Abstract
Background:
Parkinson’s disease (PD) is the second most common neurodegenerative disorder, but the disease-modifying therapies focusing on the core pathological changes are still unavailable. Rho-associated protein kinase (ROCK) has been suggested as a promising target for developing neuroprotective therapies in PD.
Objective:
We aimed to explore the promotion of α-synuclein (α-syn) clearance in a rat model.
Methods:
In a rat model induced by unilateral injection of adeno-associated virus of serotype 9 (AAV9) expressing A53T α-syn (AAV9-A53T-α-syn) into the right substantia nigra, we aimed to investigate whether Fasudil could promote α-syn clearance and thereby attenuate motor impairments and dopaminergic deficits.
Results:
In our study, treatment with Fasudil (5 mg/kg rat weight/day) for 8 weeks significantly improved the motor deficits in the Cylinder and Rotarod tests. In the in vivo positron emission tomography imaging with the ligand 18F-dihydrotetrabenazine, Fasudil significantly enhanced the dopaminergic imaging in the injected striatum of the rat model (p < 0.05 vs. vehicle group, p < 0.01 vs. left striatum in Fasudil group). The following mechanistic study confirmed that Fasudil could promote the autophagic clearance of α-syn by Becline 1 and Akt/mTOR pathways.
Conclusion:
Our study suggested that Fasudil, the ROCK2 inhibitor, could attenuate the anatomical and behavioral lesions in the Parkinsonian rat model by autophagy activation. Our results identify Fasudil as a drug with high translational potential as disease-modifying treatment for PD and other synucleinopathies.
Keywords
INTRODUCTION
Parkinson’s disease (PD) is the second most common neurodegenerative disorder, the prevalence of which will inevitably increase due to the aging of population [1]. However, the disease-modifying therapies based on the pathological changes remain unavailable [2]. The abnormal accumulation and aggregation of α-syn has been implicated in the core pathogenesis of PD, focusing therapeutic strategies on the clearance of pathological α-syn or reduction of α-syn production.
Rho-associated protein kinase (ROCK) has been suggested as a promising target for developing neuroprotective therapies in PD, basing on accumulating evidence over the past decades [3, 4]. In brain samples of PD patients, ROCK2 expression was shown to be increased in astrocytes compared to age-matched healthy controls [5]. The neuroprotective effect of Rock inhibitors was initially shown in the MPTP model showing effects on the microglial neuroinflammatory response [6, 7] or axonal protection [8]. Then the neuroprotective effect was also observed in other PD models including 6-hydroxydopamine [9], LPS [10] and α-syn over expression [3]. We previously reported the activation of autophagy by Fasudil in an in vitro model of α-syn overexpression [11]. Recently, effects on mitophagy were also reported [12]. However, the in vivo explorations on the Fasudil-promoted α-syn clearance are limited.
In the present study, we aimed to study whether Fasudil could promote the clearance of α-syn in the virus-mediated rat model of PD-alpha synucleinopathy and furthermore to determine the mechanisms involved.
MATERIALS AND METHODS
Animals
The animals used in this study were female Sprague-Dawley (SD) rats (Shanghai SLAC Laboratory Animal Company, Shanghai, China) as previously reported [13, 14], 2-months of age and weighed, on average, 233.76±10.74 g at time of surgery.
Viral vectors used and surgical delivery
The PD rat model employed was based on unilateral injection of adeno-associated virus of serotype 9 (AAV9) expressing A53T α-syn (AAV9-A53T-α-syn) into the right SNpc [13, 15] and used a pAAV-CAG-MCS with a chicken beta actin (CBA/CAG) promoter (Biowit, Shenzhen, China).
The rats were anaesthetized with 10% chloral hydrate and stereotaxically injected with 2 μl of either AAV9-empty vector (EV) or AAV9-A53T-α-syn to the SNpc (AP, –5.2 mm; ML, –2.0 mm; DV, –7.5 mm (from skull at bregma)) at a rate of 0.2 μl/min using a microinjector (Stoelting, Kiel, WI). The concentration of each vector was 5.81×1012 genomic particles (gp)/ml. Coordinates were based on the atlas of Paxinos and Watson (1997).
The rats were housed in a temperature-controlled environment with regular 12 h light/dark cycle, and allowed food and water ad libitum. All procedures were conducted in accordance with the guidelines of the International Council for Laboratory Animal Science. The study was approved by the Ethics Committee of Department of Laboratory Animal Science, Fudan University, Shanghai, China. The approval number from IRB was “20171552A498”.
Fasudil treatment
Fasudil was dissolved in normal saline at the concentration of 1.25 mg/mL before injection. Since Day 14 after the surgery, the rats were intraperitoneally injected twice with Fasudil (5 mg/kg rat weight/day) or vehicle (normal saline), and the injection lasted for the following 8 weeks (Fig. 1A).
In our study, 20 rats were included in the A53T α-syn group (vehicle/AAV9-A53T-α-syn injection), 20 rats in the Fasudil treatment group (Fasudil/AAV9-A53T-α-syn injection), 20 rats in the control group (vehicle/AAV9-empty-vector injection), and 20 rats in the Fasudil treated control group (Fasudil/AAV9-empty-vector injection).
