Abstract
Background:
Parkinson’s disease and cardiovascular disease are highly prevalent conditions in the elderly. Evidence shows inconsistent findings regarding the association between Parkinson’s disease and cardiovascular events.
Objective:
We sought to evaluate the proportion of cardiovascular adverse events among Parkinson’s disease patients included in the placebo arm of randomized controlled trials.
Methods:
For this systematic review and meta-analysis, we searched MEDLINE, EMBASE, and the Cochrane Central Register of Controlled Trials from inception to February 2017. Randomized, placebo-controlled trials in Parkinson’s disease were included. The primary outcome was the proportion of major cardiovascular adverse events, defined as myocardial infarction, stroke, peripheral artery disease, and sudden death. A random-effects meta-analysis was performed to derive pooled estimates of the proportion of adverse events and corresponding 95% confidence intervals (CIs).
Results:
236 randomized controlled trials were included, 80% (n = 189; 14704 patients) of which reported data on cardiovascular adverse events. The pooled proportion of major cardiovascular events ranged from 0.00% to 0.06% and the proportion of all cardiovascular adverse events was 3.33% (95% CI: 2.14, 4.70%), and ranged from 1.71% in de novo Parkinson’s disease patients to 4.56% in patients receiving levodopa as their only antiparkinsonian medication. The most common adverse events were hypertension and orthostatic hypotension.
Conclusions:
These results suggest that the proportion of major cardiovascular adverse events is low and that blood pressure abnormalities are the most frequent cardiovascular adverse event.
Keywords
INTRODUCTION
Parkinson’s disease (PD) is a common late-life neurodegenerative disorder that affects about 1-2/1000 of the population worldwide, with a prevalence that increases with age [1]. Cardiovascular disease (CVD) is another common condition that is also more prevalent with advancing age [2]. Additionally, PD patients are vulnerable to autonomic dysfunction and adverse events of antiparkinsonian drugs and those factors can modulate cardiovascular risk in PD patients [3].
The association of PD with cardiovascular events is still poorly understood [4]. There are inconsistent findings in the published literature concerning the association between PD and cardiovascular disease risk [5–12]. A recent systematic review of observational studies showed a putative increased risk of stroke in PD patients but did not find an increased risk of myocardial infarction or cardiovascular mortality [13, 14]. However, the studies that were reviewed failed to control for cardiovascular risk factors, which could lead to biased conclusions.
In the absence of large and well-characterized prospective cohort studies designed to investigate CVD in PD, we have the possibility of looking toward randomized controlled trials (RCTs) to obtain and analyze data on cardiovascular adverse events in patients who are closely followed for defined periods.
We, therefore, conducted a systematic review and meta-analysis of RCTs to evaluate the proportion and typology of cardiovascular adverse events among patients with PD.
MATERIALS AND METHODS
This systematic review is reported according to the PRISMA guidelines [15]. We included all double-blind RCTs with parallel or crossover designs and at least one active treatment arm from inception to February 2017.
Patients required a diagnosis of PD according to the criteria applied in the clinical trial protocol.
Studies were included regardless of PD stage, clinical indication, comorbid conditions, previous medication, or age.
Data sources and search method
We searched bibliographic databases including MEDLINE, EMBASE and the Cochrane Central Register of Controlled Trials (CENTRAL)combining the terms (placebo OR sham OR dummy) and (Parkinson* OR Parkinsonism).Additionally, we searched clinical trials registries, the WHO International Clinical Trials Registry Platform, and clinicaltrials.gov. Finally, the reference lists from the identified articles were crosschecked in order to identify any further potentially eligible studies. The search method has been previously published in a study that aimed to estimate the magnitude of the nocebo response in PD [16].
Data extraction, evaluation, and synthesis
Three reviewers (MR, GD, and ANF) independently screened the titles and abstracts retrieved during the search according to the inclusion criteria. Two reviewers (MR and ANF) independently read the full-text reports and determined whether these met the inclusion criteria.
Two reviewers extracted data from the individual studies identified for inclusion in a pre-piloted Excel document. The information obtained from the manuscripts included: participant demographics and baseline characteristics (including PD stage and baseline medication); the number of randomized participants to the placebo arm; cardiovascular adverse events in the placebo arm.
The primary outcome was the proportion of major cardiovascular (CV) adverse events (AEs) and the secondary outcome was the proportion and typology of all CV AE reported in the placebo arms of RCTs.
