Abstract
Background:
Poor olfaction is a prodromal symptom of Parkinson’s disease (PD); however, self-reported sense of smell is often dismissed as unreliable.
Objective:
To assess self-reported and objectively assessed sense of smell, independently and jointly, in relation to future risk for PD.
Methods:
We conducted a prospective analysis using data from 2,424 participants, ages 71–82 at baseline, from the Health, Aging, and Body Composition study. Exposures were self-reported poor sense of smell or taste and the objectively measured 12-item Brief Smell Identification Test score. The outcome was incident PD, analyzed using Cox proportional hazard models adjusted for age, sex, race, and cognitive function.
Results:
After approximately 10 years of follow-up, both self-reported and objectively tested poor sense of smell were independently associated with a higher risk of developing PD: the hazard ratios (95% confidence interval) were 2.8 (1.3, 5.9) and 4.0 (2.1, 7.5), respectively. When analyzed jointly, compared with participants who reported and tested normal, the hazard ratio was 2.2 (1.0, 4.6) for those reported poor sense of smell but tested normal, 3.6 (1.9, 6.9) for reported normal but tested poor, and 7.8 (3.2, 19.4) for both reported and tested poor. We did not find significant interactions between self-reported and objectively tested sense of smell in predicting PD risk.
Conclusion:
This study provides preliminary evidence that self-reported poor sense of smell or taste should not be simply dismissed as useless in predicting risk of PD. Future studies should confirm our finding and evaluate whether structured questionnaires may further improve the predictability.
INTRODUCTION
Poor sense of smell is one of the earliest symptoms of Parkinson’s disease (PD) and an important part of the newly proposed research criteria for prodromal PD [1]. While in the general population, self-recognition occurs when symptoms are first noticed, subjective complaints about poor sense of smell are often dismissed as unreliable when compared with an objective olfaction test [2, 3]. However, this self-report may contain information that is difficult to capture with a single snapshot of olfaction test [4–7], for example perceived changes in sense of smell over years. To our knowledge, no study has prospectively assessed whether self-reported poor sense of smell predicts future PD risk, with or without an objective olfaction test. Therefore, we evaluated individual and joint effects of self-reported versus objectively assessed sense of smell in relation to PD risk in the well-established Health, Aging, and Body Composition (Health ABC) study.
MATERIALS AND METHODS
Study population
The Health ABC is a community-based biracial cohort that studies changes in body composition in the context of ageing, and their functional impacts on the health of older adults. Details of the study were published elsewhere [8]. Briefly, in 1997-1998, the study enrolled 3,075 community-dwelling subjects (48.4% men and 41.6% black), aged 70–79 years, from Pittsburgh, Pennsylvania, and Memphis, Tennessee. Eligibility criteria included no difficulty in walking a quarter mile, climbing 10 steps, or performing activities of daily living; no active cancer treatment in the previous 3 years; and no plan to move away from the study area in the next 3 years. Participants were followed with clinical exams or home visits in study years 1 to 6, 8, 10, 11, and 16. In addition, regular phone calls were made to update their contact and health status every 6 months until year 15 and then quarterly through year 17. The cohort evaluated the sense of smell of 2,544 participants at year 3 clinical exam (1999-2000, referred as baseline) with the 12-item Brief Smell Identification Test (BSIT) (known as Cross-Cultural Smell Identification Test, Sensonics, Haddon Heights, NJ, USA) [9]. After excluding participants with invalid BSIT score or missing in self-reported olfaction or taste (N = 51), missing PD status (N = 50), and all prevalent PD cases (N = 19), our analyses included 2,424 participants who were followed from baseline until the date of PD diagnosis, death, last contact, or August 31, 2012, whichever came first. The study protocol was approved by the institutional review boards at the University of Pittsburgh, the University of Tennessee, and the University of California–San Francisco. All participants signed informed consent at enrollment.
PD ascertainment
We previously published details of PD identification and adjudication in this cohort [10]. Briefly, we identified a total of 156 potential PD cases from the cohort’s health follow-up based on self-reported diagnosis at enrollment and year 13, medication usage details over time (e.g., drug name, dose, frequency, and reason for use), adjudicated causes for all hospitalizations during the follow-up, and adjudicated causes of death for deceased participants. Based on these data, we conducted a retrospective case adjudication in 2015. Two experienced movement disorder specialists independently reviewed all available data, accounting for each information source that indicated a PD diagnosis, consistencies across sources and over time, and evidence against PD diagnosis [10]. They finally adjudicated 81 PD diagnoses by consensus. After excluding 19 prevalent cases and 20 potential cases with missing sense of smell data, the final analysis included 42 incident PD cases.