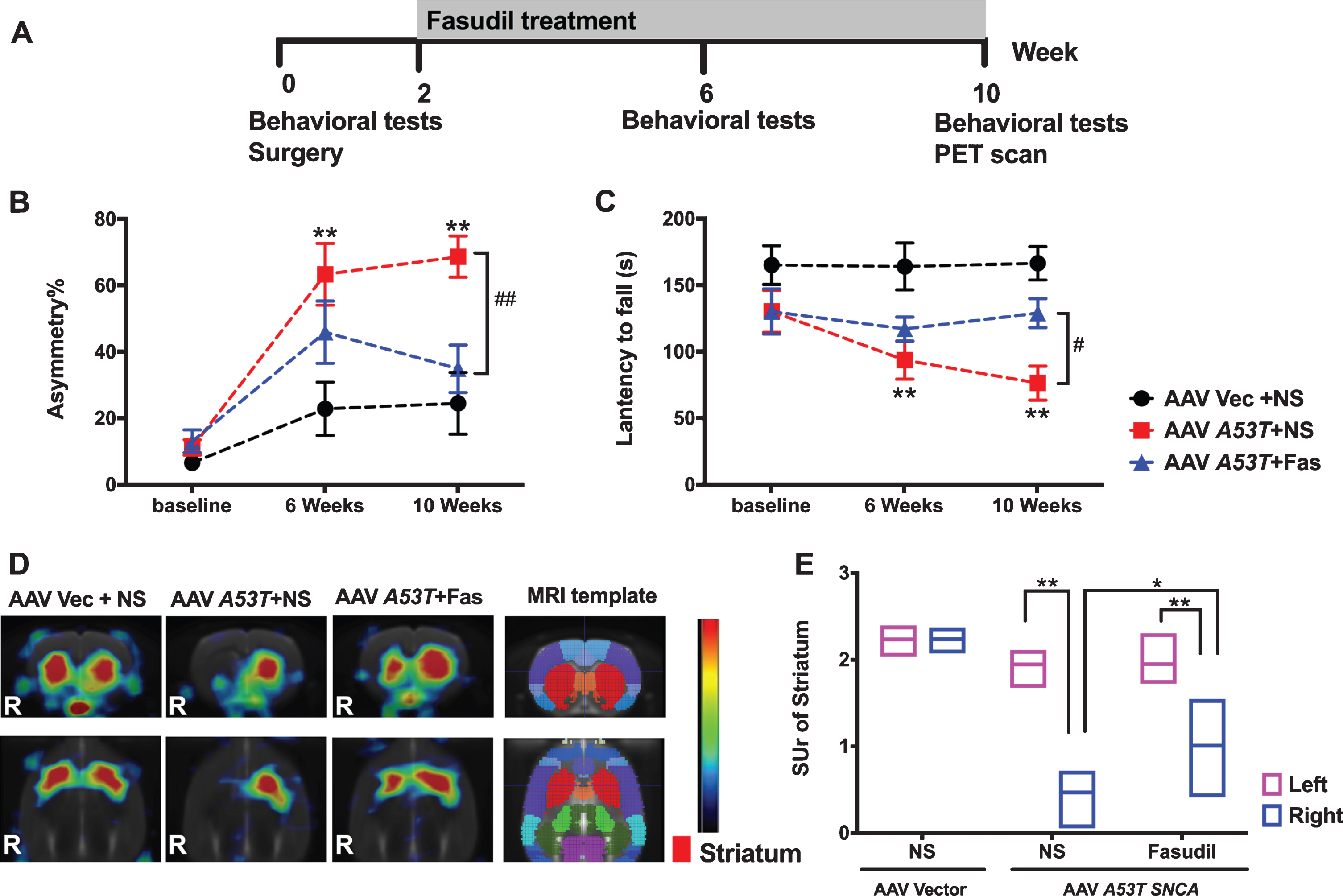
Fasudil attenuated the motor impairments and brain lesions induced by A53Tα-syn. A) The workflow of the present study. B) The asymmetry frequencies detected in the cylinder tests increased significantly 6 weeks and 10 weeks after the AAV-A53T-α-syn injection. The treatment of Fasudil significantly improved the A53Tα-syn induced asymmetry at Week 10. C) The latencies to fall in the Rotarod tests decreased significantly in the A53T-α-syn model group at Week 6 and Week 10, and the treatment of Fasudil greatly increased the latencies to fall at Week 10. (**p < 0.01 vs. control group. # #p < 0.01 vs. A53T-α-synmodel group. #p < 0.05 vs. A53T model group). D) The VMAT2 bindings in A53T-α-synmodel group decreased greatly in the PET imaging, and Fasudil treatment greatly attenuated the VMAT2 bindings in the AAV-A53T-α-syn injected striatum. (R, Right side) E) The quantitative comparation of the VMAT2 bindings in the striatums of different groups (**p < 0.01 vs. the left side without AAV injection. *p < 0.05 Fasudil treated vs. Fasudil untreated AAV-A53T-α-syn injected striatum). AAV, adeno-associated virus. Vec, vector; NS, normal saline; Fas, Fasudil.
Behavioral analysis
The behavioral tests (Rotarod test and Cylinder test) were performed at baseline just before surgery, 6 weeks and 10 weeks after surgery (Fig. 1A). The Cylinder tests were performed and calculated as described previously [13]. The Rotarod tests were performed as described in previous work [16]. Briefly, the rats were placed on a rod accelerating smoothly from 4 to 40 rpm throughout over 5 min. The latencies to fall were recorded and evaluated among different groups.
