Abstract
Background:
Previous studies have identified low serum uric acid (SUA) levels as a risk factor for the development of Parkinson’s disease (PD). Prodromal PD mainly manifests as a complex of non-motor features, but the association between SUA levels and nonmotor symptoms (NMS) burden level in advanced PD patients is poorly studied.
Objective:
To determine the association between SUA levels and NMS in PD patients.
Methods:
Data were gathered from an open label, cross sectional, study with analysis of SUA levels in 87 PD patients and were correlated to NMS through the NMS scale (NMSS). In addition, we examined the possible relation between SUA and NMS burden levels and motor scores.
Results:
There was a moderate negative association between SUA levels and NMSS total score (ρ= –0.379, p < 0.001). In line with this, we observed that higher NMS burden was associated with lower SUA levels (p < 0.001). Within individual NMSS domains, a moderate negative correlation was observed between SUA levels and the cardiovascular/falls (ρ= –0.285, p = 0.008), sleep/fatigue (ρ= –0.299, p = 0.005), and miscellaneous domains (ρ= –0.318, p = 0.003).
Conclusion:
In this observational study we observed that SUA levels were negatively associated to NMS burden in PD patients with a specific link to miscellaneous, sleep/fatigue and cardiovascular domains of the NMSS. Interestingly, we did not find a clear relation between SUA and motor scores. Future large-scale prospective studies in de novo and advanced PD are needed to evaluate and establish these associations.
INTRODUCTION
Serum uric acid (SUA) was first described as being abnormally elevated among Parkinson’s disease (PD) patients receiving levodopa treatment almost 50 years ago [1]. An association between SUA levels and the development of PD was reported in 1996 in the Honolulu epidemiological study which involved 7,968 healthy men followed up over 30 years, 92 of them developing PD [2]. In another study (the Rotterdam study) 4,695 healthy participants over 54 years of age were followed-up over 9.4 years and were found to have a significantly decreased PD risk when they had higher SUA at baseline [3]. The largest prospective epidemiological study to date followed 18,018 healthy men from the Health Professional Follow-Up Study over a period of five to seven years and found lower SUA levels among the 84 individuals who developed PD over time, when compared to healthy controls [4, 5]. SUA can therefore be considered as a candidate serum biomarker in PD [6, 7], although altered levels of SUA are not specific to PD and have been described in a range of other medical conditions, including Alzheimer’s disease (AD) and amyotrophic lateral sclerosis (ALS). A meta-analysis by Du et al. showed an inverse relationship between elevated SUA levels and a decreased risk of developing AD [8]. In PD an association with non-motor symptoms (NMS), such as cognition, sleep and dysautonomia as well as apathy and pain have been described [9]. In ALS, there was a negative association between SUA levels and mortality risk among ALS patients and randomised studies are required [10]. In otherwise healthy subjects, higher SUA levels were associated with white matter atrophy in the brain, as well as cognitive impairment [11].
NMS have become a cornerstone in the definition of prodromal PD [12, 13] as well as being a key component of the syndromic endophenotypes of PD [14]. The range of NMS that occurs in PD is broad [15] and the overall burden of NMS impacts adversely on quality of life (QoL) in PD [16]. Specific subsets of NMS such as cognition, depression, pain, sleep, dysautonomia also impart a differential but negative impact on QoL in PD as has been shown in large scale validation and cohort studies [17]. Currently, studies which have specifically addressed the relationship between SUA level and NMS have mostly examined de novo drug naïve patients and only small studies included advanced patients [9, 18], where it was concluded that the effects of SUA on advanced PD patients remain unknown. There are currently efforts to identify NMS and some motor symptoms as biomarkers to predict the risk of developing in life manifest motor PD [19]. The latest studies on this topic have suggested an inverse correlation between SUA and NMS exists; however, the results were not uniform [20, 21].
In the current study, we therefore set out to correlate SUA with NMS burden in a larger and more diverse cohort of PD patients, using structured outcomes for NMS and motor scores.
