Abstract
Background:
Pain is a disabling and often underestimated non-motor symptom (NMS) detrimentally affecting the quality of life of patients with Parkinson’s disease (PD).
Objective:
Here, we conducted a cross-sectional, observational international study on 167 patients with idiopathic PD in order to analyze the potential relationship between pain and other NMS.
Methods:
Subjects were assessed with the Unified Parkinson’s Disease Rating Scale (UPDRS) part III, Hoehn and Yahr (H&Y) stage, King’s Parkinson’s Disease Pain Scale (KPPS), Brief Pain Inventory (BPI), Non-Motor Symptoms Scale (NMSS), and Beck Depression Inventory (BDI). Spearman’s rank correlation coefficient, multiple regression and multiple index-based clustering algorithms were used for data analysis.
Results:
The prevalence of pain was 88.6%, was not correlated with age, motor severity (UPDRS part III) or disease duration, whereas a weak correlation with female gender and H&Y stage >2.5 was found. Multiple NMS correlated significantly with pain. Specifically, sleep disturbance had the strongest correlation with pain, followed by depression, gastrointestinal and cardiovascular disturbances. Further analyses showed that sleep and cardiovascular disturbance were independently associated with pain, and that these symptoms clustered together in a subset of PD patients. The relationship between pain, sleep and dysautonomia persisted independently from dopamine replacement therapy.
Conclusion:
Our study suggests that sleep disruption and cardiovascular disturbance are associated with pain in PD, and possibly identifies a specific subtype within PD patients with pain. Our data also indicate that sleep disruption, pain and dysautonomia may have a common pathophysiology, possibly involving non-dopaminergic pathways.
INTRODUCTION
Pain is a disabling, often underestimated non-motor symptom (NMS) throughout the natural history of Parkinson’s disease (PD) [1–5]. Its prevalence ranges between 40– 88%, and the pain subtypes of musculoskeletal, radicular and dystonic type are reported as the most common [4, 7]. Multiple factors are associated with pain in PD, including female gender, younger age, co-morbid affective symptoms, autonomic symptoms and motor complications [4, 9]. Surprisingly, motor severity, both disease duration and stage have not shown a consistent relationship [4, 10]. Most of the treatment options, including non-steroidal anti-inflammatory drugs (NSAIDs), opioids, dopamine agonists, duloxetine and non-pharmacological treatments have demonstrated inadequate relief or require further investigation [7, 12]. Recently, the International Parkinson and Movement Disorder Society Evidence-Based Medicine Committee’s recommendations classified the use of oxycodone/naloxone prolonged release as “possibly useful” for central, musculoskeletal and nocturnal pain in PD, whereas rotigotine has been suggested for non-motor, painful fluctuations [3, 13].
Although some pain subtypes are exacerbated by motor symptoms, growing evidence suggests that impaired central nociceptive processing plays an important role in pain pathogenesis in PD [5, 14]. Interestingly, pain may cluster with other non-motor symptoms (NMS), possibly implicating a common pathophysiology involving extra-dopaminergic pathways [8, 15–17]. Yet, recent studies suggest that pain may be the phenotypic hallmark of the “Park-Pain non-motor endophenotype” [17], raising the possibility that pain identifies a specific subgroup of patients, which require specific and personalized therapeutic approaches.
In this study, we took advantage of the King’s Parkinson’s Disease Pain Scale (KPPS) [18], which allows for more disease-specific clinical discernment of different pain subtypes [8]. Together with the Brief Pain Inventory (BPI), we aimed at characterizing pain and assessing the potential relationships with main clinical features and other NMS of PD in a large, dual-center population.