PET/CT imaging
We tested the synaptic disruption in vivo by analyzing the binding of 18F-dihydrotetrabenazine (DTBZ) to the vesicular monoamine transporter 2 (VMAT2) with positron emission tomography (PET) imaging at Week 10. As described in a previous study [17], the PET scan was performed 60 min after the intravenous18F-DTBZ (approximately 37–55.5 MBq) in each rat 10 weeks after the surgery. The quantitative analysis of 18F-DTBZ binding was analyzed with PMOD software (version 3.2, PMOD Technologies, Zurich, Switzerland). The brain regions including ipsilateral striatum (I-St), contralateral striatum (C-St), and cerebellum (CB), were first shown as standardized uptake value (SUV) by the formula: SUV measured tissue activity [Bq/ml]/(injected dose [Bq]/body weight [g]). The specific uptake ratio (SUr) of striatum was calculated as [(uptake in St-uptake in CB)/(uptake in CB)]. The percentage of SUr reduction at each region of interest (ROI) was calculated as [(SUr of control group-SUr of interested group)/SUr of Control group]×100%.
Postmortem measures
Ten weeks after surgery, the rats were sacrificed by administering an overdose of chloral hydrate (1.0 ml of 240 mg/ml, i.p.), and killed by exsanguination by transcardial perfusion with saline followed by 4% paraformaldehyde. The brains were then removed and fixed overnight with 4% paraformaldehyde in phosphate-buffered saline (PBS), cryoprotected in 10%, 20%, and 30% sucrose, embedded in OCT, and cryostat sectioned at 20 μm.
Immunofluorescence
Tissue sections were blocked with 3% bovine serum albumin (BSA) to eliminate non-specific binding, and permeabilized with 0.3% Triton X-100 in 1% BSA-PBS for 30 min. The sections were incubated with the following primary antibodies, rabbit anti-TH (sc-sc-73152, 1 : 250, Santa Cruz, USA) and anti-α-syn (ab138501, 1 : 200, Abcam, USA) at 4°C overnight, followed by a series of washes and incubation with goat anti-mouse IgG(H + L) Highly Cross-Adsorbed Secondary Antibody, Alexa Fluor 594 (#A-21145, 1 : 3000, Thermofisher, USA) and goat anti-rabbit IgG(H + L) Alexa fluor 488 Conjudate (#A-11034, 1 : 3000, Thermofisher, USA) at room temperature for 2 h. All sections were counterstained with DAPI to reveal the nucleus. Negative control sections were treated equally only without primary antibodies. For each animal, ten sections were examined in a double-blind manner.
Immunohistochemistry
The immunochemistry analysis was performed as we previously reported [13, 14]. A single series of sections was processed for visualization of tyrosine hydroxylase (TH) and α-syn via thebiotin-labeled antibody procedure. Briefly, following several washes in a PBS-T solution (0.05% tween 20 in PBS), antigens were repaired with 0.01 M citrate buffer to at 95–100°C for 10 min, endogenous peroxidase was then quenched in a 0.3% hydrogen peroxide solution containing 0.2% TritonX-100 (in TBST) for 30 min, and background staining was then inhibited in 2% bovine serumalbumin solution. Tissue was then incubated with primary antibodies overnight: rabbit anti-TH antibody (1 : 500, polyclonal, #NB300-109,Chemicon, USA) or rabbit anti-α-syn antibody [MJFR1] (1 : 500, monoclonal, #ab138501, Abcam, USA). After three washes in PBS-T, sections were sequentially incubated in biotinylated goat anti-rabbit IgG (1 : 300; Vector, Burlingame, CA) for 1 h and the Eliteavidin-biotin complex (ABC Kits; Vector, Burlingame, CA) for 45 min separated by three washes in PBS. Immunological staining was observed after reaction with DAB solution (0.05% DAB + 0.03% H2O2-Tris-Cl pH7.6)(Sigma). Then dehydrated with gradedethanol (75% –85% –95% –100%) each for 5 min, then in ethanol xylene, xylene each for 5 mins. Last, the resin mounted on glass slides. In the Nissl staining, the sections reacted with DAB peroxidase substrate (SK-4100, Vector) first and then they were counterstained with Nissl (C0117, Beyotime, Shanghai, China).
Stereology
The stereological estimation of DA neuron numbers in the SN was performed using optical fractionator (StereoInvestigator, v. 7, MBF Biosciences, Williston, VT) as described before [13]. Ten sections spanning the entire anterior/posterior extent of the SN were used for counting. All TH-ir neurons of the SN were included within each contour of each section. Parameters used were grid size, 300 μ×300 μm; counting frame, 80 μ×80 μm; and 2 μm guard zones. Tissue thickness was determined by the user at each counting site. All final values represent estimated total by number weighted section thickness and were only included if their Gunderson coefficient of error (m = 1) was less than 0.09. The process was performed in a blinded manner using coded slides.