MATERIALS AND METHODS
This was a cross-sectional, audit-based, collaborative study performed at King’s College Hospital in London and the Pirogov Russian National Research Medical University, Moscow, Russian Federation. Patients diagnosed with idiopathic PD who were seen between January 2017 and October 2018, who participated in the Non-motor International Longitudinal Study (NILS, NRES SouthEast London REC3, 10084, 10/H0808/41; http://parkinsons-london.co.uk/europar) [22] and for whom SUA levels were available as part of yearly standard lab testing in the outpatient clinic were included in the study.
Anonymised data from a total of 151 non-demented PD patients were included. All included patients satisfied the UK PD Brain Bank diagnostic criteria for PD. All SUA laboratory results were obtained from the electronic patient record systems. All patients gave written informed consent and all study procedures for the NILS study were performed in line with the Declaration of Helsinki. The main inclusion criterion was a diagnosis of idiopathic PD according to the UK Brain Bank criteria. Exclusion criteria were (1) diagnosis of Parkinsonism different to idiopathic PD; (2) dementia (defined by an MMSE score of less than 26) [23]; (3) inability for giving consent to participate in the study. The chart-review for obtaining the SUA levels was a registered audit. The data were entered into an anonymised and securely stored database (Excel 2016, ICO Data Protection License Number Z6614305) and saved on secure local hospital computers at King’s College Hospital following GDPR recommendations.
For all patients the following data were recorded: socio-demographic and historical information, including Levodopa Equivalent Daily Dose (LEDD) and Hoehn and Yahr (HY) stage. Patient-reported outcomes consisted of Mini Mental State Examination (MMSE) scores [24], Hospital Anxiety and Depression Scale (HADS) [25], PDQ-8 [26], Parkinson’s Disease Sleep Scale-version 1 (PDSS) [27], and clinician-based evaluations included Hoehn and Yahr (HY) staging [28], Scales for Outcomes in PD (SCOPA) [29], and NMSS total and domain scores [30]. The primary outcomes consisted of an association between SUA levels and NMSS scores and the relationship with NMS burden. For the latter we used four categories based on NMSS total scores: mild (1–20), moderate (21–40), severe (41–70), and very severe (>70) as per previous grading system [31]. SCOPA-Motor Scale score and SCOPA-Motor Scale domains were defined as secondary outcome variables.
Kolmogorov-Smirnov test was used for checking the distribution of data while the Spearman rank correlation coefficient was used to determine the association between SUA levels and NMS. Finally, Kruskal-Wallis test was used to assess group differences in NMS burden. Statistical significance values were set at p < 0.05 and correlation magnitude levels were defined as weak (<0.3), moderate (0.3–0.59), and strong (≥0.6) as used by Storch et al. [32]. In case of multiple testing we applied a Benjamini-Hochberg correction. IBM SPSS Version 25 (IBM SPSS Statistics for Windows, Version 25.0. Armonk, NY: IBM Corp.) was used for all statistical analyses. Data are represented as mean±standard deviation, median (25th–75th percentile) or number (percentage), unless otherwise indicated.
RESULTS
A total of 151 PD patients were considered and eligible for this study. After exclusion of participants with incomplete data, we included a total of 87 patients. The cohort of 87 PD patients consisted of 73.6% male and 26.4% female patients with a mean age of 68.34±10.34 years, disease duration of 11.92±6.50 years, median HY stage of 3.0 (25th–75th percentile 2.0–3.0), and mean LEDD of 634.97±542.25 mg. None of the patients in our cohort used urate lowering medications. In this cohort mean SUA levels were 296.99±75.86μmol/l. Further demographic data are provided in Table 1.
