Abstract
Background:
Orthostatic hypotension (OH) at an early stage of Parkinson’s disease (PD) predicts poor prognosis, which may suggest degeneration of dopaminergic neurons affects sympathetic function, causing OH.
Objective:
We tested the hypothesis that striatal dopaminergic depletion is associated with OH in PD.
Methods:
Out of 99 patients with newly diagnosed untreated PD, 81 patients were enrolled according to our selection criteria. All patients underwent head-up tilt-table testing and striatal 123I-2β-carbomethoxy-3β-(4-iodophenyl)-N-(3-fluoropropyl) nortropane (123I-FP-CIT) dopamine transporter single photon emission computed tomography (DAT-SPECT). DaTQUANT software (GE Healthcare) was used as a semi-quantitative tool to analyze DAT-SPECT data. The association between hemodynamic changes and 123I-FP-CIT uptake was examined.
Results:
123I-FP-CIT uptake in the putamen, especially the anterior part and left side, was related not only to motor severity but also to OH. Change in systolic blood pressure correlated negatively with 123I-FP-CIT uptake in bilateral anterior putamen (left: p < 0.01, right: p < 0.05) and left posterior putamen (p < 0.05). Patients with OH had more severe dopamine depletion in left anterior (p = 0.008) and posterior (p = 0.007) putamen at a similar motor severity than did patients without OH even though both groups have similar baseline characteristics. An analysis of asymmetry index showed patients with OH had symmetrically decreased dopamine levels in anterior putamen when compared to those without OH (p = 0.024).
Conclusion:
OH is closely related to striatal dopamine depletion in PD. This relation may help to account for the prognostic value of OH.
Keywords
INTRODUCTION
Symptoms of Parkinson’s disease (PD) include not only motor dysfunction but also non-motor symptoms. Sympathetic cardiovascular failure such as orthostatic hypotension (OH) is clinically important because it is present at the pre-motor phase and associated with future declines in motor and cognitive function and mortality [1–4]. Nigrostriatal denervation reflected in 123I-ioflupane dopamine transporter single photo emission computed tomography (DAT-SPECT) is also present at pre-motor phase in susceptible individuals who later develop PD [5]. In addition, a recent clustering-derived subtyping study has identified OH at an early stage of disease as a risk for rapidly progressive subtype (also named “diffuse malignant”) [6]. This finding suggests patients with OH would more likely have widespread alpha-synuclein involvement of both dopaminergic and sympathetic noradrenergic structures. However, it may also suggest that degeneration of dopaminergic neurons affects sympathetic function, causing OH. Direct evidence supporting this hypothesis is lacking.
Striatal dopamine depletion, particularly in anterior putamen, at an early stage of disease is associated with a higher risk for motor complication and dementia conversion [7]. We wondered if patients with OH already have dopamine depletion in such vulnerable striatal subregions at an early stage of PD.
We hypothesized that change in hemodynamics during orthostatic stress is associated with dopaminergic depletion in specific striatal subregions. We therefore investigated the relation between OH and striatal DAT uptake.
METHODS
Patients
A total of 99 patients with newly diagnosed un-treated PD were entered into a research database at The Jikei University School of Medicine from February 2014 through April 2019. PD was diagnosed according to the clinical diagnostic criteria of the UK Parkinson’s Disease Society Brain Bank [8], and we confirmed the presence of a PD drug response for longer than 1 year during the follow-up period. The clinical diagnosis was also supported by DAT-SPECT using 123I-2β-carbomethoxy-3β-(4-iodophenyl)-N-(3-fluoropropyl)nortropane (123I-FP-CIT), which revealed that all patients had decreased dopamine transporter uptake in the striatum, mainly in the posterior putamen on visual analysis.