Self-reported versus objectively tested sense of smell
In this cohort, olfaction was only assessed once at the year 3 clinical visit. Before taking the BSIT test, participants were asked “Do you suffer from smell and/or taste problems?” This question did not differentiate olfactory from taste deficit by design. In contrast to the sense of smell, taste perception refers specifically to the detection of modalities of sweet, salty, sour, bitter, umami, and lipids [11]. However, most people cannot effectively differentiate and report these two senses without objective testing, and what people perceive as taste is mostly flavor which is captured dominantly by the olfactory nerve as part of the sense of smell. Therefore, in this study, we considered answer to this question to reflect the self-perception of olfactory function.
Participants took the BSIT test immediately after answering this question. The BSIT is a short version of the 40-item Pennsylvania Smell Identification Test and has been widely used in clinical and epidemiologic studies [10, 12–14]. The test asks participants to scratch and smell 12 odorants experienced in daily life, one at a time, and to identify the correct odorant from four possible answers in a multiple-choice format. One point was given for each correct answer with a total score ranging from 0 to 12, with a higher score indicating a better sense of smell. We have previously reported in Health ABC study that this BSIT-assessed poor sense of smell was highly predictive of PD risk [10].
Statistical analysis
In the analysis, we defined both self-reported and BSIT-assessed sense of smell as dichotomous variables. For BSIT, a score of ≤8 was defined as poor sense of smell, consistent with the definition of prior studies [9, 14] and corresponding to about a third of our study participants. When self-reported and BSIT tested sense of smell were analyzed jointly, we used participants who both reported and tested normal as the reference for all other groups including self-reported poor but tested normal, self-reported normal but tested poor, and both self-reported and tested poor. We used Cox proportional hazards models for multivariate modeling and reported hazard ratios (HR) and 95% confidence intervals (CI), first adjusting for age, then adding sex and race, and finally additionally for cognitive function which was assessed using the modified Teng Mini-Mental State Exam. Further adjustment for other covariates such as smoking and self-reported health status barely changed risk estimates and we therefore presented results from simpler models to maintain statistical power. We further examined both multiplicative and additive interactions between self-reported and BSIT-tested olfaction using Cox models [15]. To further evaluate whether self-report improve BSIT-tested poor olfaction in predicting future PD diagnosis, we constructed receiver operating characteristic (ROC) curves using logistic regression models and tested group differences using Chi-square test. All statistical tests were two-sided with a significance level of 0.05.
RESULTS
Of the 2,424 eligible participants, 31% scored ≤8 on BSIT, but only 9.5% reported a poor sense of smell. In this older adult population, these two measures were only modestly correlated. When compared with BSIT performance, self-reported poor sense of smell had a low sensitivity (15%) but a high specificity (93%). The positive predictive value was only 49% as compared to a modest negative predicative value of 71%.
Overall, compared to participants with normal BSIT, those who performed worse on BSIT were more likely to be older, male, black race, low cognitive scores, current smokers, former alcohol drinking, to have a lower BMI, and to report lower education, less coffee drinking and fair-to-poor health status (Table 1). However, none of these factors were statistically associated with self-reported poor sense of smell.
Baseline population characteristics by objectively tested and self-reported olfactory impairment
*Age and 3MS are presented as mean (SD) and other covariates as frequency (percentage); P values were obtained from Chi-square test for sex, race, education, BMI, smoking status, coffee/alcohol drinking, health status and from two-sample t test for age and 3MS. BSIT, Brief smell identification test, score range 0–12. 3MS, Teng Mini-Mental State Exam, score range 0–100.
When analyzed independently in separate models, both BSIT-tested and self-reported poor sense of smell were strongly associated with PD risk (Table 2). After adjusting for age, sex, race, and cognition, the HR and 95% CI was 4.0 (2.1, 7.5) for tested poor olfaction and 2.8 (1.3, 5.9) for self-reports. When analyzed jointly, compared with participants who reported and tested normal sense of smell, the HR was 2.2 (1.0, 4.6) for those who reported poor but tested normal, 3.6 (1.9, 6.9) for those who reported normal but tested poor, and 7.8 (3.2, 19.4) for those who both reported and tested poor. Further adjusting for smoking, education, BMI, alcohol drinking, coffee consumption and general health status did not change the estimates, and the corresponding HRs were 1.9 (0.9, 4.1), 4.0 (2.1, 7.6), and 7.5 (3.0, 18.6) respectively. However, we did not find any statistically significant interactions on a multiplicative (p = 0.46) or an additive (p = 0.78) scale, neither did we find any statistically significant difference in the areas under the curves between the model with BSIT-tested olfaction only and the model with both self-reported and BSIT-tested olfaction (p = 0.39, Fig. 1).
Hazard ratios and 95% confidence intervals of PD by objectively tested and/or self-reported olfactory impairment
Note: Model 1: Adjusted for age only. Model 2: Further adjusted for sex and race. Model 3: Further adjusted for cognitive score.