MATERIALS AND METHODS
Study participants
This is a double-center international, observational, cross-sectional study of consecutive PD patients enrolled at the University of Rome “Tor Vergata” (URTV), Italy and the George Washington University (GW) in Washington DC, US, (April 2017-June 2018). A letter of agreement was signed between URTV School of Medicine and Surgery and GW in 2014. Patients met the 2015 Criteria of Movement Disorders Society [19]. Exclusion criteria were atypical/secondary parkinsonism, cognitive impairment (Mini-Mental Status Examination, MMSE ≤ 24/30), severe disability as indicated by Hoehn and Yahr stage (H&Y ≥4) in the ON-medication state. To avoid any bias due to other conditions causing pain independently from PD, patients with rheumatologic/orthopaedic diseases, polyneuropathy, spine injuries, cancer, were excluded. Subjects were evaluated by a neurologist with expertise in movement disorders. Levodopa equivalent daily dose (LEDD) was calculated. The study was approved by the institutional review boards (Ethics Committee URTV Hospital, Protocol 212/16; GW Institutional Review Board, Protocol 121637). All participants gave their informed consent.
Assessments
Socio-demographic and clinical history data (age, gender, disease duration, H&Y stage, LEDD, and pain treatment) were collected from all patients. The following instruments and questions were used: Motor severity: UPDRS part III. Pain: King’s PD Pain Scale (KPPS); Brief Pain Inventory (BPI). NMS: NMS Scale (NMSS); the Beck Depression Inventory (BDI). Dysautonomia: questions from the NMSS that directly pertained to autonomic dysfunction, including swallow dysfunction (NMSS_q20), drooling (NMSS_q19), unintended weight loss (NMSS_q29), constipation (NMSS_q21), urinary urgency/incontinence (NMSS_q22), urinary frequency and nocturia (NMSS_q23, NMSS_q24), orthostasis (NMSS_q1, NMSS_q2), sweating (NMSS_q30), erectile dysfunction, vaginal dryness, and difficulty with orgasm (NMSS_q26). Specific aspects of sleep disturbance were assessed by individual questions from the NMSS that pertained to different types of sleep disturbance, including excessive daytime somnolence (NMSS_q3), fatigue (NMSS_q4), insomnia (NMSS_q5) and restless leg syndrome (NMSS_q6).
Data analysis
Descriptive statistics of clinical history and socio-demographic variables were first calculated and reported using basic statistical summary methods. Univariate analysis between variables of interest and pain presence (KPPS total) was performed by way of Spearman’s correlation coefficient, r, to determine monotonic associations. Further univariate analysis was performed using Chi-Square, Fisher’s exact test, independent samples t-test, and the Kruskal-Wallis test where appropriate.
In order to identify variables with significant independent associations with pain, variables with a statistically significant unadjusted association with pain were entered into a generalized linear regression model with dependent variable KPPS total. Multicollinearity of covariates was assessed in the multivariable model by way of a variance inflation factor (VIF) analysis and the condition index. VIF <2 was considered acceptable and detected collinearity was resolved by way of purposeful selection methods in conjunction with factor analysis.
Cluster analysis was performed using multiple index-based clustering algorithms to explore possible symptom clusters. The Euclidean distance-based Ball-Hall algorithm in conjunction with optimal Schwarz Bayesian information criterion was used for proper selection of number of clusters, k. Variables of interest were compared across clusters using the aforementioned further univariate analysis methods. All statistical analyses were performed using SAS version 9.4 (SAS Institute Inc., Cary, NC) and R version 3.5.3 (R Foundation for Statistical Computing, Austria) with p < 0.05 considered statistically significant.
RESULTS
A total of 167 subjects were included in our analysis. Descriptive data are reported in Table 1. 88% of subjects (n = 148) declared some level of pain (KPPS total>0), despite optimal dopaminergic treatment, and were referred to as the PD-pain cohort in subsequent analyses. Musculoskeletal pain was the most prevalent (79.7%), followed by radicular (43.2%), fluctuation-related pain (41.9%) and nocturnal pain (40.5%) (Fig. 1). In principle, more than one single type of pain, in more than a single site might be present, a condition defined as widespread pain [20]. However, our cohort did not exhibit these features. The average total KPPS pain score was 14.4±13.2 and the average BPI interference score was 1.8±2.6. 54% of subjects reported that pain interfered with their daily activity, as indicated by a score greater than 0 on the BPI, items 9A-G. Pain had a weak, though significant, correlation with female gender (r = 0.20, p < 0.05) and H&Y stage >2.5 (r = 0.25, p < 0.05). Age, motor severity (UPDRS part III) and disease duration did not correlate with pain. In our cohort, there was no significant correlation between pain and motor subtypes of tremor-predominant, postural instability gait disturbance or indeterminate.
