Abstract
Mutations in
Keywords
INTRODUCTION
Parkinson’s disease (PD) is a common neurodegenerative disorder that causes a progressive loss of dopaminergic neurons in the substantia nigra pars compacta, resulting in the depletion of striatal dopamine [1, 2]. This decrease in dopamine levels primarily causes resting tremor, muscle rigidity, and bradykinesia, and frequently postural instability [1, 2]. PD is the second most prevalent neurodegenerative disorder, affecting 1–2% of people over the age of 65 worldwide [1, 2]. Currently, therapies for PD patients alleviate motor symptoms but do not slow disease progression [1, 2]. The etiology of PD is unknown and the molecular pathways that precipitate disease onset and contribute to disease progression remain incompletely understood. Identifying and characterizing these pathways has been the focus of PD research for the last 20 years, as this is one of the most promising strategies for developing effective disease-modifying treatments. A growing body of research indicates that risk for developing PD is influenced by a complex interaction between genetic and environmental factors together with age. Many genes have been linked to PD, providing insight into the cellular mechanisms that may underlie neuronal pathology. Most of the monogenic causes of PD lead to early-onset autosomal recessive PD or parkinsonism (
LRRK2
Mutations in
LRRK2 is a relatively large multidomain protein (2527 amino acids) that is expressed, at varying levels, in many diverse tissues and cell types. At its N-terminus, LRRK2 contains armadillo repeat, ankyrin repeat and leucine-rich repeat domains. The enzymatic domains of LRRK2 are located in the center of the protein; they include a Ras-of-Complex (ROC) GTPase, a C-terminal-of-Roc (COR) and kinase domain, followed by WD40 repeats at its C-terminus (Fig. 1). LRRK2 has been shown to form homodimers, and dimerization is likely important for regulating LRRK2 activity [52–54]. PD-linked familial mutations cluster within the central enzymatic domains (N1437H, R1441C, R1441G, R1441H, Y1699C, G2019S, and I2020T) [55–60], and enhance LRRK2 kinase activity [61–64]. Familial mutations commonly increase LRRK2 autophosphorylation at Ser1292 and the LRRK2-mediated phosphorylation of substrates that include a subset of Rab GTPases, suggesting that sustained increases in LRRK2 kinase activity can contribute to the initiation and progression of PD [61, 64–66].
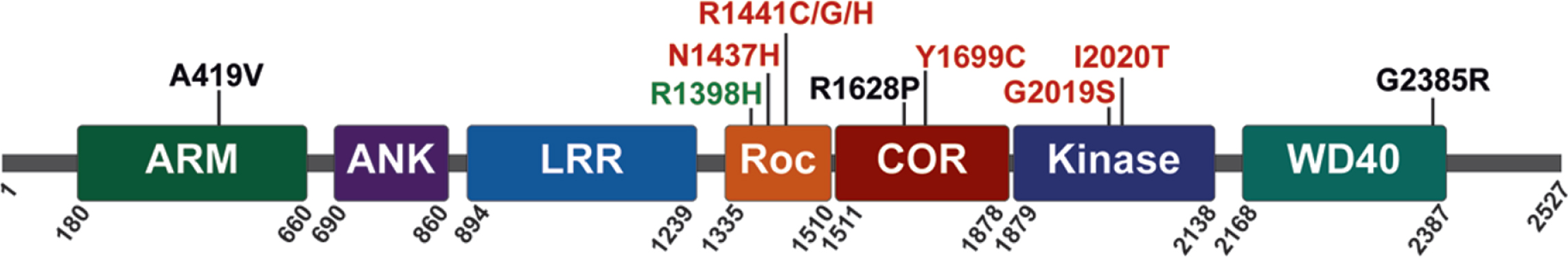
LRRK2 protein domain architecture with PD-linked familial mutations (red), sporadic PD risk variants (black), and sporadic PD protective variants (green), as indicated.
