Abstract
Background:
Striatal dopamine deficits play a key role in the pathogenesis of Parkinson’s disease (PD), and several non-motor symptoms (NMSs) have a dopaminergic component.
Objective:
To investigate the association between early NMS burden and the patterns of striatal dopamine depletion in patients with
Methods:
We consecutively recruited 255 patients with drug-naïve early-stage PD who underwent 18F-FP-CIT PET scans. The NMS burden of each patient was assessed using the NMS Questionnaire (NMSQuest), and patients were divided into the mild NMS burden (PDNMS-mild) (NMSQuest score <6;
Results:
Patients in the PDNMS-severe group had more severe parkinsonian motor signs than those in the PDNMS-mild group, despite comparable DAT activity in the posterior putamen. DAT activity was more severely depleted in the PDNMS-severe group in the caudate and anterior putamen compared to that in the PDMNS-mild group. The inter-sub-regional ratio of the associative/limbic striatum to the sensorimotor striatum was lower in the PDNMS-severe group, although this value itself lacked fair accuracy for distinguishing between the patients with different NMS burdens.
Conclusion:
This study demonstrated that PD patients with severe NMS burden exhibited severe motor deficits and relatively diffuse dopamine depletion throughout the striatum. These findings suggest that the level of NMS burden could be associated with distinct patterns of striatal dopamine depletion, which could possibly indicate the overall pathological burden in PD.
INTRODUCTION
Parkinson’s disease (PD) is characterized by cardinal motor signs including bradykinesia, rigidity, and tremor, but it is also frequently accompanied by several non-motor symptoms (NMSs) [1]. The NMSs occur not only in the advanced stages but also in the early stages of PD, even before the manifestation of the motor symptoms [1, 2] and adversely affect the health-related quality of life [3]. Moreover, non-motor manifestations have now been incorporated into the clinical diagnostic criteria for PD [4]. Although the neuropathology underlying early NMSs in PD is still unclear, the dysregulation of multiple neurotransmitter systems has long been proposed as an important contributor [5–8]. In particular, striatal dopamine deficits are known to play a key role in the pathogenesis of PD, and a range of NMSs also appear to have a dopaminergic component [8].