Transmission electron microscope (TEM) imaging
As previously reported [11, 18], the autophagosome was assessed with TEM. We randomly selected 3 rats from each group for TEM imaging with a computer-assistant random number table. The brain slides from different groups were fixed in ice-cold 2.5% glutaraldehyde in 0.1 mol/L PBS, post fixed for 1 h in 1% osmium tetroxide in the same buffer, and dehydrated in graded alcohols and acetones. After perfusion, the sections containing SNpc were collected according to the brain atlas of rat, and finally a total of 2 mm3 tissue from the middle part of SNpc in right hemisphere was adopted. And at least 3 ultrathin sections of 80 nm thick for one tissue sample were prepared with an ultra microtome, in the middle and near the two sides of this sample. The sections were then stained with 3% uranyl acetate and lead citrate, and examined with TEM (PHILIPS CM-120, Netherlands). After glancing over the whole section, four fields with dopaminergic neurons were picked in each ultrathin section. Typical changes of autophagosomes were selected, but the number was not quantified in the current study.
Cell culture and cycloheximide (CHX) intervention
The SH-SY5Y cells overexpressing A53T-α-syn were constructed as previously described [11]. The SH-SY5Y-A53Tcells were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM, Invitrogen, USA) supplemented with 10% (v/v) fetal bovine serum (HyClone, USA) and Geneticin (200 μg/ml, Invitrogen, USA). The medium was replaced every 2 days and cultures were maintained in an atmosphere of 5% CO2 and 95% O2 humidified air at 37°C.The cells were treated with cycloheximide (CHX, 25 μg/mL) first to inhibit protein synthesis, and then treated with Fasudil (15 μg/ml) for 24 h, 36 h, and 48 h separately.
Western blot analysis
Following transcardial perfusion the brain was rapidly removed and the SN fresh dissected and frozen on dry ice. SN tissues were prepared for western blot analysis. Briefly, tissue was homogenized in ice-cold lysis buffer (1×RIPA, Thermo Fisher Scientific), supplemented with protease inhibitors by a microcontent motor-operated tissue homogenizer (Kimblekonts, USA). Lysates were then centrifuged at 10,000 g for 20 min at 4°C, and the supernatants were collected for western blot. Equal amounts of protein (20 μg) were subjected to SDS-PAGE, transferred to PVDF membrane, blocked, and probed overnight at 4°C with the primary antibody of either p-MYPT (Thr696) (rabbit anti-human, 1 : 1000, monoclonal, #5163, CST, USA), VMAT2 (rabbit anti-human, 1 : 1000, polyclonal, #ab70808, Abcam, USA), TH (rabbit-anti-human, 1 : 1000, polyclonal, #ab112, Abcam, USA), α-syn (rabbit-anti-human, 1 : 1000, monoclonal, #ab138501, Abcam, USA), LC3-I/II (rabbit anti-human, 1 : 1,000, polyclonal, #NB100-2200, NOVUS, China), α-synSer129(rabbit anti-human, 1 : 1,000, polyclonal, #ab51253, Abcam, USA), p-JNK 1(Thr183/Tyr185) (rabbit anti-human, 1 : 1,000, polyclonal, #9251, CST, USA), Blc-2 (rabbit anti-human, 1 : 1,000, monoclonal, #15071, CST, USA), p-Bcl-2 (Ser70) (rabbit anti-human, 1 : 1,000, monoclonal, #2827, CST, USA), Beclin-1(rabbit anti-human, 1 : 1,000, monoclonal, #3495, CST, USA) or 6X His tag (mouse, 1 : 1,000, monoclonal, #ab18184, Abcam, USA).
To control for protein loading, GAPDH (mice anti-human, 1 : 1000, monoclonal, #60004-1-Ig, Proteintech, USA) was used. After washing in TBST, secondary antibody (goat anti-rabbit or goat anti-mice IgG-HRP, 1 : 3000, Jackson, USA) was added and developed with enhanced chemiluminescence as previously described [19]. The blots were finally visualized and quantified with Quantity Software (Bio-Rad, CA). Quantitative results were obtained from six rats in each group.
Statistical analysis
All values were presented as mean±SEM. Analyses were performed using SPSS 22.0 (SPSS Inc., Chicago, IL) and graphs were visualized using Graph Pad Prism 7 (San Diego, CA, USA) software. One-way ANOVA was applied for the experiments involving three or more experimental groups, followed by Tukey’s multiple comparisons. Results with comparisons that produced P values <0.05 were considered statistically significant.
RESULTS
Treatment with Fasudil attenuates the behavioral impairments induced by AAV9 A53T α-syn
In rats receiving AAV9 A53T α-syn, a significant motor impairment was produced when assessed at Weeks 6 and 10 (Fig. 1A–C). The intraperitoneal injection of Fasudil (5 mg/kg rat weight) greatly attenuated the behavioral impairments induced by AAV9-A53T-α-syn. Specifically, AAV9-A53T-α-syn rats showed greater forelimb use asymmetry compared to empty vector controls when measured at Weeks 6 and 10 (63.24% and 68.55%, respectively) (Fig. 1B). The treatment of Fasudil improved the forelimb use asymmetry to 45.81±9.38% at Week 6 (vs. vehicle /AAV9-A53T-α-syn group at Week 6, p = 0.08) and 34.80±7.15% at Week 10 (vs. vehicle/AAV9-A53T-α-syn group at Week 10, p < 0.01) (Fig. 1B). In the Rotarod tests, the average latency to fall in the A53T α-syn group decreased to 93.35±14.28 s at Week 6 (vs. vehicle/AAV9-empty-vector group at Week 6, p < 0.01) and decreased to 76.08±12.80 s at Week 10 (vs. vehicle/AAV9-empty-vector group at Week 10, p < 0.01) (Fig. 1C). The treatment of Fasudil improved the latency to fall to 116.75±9.06 s at Week 6 (vs. vehicle/AAV9-A53T-α-syn group at Week 6, p = 0.25) and 128.76±10.93 s at Week 10 (vs. vehicle/AAV9-A53T-α-syn group at Week 10, p < 0.05) (Fig. 1C). All data were compared with the rats injected with empty vector.