None of the patients had received levodopa, other anti-Parkinson drugs, or treatment for orthostatic hypotension (OH). The motor severity of PD was assessed according to the Unified Parkinson’s Dise-ase Rating Scale (UPDRS) part III scores. The pat-ients were divided into tremor-dominant type (TDT), akinetic-rigid type (ART), and mixed type (MXT) subgroups on the basis of the tremor and non-tremor scores, estimated with the use of part III of the UPDRS, as described by Spiegel and colleagues [9]. If patients were already receiving antihypertensive dr-ugs, such drugs were withdrawn at least 48 hours before orthostatic hypotension was evaluated. All pat-ients underwent evaluation of their olfactory function and Mini-Mental State Examination (MMSE). Olfactory function was assessed by the odor stick ide-ntification test for the Japanese (OSIT-J) (Daiichi Yakuhin Sangyo Co. Ltd., Tokyo, Japan) according to our previous report [10]. Symptom duration was measured from the onset of one of four hallmark symptoms of PD. This study was approved by the Ethics Committee of Jikei University School of Medicine, and all subjects gave written informed consent before enrollment.
We excluded 18 patients because they met any of the following exclusion criteria: 1) Patients who had lesions including atrophy on brain magnetic res-onance imaging of the striatum, brainstem, or cerebellum. 2) Patients with overt diabetes, cirrhosis, end-stage renal failure, anemia, dehydration on blood tests and relevant cardiac disease or any abnormalities on routine chest radiography or electrocardiography. 3) Patients with cognitive dysfunction as indicated by MMSE score less than 24. 4) Patients who were receiving medications known to interact with the striatal binding of 123I-FP-CIT [11] and to affect autonomic function, such as beta-blockers and anticholinergic agents. 5) Patients aged less than 50 years and over 85 years.
Consequently, we retrospectively analyzed data from 81 patients with newly diagnosed untreated PD.
123I-FP-CIT SPECT
SPECT was performed using a dual-head scintillation camera (Discovery NM 630; GE Healthcare) with a low energy-high resolution collimator, 4 h after a bolus injection of 167 MBq of 123I-FP-CIT (DaTSCAN; Mediphysics, Tokyo, Japan). The imaging time was 32 min. The parameters of acquisition were as follows: rotation radius 15 cm, circular orbit; zoom 1.33; matrix size 128×128 pixels, pixel size 3.32 mm2; acquisition time 4 rotations, 8 m per rotation, 120 projections per rotation; and energy window 159 KeV±10%. Data were reconstructed by the ordered-subset expectation maximization (OSEM) method (iteration: 2, subset: 10) using a Butterworth filter setting with a critical frequency of 0.7 and a power of 10.0. Semi-quantitative calculations of str-iatal uptake were performed with the use of DaT-QUANT software (GE Healthcare). This software package calculated the patient’s uptake automatically. The volumes of interest (VOIs) of DaTQUANT fixed-size were semi-automatically placed over the right and left putamen and caudate nucleus in the transaxial slice showing the most intense tracer uptake and adjusted manually if necessary. Another VOI was placed over the occipital lobe representing cortical background. The software automatically placed the same VOIs in the adjacent previous and following slices so that data from three contiguous slices were used. Patient’s uptake was calculated for the posterior putamen (PP), anterior putamen (AP), and caudate nucleus (CA) by dividing the mean counts per pixel in the corresponding VOI by the mean count per pixel in the occipital lobe VOI. The software also pr-ovided a percent deviation of patient’s uptake from the age-matched, mean value of the normal database (118 healthy volunteers) that the patient’s measured value represents [12, 13]. Asymmetry index for the PP, AP, and CA was calculated according to the formula: [(a-b)/(a+b)]×2×100, where a and b represent the two different side [14].