CV AEs were defined according to how they were reported in the main clinical trial publications. We collected data regarding cardiac diseases (coronary heart disease, pericarditis, rhythm abnormalities, valvulopathy, and sudden death), vascular diseases (artery peripheral disease and blood pressure abnormalities), syncope, cerebrovascular diseases (ischemic stroke, transient ischemic attack (TIA), and hemorrhagic stroke), and thromboembolic events (deep vein thrombosis and pulmonary embolism). Major cardiovascular events combined myocardial infarction, stroke, peripheral artery disease, and sudden death.
Unspecified symptoms such as fatigue, peripheral edema, back pain, palpitations, abnormal lipid profile, dizziness/lightheadedness, dyspnea, vertigo, epigastric discomfort, chest pain/discomfort/tightness, and falls were not considered CV AEs. For this study these symptoms were only considered when reported together with CV AEs (e.g., “chest pressure or angina” or “orthostatic hypotension/dizziness”). Postural dizziness was included when reported together with orthostatic hypotension. When symptomatic or asymptomatic orthostatic hypotension was specifically reported, we only considered the symptomatic events. Hypotension and orthostatic hypotension were considered separate events. AEs reported broadly as “cardiovascular”, “cardiac”, and/or “vascular” without further definition were considered as unspecified CV AEs.
Patients were classified as de novo PD patients if not previously treated for PD. Early-stage was included de novo PD patients and/or patients without motor fluctuations. Advanced/late stage was considered when motor fluctuations were present.
Two authors (MA and BM) independently assessed the risk of bias, using the Cochrane Collaboration’s risk of bias tool [17]. Disagreements were resolved by a third reviewer (GSD). We rated studies as having a high overall risk of bias if at least one domain was rated as having a high risk or if three or more domains were rated as having an unclear risk of bias. The remaining options were rated as overall low risk.
Statistical analysis
Publicly-accessible meta-analysis software was used to synthesize the results and to determine the pooled estimated proportion of CV AEs in PD patients [18].
The results of the individual and pooled studies were expressed in percentages (proportion) and 95% -confidence intervals (CI). Freeman-Turkey transformation (double arcsine transformation) was used to adjust the dataset to estimate the proportion of the events, limiting the CI among 0–100% [19]. As considerable heterogeneity between studies was expected due to the wide clinical range of PD patients included,the random-effects model of DerSimonian and Laird [20] was used by default as this approach is the simplest and most commonly used method for fitting the random-effects model and is particularly useful for larger samples [21]. Statistical heterogeneity was assessed using I2 (based on chi-squared statistic and its degrees of freedom), which describes the percentage of the variability in effect estimates that is due to heterogeneity rather than to chance [22].
We also performed subgroup analyses for early and advanced/late-stage PD, de novo PD (no previous treatment with antiparkinsonian medication) and patients whose only antiparkinsonian medication was levodopa.
Following a conservative approach, unspecified AEs reported broadly as “cardiovascular”, “cardiac”, and/or “vascular” were assumed as null when quantifying the proportion of each CV AE.
RESULTS
The 236 trials included (Fig. 1) enrolled17167 PD patients with a mean age of 64.9 (±4.3) years and mean disease duration of 6.40 years (range 0 to 10 years). The mean follow-up time was 25 weeks (range 1 day to 261 weeks).
