Abstract
Background:
More than 75% of Parkinson’s disease (PD) patients will develop dementia. Previous studies on the cognitive predictors of dementia in PD had some methodological limitations and the cognitive tests identified as good predictors vary greatly.
Objective:
This prospective cohort study aims to identify the optimal cognitive predictors of dementia in PD using complementary statistical methods.
Methods:
Eighty PD patients without dementia underwent polysomnographic recording, a neurological examination, and a complete neuropsychological assessment at baseline. They were then followed for a mean of 4.3 years. Baseline group comparisons and survival analyses were used to identify optimal cognitive predictors. Moreover, patients who developed dementia were pair-matched at baseline according to age, sex, and education to healthy controls (2 : 1), and receiver operating characteristic curves were calculated for cognitive tests.
Results:
At follow-up, 23 patients (29%) developed dementia. PD patients who developed dementia had poorer baseline performance and a higher proportion of clinically impaired performance on several cognitive tests. Impaired baseline performance on the Block Design subtest was the best independent predictor of dementia (HR = 8). Moreover, the Trail Making Test part B (time) and Verbal Fluency (semantic) had the best psychometric properties (area under the curve >0.90) for identifying PD patients at risk of dementia.
Conclusion:
The present study identified three cognitive tests as the most accurate to detect individuals with PD at high risk of developing dementia.
INTRODUCTION
Cognition in Parkinson’s disease (PD) is heterogeneous, with some patients having normal cognition, and others presenting either mild cognitive impairment (MCI) or dementia [1,2, 1,2]. The risk of developing dementia is six times higher in PD patients compared to healthy individuals [3]. Moreover, approximately 75% of PD patients will develop dementia during the course of the disease [4]. Investigative biomarkers of dementia in PD include structural, functional and molecular neuroimaging markers, as well as cerebrospinal fluid based markers [5]. However, these techniques can be invasive, expensive, and difficult to access. Therefore, there is still a need for sensitive tools accessible in a clinical setting, research, or intervention trials that could detect PD patients at high risk of dementia.
Longitudinal studies have investigated the cognitive markers of dementia in PD [6–16]. However, the cognitive tests identified as good predictors of dementia in PD vary greatly across studies (see Supplementary Table 1). This could be explained by differences and heterogeneity in study samples, design, and statistics (for example, the confounding variables selected) but also by some methodological limitations, notably heterogeneous criteria for identifying dementia in PD or a limited number of cognitive tests used [6–16], relatively small sample size at baseline [7, 11], low follow-up rate with possible heterogeneous attrition [12–14,16, 12–14,16], and absence of a healthy control group to enable comparison with normal values [8–10,12–14, 8–10,12–14] (see Supplementary Tables 1 and 2). Furthermore, the optimal cut-off values and other psychometric properties for different neuropsychological tests to identify patients at high risk for dementia in this population need to be investigated.
In the present study, we aimed to identify new and optimal cognitive predictors of dementia in PD patients by combining complementary statistical methods, including baseline group comparisons, survival analyses, and receiver operating characteristic curves with normal values for comparisons.