Head-up tilt-table testing (HUT)
All subjects underwent HUT in a silent room, maintained at an ambient temperature of 23°C to 26°C. After an overnight fast, the subjects comme-nced the study between 9:00 am and 10:00 am. After 20 min of rest in the supine position, the subject was tilted to a 60° upright position within 15 s by means of a head-up tilt table. Brachial systolic and diastolic blood pressures were meaured with an automated sphygmomanometer after 20 min of rest in the supine position (SBPsupine, DBPsupine) and every 1 min after the subject was tilted for up to 10 min. The maximum falls in systolic blood pressure (⊿SBP), diastolic blood pressure (⊿DBP) and mean arterial pressure (MAP) defined as (SBP+2 x DBP)/3 in a tilt-up position were evaluated. Supine hypertension was defined as SBPsupine≥140 mmHg and/or DBPsupine≥90 mmHg. OH was defined as ⊿SBP≥20 mmHg and/or ⊿DBP≥10 mmHg in patients without supine hypertension, or as ⊿SBP≥30 mmHg in patients with supine hypertension [15–17].
Cardiac MIBG scintigraphy
To evaluate cardiac sympathetic denervation, cardiac MIBG scintigraphy was performed. The ratio of the average pixel count in the heart (H) to that in the mediastinum (M) (H/M ratio) 15 min (early) and 3 hours after (delayed) of injection of 111 MBq 123I-MIBG (FUJIFILM RI Pharma Co., Ltd. Tokyo, Japan) was calculated. The lower limit of normal ratio is 2.3 for early H/M and 2.1 for delayed H/M [18].
Statistical analyses
Statistical analyses were performed using a statistical data analysis system (Esumi Co., Ltd., Tokyo, Japan). One-way analyses of variance (with Bonferroni post hoc testing) were used to compare groups, and χ2 tests were used to compare frequencies for categorical variables. Relations between sympathe-tic cardiovascular variables and 123I-FP-CIT uptake were tested by Pearson product-moment. Associations of sympathetic cardiovascular variables with 123I-FP-CIT uptake were also examined by multiple regression analysis or analysis of covariance with adjustment for covariables. p values of < 0.05 were considered to indicate statistical significance.
RESULTS
Baseline results
Among the 81 patients, 43 were men (53.1 %). Mean age (±SD) and symptom duration were 70.6±8.9 years and 1.5±1.5 years, respectively. UPDRS-III scores averaged 20.0±9.7. Sixty-one (75.3%) patients had ART PD. OSIT-J scores and MMSE averaged 4.8±2.8 and 28.4±1.9, respectively. Thirty-five patients had hypertension and were receiving antihypertensive drugs. Thirty-three patients had OH. Of these patients, 28 patients had OH within 3 min of upright tilt-table testing (classic-al OH) and 5 patients had OH beyond 3 min (delayed OH).
Clinical characteristics of patients with and without OH were summarized in Table 1. Age, sex, sym-ptom duration, motor subtype and MMSE in the two groups did not differ statistically. The two groups had similar UPDRS-III scores. Olfactory function and cardiac uptake of MIBG tended to be lower in patients with OH than in those without OH [olfactory function: 4.1±2.8 vs. 5.2±2.8, p = 0.108, cardiac uptake of MIBG (delayed): 1.35±0.33 vs. 1.48±0.41, p = 0.062].
Comparison of clinical characteristics between PD patients with and without OH a
PD, Parkinson’s disease; OH, orthostatic hypotension; UPDRS-III, Unified Parkinson’s Disease Rating Scale part III; HT, hypertension; NGSP, National Glycohemoglobin Standardization Program, BMI, body mass index; ART, akinetic-rigid type; TDT, tremor-dominant type; MT, mixed type; OSIT-J, odor stick identification test for the Japanese; MMSE, Mini-Mental State Examination; SBP, systolic blood pressure; DBP, diastolic blood pressure; ⊿SBP, difference in systolic blood pressure between two positions; ⊿DBP, difference in diastolic blood pressure between two positions; MAP, mean arterial pressure; MIBG, 123I-metaiodobenzylguanidine; H/M, heart-to-mediastinum ratio. †Analyses were performed by analysis of covariance (ANCOVA) test controlling for age, sex, symptom duration, motor subtype, UPDRS-III scores and cardiac uptake of MIBG(delayed).