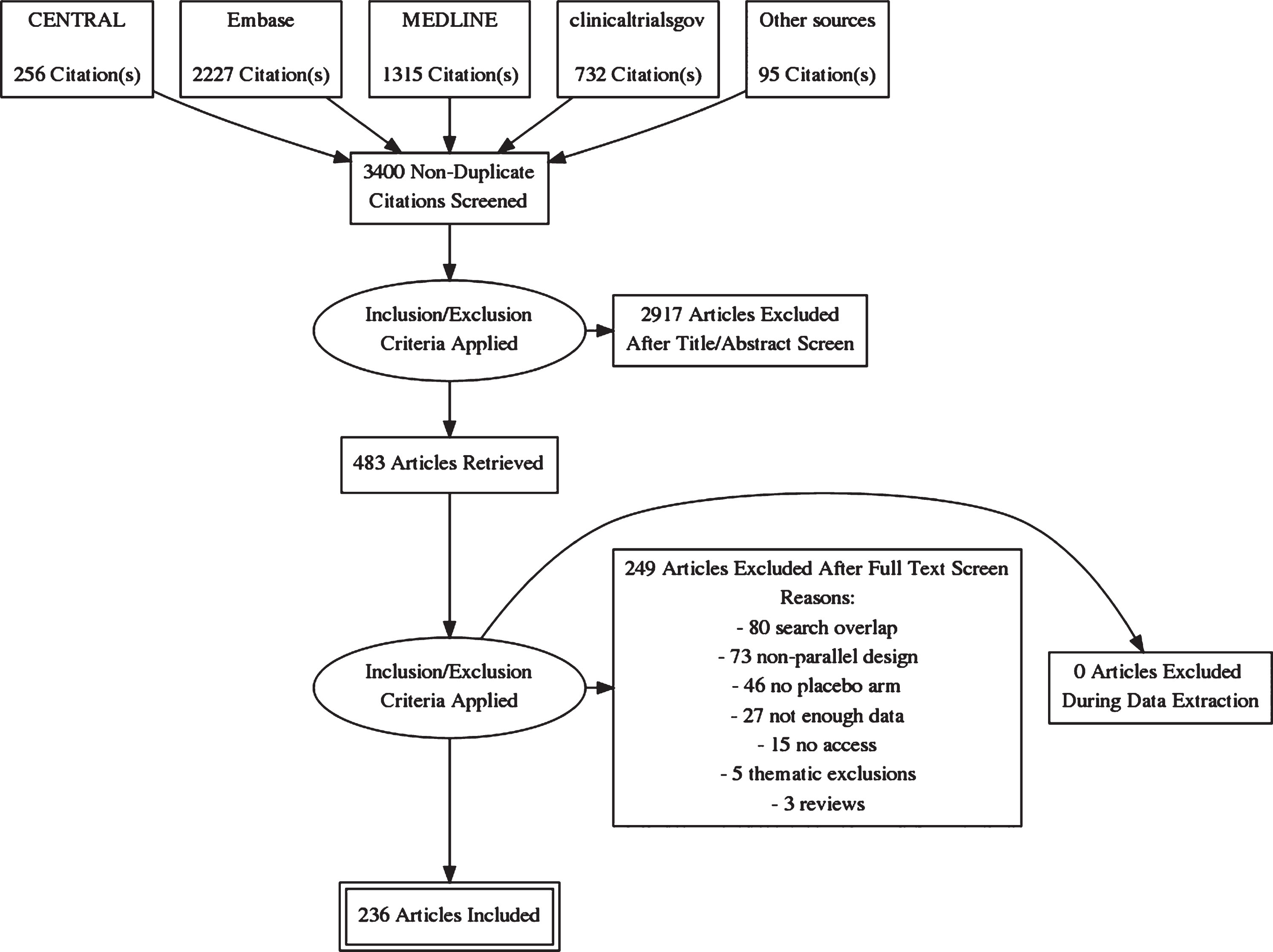
Flowchart.
Eighty percent of the trials (n = 189) reported the presence or absence of CV AEs. The remain 20% of the RCT did not report data of CV AEs on the main file of the trial. Table 1 summarizes the patients’ characteristics in the placebo arms of the trials with data on CV AEs.
Baseline characteristics of Parkinson’s disease patients in randomized controlled trials which report the presence or absence of cardiovascular adverse events
PD, Parkinson’s disease; RCT, randomized controlled trials; IQR, interquartile range; SD, standard deviation.
Considering only the trials with data on CV AEs (n = 189), the pooled proportion of major CV events was low, ranging from 0.00% to 0.06% (Table 2).
Pooled proportion of main cardiovascular adverse events reported among patients with Parkinson’s disease in the placebo arms of randomized controlled trials
CV AE, cardiovascular adverse events; MI, myocardial infarction; RCT, randomized controlled trial; CI, confidence interval. Full table with all CV AE is available in Supplementary File 2.
The pooled proportion of overall CV AEs was 3.33% (95% CI: 2.14, 4.70%, I2 92%; 189 RCT; n = 14704; Supplementary File 1). The proportion of CV AEs in the early stage patients was 3.26% (95% CI: 0.93, 6.56; I2 97%; 57 RCT; n = 6101; Fig. 2). Regarding advanced/late stage patients, the estimated proportion of CV AEs was 2.36% (95% CI: 1.39, 3.51; I2 74% : 83 RCT; n = 5812; Fig. 3).