METHOD
Participants
This study is part of our ongoing longitudinal study on sleep and cognition in PD. A total of 100 PD patients were recruited from the Movement Disorders Clinic at the Montreal General Hospital and the Unit
Procedure
Baseline assessment
At baseline, all participants underwent one-night polysomnographic (PSG) recording, as well as clinical, neurological, and neuropsychological assessments. PSG recording included at least two standard electrode derivations for monitoring EEG activity during sleep (C3–A2 and O2–A1), left and right electrooculograms, and submental EMG [19, 20]. Respiration was monitored using a nasal canula or a nasal/oral thermistor with thoracic and abdominal strain gauges, and pulse oximetry was performed. Sleep stages were recorded according to standard methods [21, 22]. PSG variables included sleep latency and efficiency, REM sleep latency and efficiency, total sleep time, sleep stage percentage, apnea-hypopnea index, and mean O2 saturation. The severity of motor symptoms and PD were evaluated with the Unified Parkinson’s Disease Rating Scale part III (UPDRS-III) and the Hoehn and Yahr Scale [23, 24]. Participants completed questionnaires assessing the severity of symptoms of depression (Beck Depression Inventory, Second Edition), anxiety (Beck Anxiety Inventory), daytime sleepiness (Epworth Sleepiness Scale), and insomnia (Insomnia Severity Index) [25–28]. Patients also completed tests for olfaction (University of Pennsylvania Smell Identification Test or UPSIT) and color discrimination (Farnsworth-Munsell 100-Hue Test or FM-100) [29, 30]. Patients were instructed to take their usual medication as prescribed during the study. Two cognitive screening tests were administered: the Mini-Mental State Examination (MMSE) and the Montreal Cognitive Assessment (MoCA) [31, 32]. Participants underwent a complete neuropsychological assessment administered by a neuropsychologist according to standard procedure [33]. Five cognitive domains were assessed with the following tests: (1) attention [Trail Making Test (TMT) part A (time), Digit Span (scaled score) subtest from the Wechsler Adult Intelligence Scale – Third Edition (WAIS-III), Stroop Color Word Test (SCWT) part III–II (time and error)]; (2) executive functions [SCWT part IV–III (time and error), TMT part B (time) and TMT part B – A (time), Verbal Fluency, phonetic (letters P, F, L in French or F, A, S in English, one minute each); and Verbal Fluency, semantic (animals or fruits/vegetables, one minute each)]; (3) learning and memory [Rey Auditory-Verbal Learning Test (RAVLT), sum of trials 1 to 5, List B, immediate and delayed recalls, and recognition; and Rey-Osterrieth Complex Figure test (ROCF), immediate and delayed recalls]; (4) visuospatial abilities [copy of the ROCF, Block Design (raw and scaled scores) subtest from the WAIS-III, Bells test (omissions)]; and (5) language [MMSE language section, Boston Naming Test 30 items] [19]. Normative data used to calculate z-scores or to determine normal vs. impaired performance were published previously, except for the BNT (in-house norms) [19, 34].
Cognitive status was determined by consensus between the neuropsychologist (JFG) and neurologist (RBP) as: normal cognition, MCI or dementia. The diagnosis of dementia was based on the proposed MDS Task Force Level II criteria, and defined as impaired performance in at least two cognitive domains on neuropsychological testing, along with evidence of significant functional impact of cognitive deficits on daily living activities [1]. Daily functioning impairment was determined during the interview with participants and, when available, their relatives. We determined whether new significant difficulties (i.e., patient can no longer execute the task or needs help from others) were present in several activities, such as cleaning the house, managing finances, shopping, performing chores, driving the car, using public transportation, or preparing meals. In order to assess mental organization and ability to function in daily living, patients were also questioned about their use of medications, including the ability to clearly describe their medication (i.e., drug, dose, and timing) and whether they can organize and take it independently (questions were derived from the Pill Questionnaire appended to Dubois et al. 2007) [1]. MCI diagnosis was based on a modified version of the proposed MDS Task Force Level II criteria and defined as: 1) impaired performance (at least 1.5 SD below the normative data or, depending upon the norms, a scaled score ≤6 or percentile range ≤10) on at least two neuropsychological tests in the same cognitive domain; 2) subjective cognitive complaint at the clinical interview noted by the clinician or reported by either the participant or relatives; 3) absence of significant impairment of daily living activities; and 4) absence of other conditions (medical, psychiatric) that could better explain the cognitive decline [2, 35]. MCI subtypes were defined as: single domain (impaired on at least two tests in the same cognitive domain) or multiple domain (impaired on at least two tests in at least two distinct cognitive domains).
Follow-up assessment
All PD patients were offered a neurological exam and a shorter version of the neuropsychological assessment that was previously described: (1) attention (TMT, Digit Span, and SCWT); (2) executive functions (TMT, SCWT, and Verbal Fluency); (3) learning and memory (RAVLT and ROCF); and (4) visuospatial abilities (ROCF). If the patient refused the neuropsychological assessment, the neurologist administered the cognitive screening tests previously mentioned (MMSE or MoCA). If the patient was unable to come to be assessed in person (e.g., severe dementia and/or disability), the neurologist conducted a telephone interview with the patient, relatives and/or treating physician, completed by a clinical chart review. The presence or absence of dementia was determined by consensus between the neuropsychologist and the neurologist based on these examinations [1, 2]. Follow-up duration was defined as the time between the baseline assessment and the last cognitive status diagnosis.