Recent studies suggest increased LRRK2 kinase activity and endolysosomal pathology in nigral dopa-minergic neurons from brains of sporadic PD subjects, including the accumulation of Rab5-positive early endosomes, depletion of late endosomes, depletion of lysosomes, and depletion of lysosomal GCase [67, 68]. Whether these vesicular abnormalities are dependent on LRRK2 activity is uncertain. In experimental neurotoxin models, systemic rotenone exposure in rats elevates the kinase activity of endogenous LRRK2, induces nigral dopaminergic neurodegeneration, accumulation of phosphorylated
THE ENDOLYSOSOMAL PATHWAY
The endolysosomal pathway is a multifaceted, highly regulated process that is critically important for many cellular functions including proteostasis, extracellular and intracellular signaling, organelle homeostasis, and membrane organization. The canonical endosomal pathway initiates with clathrin-mediated endocytosis, and vesicle uncoating followed by fusion of newly formed endocytic vesicles with early endosomes. Early endosomes are mildly acidic, which facilitates the uncoupling of ligands and membrane-bound receptors for recycling. The early endosome also acts as a sorting station, targeting cargo towards one of three destinations: 1) back to cell surface membranes, directly or via recycling endosomes, 2) for degradation through the endosomal sorting complexes required for transport (ESCRT) pathway or 3) for retrograde sorting to the
The endolysosomal pathway also intersects with the phagocytosis and autophagy pathways. Phagocytosis occurs when immune-related cells engulf extracellular material such as apoptotic cells, cellular debris, or infectious agents. Cargo in the phagocytic pathway is recycled or degraded via the endolysosomal pathway when phagosomes fuse with late endosomes or lysosomes. Autophagy occurs when cells digest intracellular components through one of three pathways: 1) macroautophagy, 2) microautophagy, or 3) chaperone-mediated autophagy. Macroautophagy is regulated by ULK1, which induces the formation of a double-membraned autophagosome, around large intracellular components, that are marked for degradation. After formation, autophagosomes undergo a series of maturation steps, including lipidation of LC3-I to LC3-II before fusing with late endosomes or lysosomes. Microautophagy occurs through the direct lysosomal engulfment of small, unlabeled cellular components. Chaperone-mediated autophagy is a process through which chaperone proteins, such as Hsc70, deliver small cellular components that are marked for degradation, usually by ubiquitination, in cooperation with the LAMP2A receptor to lysosomes for degradation. Phagocytosis, autophagy and vesicle trafficking through the endolysosomal pathway are highly coordinated processes that are regulated by many factors (for review, see [71–73]).