Differences in striatal 123I-FP-CIT uptake between patients with and without OH
The comparison of striatal 123I-FP-CIT uptake between patients with and without OH were also summarized in Table 1. Patients with OH had significantly lower 123I-FP-CIT uptake in left PP and AP than did those without OH after adjusting for age, sex, symptom duration, motor subtype, UPDRS-III score and cardiac uptake of MIBG (delayed) (Patient’s uptake: left PP; p = 0.007, left AP; p = 0.008, Percent deviation: left PP; p = 0.002, left AP; p = 0.002).
Correlations of 123I-FP-CIT uptake in striatal subregions with hemodynamic changes during HUT
There was no correlation between supine blood pressure and 123I-FP-CIT uptake in any striatal subregions (Table 2). In contrast, there was a significant correlation between the degree of OH and 123I-FP-CIT striatal uptake.
No correlation of striatal 123I-FP-CIT uptake with supine blood pressure
123I-FP-CIT, 123I-2β-carbomethoxy-3β-(4-iodophenyl)-N-(3-fluoropropyl)nortropane; SBPsupine, systolic blood pressure after 20 min of rest in the supine position; DBPsupine, diastolic blood pressure after 20 min of rest in the supine position; PP, posterior putamen, AP, anterior putamen; CA, caudate. *This value is percentage of deviation from age-matched, mean value of normal database from the Parkinson Progression Makers Initiative. c p > 0.05. † p value after adjusted for age, sex, symptom duration, motor subtype, UPDRS-III scores and cardiac uptake of MIBG (delayed).
Correlations of 123I-FP-CIT uptake in striatal subregions with the changes in blood pressure during HUT are summarized in Table 3. 123I-FP-CIT uptake in the putamen, especially anterior part and the left side, was negatively associated with ⊿SBP (Patient’s uptake: left PP; p = 0.013, left AP; p = 0.009, right AP; p = 0.048, Percent deviation: left PP; p = 0.006, left AP; p = 0.006, right AP; p =0.041) and ⊿DBP (Patient’s uptake: left PP; p =0.017, left AP; p = 0.007, Percent deviation: left PP; p = 0.022, left AP; p = 0.013), and positively associated with MAP in a tilt-up position (Patient’s uptake: left PP; p = 0.031, left AP; p = 0.015, right PP; p = 0.037, right AP; p = 0.032, Percent deviation: left PP; p = 0.028, left AP; p = 0.014, right PP; p = 0.041, right AP; p = 0.043) after adjustment for age, sex, symptom duration, motor subtype, UPDRS-III scores and cardiac uptake of MIBG (delayed) on multiple regression analysis. Multiple regression an-alysis also revealed that 123I-FP-CIT uptake in all of these regions was associated with UPDRS-III scores as well as hemodynamic changes [Left PP: UPDRS-III scores; β=–0.255, p = 0.040, ⊿SBP; β=–0.285, p = 0.013, R2 = 0.169, Left AP: UPDRS-III scores; β=–0.285, p = 0.015, ⊿SBP; β=–0.279, p = 0.009, R2 = 0.270 (Fig. 1), Right AP: UPDRS-III scores; β=–0.269, p = 0.018, ⊿SBP; β=–0.206, p = 0.048, R2 = 0.309].
Correlation of striatal 123I-FP-CIT uptake with change in hemodynamics during head-up tilt-table testing
123I-FP-CIT, 123I-2β-carbomethoxy-3β-(4-iodophenyl)-N-(3-fluoropropyl)nortropane; ⊿SBP, difference in systolic blood pressure between two positions; ⊿DBP, difference in diastolic blood pressure between two positions; MAP, mean arterial pressure; PP, posterior putamen, AP, anterior putamen; CA, caudate. *This value is percentage of deviation from age-matched, mean value of normal database from the Parkinson Progression Makers Initiative. †p value after adjusted for age, sex, symptom duration, motor subtype, UPDRS-III scores and cardiac uptake of MIBG(delayed). ap < 0.05; bp < 0.01.