Socio-demographic characteristics of the Parkinson’s disease cohort
NMSB, Non Motor Symptom Burden; LEDD, Levodopa Equivalent Daily Dose; SCOPA, Scales for Outcomes in Parkinson’s disease; NMSS, Non Motor Symptoms Scale; HADS, Hospital Anxiety and Depression Scale; MMSE, Mini-Mental State Examination; PDSS, Parkinson’s Disease Sleep Scale; PDQ-8, Parkinson’s disease Quality of life Questionnaire 8; mg, milligram; L, litre; aKruskal-Wallis test; bChi-Square test; *corrected p-value (Benjamini-Hochberg correction for multiple testing). Data is presented as mean±standard deviation, median (25th–75th percentile), or number (percentage)
Spearman rank correlation analysis showed a moderate negative association between SUA level and NMSS scores (ρ= –0.379, p < 0.001) (Fig. 1). As we also observed significant associations between SUA levels and LEDD (p = 0.019) and SCOPA complications scores (p = 0.042), we performed a multiple regression analysis with these factors and NMSS score as predictors. In this model (R2 = 0.244, p = 0.005), the only significant predictor of SUA levels were NMSS scores (β= –0.312; p = 0.027). Moreover, moderate negative associations were found between the following NMSS domains and SUA levels: cardiovascular/falls (ρ= –0.285, p = 0.008), sleep/fatigue (ρ= –0.299, p = 0.005), and miscellaneous (ρ= –0.18, p = 0.003) (Fig. 1). We did not observe significant correlations between SUA levels and the remaining NMSS domains, nor with HADS, PDSS, MMSE, and PDQ-8 scores (p≥0.10).
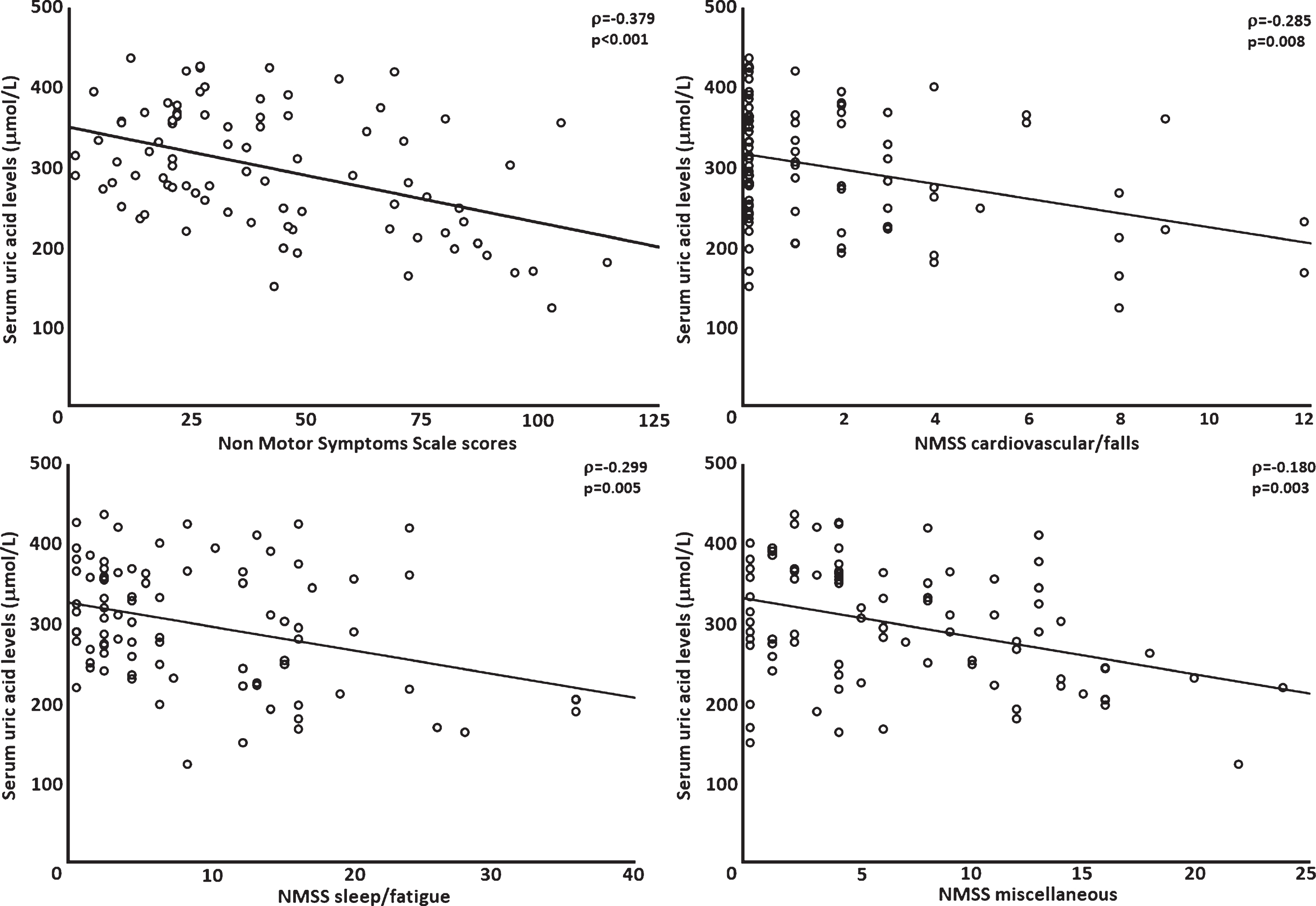