Recent findings show that LRRK2 physically or functionally interacts with numerous proteins throughout the endolysosomal pathway, and that manipulating LRRK2 expression levels or kinase activity leads to pathological disruptions in vesicle trafficking and protein degradation (Fig. 2; Table 1). Additionally, brain tissue from sporadic PD subjects has shown accumulation of LRRK2 in enlarged granules that colocalizes with the late endosome protein Rab7b and lysosomal protein LAMP2 [74]. Disrupted trafficking or degradation of LRRK2 in the brain of sporadic PD subjects highlights the endolysosomal system in PD pathogenesis and suggests that LRRK2 may contribute to this pathology.
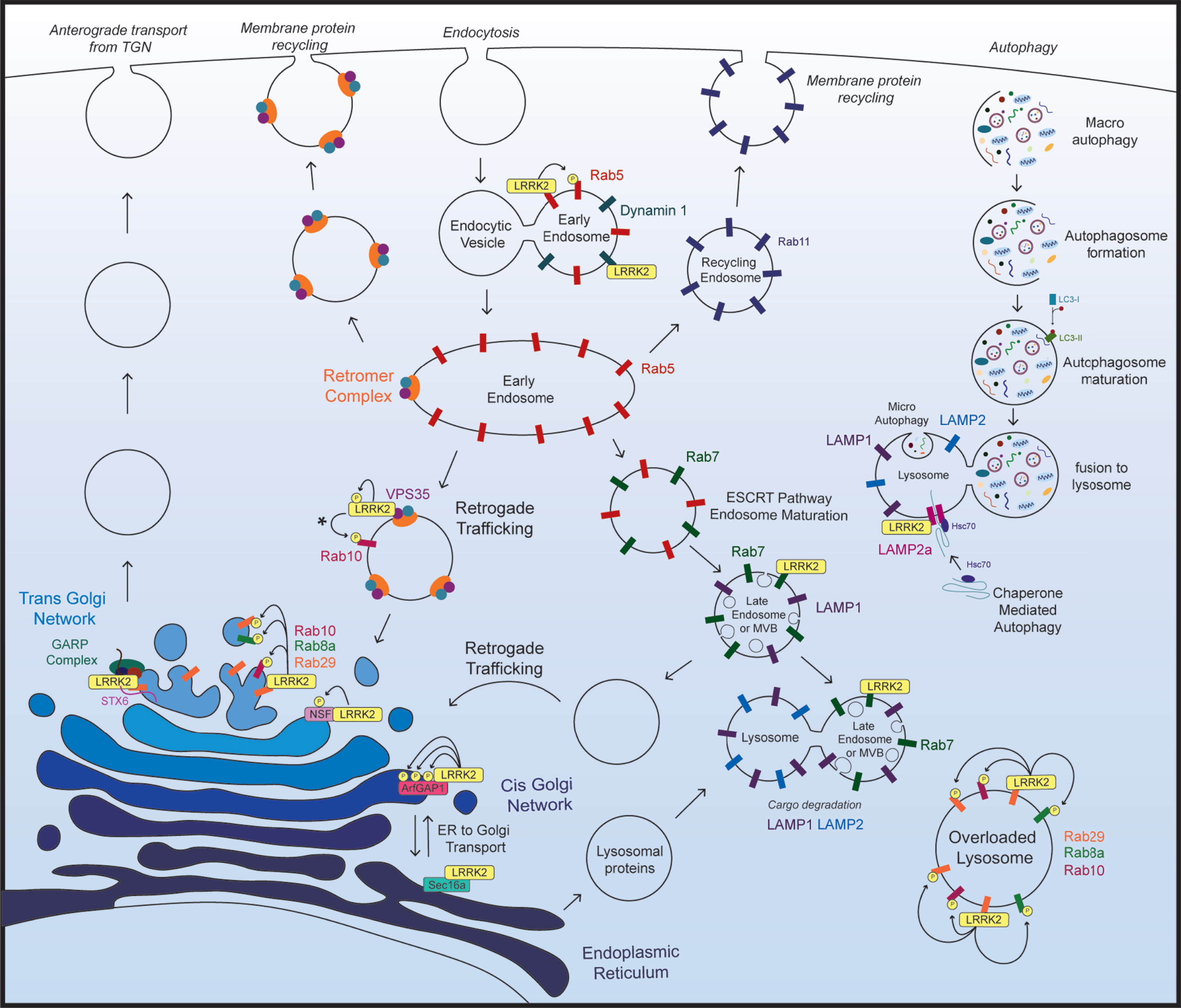
LRRK2 in the endolysosomal pathway. Key aspects of the endolysosomal pathway highlighting LRRK2-interacting proteins and kinase substrates. *The specific subcellular location of VPS35-induced activation of LRRK2 and subsequent Rab10 phosphorylation not been established. As VPS35 resides in the retromer complex, and the retromer complex primarily localizes to endosomal and vesicular membranes in the retrograde trafficking pathway, it most likely occurs on endosomal membranes, however this has not been directly assessed.
LRRK2-interacting proteins in the endolysosomal pathway
LRRK2 regulates the endolysosomal pathway at multiple steps, through mechanisms that are not yet well defined. One way that LRRK2 likely impacts this pathway is through interaction with or direct phosphorylation of multiple Rab proteins localized throughout the endolysosomal system [64, 75] (Table 2).
Rab proteins that interact with LRRK2
LRRK2 AND RABS
Rabs are small GTPases that reside on specific intracellular compartments, many of which are in the endolysosomal pathway. The highly specific subcellular localization of Rabs is important for their regulatory functions in this pathway and is controlled by several distinct Rab-interacting proteins. For example, binding to Rab escort proteins (REPs) promotes prenylation of Rabs via geranylgeranyl transferase, which subsequently tethers Rabs to compartment-specific membranes. Membrane-bound Rabs can then be activated by guanine nucleotide exchange factors (GEFs) that catalyze the exchange of GDP for GTP. GTP-bound active Rabs recognize specific effector proteins to facilitate docking and fusion of Rab-positive membranes to target membranes. Rabs are also regulated by GTPase-activating proteins (GAPs) that catalyze the hydrolysis of GTP to GDP, switching their target Rabs to an inactive state. GDP dissociation inhibitor (GDI) proteins bind inactive Rabs and remove them from membranes. Individual Rabs move onto and off specific subcellular membrane compartments throughout the endolysosomal pathway, in a highly controlled manner to regulate signaling and sorting throughout this pathway (for review, see [69, 77]).