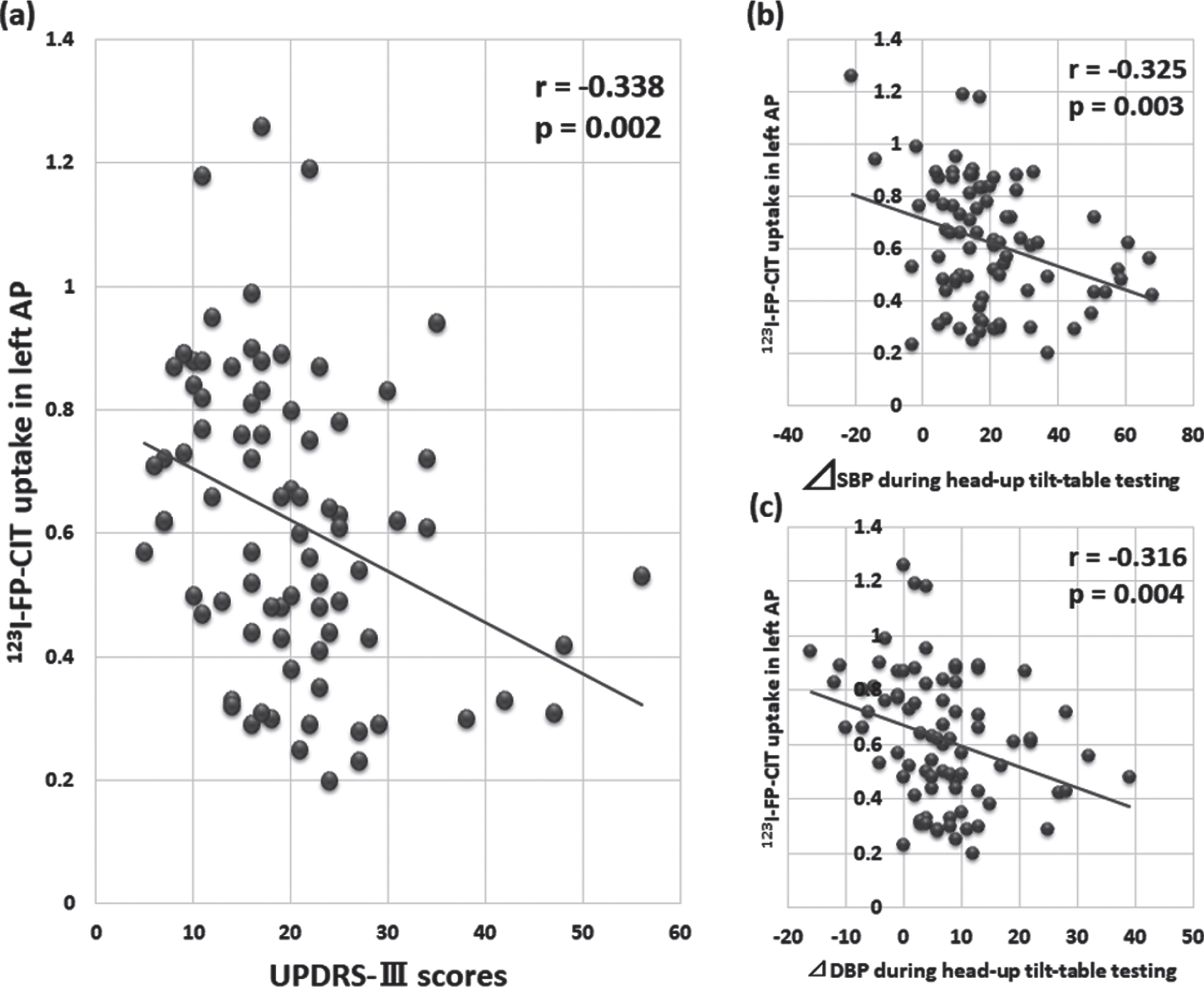
123I-FP-CIT uptake in left anterior putamen correlated negatively with UPDRS-III scores (a), fall in systolic blood pressure (b) and that in diastolic blood pressure (c).