We have previously tested the hypothesis that patients with PD with a greater burden of NMSs might have greater striatal dopamine loss compared to those with fewer NMSs [9]; we failed to reveal the association between early NMS burden and the patterns of striatal dopamine depletion. However, our previous work was critically limited by the fact that the severity of NMSs was not assessed with an initial diagnostic workup, i.e., the gap between the time of dopamine transporter (DAT) scans and assessment of NMSs exceeded 3 years. Moreover, all patients were medicated at the time of NMSs assessment, and the confounding effect of dopaminergic medications could not be excluded. Hence, the present study reinvestigated whether early NMS burden was associated with different patterns of striatal dopamine depletion in early-stage PD. To overcome these limitations, the severity of NMSs was simultaneously assessed at the timing of DAT scans, at a drug-naïve status, using a simple assessment tool, the NMS Questionnaire (NMSQuest) [10].
METHODS
Participants
We reviewed the medical records of 255 consecutive patients with
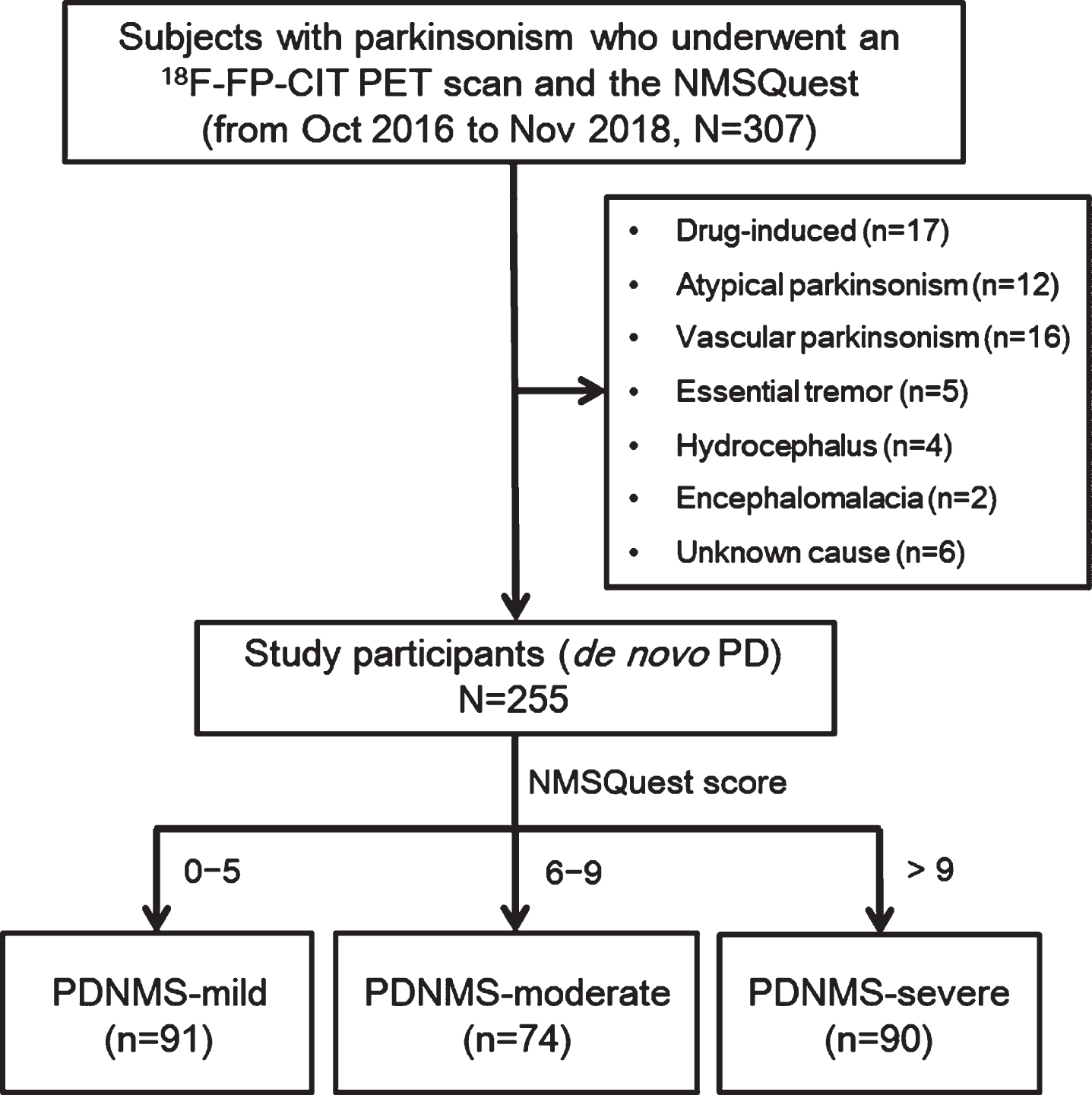
Flow diagram of participant enrollment. NMSQuest, non-motor symptoms questionnaire; PD, Parkinson’s disease; PDNMS-mild, a PD group with mild non-motor symptom burden; PDNMS-moderate, a PD group with moderate non-motor symptom burden; PDNMS-severe, a PD group with severe non-motor symptom burden.
Assessment of non-motor symptom burden
We translated and used the NMSQuest [10] to assess the NMS burden of each patient with PD at the initial diagnostic workup. The NMSQuest is a validated self-report instrument, which is widely used for screening NMS in movement disorders clinics [14], and the score ranges from 0 to 30 (Supplementary Methods). Based on the grading system proposed by Chaudhuri et al. [15], we classified the patients into three groups according to the NMSQuest scores: a PD group with mild NMS burden (PDNMS-mild) (NMSQuest score, 0–5;
Acquisition of the 18F-FP-CIT PET images
18F-FP-CIT PET imaging was performed using the Discovery 600 (GE Healthcare, Milwaukee, WI, USA) device, which acquires images with a three-dimensional resolution of 2.3 mm full-width at half-maximum. After a 6 h fast, the patients were intravenously injected with 5 mCi (185 MBq) of 18F-FP-CIT. Ninety minutes after the injection, PET images were acquired for 20 min in three-dimensional mode at a power of 120 kVp and a current of 200 mA [16].