LRRK2 complexes with, or directly phosphorylates multiple Rab proteins in the endolysosomal pathway, including Rab29 [66, 79] (Table 2). Recent studies have found that Rab29 can recruit LRRK2 to specific cellular compartments, including the TGN and overloaded lysosomes [79–84]. Recruitment of LRRK2 by Rab29 subsequently enhances LRRK2 kinase activity and promotes LRRK2-mediated recruitment of additional Rab proteins [79–84]. Surprisingly, LRRK2 kinase activity is also enhanced by Rab8a, Rab12 and Rab38, but not to the extent of LRRK2 activation by Rab29 [82]. LRRK2 activation by other Rabs has not yet been fully investigated, and the functional implications of these pathways are currently unclear. However, emerging data suggest intriguing roles for LRRK2-mediated phosphorylation of Rab8a and Rab10 in regulating ciliogenesis and centrosomal cohesion [66, 85–87]. LRRK2-Rab interactions will be further discussed in the following sections focusing on individual components of the endolysosomal pathway.
LRRK2 DELETION DISRUPTS THE ENDOLYSOSOMAL PATHWAY
LRRK2 knockout in rodent models
LRRK2 is expressed in many tissues and cell types, with highest expression in lung, kidney, and immune cells and relatively low expression in the brain [88]. LRRK2 knockout mice and rats have discolored kidneys with lysosomal pathology that progresses with age [88–94]. For example, LRRK2 knockout leads to the formation of large vacuoles, the accumulation of lipofuscin granules, and formation of high-molecular weight ubiquitin-positive protein aggregates in the kidneys [88–93]. Specific perturbations in the endolysosomal pathway have also been observed in LRRK2 knockout kidneys including increased levels of lysosomal proteins LAMP1, LAMP2 and cathepsin B, as well as enlarged LAMP1-positive vesicles [89, 94]. Additionally, LRRK2 knockout mice are more susceptible to lysosomal overloading, specifically in kidney tissue, in response to intraperitoneal chloroquine injections, exhibiting increased LAMP1 staining, vacuolization, and lipofuscin in renal proximal tubule cells [80]. Perturbations in autophagy have also been observed in LRRK2 knockout kidneys, as changes in LC3-II/LC3-I protein levels, however it is currently unclear whether LRRK2 knockout enhances autophagosome formation or impairs autophagic flux in kidney tissue [88, 92].
LRRK2 knockout also causes lysosomal pathology in lung and liver tissues, manifesting as vacuolization in both tissues, accumulation of lamellar bodies in type II pneuomocytes in the lungs and formation of lipid droplets in the liver [89, 90]. Notably, tissues that express lower levels of LRRK2, including the heart and brain, exhibit no apparent endolysosomal pathology in LRRK2 knockout animals [88–92]. LRRK2 knockout causes mild behavioral changes in mice without causing dopaminergic neuronal degeneration or overt neuropathology [88, 90–92]. LRRK2 knockout may be compensated for by the expression of its homolog LRRK1 in the brain. Interestingly, LRRK1/2 double knockout mice have shortened lifespan, as well as modest age-dependent dopaminergic neuronal degeneration and formation of large autophagic vacuoles in the SN [95].
LRRK2 knockout in non-mammalian models
In non-mammalian models and primary cell cultures, depletion of LRRK2 disrupts endocytosis, lysosomal function, and autophagosome formation [91, 97]. In
LRRK2 also regulates lysosome function and morphology in primary cultures. In primary astrocytes,
LRRK2 knockout disrupts synaptic vesicle endocytosis
LRRK2 knockout or knockdown also disrupts synaptic vesicle endocytosis (SVE) in animals and primary neuronal cultures [19, 100–104]. Knockout of the
As previously noted,
LRRK2 AND LYSOSOMES
Expression of familial PD LRRK2 mutations alters lysosome morphology, distribution, pH and degradative capacity in many distinct cell types including fibroblasts, striatal neurons, primary neurons, and primary astrocytes [79, 106]. Fibroblasts derived from
The effects of G2019S LRRK2 expression in primary neurons appear to vary depending on timing and LRRK2 protein levels. Similar to primary astrocytes, transient transfection of G2019S LRRK2 in primary cortical neurons causes lysosomal swelling [79]. In these neurons, G2019S LRRK2 overexpression also leads to a reduction of mannose-6-phosphate receptor (M6PR) at lysosomes and at the Golgi, which could indicate a defect in M6PR endosomal sorting [79]. Conversely, primary cortical neurons from G2019S LRRK2 knockin mice experience an opposing shift in lysosome morphology, generating both smaller lysosomes and an increase in total lysosome number without perinuclear clustering [107]. Like G2019S LRRK2-expressing primary astrocytes, G2019S LRRK2 knockin neurons have decreased LAMP1 protein levels and increased lysosomal pH [96, 107]. G2019S LRRK2 knockin mice likely express lower levels of pathogenic LRRK2 compared to transiently transfected neurons. Additionally, germline expression of G2019S LRRK2 could potentially result in developmental compensation within the endolysosomal pathway that modulates the toxicity of this pathogenic LRRK2 variant. These putative compensatory pathways are unlikely to be engaged in experiments where G2019S LRRK2 is transiently expressed for short periods and could contribute to the differences observed in lysosomal morphology.