Analysis of covariance showed that patients with OH had more severe dopamine depletion in the left PP and AP at a similar UPDRS-III scores than did patients without OH after adjusting post hoc for age, sex, symptom duration, motor subtype, UPDRS-III score and cardiac uptake of MIBG [Patients’ uptake: left PP; p = 0.007, left AP; p = 0.008, Percent deviation; left PP; p = 0.002, left AP; p = 0.002 (Fig. 2a)]. Dopamine depletion in the right AP was marginally significant [Patients’ uptake: p = 0.064, Percent deviation; p = 0.055 (Fig. 2b)].
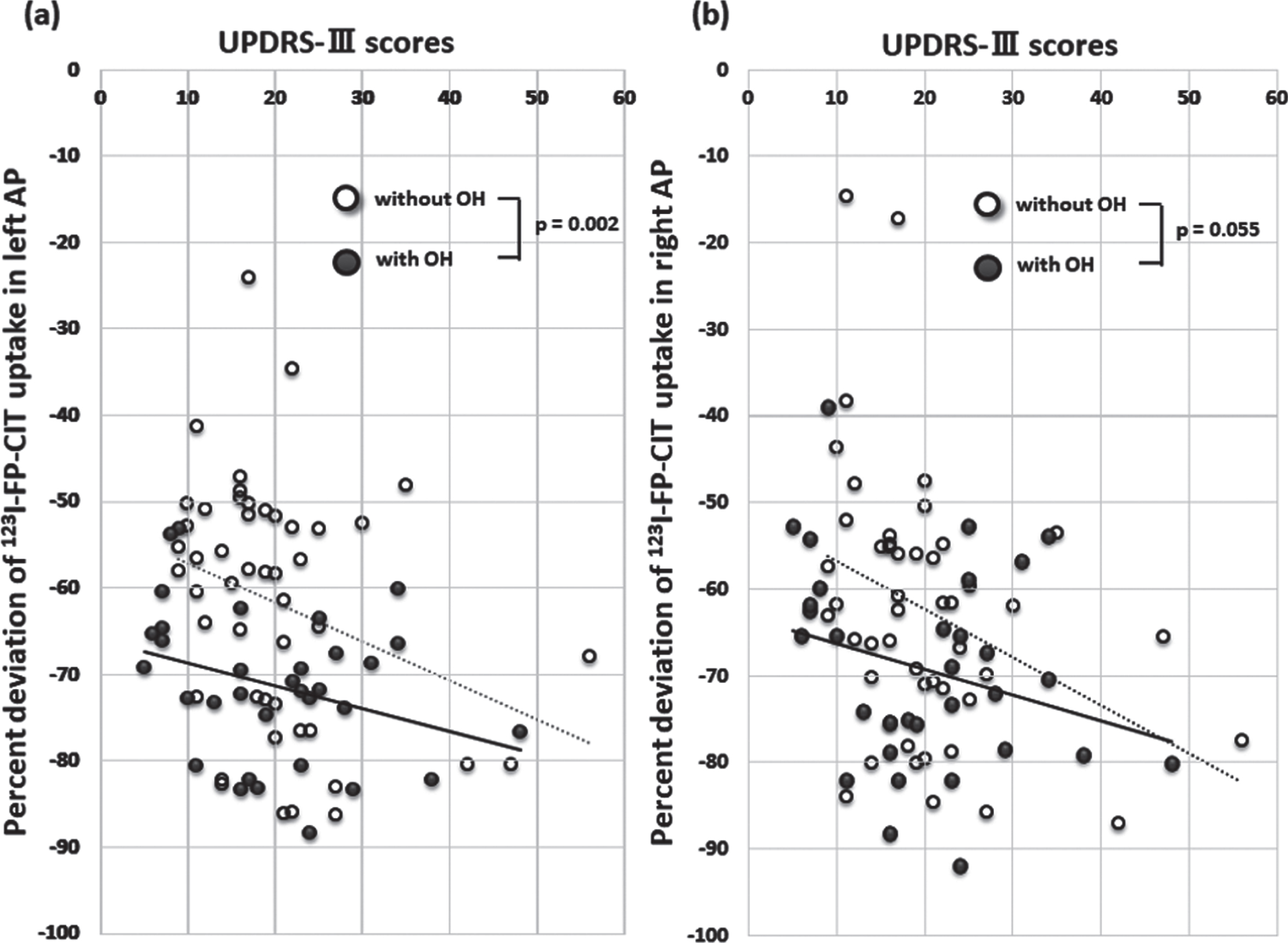
Patients with OH had a significantly lower 123I-FP-CIT SPECT uptake in left (a) and a marginally significant lower uptake in the right (b) anterior putamen at a similar UPDRS motor score than did patients without OH after controlling for age, sex, symptom duration, motor subtype, UPDRS-III scores and cardiac uptake of MIBG. a solid line, patients with OH; a dotted line, patients without OH.