Quantitative analyses of the 18F-FP-CIT PET
Image processing was performed using MATLAB (The MathWorks, Inc, Natick, MA, USA) software for statistical parametric mapping (SPM8) and ITK-SNAP (http://www.itksnap.org). All reconstructed 18F-FP-CIT images were normalized to the 18F-FP-CIT template, which was created using the 18F-FP-CIT PET images and T1-weighted MR images of 40 healthy controls [17]. None of the healthy controls, whose images were used for 18F-FP-CIT template creation had any previous history of neurological or psychiatric illness. They showed normal cognitive function on all neuropsychological tests and exhibited normal findings on neurologic examination, structural MRI, and 18F-FP-CIT PET. Twelve volumes-of-interest (VOIs) were drawn on the 18F-FP-CIT template, as described previously [11]. The striatum was divided into dorsal and ventral portions, along the transverse plane, using the anterior-posterior commissure line. The ventral portion comprised two sub-regions: ventral striatum and ventral putamen. Subsequently, the dorsal portion was divided into the following anterior and posterior sub-regions along the coronal anterior commissure plane: the anterior caudate, posterior caudate, anterior putamen, and posterior putamen. DAT activity in each VOI was estimated using the specific/nonspecific binding ratio as a surrogate, which was defined as follows: (mean standardized uptake value of the striatal sub-region VOIs-mean standardized uptake value of the occipital VOI)/mean standardized uptake value of the occipital VOI. The inter-sub-regional ratio (ISR) was calculated by dividing the DAT activity in the striatal sub-regions excluding the sensorimotor striatum (i.e., associative striatum [anterior and posterior caudate and anterior putamen] and limbic striatum [ventral striatum]) by that in the sensorimotor striatum (i.e., posterior and ventral putamen).
Predictive ability of the striatal dopamine depletion patterns for the non-motor symptom burden
The concordance index (or c-index) [18] was calculated to assess the accuracies of the striatal dopamine depletion pattern (i.e., ISR of the associative striatum to the sensorimotor striatum, which significantly differed between the PDNMS-mild and PDNMS-severe groups) to predict the NMS burden (i.e., total NMSQuest scores, sub-scores of the non-motor items with a dopaminergic basis, and sub-scores of the non-motor items with a non-dopaminergic basis). The non-motor items with a dopaminergic basis were determined according to a previous systematic review [19]. The bootstrap method was used to compare the predictive ability of ISR for determining the NMS burden.
Statistical analyses
To compare the baseline demographic characteristics and NMSQuest scores between the PD groups, Student’s
RESULTS
Baseline clinical characteristics and overall non-motor symptom burden
The baseline clinical characteristics and the NMSs burden assessed by the NMSQuest are summarized in Table 1. Patients in the PDNMS-severe group exhibited more severe parkinsonian motor signs (UPDRS-III, 25.31±9.34) than those in the PDNMS-mild group (20.60±8.00;
Demographic characteristics and non-motor symptoms of the PD groups according to the non-motor burden
The values are expressed as the mean±standard deviation. PDNMS-mild, a PD group with mild non-motor symptom burden (NMSQuest score, 0–5); PDNMS-severe, a PD group with severe non-motor symptom burden (NMSQuest score >9). PD, Parkinson’s disease; UPDRS-III, the Unified Parkinson’s disease Rating Scale Part III; K-MMSE, the Korean version of the Mini-Mental State Examination; CCSIT, the cross-cultural smell identification test; BDI, Beck Depression Inventory; RBD, rapid eye movement behavior disorder; NMSQuest, non-motor symptoms questionnaire. aNumber of positive answers classified by NMSQuest domains.
Striatal DAT availability in the PDNMS-mild and PDNMS-severe groups
The PDNMS-severe group exhibited greater decreases in the DAT activity in the anterior caudate (
DAT availability of the striatal sub-regions in the PD groups according to non-motor burden
PDNMS-mild, a PD group with mild non-motor symptom burden (NMSQuest score, 0–5); PDNMS-severe, a PD group with severe non-motor symptom burden (NMSQuest score >9). aFalse discovery rate (FDR)-controlling method was used for multiple comparison correction.