Baseline characteristics of the study population (n = 167)
Reported as # (%), mean±standard deviation, or median (interquartile range). H&Y, Hoehn and Yahr scale; UPDRS III, Unified Parkinson’s disease Rating Scale Part III (motor examination); MMSE, Mini-Mental State Examination; KPPS, King’s Parkinson’s Disease Pain Scale; BPI, Brief Pain Inventory; NMSS, Non-Motor Symptoms Scale for Parkinson’s Disease; BDI, Beck Depression Inventory.
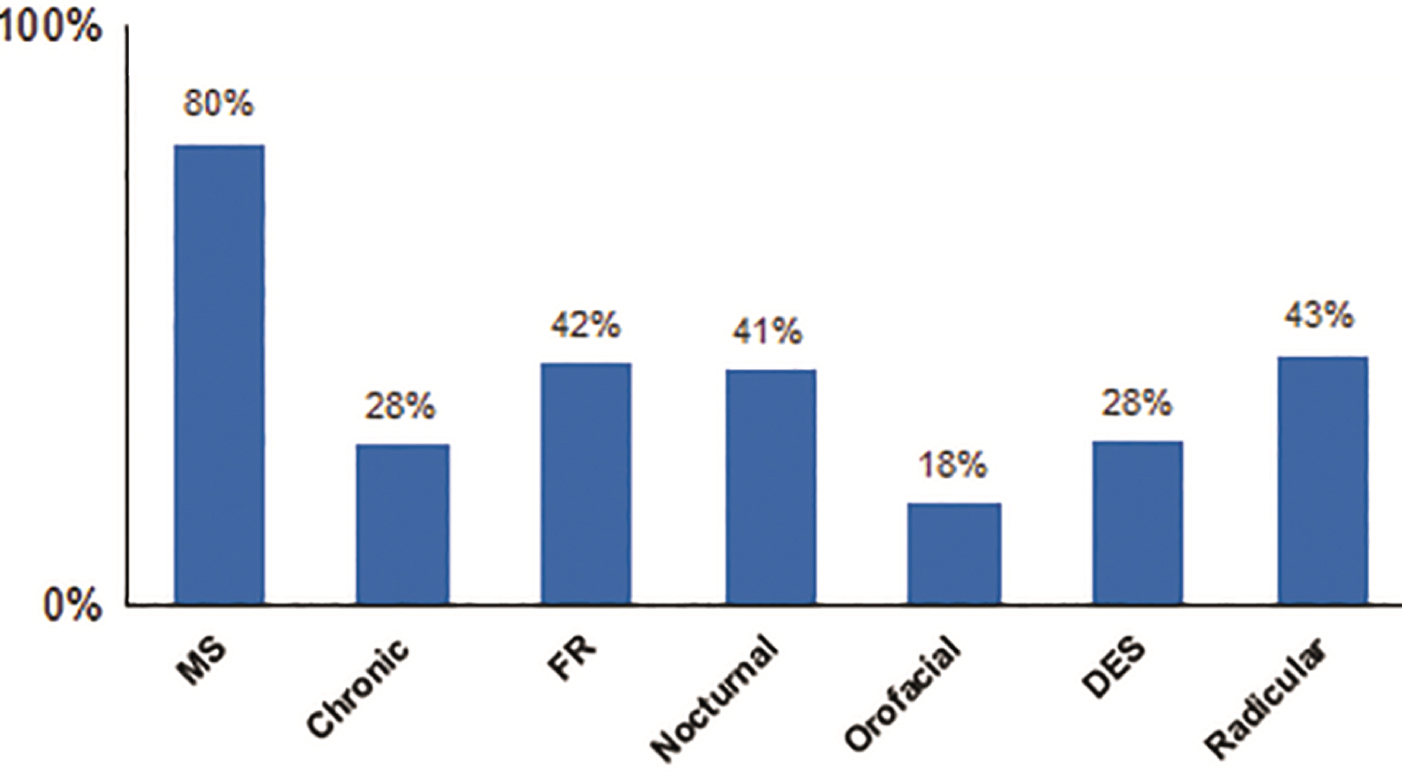
Prevalence of KPPS pain subtypes. Among patients who declared some level of pain (KPPS total > 0), musculoskeletal pain was the most prevalent (79.7%), followed by radicular (43.2%), fluctuation-related pain (41.9%) and nocturnal pain (40.5%). MS, musculoskeletal; FR, fluctuation-related, DES, discoloration/edema swelling.
