkamp, Heike Kuzan, and Inka Geurink for expert technical assistance, as well as Dr. Sabine Jedtberg (UKSH, Kiel) for the helpful discussions during the elaboration of the manuscript. This study has been funded by grants from NanoString Technologies (Seattle, USA) to FC, the Faculty of Medicine, Kiel University (F359909) to FC and NGM, the Family Mehdorn Foundation to FC, and the German Research Society (Deutsche Forschungsgemeinschaft, DFG WE 2366/4-3) to TW.