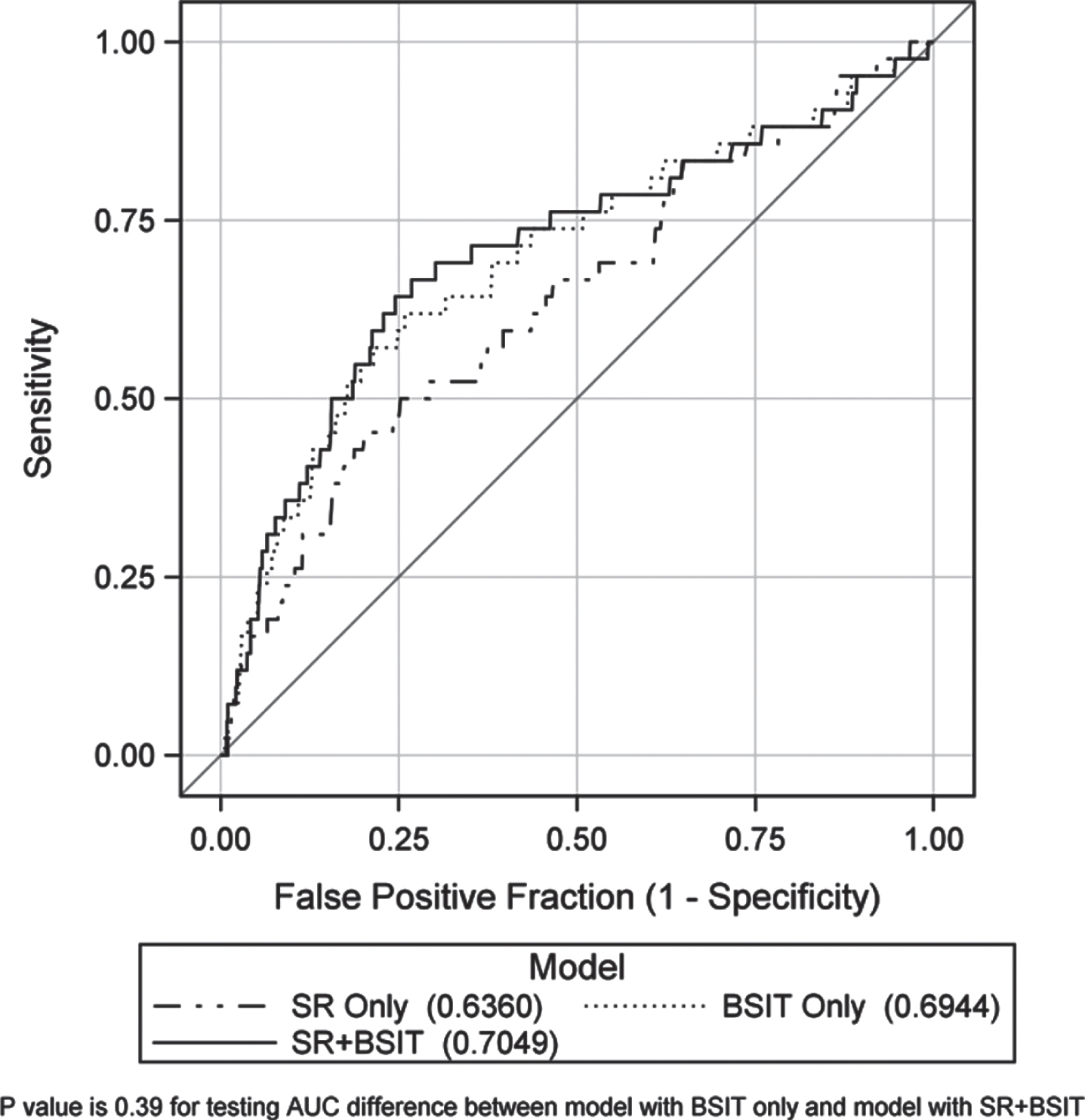
Receiver Operating Characteristic (ROC) Curves Across Three Models.
DISCUSSION
To our knowledge, this is the first study that has evaluated the usefulness of self-reported poor sense of smell along with an objective test in predicting future PD risk. When presented alone, self-reported poor olfaction doubles the risk for future PD diagnosis, and when presented along with an objectively tested deficit, the risk rises to about eight times high, nearly doubling the previously reported risk estimate solely based on an objective test [10, 17]. Although our analyses were based on small numbers and the ROC curve did not show that adding self-report significantly improved the predictability beyond a BSIT test, self-report by itself strongly predicted future PD risk and can be useful in settings where an objectively tested olfaction is not readily available.