Pooled proportion of cardiovascular adverse events in 57 RCTs enrolling 6101 patients with early stage Parkinson’s disease –3.26% (95% CI: 0.93, 6.56).

Pooled proportion of cardiovascular adverse events in 83 RCT enrolling 5812 patients with Parkinson’s disease with late stage Parkinson’s disease –2.36% (95% CI: 1.39, 3.51).
The proportion of CV AEs in de novo PD patients was 1.71% (95% CI: 0.47, 3.47%; I2 71%; 15 RCT; n = 1870; Fig. 4). While the proportion in patients whose only antiparkinsonian medication was levodopa presented was 4.56% (95% CI: 1.37, 9.01%; I2 89%; 19 RCT; n = 1618; Fig. 5).
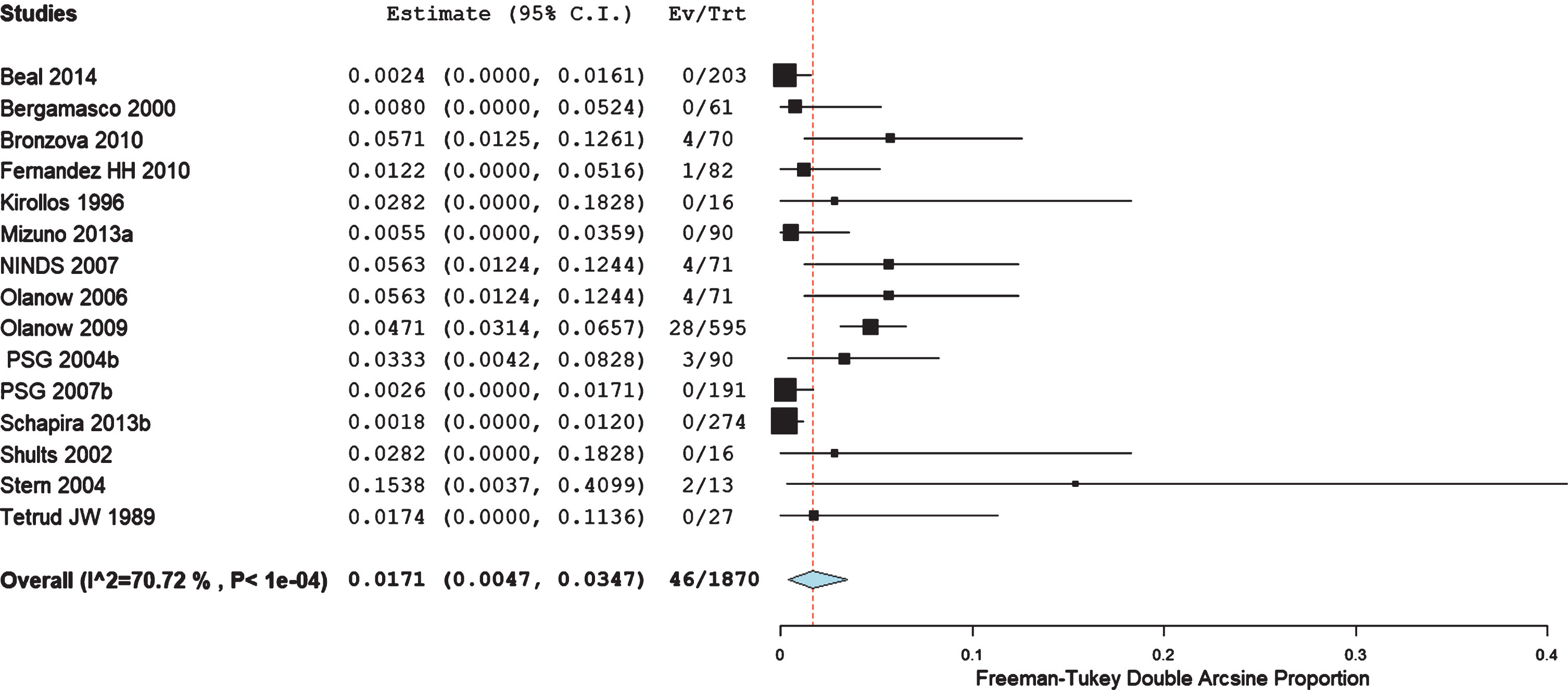
Pooled proportion of cardiovascular adverse events in 15 RCT enrolling 1870 de novo Parkinson’s disease patient’s –1.71% (95% CI: 0.47, 3.47%).