Treatment with Fasudil attenuates deficits in striatal VMAT2 (DBTZ-PET)
Using Micro-PET/CT imaging, the unilateral injection of AAV9 A53T α-syn to the right SN significantly decreased VMAT-2 binding by 75.55±7.50% compared to the contralateral side 10 weeks after AAV surgery (p < 0.01) (Fig. 1D, E). In animals treated with Fasudil, deficits in VMAT-2 binding was significantly reduced to 48.25±14.53% (p < 0.05 vs. right striatum in vehicle/AAV9-A53T-α-syn group, p < 0.01 vs. left striatum in Fasudil/AAV9-A53T-α-syn group) (Fig. 1D, E).
Fasudil treatment reduces TH-positive neuronal loss and accumulation of α-syn in the SN
A significant loss of tyrosine-hydroxylase-immunoreactive (TH-ir) neurons in the SN was observed in rats having received AAV9-A53T-α-syn (Fig. 2A–C; 45.58±6.66% of the empty vector controls, p < 0.01). Treatment with Fasudil significantly protected against this DA neurons loss (Fig. 2A–E) and decreased the α-synuclein-induced ROCK activation (Fig. 2A, F) by recovery to 73.94±4.02% DA neuronal survival of the empty vector controls (Fig. 2C).
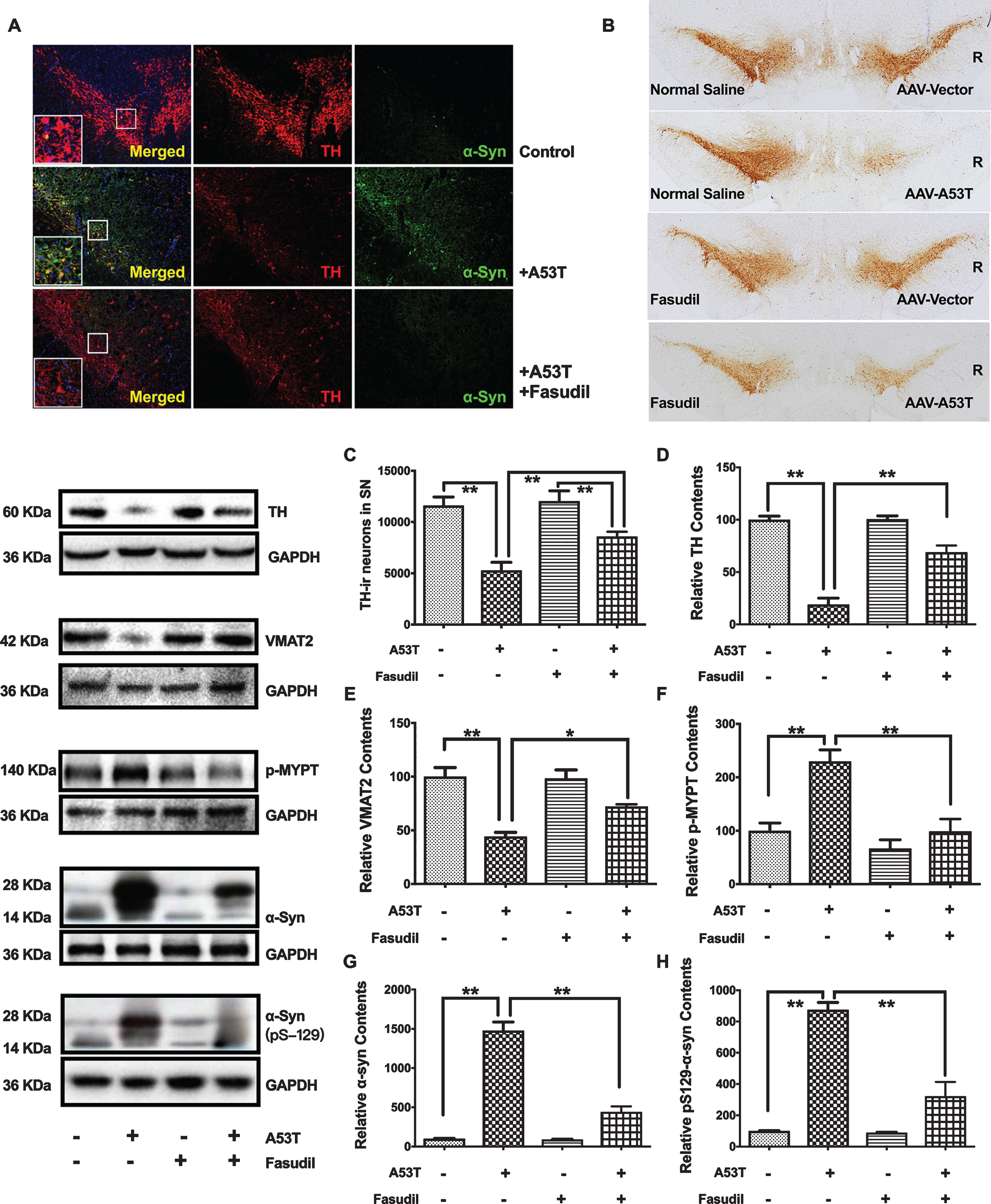
Fasudil greatly improved the TH-positive neuronal survival and inhibited α-syn accumulation. A) Treatment of Fasudil for ten weeks greatly improved the TH-positive (Red) staining and decreased the α-syn (Green) staining in the immuno fluorescence staining of the right SN injection of AAV9-A53T α-syn. B) Treatment of Fasudil for ten weeks significantly improved the neuronal loss induced by the right SN injection of AAV9-A53T α-syn. C) Stereological counting of TH-ir neurons in the right SN. **p < 0.01 vs. the SN injection of AAV9-A53T α-syn. D-H) The TH, VMAT2, p-MYPT, α-syn andα-synSer129expression in the western blotting. Quantitative results were obtained from among at least six rats. **p < 0.01 compared with vs. the SN injection of AAV9-A53T α-syn. *p < 0.05 compared with vs. the SN injection of AAV9-A53T α-syn.