However, the interaction effect between the presence of OH and UPDRS-III scores on 123I-FP-CIT uptake or percent deviation in left PP and bilateral AP was not significant (left PP and bilateral AP: p > 0.05).
Effect of OH on asymmetry index in striatal subregions
The results were shown in Table 4. Asymmetry index of patient’s uptake in PP, AP and CA was not significantly different between patient with and without OH. However, AP asymmetry index of percent deviation from age-matched, mean value of normal database was significantly decreased in patients with OH than in those without OH after adjusted for age, sex, symptom duration, motor subtype, UPDRS-III score and cardiac uptake of MIBG (null p < 0.001, adjusted p = 0.024).
The association of orthostatic hypotension with asymmetry index in striatal subregions
OH, orthostatic hypotension; PP, posterior putamen; AP, anterior putamen; CA, caudate. *This value is percentage of deviation from age-matched, mean value of normal database from the Parkinson Progression Makers Initiative. †Analyses were performed by analysis of covariance (ANCOVA) test controlling for age, sex, symptom duration, motor subtype, UPDRS-III scores and cardiac uptake of MIBG (delayed).
DISCUSSION
To the best of our knowledge, this study is the first to demonstrate an association between OH and striatal dopamine depletion in untreated early-stage patients with PD. Patients with OH had more severe dopamine depletion in left PP and AP at a similar UPDRS-III scores than did patients without OH even though two groups have similar baseline characteristics such as age, UPDRS-III scores. Dopaminergic depletion in the putamen, especially the anterior part and left side and, was closely related not only to motor severity but also to hemodynamic changes during ort-hostatic stress, whereas it was not associated with the degree of SH. It correlated negatively with cha-nges in blood pressure during orthostatic stress. In addition, an analysis of asymmetry index showed pat-ients with OH had symmetrically decreased dopa-mine levels in AP when compared to those without OH.
OH is attributed to combined failure or dysregulation of both central and peripheral parts of the autonomic nervous system. As for central regions, the degeneration of noradrenergic periacqueductal ductal grey neurons, rostral ventrolateral medulla, dorsolateral vagal motor nucleus and nucleus ambiguus is, in particular, associated with OH. These structures connect with the anterior cingulate cortex, insular cortex, amygdala, hypothalamus and thalamus, and control heart rate and cardiac contractility under orthostatic or emotional stress [19–22]. Increasing evidence also suggests that the putamen participates in autonomic activities along with other brain regions, such as the brainstem, accumbens, thalamus, amygdala, insula, and anterior cingulate cortex [23, 24]. One study reports that patients with OH had reduced brainstem functional connectivity to the putamen during Valsalva maneuvers [23], and another study reports the right and left putamen volume positively correlate with parasympathetic cardiac vagal tone [24]. These previous findings seem to support our findings that dopaminergic depletion in the putamen is associated with autonomic activity during orthostatic stress.