Several recent studies have shown that LRRK2 is recruited to vesicular compartments in the endolysosomal pathway, often in response to stress [80, 109]. LRRK2 subsequently phosphorylates and stabilizes Rab proteins at these compartments, which then recruit downstream effector proteins [80, 109]. Eguchi et al. found that Rab29 recruits LRRK2 to enlarged lysosomes in response to chloroquine treatment, and that LRRK2 recruitment counteracts chloroquine-induced lysosomal overloading in RAW264.7 mouse macrophages and in HEK-293 cells [80]. LRRK2 recruitment initiates the recruitment of four additional Rab proteins, two of which, Rab8a and Rab10, are well-characterized LRRK2 kinase substrates [64, 80]. Phosphorylation of Rab8a and Rab10 by LRRK2 subsequently stabilizes these proteins on lysosomal membranes allowing them to recruit EHBP1 and EHBP1L1, which promote lysosomal secretion and counteract lysosomal overloading [80]. Interestingly, LRRK2 kinase inhibition or LRRK2 knockdown exacerbates lysosomal enlargement in chloroquine-treated cells, and overexpression of PD-linked LRRK2 mutants protects cells from chloroquine-induced lysosomal enlargement [80]. Upregulation of this pathway may have beneficial effects in tissues that express high levels of LRRK2, such as lung and kidney, but could have adverse effects in cells that express low levels of LRRK2. For example, upregulating LRRK2-mediated lysosomal secretion in neurons could increase extracellular levels of proteins that would otherwise be degraded by the lysosomal pathway, such as
LRRK2 is also recruited to late endosomes and lysosomes in RAW264.7 mouse macrophages in response to endolysosomal membrane damage via pathogen infection or L-leucyl-L-leucine methyl ester (LLOME) treatment [108]. At damaged endolysosomal membranes LRRK2 phosphorylates Rab8a and stabilizes it on these membranes [108]. Phosphorylated Rab8a then recruits the membrane damage marker Galectin-3 and the ESCRT-III pathway component CHMP4B [108]. LRRK2 KO or Rab8a KO enhances lysosomal membrane damage and increases lysophagy in response to LLOME treatment, indicating that this LRRK2-Rab8a pathway stabilizes endolysosomal membrane integrity and promotes endolysosomal membrane repair through the ESCRT pathway [108]. LRRK2 is also recruited to late phagosomes in activated human iPSC-derived macrophages [109]. Like LRRK2 recruitment to endolysosomal membranes, LRRK2 recruitment to late phagosomes promotes the recruitment, phosphorylation and stabilization of Rab8a and Rab10 [109]. LRRK2 does not appear to affect initiation or rate of phagocytosis in these cells, and the downstream effects of LRRK2-Rab recruitment to late phagosomes is currently unknown [109]. Likewise, whether Rab29 is required for LRRK2 recruitment to damaged endolysosomal membranes or late phagosomes has not yet been assessed.