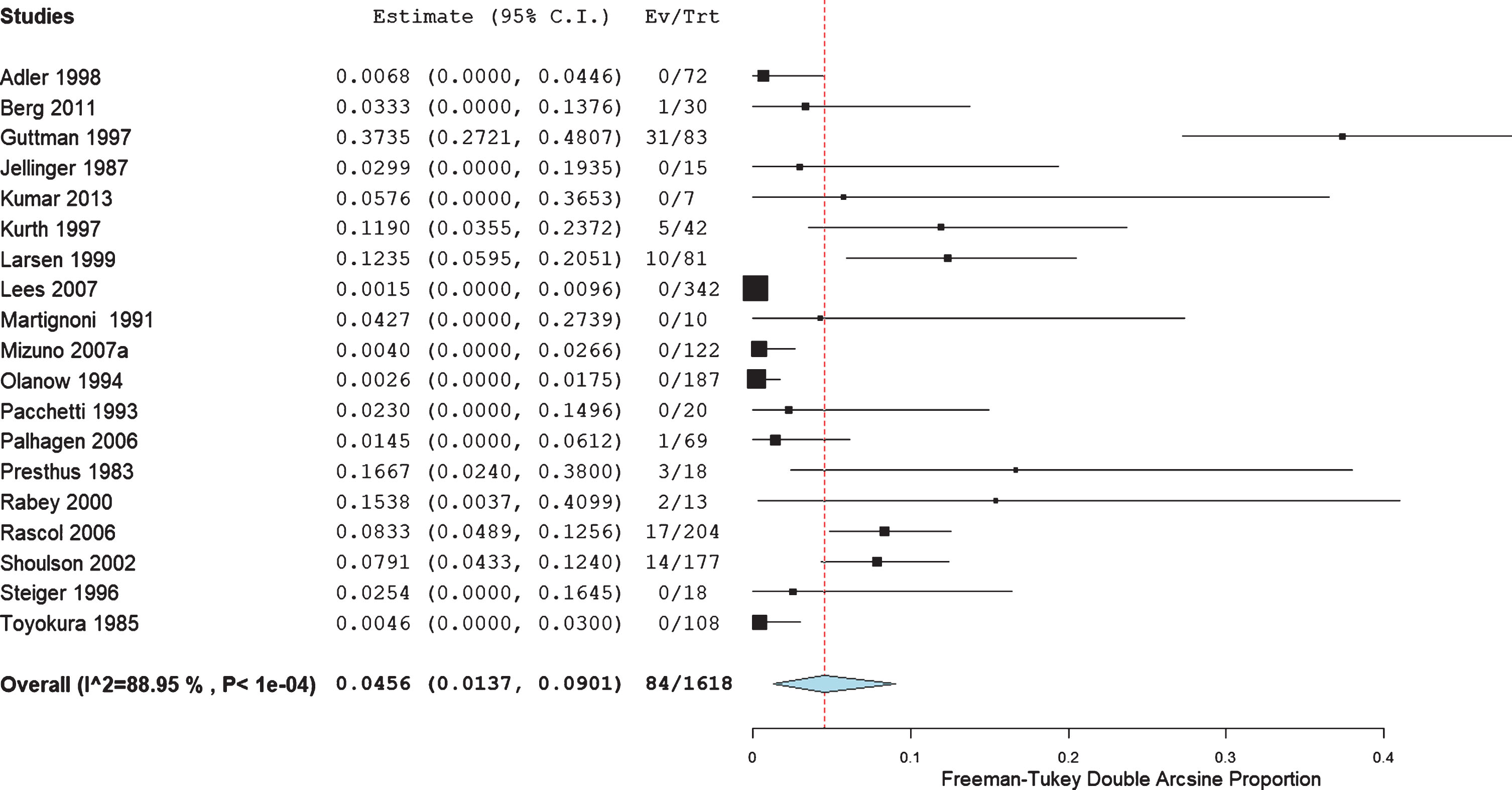
Pooled proportion of cardiovascular adverse events in 19 RCT enrolling 1618 patients with levodopa as the only antiparkinsonian medication –4.56% (95% CI: 1.37, 9.01%).