Scatter plot and regression line representing the relationship between serum urate levels and Non-Motor Symptom Scale (NMSS) total scores, and specific NMSS domains, in Parkinson’s disease patients (n = 87).
When looking at the link between SUA levels and NMSB, we observed that SUA levels were highest (314.55±53.76μmol/L) in the group of patients with mild NMSB, and lowest (230.26±67.79μmol/L) in patients with very severe NMSB (p < 0.001; Table 1; Fig. 2). The groups were well-matched for baseline demographics (p≥0.10), except for SCOPA motor and activities of daily living scores (p = 0.015; Table 1).
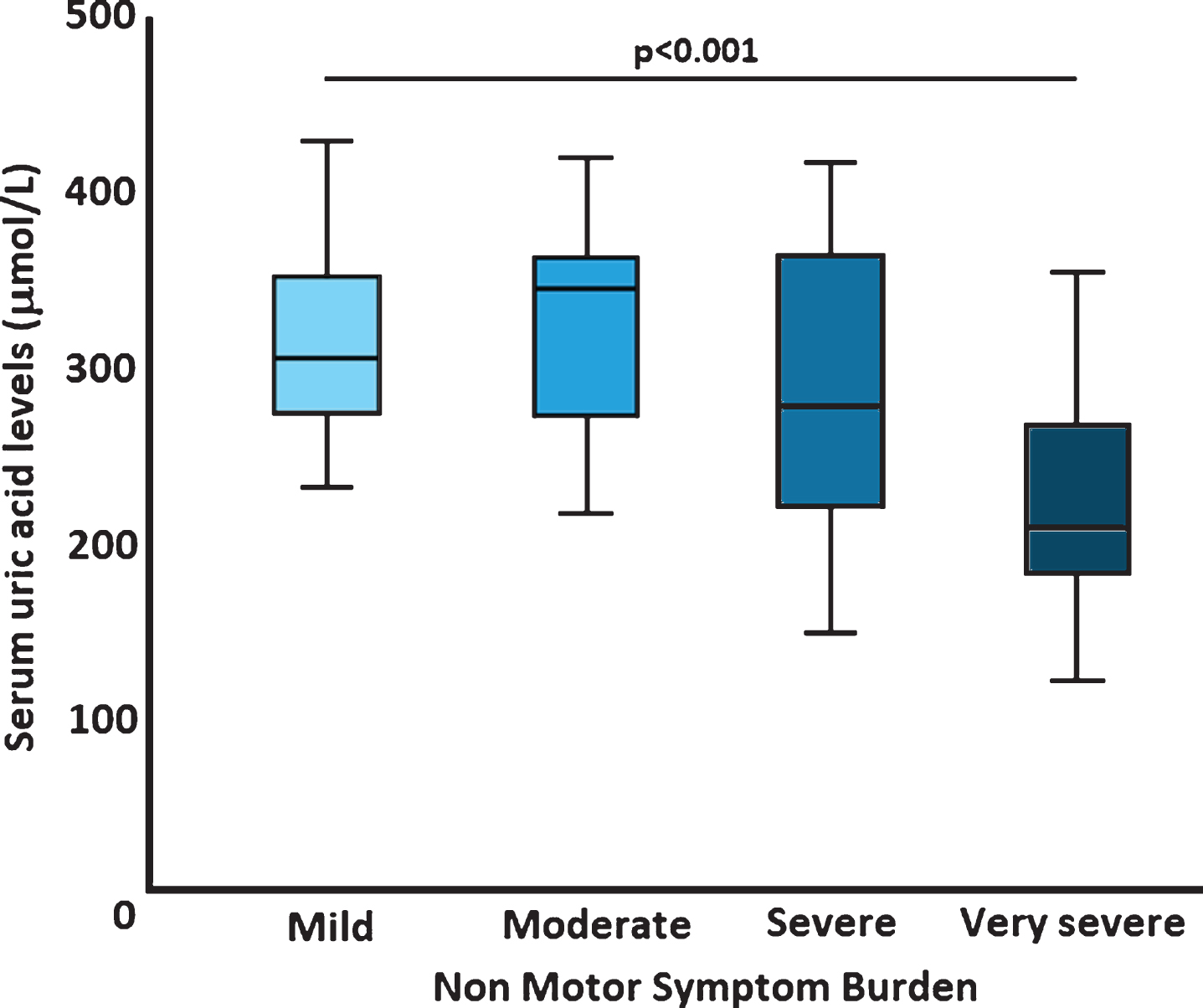
Serum uric acid levels across different non motor burdens in Parkinson’s disease patients (n = 87). Horizontal bars represent mean and error bars represent 95% confidence intervals.
DISCUSSION
In this study, we systematically correlated SUA levels with a range of NMS as well as NMS burden in a cohort of PD patients across all stages of the disease. A moderate inverse correlation was observed between SUA levels and the NMSS total score and specific correlations with the cardiovascular/falls, sleep/fatigue, and miscellaneous domains of the NMSS. In the latter domain, changes in the ability to smell, as well as suffering from pain not explained by other conditions showed moderate negative associations with SUA levels. These links have not been described before, and, as such, our findings provide novel areas to explore in future studies.