Expression of PD-linked LRRK2 variants also appears to impair lysosomal function by negatively regulating lysosomal glucocerebrosidase (GCase) activity [110, 111]. iPSC-derived dopaminergic neurons from PD subjects harboring
LRRK2 AND SYNAPTIC VESICLE ENDOCYTOSIS
G2019S LRRK2 overexpression impairs SVE in primary midbrain neurons, primary hippocampal neurons and in
Auxilin regulates a key step in SVE by recruiting Hsc70 to clathrin-coated synaptic vesicles to initiate their uncoating, which is followed by vesicle repackaging and recycling [114]. LRRK2 is suggested to phosphorylate auxilin at Ser627 within its clathrin-binding domain [110]. Nguyen and Krainc show that phosphorylation status at S627 regulates auxilin-clathrin binding and propose that 1) LRRK2-mediated phosphorylation of auxilin at this residue inhibits its binding to clathrin and 2) inhibiting auxilin-clathrin interaction impairs recycling of synaptic vesicles, causing a reduction in synaptic vesicle number and an increase in cytosolic dopamine levels [110]. This hypothesis is supported by the observation that overexpression of wild-type auxilin partially protects
Dynamins are small GTPases that participate in clathrin-mediated endocytosis and intracellular vesicle budding by promoting membrane scission [118, 119]. Mammals have 3 Dynamin genes (Dynamin 1, 2 and 3), all of which are expressed in the brain [118, 119]. Dynamin 1 is highly enriched in neurons; Dynamin 2 is ubiquitously expressed, and Dynamin 3 is expressed in the brain but at lower levels than Dynamin 1 [118, 119]. All 3 Dynamins participate in SVE, with Dynamin 1 playing the most prominent role in this process [118]. LRRK2 complexes with Dynamin 1, 2 and 3 in HEK-293 cells, and with Dynamin 1 in the mouse brain [115]. LRRK2 and Dynamin 1 also colocalize on early endosomes in SH-SY5Y cells [115]. Interestingly, G2019S LRRK2 overexpression in these cells enhances LRRK2-Dynamin 1 colocalization and reduces levels of Dynamin 1 at early endosomes [115]. Additionally,
Synaptojanin-1 (
LRRK2 AND THE GOLGI NETWORK
LRRK2 has a robust effect on Golgi integrity and vesicle sorting via the Golgi. Overexpression of familial LRRK2 mutants induces Golgi fragmentation in cell lines, primary neurons, and striatal neurons [78, 126]. Surprisingly, LRRK2 depletion has a similar effect, leading to Golgi fragmentation and defective Golgi sorting of lysosomal hydrolases [127].
When transiently overexpressed in cultured cells, Rab29 recruits LRRK2 to the TGN, where it activates LRRK2, enhancing LRRK2 autophosphorylation and substrate phosphorylation [79, 81–84]. Rab29-mediated recruitment of LRRK2 to the TGN promotes LRRK2-mediated phosphorylation of Rab29 and recruitment and phosphorylation of 2 additional LRRK2 substrates, Rab10 and Rab8a to the TGN [79, 81–83]. This process requires Rab29 prenylation and membrane association but does not rely on other proteins that are specifically enriched on TGN membranes, as artificially targeting Rab29 to mitochondrial membranes initiates this cascade at the mitochondria [84]. Guanine nucleotide-binding of Rab29 and LRRK2 is required for this process, but Rab29 recruitment of LRRK2 does not require LRRK2 kinase activity [82, 83]. This pathway is likely important for maintaining Golgi integrity, as Rab29 depletion or expression of Rab29 dominant-negative or phospho-mimetic mutants also causes Golgi fragmentation [78, 128]. In addition to complexing with Rab29, LRRK2 also binds BAG5 and GAK proteins in cultured cells and mouse brain [126]. Overexpression of any of these individual proteins causes Golgi fragmentation which can be blocked by concurrent knockdown of any of the other proteins individually [126]. These results implicate all four complex components in Rab29/LRRK2-mediated Golgi maintenance [126].