The most common AEs reported were hypertension and orthostatic hypotension (Table 2 and Supplementary File 2).
Risk of bias
The reported risk of bias refers to the main outcome of clinical trials included, which focus on efficacy rather than safety.
We indicated 61 (25.8%) trials as having a low overall risk of bias based on defined criteria. Figure 6 displays a detailed overview of the risk of bias across studies. The complete summary of bias for all studies can be found in Supplementary File 3.
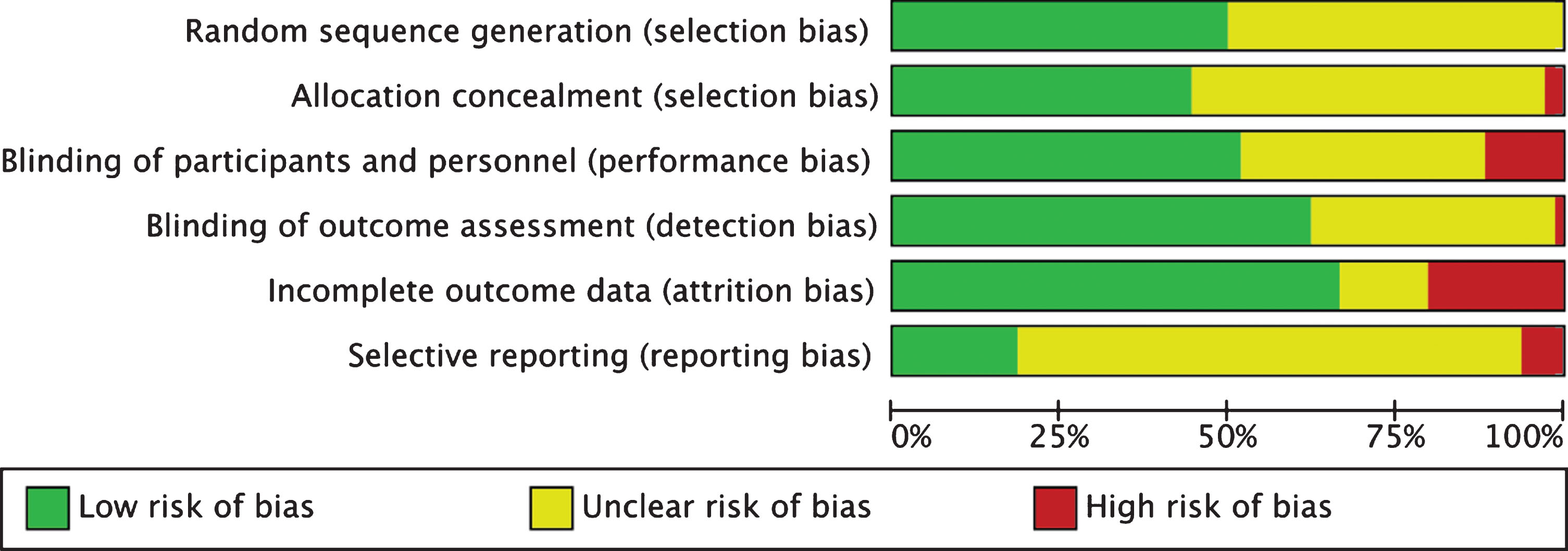
Risk of bias across domains graph.
DISCUSSION
In this systematic review of RCTs, the proportion of major CV AEs in placebo arms of PD patients ranged from 0.0 to 0.20%. The proportion of all CV AEs ranged from 1.71% and 4.56% and blood pressure abnormalities were the most frequent AE.
Although the proportion of all CV AEs in PD patients included in RCTs was higher than in RCTs on restless legs syndrome (0.61%), the proportion of major CV AEs was very low compared to the same study (0.49%). Moreover, the patients included in those trials were younger (54.9±11.9 years) and followed up for a shorter time frame (median 12 weeks) [23].
The comparison with other diseases would be helpful in the interpretation of our data, however, to the best of our knowledge, the previous study on restless legs syndrome is the only study using a similar methodology to estimate the proportion of CV AEs in RCT.
In our study, most CVAEs were related to blood pressure abnormalities. Our results demonstrate that orthostatic hypotension was more frequent in later disease stages, which is consistent with previously published studies [24].