Predictive ability of the striatal dopamine depletion patterns for the non-motor symptom burden
The estimated accuracy (i.e., c-index) of the ISR value (i.e., DAT activity ratio of the associative/limbic striatum to the sensorimotor striatum) for predicting the total NMSQuest score was 0.541 (95% confidence interval [CI], 0.504–0.584;
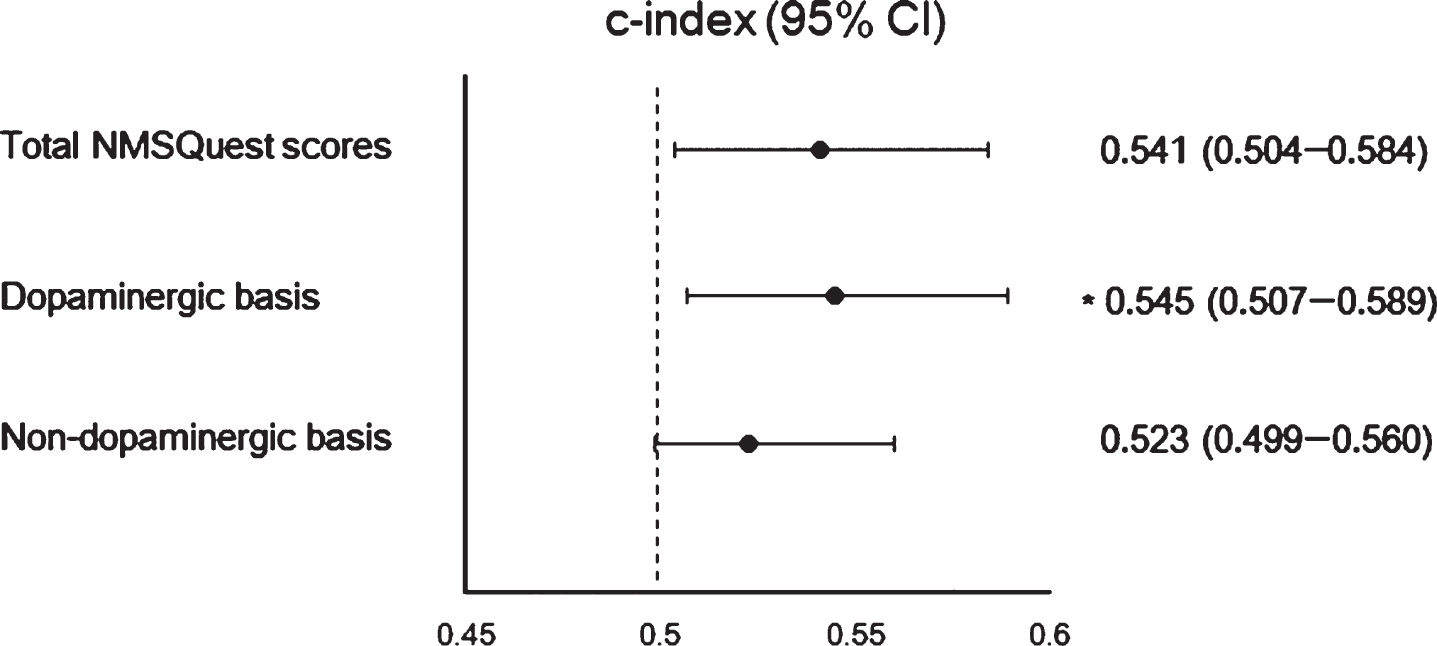
Predictive ability of the striatal dopamine depletion patterns for the non-motor burden. The dopamine transporter activity ratio of the associative striatum to the sensorimotor striatum lacked fair accuracy for differentiating between the patients with different non-motor burdens (total NMSQuest scores, c-index 0.541; sub-scores of non-motor items with a dopaminergic basis, c-index 0.545; sub-scores of non-motor items with a non-dopaminergic basis, c-index 0.523). NMSQuest, non-motor symptoms questionnaire; CI, confidence interval. *
DISCUSSION
The major findings of this study were as follows: 1) Patients in the PDNMS-severe group had more severe parkinsonian motor signs (i.e., higher UPDRS-III scores) than those in the PDNMS-mild group, despite comparable DAT activity in the sensorimotor striatum between the groups. 2) The PDNMS-severe group exhibited greater decreases in DAT activity in the associative striatum than that in the PDNMS-mild group. 3) The ISR of the associative/limbic striatum to the sensorimotor striatum was lower in the PDNMS-severe group. These findings suggest that early NMSs could be associated with diffuse dopamine depletion in the striatal sub-regions other than the sensorimotor striatum in patients with
The neural substrates for NMSs in PD remain incompletely understood, although widespread pathologic changes in the central and peripheral nervous systems are thought to be the basis for these non-motor features [20, 21]. The fact that NMSs can arise in the early or premotor stages of PD has received wider recognition [1, 2], which corresponds to the pathologic progression of PD from the non-dopaminergic structures [22–24]. According to the Braak staging [22], Lewy pathology arises from the olfactory complex and lower brainstem (stages 1 and 2). The changes in these non-dopaminergic structures could cause several NMSs including hyposmia, RBD, and dysautonomia [25]. Moreover, Lewy pathology appears to occur peripherally in the gastrointestinal tract or autonomic neurons in the premotor phase [23, 24], which might cause constipation [26]. Subsequently, several animal studies [27–29] and
Recently, we published two studies that whether the patterns of striatal dopamine depletion could 1) provide prognostic information on the clinical profiles of early-stage PD [51] and 2) contribute to the early burden of NMSs in PD [9], assuming that the degree of unevenness of striatal dopamine loss would possibly define heterogeneous PD subtypes. However, the former study [51], which retrospectively enrolled a total of 634 patients with
In accordance with our initial hypothesis, PD patients with a greater NMS burden had greater dopamine depletion in the associative striatum than those with a lower NMS burden. Moreover, the motor deficits in patients in the PDNMS-severe group were more severe than those in the PDNMS-mild group, despite the comparable DAT activity in the sensorimotor striatum. The PDNMS-severe group also had a higher frequency of RBD cases than the PDNMS-mild group, where RBD is known to be associated with increased severity and frequency of other non-motor features such as sleepiness, depression, and cognitive impairment [57]. These findings suggest that overall non-motor burden can be a clinical marker of diffuse striatal dopamine deficits and can further redefine PD subtypes (e.g., subtype with mild clinical symptoms with selective putaminal involvement vs. subtype with severe clinical symptoms with diffuse striatal dopamine loss). The concept that NMSs should also be considered when identifying the heterogeneity of PD is gradually gaining recognition, and early NMS burden may be informative from a prognostic prospective [9, 55]. In line with these previous observations, the present study also demonstrated the possible role of striatal dopamine loss in mediating the relationship between