Although findings from individual studies yielded conflicting results regarding the question whether SUA levels are decreased or not in PD, a recent meta-analysis of 13 studies with over 4,500 participants convincingly showed that SUA levels are significantly reduced in PD patients, and, moreover, the levels decrease further with increasing disease progression [33]. Whether or not decreased SUA levels are also associated with non-motor features of PD remains relatively under studied. The available evidence so far shows that SUA may be protective against the development of PD dementia in male patients [34]; in PD, SUA levels appear to influence specific cognitive tasks, such as verbal fluency and vigilance [18], and SUA levels are linked to the development of apathy in early stage PD patients [21]. More recent studies in PD have, in addition, shown effects on other NMS. Huang et al. showed that higher SUA levels were associated with less fatigue [20], in line with our findings of an inverse association between NMSS sleep/fatigue scores and SUA levels. In a similar study to ours, but performed in a cohort of de novo PD patients, Moccia et al. showed that lower SUA levels were associated with higher NMSS scores for the attention/memory, mood/cognition, and cardiovascular domains [9]. The differences with our cohort, where SUA levels were associated with sleep/fatigue and the miscellaneous domains, could be explained by the differences in disease duration and the use of dopaminergic medication. On the other hand, in line with our findings, it was reported that the lowest tertile of patients with mild NMS progression presented higher SUA levels compared to those with greater NMS progression [9]. Moreover, in a recent study by Papandreou et al. in a large elderly population (n = 1842) lower SUA levels were associated with longer sleep duration [35].