Examination of the differences between the American and Italian subjects showed that the two cohorts had similar disease stages and prevalence of pain as measured by a KPPS > 0. However, the American cohort was older, had higher LEDD, higher motor scores, more severe pain scores on BPI and higher NSAID use. The experience of non-motor symptoms across the majority of domains was similar between groups, with the exception of autonomic symptoms, which were more prevalent in the American cohort (Supplementary Table 1).
Eighty-five percent of all PD-pain subjects were taking dopaminergic medications. There was a weak but significant correlation between higher LEDD and higher pain scores (r = 0.18, p < 0.05). Nearly 32% of PD-pain subjects reported taking NSAIDs, 32% were on antidepressants (SSRIs), 3.4% were taking a steroid, 2.0% were on opioid medication and 40% were enrolled in a motor rehabilitation program (Fig. 2). PD-pain subjects taking NSAIDs had significantly higher pain scores on the KPPS and higher cardiovascular scores than PD-pain subjects who were not taking analgesics (p = 0.0073) (Table 2, Supplementary Table 2). There was no significant difference for those enrolled in rehabilitation programs.
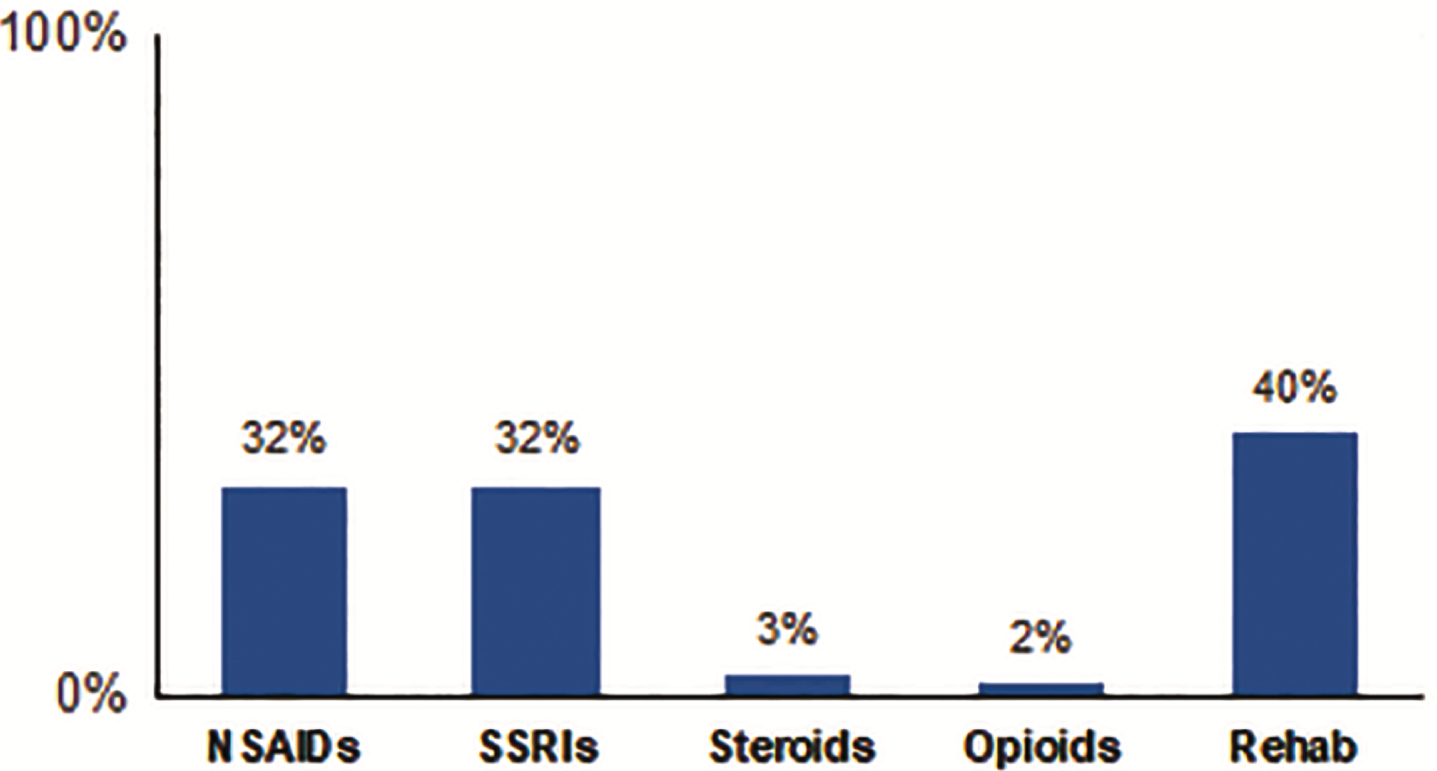
Pain management in PD-pain subjects. Nearly 32% of PD-pain subjects reported taking NSAIDs, 32% were on antidepressants (SSRIs), 3.4% were taking a steroid, 2.0% were on opioid medication and 40% were enrolled in a motor rehabilitation program. NSAIDs, non-steroidal anti-inflammatory drugs; SSRIs, selective serotonin reuptake inhibitors.
Pain scores in PD-pain patients who did or did not receive an intervention for pain