The potential for Fasudil to modify α-syn accumulation in the AAV9 A53T α-syn rat model was evaluated by double label immunofluorescence and western blot. Figure 2A shows the co-localization of α-syn (red) and TH-ir neurons (green) in the SN, and Fig. 2G and 2H shows the increase of α-syn protein by western blot, both as a result of AAV9 driven overexpression of α-syn. Compared with vehicle/AAV9-A53T-α-syn rats, Fasudil significantly decreased α-syn levels in the right SN (Fig. 2G, H). In support of these findings in vivo, SH-SY5Y cells (pre-treated with CHX) Fasudil treatment significantly promoted α-syn degradation (Fig. 3).
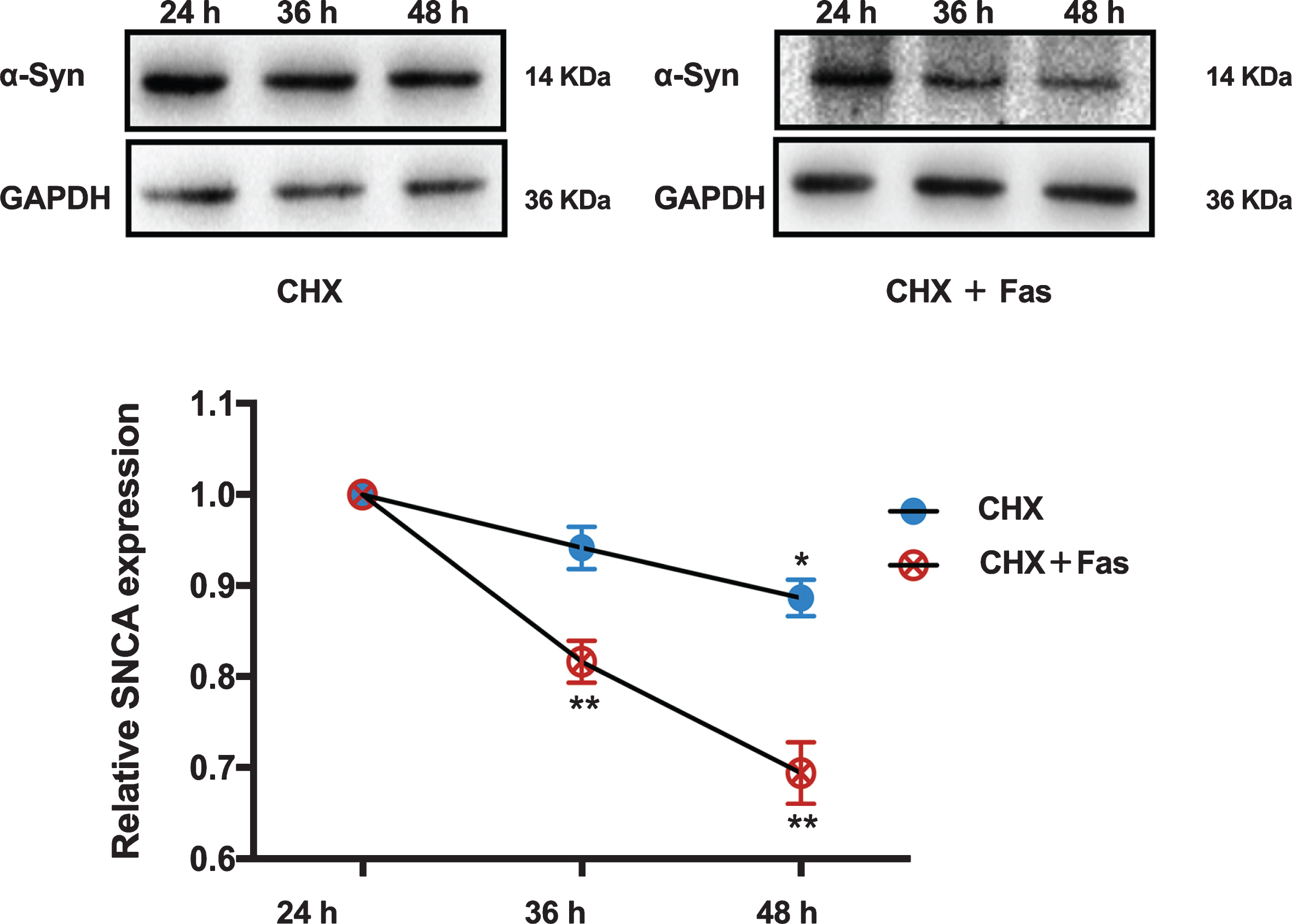
Fasudil promoted the α-syn clearance in the SH-SY5Y cells overexpressing A53Tα-syn. Quantitative results were obtained from at least six samples. *p < 0.05 vs. the SH-SY5Y cells treated with CHX for 24 h. **p < 0.01 vs. the SH-SY5Y cells treated with CHX and Fasudil for 24 h.