The Rab29-LRRK2 pathway is also important for retrograde trafficking as knockout or dominant-negative Rab29 expression disrupts retrograde trafficking of M6PR, Sortilin and Furin from endosomes and lysosomes to the TGN [79, 128]. Beilina et al. has recently reported a novel interaction between LRRK2, Rab29 and the Golgi-associated retrograde protein (GARP) complex [94]. Specifically, LRRK2 and Rab29 interact with the GARP complex component VPS52 at the TGN. Overexpression of Rab29 recruits VPS52 to the TGN, and this effect is enhanced by co-expression of LRRK2 [94]. Conversely, knockdown of VPS52, or other GARP complex components (VPS53 or VPS51), inhibits Rab29-mediated recruitment of LRRK2 to the TGN, indicating a cooperative stabilization of the Rab29:LRRK2:GARP complex that requires each component [94]. LRRK2 and VPS52 both complex with VAMP4 and syntaxin-6, SNARE proteins that localize to the TGN, suggesting that LRRK2 may stabilize GARP complex interactions with SNARE proteins at the TGN to promote membrane fusion and vesicle trafficking [94]. This hypothesis is supported by the observation that LRRK2 or Rab29 knockdown destabilizes the VPS52-syntaxin-6 interaction [94]. Knockdown of Rab29, LRRK2 or VPS52 decreases retrograde trafficking of M6PR or Cholera toxin B from endosomes to the TGN and impairs anterograde trafficking of GPI proteins from the TGN to the plasma membrane [94]. Conversely, primary astrocytes from R1441C LRRK2 knockin mice exhibit a kinase-dependent increase in retrograde and anterograde vesicular trafficking [94].
This study provides valuable mechanistic insight into the role of LRRK2 in vesicle trafficking to and from the TGN. Because the GARP complex has not been shown to directly regulate anterograde trafficking, the precise mechanisms through which LRRK2 modulates this pathway are currently unclear. The effects of LRRK2 on anterograde trafficking from the TGN may be an indirect result of modulating retrograde trafficking through the GARP complex, or could be caused by direct interactions between LRRK2 and the anterograde trafficking pathway. Further examination of anterograde trafficking components and LRRK2 will be required to distinguish between these two possibilities. How chronic LRRK2-mediated enhancement of retrograde trafficking affects the endolysosomal system
ArfGAP1 is a GAP for Arf1, a cis-Golgi protein that functions in the secretory pathway and regulates Golgi organization and lipid homeostasis in this organelle [129, 130]. ArfGAP1 was initially identified as a LRRK2 modifier in yeast where deletion of the ArfGAP1 ortholog,
Consistent with the observation that
LRRK2-mediated effects on Golgi integrity may also involve the N-ethylmaleimide-sensitive fusion (NSF) protein. NSF primarily functions as a SNARE complex chaperone, facilitating vesicle fusion between intracellular membranes and SNARE complex disassembly for recycling [133]. It was originally identified as an essential factor for vesicle trafficking in the Golgi [134]. NSF has also been shown to facilitate vesicle trafficking from the endoplasmic reticulum to the Golgi, and Golgi reassembly following mitosis [135, 136]. LRRK2 complexes with NSF in mammalian cells and phosphorylates NSF
In addition to maintaining Golgi integrity, LRRK2 has also been implicated in the maintenance of Endoplasmic Reticulum Exit Sites (ERESs) through interaction with the ERES protein Sec16a [138]. Sec16a recruits and stabilizes protein complexes to ERESs that facilitate the formation of COPII-coated vesicles [139]. Knockdown or overexpression of Sec16a results in secretory pathway disruption and disorganization of ERESs [139]. LRRK2 complexes with Sec16a in mammalian cells and in the mouse brain, and colocalizes with Sec16a at ERESs in primary hippocampal neurons [138].
LRRK2 interacts with multiple proteins resident at the Golgi and endoplasmic reticulum to regulate Golgi integrity, and Golgi network anterograde or retrograde trafficking. Additionally, modulating the expression of individual LRRK2-interacting proteins in these pathways can prevent LRRK2-induced Golgi fragmentation (Rab29, BAG5, GAK) or LRRK2-induced toxicity (ArfGAP1) [125, 126]. Most studies examining the effects of LRRK2 on the Golgi network have been performed in cell culture models with overexpression or depletion of LRRK2. Examining Golgi integrity and vesicle trafficking in knockin rodent models where familial LRRK2 mutants are expressed at endogenous levels will be important for evaluating 1) whether the Golgi network is disrupted by familial LRRK2 mutations
LRRK2 AND THE RETROMER
The retromer complex regulates retrograde trafficking and retrieval of transmembrane protein cargo from early endosomes to the TGN or cell surface membrane for recycling [140–142]. The retromer complex has two components, the cargo-selective complex (CSC) and a sorting nexin (SNX) dimer. VPS35 is the largest component of the CSC and is essential for retromer-mediated cargo sorting, particularly for cargo recognition, which is critical for cellular function. Recently, VPS35 has been identified as a familial PD gene, with a missense mutation (D620N) causing late-onset, autosomal dominant familial PD [143, 144]. The precise effects of the D620N mutation on VPS35 function, and the molecular mechanisms that underlie the pathogenicity of this mutation, are currently unclear [145]. The dominant inheritance pattern of PD associated with D620N VPS35 suggests that this mutation most likely manifests disease via 1) a partial dominant-negative effect on VPS35 function or 2) a toxic or normal gain-of-function effect.