Reported as mean±standard deviation and median (interquartile range). KPPS, King’s Parkinson’s Disease Pain Scale; NSAIDs, Non-steroidal anti-inflammatory drugs; SSRIs, Selective serotonin reuptake inhibitors; Rehab, rehabilitation.
Ninety-eight percent of all subjects reported at least 1 NMS (NMSS>0). Multiple NMS correlated significantly with pain (Table 3). Sleep disturbance had the strongest correlation with pain (r = 0.42, p < 0.001), followed by depression (r = 0.36, p < 0.001), GI disturbance (r = 0.35, p < 0.001), and cardiovascular disturbance (r = 0.27, p < 0.001). Distinct subtypes of pain had significant relationships with NMS (Supplementary Table 3). Fluctuation-related pain was associated with sleep disturbance (r = 0.275, p < 0.0024), cardiovascular disturbance (r = 0.028, p = 0.0005) and GI disturbance (r = 0.27, p = 0.0006). Nocturnal pain also had a moderate correlation with sleep disturbance (r = 0.38, p < 0.0001) and GI disturbance (r = 0.25, p = 0.0022), while musculoskeletal pain correlated significantly with the presence of depression (r = 0.21, p = 0.0089).
Correlation between presence of pain (KPPS > 0) and other factors
LEDD, Levodopa equivalent daily dosage; H&Y, Hoehn and Yahr scale; UPDRS, Unified Parkinson’s disease Rating Scale; NMSS, Non-Motor Symptoms Scale for Parkinson’s Disease; BDI, Beck Depression Inventory.
Multiple regression analysis (F = 19.95; R2 = 0.42; p < 0.001) of all subjects (n = 167) showed an independent association between pain and female gender (p = 0.0027), cardiovascular disturbance (p = 0.0434) and sleep issues (p < 0.001) (Table 4). The “miscellaneous” questions from domain 9 of the NMSS also had an independent association with pain, but this relationship was confounded since one of the questions in that domain specifically addressed pain.