Patients with PD exclusively treated with levodopa presented the highest proportion of major CV AEs, mainly due to the higher proportion of coronary heart disease (0.20%). Levodopa is known to increase homocysteine which may be associated with increased proportion of coronary artery disease [3, 25]. However, current guidelines do not recommend routine assessment of homocysteine for refinement of CVD risk stratification [26]. Additionally, this subgroup of patients also presented a higher proportion of orthostatic hypotension (3.07%), which could also confer an increased risk of coronary heart disease by itself [27–29]. We can also consider the possibility of selection bias of older and more advanced patients in this subgroup. However, Table 1 shows similar age and disease duration and at least one-third of patients were classified as early stage.
Other therapeutics used in PD have been studied due to increased cardiovascular risk [4, 30].
It is also possible that this putative increased risk results from many factors together, since the presence of dysautonomia and advanced age are important risk factors for the development of iatrogenic cardiac events [31].
A large cohort study from a Taiwanese insurance database showed that PD patients in the first three years after diagnosis, who were on at least one antiparkinsonian medication (including levodopa), presented a higher risk of acute myocardial infarction compared with matched controls [9]. However, the same study reported that the same group of patients also presented a higher risk of stroke [8], which was an extremely rare event in our review.
In the systematic review of observational studies, the overall analysis of myocardial infarction risk was not significantly increased, however a subgroup analysis considering only prospective studies found a significantly higher risk (OR 1.59; 95% CI 1.14, 2.22; I2 = 48%) [13].
Although restrictive valvular heart disease has been related to dopamine-agonist treatment in observational studies [32], there were no reports of valvopathy in placebo arms of clinical trials.
Our study has several limitations. In many studies included in this meta-analysis, the search for AEs was open and thus not systematically collected. However, it is expected that CV events, particularly major events, would be reported as AEs. Despite de novo events were reported, chronic conditions like angina or peripheral vascular disease are likely to be underreported. However due to its severity, it is probable that the worsening of these chronic cardiovascular baseline disease would be reported. Following this idea, the lack of information regarding risk factors of CVD would also be important to better estimate/understand the prevalence of CVD in PD. Additionally, we should stress that our analysis depends on the way AEs were reported in the main publication of the included clinical trials. Another source of bias can be related to the reporting, particularly if the event is rare since some trials only report AEs with a proportion above a specific threshold. Finally, we analyzed selected populations of PD patients who fit RCT inclusion criteria, which could lead to selection bias with their severe comorbidities, including CV disorders. However, the high spectrum of disease status included and the large pooled sample suggest that a broad group of PD patients were included.
In conclusion, our meta-analysis suggests that the proportion of major CV AEs in patients with PD is low and lower than in restless legs syndrome patients. The most frequent CV AE was orthostatic hypotension and is related with disease progression. Finally, we cannot exclude a potential risk of levodopa on coronary heart disease.
CONFLICT OF INTEREST
Dr. Alves reports participation in conferences with Boehringer-Ingelheim, AstraZeneca, Bayer, Bristol-Myers-Squibb, Grünenthal, Tecnimede, and Merck Sharp & Dohme. Prof. Caldeira reports participation in conferences with Bristol-Myers-Squibb, Bayer, Boheringer Ingelheim, Daiichi-Sankyo, Merck-Serono, Ferrer, Pfizer, Novartis, and Roche. Prof. Ferro reports honoraria for speaker, clinical trial coordinator and advisory board from Boehringer-Ingelheim. Prof. Ferreira reports personal fees from Grunenthal, Fundação MSD (Portugal), TEVA, MSD, Allergan, Medtronic, GlaxoSmithKline, Novartis, Lundbeck, Solvay, BIAL, Merck-Serono, Merz, Ipsen, Biogen, Acadia, Allergan, Abbvie, and Sunovion-Pharmaceuticals. Dr. Leal Rato, Dr. Duarte, and Dr. Nunes Ferreira have nothing to report.
Footnotes
ACKNOWLEDGMENTS
M. Alves was awarded a scholarship from Fundação para a Ciência e Tecnologia (SFRH/BD/129700/2017).
IMM – UID/BIM/50005/2019, project funded by Fundação para a Ciência e a Tecnologia (FCT)/ Ministério da Ciência, Tecnologia e Ensino Superior (MCTES) through Fundos do Orçamento de Estado.