NMS burden and PD subtyping. However, it should be noted that the pattern of striatal dopamine loss (i.e., ISR value of the associative striatum to the sensorimotor striatum) did not accurately predict the overall NMSs burden in this study. Moreover, partial correlation analyses did not reveal a correlation with a sufficiently large effect size between the striatal DAT activity and NMSs (correlation coefficients <0.2), after adjusting for age, sex, and disease duration (see Supplementary Table 2), probably because the dopaminergic contribution to the NMSs in PD is not absolute, even for some NMSs that are commonly known to have a dopaminergic basis [19]. In other words, the association between NMS burden and striatal dopamine depletion pattern may not indicate a causal relationship. Instead, a greater NMS burden may indicate a greater pathological burden, including diffuse dopaminergic dysfunction and multiple neurotransmitter deficits and pathological changes. Furthermore, the discrepancies with the results of previous studies (i.e., lack of association between striatal dopamine dysfunction and NMS burden) [9, 50] may be attributed to the methodological differences, including the participants (drug-naïve or not), statistical approach, DAT scan modalities, and NMS assessment tools.
Our study had some limitations. First, the NMSQuest scores do not specifically measure the intensity of each NMS, but only reflect a summation of the reported range of NMS; however, the number of NMSs declared by the NMSQuest is highly correlated with the level of the burden of NMSs assessed by the Non-Motor Symptoms Scale [15, 58], which can provide information on the severity and frequency of each specific NMS [59]. Second, grouping according to the NMSQuest scores may have been a little arbitrary, although the cut-off criteria has been validated [15]. However, grouping has some advantages that make it easy to perform comparative analyses and explore the association between variables. Moreover, the “total” NMSQuest scores may obscure the possible role of striatal dopamine deficits in the NMS burden, although we also calculated the c-index of the striatal dopamine depletion pattern to predict the sub-scores of non-motor items with a dopaminergic basis. Third, DAT scans may not be an ideal surrogate marker for striatal dopaminergic deficits [60], since FP-CIT binding cannot detect DAT downregulation [42] and reflect the floor effect of dopamine loss, particularly in the posterior putamen [61]. Fourth, K-MMSE is neither sensitive nor specific to screen mild cognitive decline in patients with early-stage PD, and other neuropsychological scales such as the Montreal Cognitive Assessment [62] might be more useful to assess global cognitive abilities in PD. Fifth, other widely used NMS instruments such as the Parkinson Anxiety Scale [63] and Lille Apathy Rating Scale [64] were not assessed. This information is important because anxiety or apathy is frequently observed in patients with early-stage PD and can adversely affect quality of life. Finally, we estimated the disease duration as the difference between age at diagnosis and onset of parkinsonian motor symptoms. However, the duration of prodromal phase (i.e., non-motor manifestations prior to the appearance of definite motor deficits) might be longer in patients with greater NMS burden, which can bias the results. Some comorbidities unrelated to PD pathology might also affect the NMS burden in patients with PD.
In conclusion, the results of this study demonstrated that patients with PD with severe NMS burden exhibited severe motor deficits and relatively diffuse dopamine depletion throughout the striatum. These findings suggest that the overall burden of NMSs could be associated with distinct patterns of striatal dopamine depletion, which could possibly indicate the overall pathological burden in PD.
CONFLICT OF INTERST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
This research was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (grant number: NRF-2018R1D1A1B07048959). This research was also supported by the Brain Research Program through the NRF funded by the Ministry of Science, ICT and Future Planning (grant number: NRF-2018M3C7A1056898).