Multiple Regression Analysis to identify variables with significant independent associations with pain
LEDD, Levodopa equivalent daily dosage; H&Y, Hoehn and Yahr scale; NMSS, Non-Motor Symptoms Scale for Parkinson’s Disease; BDI, Beck Depression Inventory.
When comparing patients with pain (n = 148) and patients without pain (n = 19), patients with pain had significantly higher cardiovascular and sleep disturbance scores. Generally, all manifestations of dysautonomia were evident in the pain group. Depression, however, was similar between patients with pain and those without (Supplementary Table 4). In the subjects with pain on dopaminergics (n = 126), sleep disturbance continued to have an independent association with pain (p = 0.0005). Further analysis of the association between specific aspects of sleep disruption assessed with the NMSS and pain showed that fatigue had the strongest correlation with pain and correlated with multiple pain subtypes (Supplementary Table 5).
Cluster analysis revealed a k = 3 cluster model to be an optimal solution in clustering symptoms of interest with pain. Inter- and intra-cluster variable characteristics are outlined in Supplementary Table 6. Cluster 1 included the majority of patients and was characterized by shorter disease duration, moderate motor symptoms, low pain scores and a relative paucity of NMS. Cluster 2 had similar age, disease duration and motor scores compared to cluster 1, but this group had a higher NMS burden, with pain, sleep and dysautonomia scores that were significantly higher than cluster 1. Cluster 3 included older patients, with longer disease duration, a high burden of NMS, and significantly higher pain scores and dysautonomia.
DISCUSSION
Pain can occur throughout the course of PD and is associated with a reduced health-related quality of life [5]. Our data supports the existing evidence that pain is a highly prevalent NMS in PD, with musculoskeletal pain being most common [4, 8]. Consistently, the average pain severity score in our sample was mild (BPI 1.9±2.0; KPPS 14.4±13.2) [4, 15], but pain was nevertheless disruptive to daily activities in >50% of our sample [15]. The prevalence of pain was similar between the Italian and American groups, but the severity of pain was higher in the American cohort. This may be related to the fact that the American cohort had higher overall LEDD or a higher incidence of dysautonomia, both of which correlated with higher pain scores in the combined cohort. Alternatively, socio-cultural differences between groups may have accounted for this difference.
Several factors were associated with increased pain scores across all PD-pain patients, including female gender, higher LEDD and advanced stage, whereas pain did not correlate with motor severity (UPDRS part III score), supporting the notion that the mechanical influence of motor symptoms is not the only determinant in pain pathophysiology [4, 10].
Surprisingly, despite the high prevalence of pain and its interfering with daily activity, only a minority were on analgesics (Fig. 2). Of note, PD-pain patients taking NSAIDs had higher pain scores (Table 2), reflecting an inadequate treatment of pain. The efficacy of pain management relies upon a comprehensive pain assessment that should drive the therapeutic choice: NSAIDs are inappropriate for the treatment of recurrent or chronic pain of severe intensity, while SNRIs or gabapentinoids are more efficacious for the treatment of neuropathic pain [21]. Alternatively, the higher pain scores in the PD-pain patients taking NSAIDs may also be related to the more prominent cardiovascular symptoms in this group (Supplementary Table 2), which may magnify pain symptoms.
Pain correlated most strongly with sleep disturbance, depression, GI dysfunction and cardiovascular disturbance (Table 3) [8, 22]. In a sub-analysis of our cohort of patients on dopaminergics (n = 126), sleep and cardiovascular disturbance continued to correlate with pain, suggesting that the relationship between pain, sleep and dysautonomia persisted despite dopamine replacement treatment. The multivariable regression analysis revealed that cardiovascular disturbance and sleep were independently associated with pain, suggesting a possible interplay among these conditions (Table 4).