Fasudil treatment enhances autophagy
Treatment with Fasudil enhanced the formation of autophagosome in the SN of rats with AAV9-A53T-α-syn injection in the TEM analysis compared to AAV9 vehicle/AAV9-A53T-α-syn controls (Fig. 4A). Consistent with these results, the ratio of LC3-II to LC3-I and the ratio of LC3-II to GAPDH were both increased in Fasudil-treated rats (Fig. 4B), suggesting that Fasudil enhanced macroautophagic activity. These findings could be related to the clearance of abnormal α-syn accumulation. Fasudil further showed JNK 1-activation via phosphorylation (Fig. 4C) of Bcl-2 at Ser70 (Fig. 4D) and increases in Beclin 1 (Fig. 4E). The activation of JNK 1/Bcl-2/Beclin 1 could be responsible for initiating the formation of macro autophagosomes.
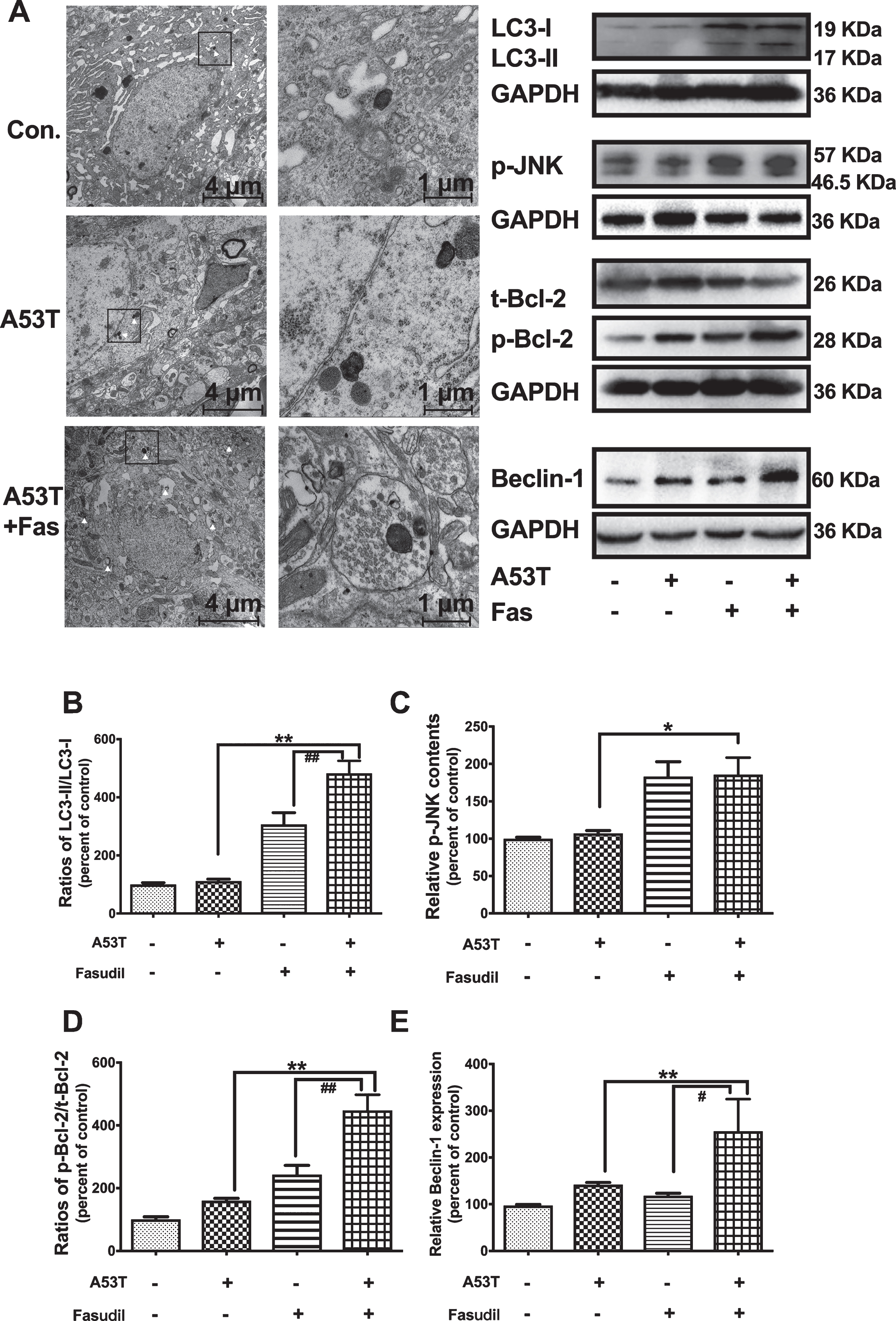
Fasudil activated the autophagic clearance of α-syn via JNK/Bcl-2/Beclin-1 pathway. A) Fasudil promoted the autophagosome formation as detected by TEM. The white arrow pointed to autophagosome. B–E) The ratio of LC3-II/LC3-I, the p-JNK expression, the ratio of p-Bcl-2/t-Bcl-2, and Beclin-1 expression was assessed by western blotting. Quantitative results were obtained from among at least six rats. **p < 0.01; *p < 0.05 compared with the SN injection of AAV9-A53T α-syn with normal saline. # #p < 0.01; #p < 0.05 compared with the SN injection of AAV9-vector and Fasudil.