As outlined above, urate appears to be linked to at least some NMS in PD, but also in other diseases similar effects have been observed [36]. How urate influences these symptoms remains unclear, but for example in PD the SCNA gene, encoding for α-synuclein, may be influenced by elevated urate levels through autophagy activation via an mTOR-dependent signalling pathway [37]. Other factors that might be influenced by urate are oxidative stress and neuroinflammation [36]. A reduction in oxidative stress by urate is believed to be induced by urate’s role as a radical scavenger for nitrogen-oxygen-based radicals [8], where at the same time it may temper increased inflammatory responses to endothelial dysfunction and vascular damage [36]. The latter, and specifically systemic C-reactive protein, have in turn been linked to white matter atrophy in the brain [38], and elevated SUA levels have been associated with brain atrophy [11].
Other mechanisms mediating the apparent protective effects of urate, are likely to involve amyloid-β, perhaps mediated via the glymphatic system [39], responsible for the removal of amyloid-β [40]. The protective effect of urate on cognitive performance, through amyloid-β, has not only been suggested for dementia in PD [41], but also in AD [42] and small vessel disease [43]. Maetzler et al. have shown that SUA levels correlated positively with amyloid-β42 levels in the cerebrospinal fluid of PD patients, and it may be assumed that SUA levels also impact on sleep, through amyloid-β. Moreover, this could provide an interesting explanation for cognitive impairments [44]. This seems to be corroborated by a recent study where researchers reported that lower SUA levels were associated with cognitive impairment, as judged by the Mini-Mental State Examination (MMSE) in PD patients [45]. Also others have reported an association of SUA levels and impaired attention and memory in PD [9]. The underlying mechanisms, as suggested before, might be amyloidergic where uric acid influences neuronal biology by altering cell viability and potentiating the proapoptotic effect mediated by amyloid-β, leading to cognitive impairment and dementia [46]. Since pathological accumulation of this protein has been considered a key player in the development of sleep disturbances in PD patients [47], the glymphatic system may also have an influence on this mechanism.
As with most research studies, there are several limitations to our study. First, this was cross-sectional, one-point-in-time, study without a control group, calling for further controlled, prospective, studies. A second limitation is that our relatively small sample size, accounting for the only moderate magnitudes found in our correlation analyses. This is also highlighted by the lack of a clear association between SUA levels and motor symptoms in our cohort of PD patients, as opposed to previous studies. Nevertheless, we feel our data are still meaningful as we used a structured approach to determine non-motor symptoms and burden through the validated NMSS and the narrow time window between the assessment of SUA levels and the non-motor assessments.
In summary, we observed that SUA levels in advanced PD patients were correlated with NMS burden. In addition, we observed moderate inverse correlations between SUA levels and the NMSS specific cardiovascular/falls, sleep/fatigue, and miscellaneous NMSS domains. These links to specific NMS have not been described before in the literature and provide novel areas to explore. Future prospective and longitudinal trials should confirm our results and compare the findings in PD patients to healthy controls subjects, as also the latter population exhibits non-motor symptoms.
CONFLICT OF INTEREST
The authors declare no conflict of interest.
Footnotes
ACKNOWLEDGMENTS
We acknowledge data collection efforts by all contributors, collaborators, and administrative staff of the NILS study, as well as to the UK NILS collaborators Dr B Kessel, Professor A Schapira, and Drs S Pal, JC Sharma, PF Worth, R Sophia, M Silverdale and Cathy Ellis for their contribution to the study. The views expressed are those of the author(s) and not necessarily those of the NHS, the NIHR or the Department of Health. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