In our cluster analysis, cluster 1 and 3 may represent the early patient with predominant motor symptoms and the advanced patient with a higher motor and non-motor burden, respectively, given the differences in disease duration. However, cluster 2, which had similar age, disease duration and motor scores compared to cluster 1, had higher pain, sleep and dysautonomia scores than cluster 1. Therefore, cluster 2 may represent a phenotypic variant of PD, characterized by more prominent symptoms of pain, sleep disturbance and dysautonomia. Non-motor endophenotypes of PD including a “Park-pain phenotype” have been proposed [17] and the combination of pain with sleep dysfunction and dysautonomia suggests an underlying serotonergic and noradrenergic PD subtype.
Sleep and pain
Sleep disorders are a common NMS in PD. We show that sleep disruption is significantly more common in PD patients experiencing pain (Tables 3 and 4). This association persisted in patients on dopaminergic medication, indicating that the sleep-pain relationship cannot be solely explained by an imbalance of dopaminergic content, nor can sleep or pain issues be adequately treated by dopaminergic drugs alone.
Our regression analysis suggests that sleep alterations could be a predictor of pain in PD, in line with other studies [8, 17]. There are several possible explanations for this relationship. Pain can alter sleep quality and sleep architecture, causing sleep fragmentation, decreased sleep efficiency, reduced REM sleep and increase stage N1 sleep [23]. There is also evidence in the other direction, that poor sleep lowers the pain threshold [23]. However, it is possible that sleep disruption and pain share a common pathophysiology. Indeed, noradrenergic input from the locus coeruleus may be implicated in the pathophysiology of both. Input from the coeruleus inhibits the pedunculopontine nucleus to induce REM sleep [15, 24]. The locus coeruleus is also involved in the medial pain pathway, modulating the transmission of ascending nociceptive information at the level of the dorsal horn. Other brainstem nuclei, including the dorsal raphe nucleus and reticular formation have also been implicated in the pathogenesis of sleep disruption and pain in PD. Thus, the co-occurrence of sleep disruption and pain might be related to spreading synucleinopathy in the early disease stages.
Our cluster analysis indicates that in a subgroup of patients, pain seems to cluster with sleep disturbances. This fits the phenotypic paradigm proposed by Sauerbier et al. [17] who proposed that the “limbic” phenotype of PD is dominated by NMS such as fatigue, pain, depression and weight loss. These clinical features are in agreement with the observation that plasma serotonin (5-HT) and 5-HT metabolite (5-HIAA) levels were significantly reduced in PD patients with pain compared to controls. The insular cortex might be involved in this phenotype, since the insula is a key structure in the pain matrix, and specifically in the sensory discrimination component of the pain pathway [17]. Consistently, reduced cortical thickness in bi-temporal poles, insula and dorsolateral pre-frontal cortex, as well as reduced connectivity between the nucleus accumbens and hippocampus was observed in a multi-modal imaging study comparing PD patients with or without pain [10]. The theory that noradrenergic and serotonergic mechanisms are key drivers of pain in PD is also supported by the association between fatigue and pain, seen in our cohorts. Fatigue is a complex phenomenon in PD that can be confounded by co-morbid depression, apathy, medication side effects, physical deconditioning and sleep disruption. Autonomic dysfunction, and specifically cardiac sympathetic denervation, is also associated with fatigue. PD patients with fatigue were found to have reduced noradrenergic tone, with decreased cardiac 123I-metaiodobenzylguanidine (MIBG) uptake [25] and reduced 5-HT transporter binding on 11C-DASB PET in the striatum, cingulate, amygdala and thalamus compared to those without fatigue [26]. Therefore, reduction of both noradrenergic and serotonergic tone may play a role in the pathogenesis of fatigue, as well as of pain, in PD.
Dysautonomia and pain
Our regression analysis demonstrated the association between cardiovascular disturbances and pain, in agreement with prior studies [16]. The connection between pain, sleep deprivation and autonomic dysfunction has been suggested in other populations as well. Schetetsky and colleagues reported that sleep deprived individuals had lower heat pain thresholds compared to those with normal sleep. The heat pain threshold also correlated inversely with sudomotor skin responses. Collectively, those with increased pain had reduced sudomotor response habituation caused by repeated pain stimuli, suggesting that there may be an over-reaction of the sympathetic cholinergic system to nociceptive stimuli [27].