DISCUSSION
The abnormal accumulation and aggregation of α-syn [20] is the pivotally implicated in the pathogenesis of PD [21]. In the present study, we reported the positive effects of Fasudil on the activation of macro autophagy, clearance of α-syn aggregation, protection of nigrostriatal function, and attenuation of the behavioral deficits in the rat model of PD with vehicle /AAV9-A53T-α-syn injection. Therefore, Fasudil is a promising medicine to be further studied in the disease-modification explorations of PD.
In this study, Fasudil could ameliorate the behavioral impairments induced by α-syn overexpression. Furthermore, such protective effects were further confirmed by the in vivo dopaminergic neuroimaging. As previously reported, in some PD models induced by MPTP or 6-hydroxydopamine, Fasudil could prevent the loss of dopaminergic neurons, and attenuate the process of neurodegeneration. In a lipopolysaccharide (LPS)-induced mice model of PD, Fasudil could inhibit the increase of α-syn induced by intranasal LPS, and attenuated the LPS-induced behavioral impairments [10]. In a transgenic mouse model of A53Tα-syn (α -synA53Tmice), Tatenhorst L reported that Fasudil treatment reduced the α-syn pathology in the midbrain of α-synA53Tmice, and improved the motor performance in Catwalk analysis [3]. All these studies suggested some neuroprotective effects of Fasudil in PD, and inspired us to further explore the possible mechanisms underlying these effects.
Although many studies supported the neuroprotective effects of Fasudil, the underlying mechanisms were still not clear, and remained to be further elucidated. Based on previous reports, the neuroprotective effects of Fasudil might be caused by the following mechanisms. First, Fasudil might promote the regeneration of dopaminergic neurons [22], thus offering a promising cell-based therapy for neuronal lesions in neurodegenerative disorders. However, the evidence is quite limited. Second, Fasudil might stabilize the damaged axon to restore the neuron function by ROCK inhibition [23]. Besides that, ROCK inhibition also promoted the neuroprotective survival cascades activation [8] and enhanced the expression of anti-oxidative factors [24]. Not limited to neurons, Fasudil intervention could also promote the shift from inflammatory M1 to anti-inflammatoryM2 microglia in experimental PD models in glia-derived mechanisms [10, 24]. In our studies, we focused mainly on the effects of Fasudil α-syn, which played an initial and core role in synucleinpathies.
In the present study, we detected the effects of Fasudil on α-syn in a rat model induced by unilateral injection of AAV-A53T-α-syn in the SN [14]. In our study, Fasudil inhibited ROCK activation and promoted the clearance of α-syn, which was caused by the induction of macroautophagy by Fasudil. The activation of macro autophagy induced by Fasudil was mediated by the Becline 1 and Akt/mTOR1 pathways. Similar activation of autophagy by Fasudil could also be supported by previous studies in myocardial cell lines [25] and glioblastoma cells [26]. In our Supplementary Material, the ROCK inhibitor Y-27632 could also promote the α-syn clearance, supporting the effects of ROCK inhibition on α-syn clearance. However, in a previous report, the more selective ROCK inhibitor Y-27632 failed to affect α-syn aggregation, suggesting the effects of Fasudil might beyond ROCK inhibition [3]. In Zhou’s reports, the inhibition of ROCK signaling pathway resulted in a substantial reduction in the expression of α-syn mRNA and protein [27]. In a study performed in α-synA53T transgenic mice, Fasudil interacted with α-syn specifically in residues Y133 and Y136, and attenuated α-syn pathology [3]. All these effects of Fasudil reflected that Fasudil might attenuate α-syn pathology by multiple targets.
As we reported, Fasudil might activate macro autophagy to deplete the aggregation prone proteins, suggesting it a promising compound in neurodegenerative disorders, such as Alzheimer’s disease, Huntington’s disease, and amyotrophic lateral sclerosis. Considering the fact that Fasudil has been approved for clinical use in many countries, it has a promising translational potential for clinical practices in neurodegenerative disorders.
Conclusions
Fasudil could promote the macro autophagic clearance of α-syn by Becline 1 and Akt/mTOR pathways. Such autophagic activation contributed to the neuroprotective effects on the parkinsonian lesions induced by α-syn accumulation, and finally improve the behavioral impairments. Our results might promote the translational therapies development in the disorders with abnormal proteins accumulation.
CONFLICT OF INTEREST
The authors declare that they have no competing of interest.
Footnotes
ACKNOWLEDGMENTS
This work was supported by the China Postdoctoral Science Foundation (grant number: 2017M610227, 2018T110350); the Ministry of Science and Technology of China (grant number: 2016YFC1306500, 2016YFC1306504); the National Nature Science Foundation of China (grant number: 81771372, 81571232, 81701250), and Shanghai Municipal Commission of Science and Technology (grant number: 2018SHZDZX01, 17JC1401400).