Recent studies of D620N VPS35 in rodent models and PD subjects indicate that this mutation does not manifest an obvious loss-of-function effect, but potentially induces PD pathology by activating LRRK2 [146–148]. Notably, although homozygous
LRRK2 may also regulate retrograde trafficking through interactions with Rab32 or Rab38. Rab32 and Rab38 both complex with LRRK2 and with sorting nexin 6 (SNX6), a SNX dimer subunit of the retromer [149, 150]. Expression of constitutively-active or inactive Rab32 impairs retrograde trafficking of the retromer cargo M6PR [149]. Whether LRRK2 modulates Rab32-mediated disruption of retrograde trafficking has not yet been tested, and it is currently not known whether Rab32 and Rab38 exist together in a complex with LRRK2 and SNX6, or independently. This putative pathway for LRRK2-retromer interaction via Rab proteins is of interest but has not yet been fully characterized. Further evaluation of LRRK2 and retromer subunit interactions will be beneficial in determining how these proteins functionally intersect and modulate each other. Evaluating whether D620N VPS35-dependent neurodegeneration can be ameliorated by LRRK2 knockout or kinase inhibition in rodent models will be important for validating the significance of their functional interaction in the context of PD-like pathology.
LRRK2 AND AUTOPHAGY
Autophagy is a critical process for maintaining protein homeostasis and preventing protein aggregation. Perturbations in autophagy have been observed in brain tissue from PD subjects and in animal models of PD. The relationship between LRRK2 and autophagy has been an area of growing interest and has been studied in many model systems. As this topic has recently been comprehensively reviewed [151], we will provide a limited discussion of LRRK2 and autophagy.
Expression of familial LRRK2 mutants in rodents, or in
Familial LRRK2 mutations can alter mitophagy and macroautophagy in diverse model systems, however, the impact of LRRK2 on these pathways varies in both magnitude and direction. These variations in effect size likely result from differences in experimental systems including cell type, cellular context, and timing and dosage of LRRK2 expression. For example, human fibroblasts derived from PD subjects harboring G2019S or R1441C LRRK2 mutations exhibit decreased mitophagy that results from enhanced phosphorylation of Rab10 [157]. Rab10 phosphorylation impairs the Rab10-optineurin interaction and prevents Rab10 from recruiting optineurin to depolarized mitochondria to initiate mitophagy [157]. Alternatively, transient overexpression of G2019S or R1441C LRRK2 in primary cortical neurons enhances calcium uptake by mitochondria, leading to mitochondrial membrane depolarization and increased mitophagy specifically in dendrites, where transient calcium influxes are highest [152, 156].
LRRK2 knockout mice exhibit altered expression of autophagy markers and formation of large autophagic vacuoles in kidneys, but lack autophagic pathology in the brain [88, 93]. This is likely due to LRRK2 being highly expressed in kidneys relative to the brain, and LRRK2 knockout in the brain may potentially be compensated for by LRRK1 expression. Interestingly, LRRK1/2 double knockout in mice causes age-dependent impairment of the autophagy pathway in the brain that results in the formation of p62-positive puncta and the accumulation of autophagic vacuoles in the substantia nigra [95]. Familial LRRK2 mutations appear to have subtle effects on macroautophagy
CONCLUSIONS
The effects of LRRK2 knockout or PD-linked familial
Lysosomes appear to be particularly sensitive to changes in LRRK2 expression and kinase activity. Impaired lysosomal degradation capacity and im-paired autophagy are emerging as recurring themes in distinct PD models. Identifying the mechanisms through which LRRK2 activity modulates lysosome homeostasis and protein degradation will be important for understanding how
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
The authors work is supported in part by grants from the National Institutes of Health (R01 NS091719, R01 NS105432, R01 NS117137, to D.J.M.) and the Parkinson’s Foundation (PF-FBS-1894, to M.E.).