In the past two decades, poor sense of smell has been increasingly recognized as an important prodromal symptom for PD and other neurodegenerative diseases. In large epidemiologic studies, a brief smell identification test such as the BSIT, Sniffin’ Sticks, San Diego Odor Identification Test, or the Odor Stick Identification Test for the Japanese, is often the choice to screen for olfaction impairment [2, 18]. These tests only take about a few minutes to do and have generated comparable results across similar population [2]. However, these are simple screening tests by design and only assess olfaction identification. Further, in most studies, participants’ sense of smell was only tested once. While this provides a reliable snapshot of the participants’ sense of smell at the time of the test, it does not reflect potential changes in their sense of smell over time.
Self-awareness is often how health concerns are first noticed and reported. Self-reported health status is associated with a wide range of adverse health outcomes including mortality, functional limitation, chronic diseases, and disability [19–21]. However, in the case of olfaction, when compared with an objective test, self-reported poor sense of smell often had a low sensitivity and a high specificity [4, 5]. Self-report has thus been considered unreliable and were often ignored when evaluating the role of olfactory impairment in health. Our analysis however showed that self-reported poor sense of smell alone predicted future PD risk. Further, theoretically people may report subtle changes in chemosensation over years which cannot be readily captured by a single objective test which relies on high concentrations of odorants and suprathreshold levels. Self-report olfaction may also capture additional aspects of central nervous system pathophysiology due to the evolutionary ancient origins of the olfactory system and its connections to several key areas of the brain [22].
In the current study, the sense of smell and taste was asked about 20 years ago using a single question which was not ideal. With the COVID-19 pandemic and anosmia as one of the presenting complaints, we expect the awareness of this sensory deficit in the general population will increase [23–25]. In addition, structured questionnaires have been developed for use in large population-based studies such as the chemosensory questionnaire by the National Health and Nutrition Examination Survey [5, 26]. This questionnaire is much more detailed, including questions to separate the sense of smell from that of taste and to assess perceived changes with age and altered sensations. These recent developments may increase the usefulness of self-evaluation in assessing olfaction status. Therefore, the value of self-evaluated sense of smell in studying PD and the health of older adults should be further evaluated in future studies.
When self-reported, it is often infeasible to differentiate the sense of smell from the perception of taste which is intimately tied to olfaction [11]. Therefore an alternative possibility is that the additional risk associated with self-reported poor sense of smell may relate to taste deficit. Empirical evidence on gustatory deficit and PD is very limited and inconsistent [27, 28]. In a retrospective analysis of medical records, 28.6% of clinically diagnosed olfactory and gustatory disorder patients later developed PD, compared to 9.9% for a pure olfactory disorder and 3.8% for a pure gustatory disorder [28]. The study was however conducted at a tertiary Smell and Taste Center and did not include participants without olfactory or gustatory deficit as the reference group. In our study, we were not able to disentangle olfactory from gustatory deficits due to the wording of our question and the lack of an objective taste testing. However, our question was asked in the way that clinicians typically ask patients about smell problems, and thus our results are clinically applicable.
Strengths of the current study include its community-based populations and a relatively long follow-up. Main limitations include the small analytic sample sizes and the lack of real-time clinical confirmations of PD diagnosis. Further, both perceived and objectively tested sense of smell was only assessed once at the Year 3 clinical visit, and therefore we were unable to assess the changes in the sense of smell in relation to PD risk. Also, due to the one-time assessment of olfaction, it did not differentiate permanent olfactory impairment from transitory ones. Another important limitation is that we only used one simple question to ask about poor sense of smell and taste. We did not differentiate these two sensory deficits and did not assess any of these sensory modalities in details. Future studies should use more structured approach to assess these two important sensory deficits as discussed above. Finally, the study did not assess the causes for poor sense of smell (e.g., chronic rhinosinusitis) and therefore we were unable to differentiate poor olfaction due to neurodegeneration from those unrelated.
In summary, this prospective study provides preliminary evidence that self-reported poor sense of smell or taste should not be simply dismissed as useless in predicting risk of PD, especially when a single objective test is not readily available. Future studies with larger sample size are needed to confirm this finding and evaluate whether structured questionnaires may further improve the predictability.
CONFLICTS OF INTEREST
The authors have no conflicts of interest relevant to this article to declare.
Footnotes
ACKNOWLEDGMENTS
This research was supported by National Institute on Aging (NIA) contracts #N01-AG-6-2101; N01-AG-6-2103; N01-AG-6-2106; NIA grant R01-AG028050; NINR grant R01-NR012459, and in part by the Intramural Research Program of the NIA/NIH. Dr. Chen is supported by a start-up fund from Michigan State University (GE100455), the Parkinson’s Foundation (Grant No. PF-IMP-1825), and the Office of the Assistant Secretary of Defense for Health Affairs, through the Parkinson’s Research Program (Award No. W81XWH-17-1-0536), and the National Institute of Environmental Health Sciences (R01ES029227). Opinions, interpretations, conclusions and recommendations are those of the author and are not necessarily endorsed by the Department of Defense or the National Institutes of Health.