As discussed above for the association between sleep and pain, noradrenergic input from the locus coeruleus plays a significant role modulating ascending nociceptive input. A dominant noradrenergic deficit is also likely to be the key of dysautonomic symptoms [28]. Indeed, reduced noradrenergic innervation disrupts reflex cardiovascular responses and is one of the causes of autonomic failure in PD patients. Moreover, Lewy body pathology in the locus coeruleus may affect the noradrenergic modulation of pain as well as the dysautonomic responses.
However, the association between dysautonomia and pain might underlie additional conditions. Actually, in PD, cardiovascular dysautonomia (orthostatic hypotension especially) is predominantly due to degeneration of post-ganglionic sympathetic neurons rather than CNS synucleinopathy [29]. In these cases, tissue hypoperfusion related to cardiovascular dysautonomia can account for pain, as it occur in patients with orthostatic hypotension that often experience the so-called “coat-hanger ache” due to muscular ischemia, induced by the drop in perfusion pressure upon standing [30]. Finally, we should also consider that pain and dysautonomia might characterize later phases of PD, thus representing features of an advanced clinical stage.
From a clinical standpoint, pain during fluctuation falls into the pattern of sensory non-motor fluctuation. The other two types of non-motor fluctuation are autonomic and cognitive [31]. Tachyarrhythmias often associated with hyperhidrosis can occur in this period as part of the autonomic sensory fluctuation and may be associated with pain. In addition, cardiovascular disturbance in PD is known to be associated with impaired cardiovascular sympathetic response, baroreflex failure, and low noradrenergic tone. The vasodilation induced by dopamine from levodopa can accentuate orthostatic intolerance in those with cardiovascular disturbance [32]. It is possible that patients with low noradrenergic tone are more sensitive to minute changes of dopamine concentration both in plasma and CSF, and are more susceptible to having non-motor fluctuations in the form of cardiovascular symptoms and sensory disturbances.
Fluctuation-related pain is also associated with patterns of dyskinesias such as diphasic or peak dose and the phenomenon may also be associated with fluctuations in blood pressure and heart rate. A number of studies analyzing the biochemistry of dopamine metabolites in the CSF of different subgroups of patients support this assumption, suggesting also an additional component determined by the biochemical pathway leading to noradrenaline metabolite production. Interestingly, along with disease progression, CSF homovanillic acid (HVA) changes do not match with dopamine level modifications, showing an increase in HVA/DA ratio in advanced and dyskinetic patients compared with younger patients, suggesting a possible non-dopaminergic metabolism in patients with fluctuations [33].
Altogether based both on our data and recent findings, we hypothesize a possible aminergic dysfunction (both dopaminergic and noradrenergic) underlying fluctuation-related pain and its association with cardiovascular disturbance [34], although the mechanistic bases remain to be elucidated.
Study limitations
There are several limitations of this study. First, the small sample size increases the possibility of a type 2 error. The lack of a healthy control group also limits the interpretation of our data, and a case-control design would help to interpret these findings. Furthermore, our assessment methods were suboptimal, as we did not use imaging or fluid biomarker measurement, or objective measures of orthostatic hypotension. In addition, cognitive functions have not been extensively evaluated and did not include specific questionnaires for sleep disturbance and dysautonomia in PD, although the NMSS has been indeed validated against sleep and autonomic tools.
Despite the limitations, we extend and confirm the relevance of pain, in our dual-center, independent cohorts. Our data show that pain is highly prevalent in PD, with musculoskeletal pain being most common. Only a minority of patients are treated, strengthening the concept that pain is not well recognised in clinical practice. We show that cardiovascular disturbance and sleep dysfunction are independently associated with pain and support the concept of pain dominant non-motor endophenotype of PD (Park-Pain). The combination of pain with sleep dysfunction and dysautonomia suggests an underlying serotonergic and noradrenergic system dysfunction.
FINANCIAL DISCLOSURE
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