Recent studies have shown patients with PD exhibit an asymmetric nigrostriatal dysfunction [25]. Therefore, we examined whether the presence of OH contributes to this asymmetry of striatal dopamine depletion. Although patients with OH exhibited left-hemispheric predominance of dopamine depletion in the putamen, asymmetry index in PP was not different between patients with and without OH. On the contrary, asymmetry index in AP of percent deviation from age-matched, mean value of normal database was significantly decreased in patients with OH than in those without OH, suggesting patients with OH have a symmetrically decreased DAT availability in AP even at early stage of disease.
The AP is a part of associative striatum and is thought to be associated with cognition in patients with PD [27]. Although many studies have suggested dopamine loss in the CA as a candidate for future cognitive impairment, it has documented that selective dopamine loss in AP at early stage of PD is associated with a higher risk for dementia conversion and motor complication [7]. Therefore, symmetrically decreased dopamine level in AP in patients with OH seems to support the association between OH and diffuse malignant subtype of PD.
One possible explanation for why the AP is specifically affected is that this region is vulnerable to early deposition of amyloid-β (Aβ). A recent paper investigating DAT activity on dementia with Lewy bodies (DLB) with and without Aβ deposition, reported a lower DAT activity in AP and ventral striatum in the subgroup with Aβ deposition [26]. This result suggests that AP and ventral striatum are the most vulnerable regions and are involved early in amyloid deposition. DLB is one of Lewy body spectrum disorders having more severe autonomic failure and cognitive dysfunction than PD. Striatal pathological changes in PD with OH might be similar to those in DLB.
On the other hand, parkinsonism with OH at an early-stage of disease may suggest multiple system atrophy with predominant parkinsonism (MSA-P) as well as PD. We confirmed, therefore, all enrolled patients had a PD drug response for longer than 1 year, and all patients with PD and OH were hyposmic (OSIT-J score 4.1±2.8 is classified into severe hyposmia [10]), which is not generally observed in patients with MSA-P who have preserved olfactory function. It has documented OSIT-J score averages 8.7±2.2 in patients with MSA-P [28].
OH in PD often occurs beyond 3 min standing, which is clinically important because it could contribute to falls and increase morbidity [29]. We therefore examined hemodynamic changes after subject was tilted for up to 10 min. Decreased cardiac uptake of MIBG reflects cardiac sympathetic dysfunction [30] and is possibly involved in the pathophysiology of OH [31]. We include, therefore, its value in the independent factors to confirm significant association between OH and striatal dopamine depletion on multiple regression analysis.
The strength of this study is that we enrolled only patients with newly diagnosed, drug-naïve PD who had never taken any drugs affecting autonomic and dopaminergic function. We tried minimizing confounding biases by excluding patients with young and very old onset PD and squaring baseline characteristics. However, our study had several limitations: First, this study focused on one subject group. We evaluated, therefore, a percent deviation of patient’s uptake from age-matched, normal database provided by the DaTQUANT software. DaTQUANT software was created in 2013 as an adjunct processing tool with advanced quantitative uptake methods and a designated normal database from the Parkinson Progression Markers Initiative (PPMI), a comprehensive study of patients with diagnosed PD, to provide a more accurate assessment of a patient’s case. The software is over 95%accurate in automatic VOI placement, with the option of manual adjustments as needed. DaTQUANT can redirect any image automatically back to the optimal orientation of the anatomy within the images, which can remove the head-tilt artifact and is related to the potential problem of inaccurate VOI placement [12, 13]. Second, this was a relatively small study with a relatively homogeneous ethnic background. Further studies are needed in patients of different ethnic origins to be able to generalize to the overall population of patients with PD worldwide.
In conclusion, this is the first study to show that hemodynamic change during orthostatic stress is closely related to striatal dopamine depletion in pat-ients with PD. Our findings may deepen the knowledge on striatal role in the sympathetic pathway, underlying mechanisms and helping also to identify PD patients for whom the most rapid progression rate could be expected.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
Special thanks to radiological technologist Takashi Oshita for his time and dedication to obtain 123I-FP-CIT SPECT data.