Statistical analysis
All statistical analyses were performed using SPSS 24.0. Measures of Skewness and Kurtosis were made to assess the normality of the variables. First, baseline sociodemographic, clinical, sleep and cognitive characteristics were compared between the two groups (dementia vs. dementia-free) using bilateral Student’s t test for independent samples (or the nonparametric tests of Mann-Whitney U when the variables were not normally distributed). Statistical significance was set at p < 0.05. The Benjamini-Hochberg procedure was used to control for the false discovery rate (FDR) resulting from the multiple statistical comparisons. We attributed a z score of – 3.5 (corresponding to a very weak performance) on a cognitive test when the patient was unable to complete it due to severe cognitive impairment. Age was added as a covariate to the cognitive variables when they were correlated together (r≥0.3). Chi-square tests (χ2) were used to compare the proportions of patients between the groups for the following variables: sex, medication, MCI, and impaired performance on cognitive tests (1.5 SD below the normative data). Then, Kaplan-Meier survival curves and Cox proportional hazard analyses were performed to assess the risk of developing dementia according to baseline performance on individual cognitive tests, controlling for age, sex, education, disease duration, severity of motor symptoms, and levodopa dose. A combined Cox regression model was generated with all the significant individual cognitive predictors, along with the covariates mentioned previously, to determine the independent cognitive predictors of dementia in PD. The time to event variable for patients who did not develop dementia was the time between the first and the last visit. For stratification, all predictive cognitive tests were dichotomized into normal/abnormal using an age, sex, and education z score of –1.5. Finally, to determine at baseline the accuracy and best threshold value of cognitive tests to predict dementia, receiver operating characteristic (ROC) curves were generated. We compared PD patients who developed dementia (disease group) to healthy controls (normal group) on cognitive tests identified as significantly affected in patients who were found to have developed dementia based on previous analyses. Characteristics identified were the area under the curve (AUC), the sensitivity, specificity, positive predictive value (PPV), negative predictive values (NPV), and the percentage of correctly diagnosed patients. Three cut-off values were generated for each cognitive test: 1) the maximum accuracy value, calculated by the Youden Index (y = sensibility + specificity –1); 2) the optimal screening value, or the lowest value with >80% sensitivity and NPV; and 3) the optimal diagnostic value, or the highest value with >80% specificity and PPV. We also generated ROC curves comparing PD patients who developed dementia to PD patients who remained dementia-free.
RESULTS
Between 2003 and 2012, 100 patients were recruited in the baseline cohort, and some of these participated in previous studies conducted by our group on other non-motor predictors of dementia in PD [19, 37]. Most of the patients (60/80) in the Anang study [36] were included in the present study: 10 patients did not meet our inclusion criteria (see above) and 10 patients were added because their follow-up information was available. Twenty patients could not be assessed at follow-up: 6 had died and 14 refused to participate at the follow-up or were unreachable. Consequently, 80 patients met inclusion criteria (view Fig. 1 for flowchart). The patients excluded were equivalent to the patients included in the study for age (68.75±10.47 vs. 66.28±8.17, p = 0.151), proportion of men (50% vs. 69%, p = 0.116), education (15.00±4.21 vs. 14.54±3.50, p = 0.753), disease severity on the Hoehn & Yahr Scale (2.57±0.98 vs. 2.30±0.88, p = 0.450), PD duration in years (5.11±4.11 vs. 4.00±3.50, p = 0.381), and MMSE score (28.17±1.89 vs. 28.38±1.64, p = 0.770). At the last follow-up, 46 patients underwent a neuropsychological assessment and neurological exam, 13 had office-based cognitive testing (MMSE or MoCA) and a neurological exam, and 21 were followed by telephone by the neurologist (interview with the patient, relatives and/or treating physician, completed by a clinical chart review) to assess the presence or absence of dementia. After a mean follow-up of 4.3 years (1–10 years), 23 patients (29%) had developed dementia.
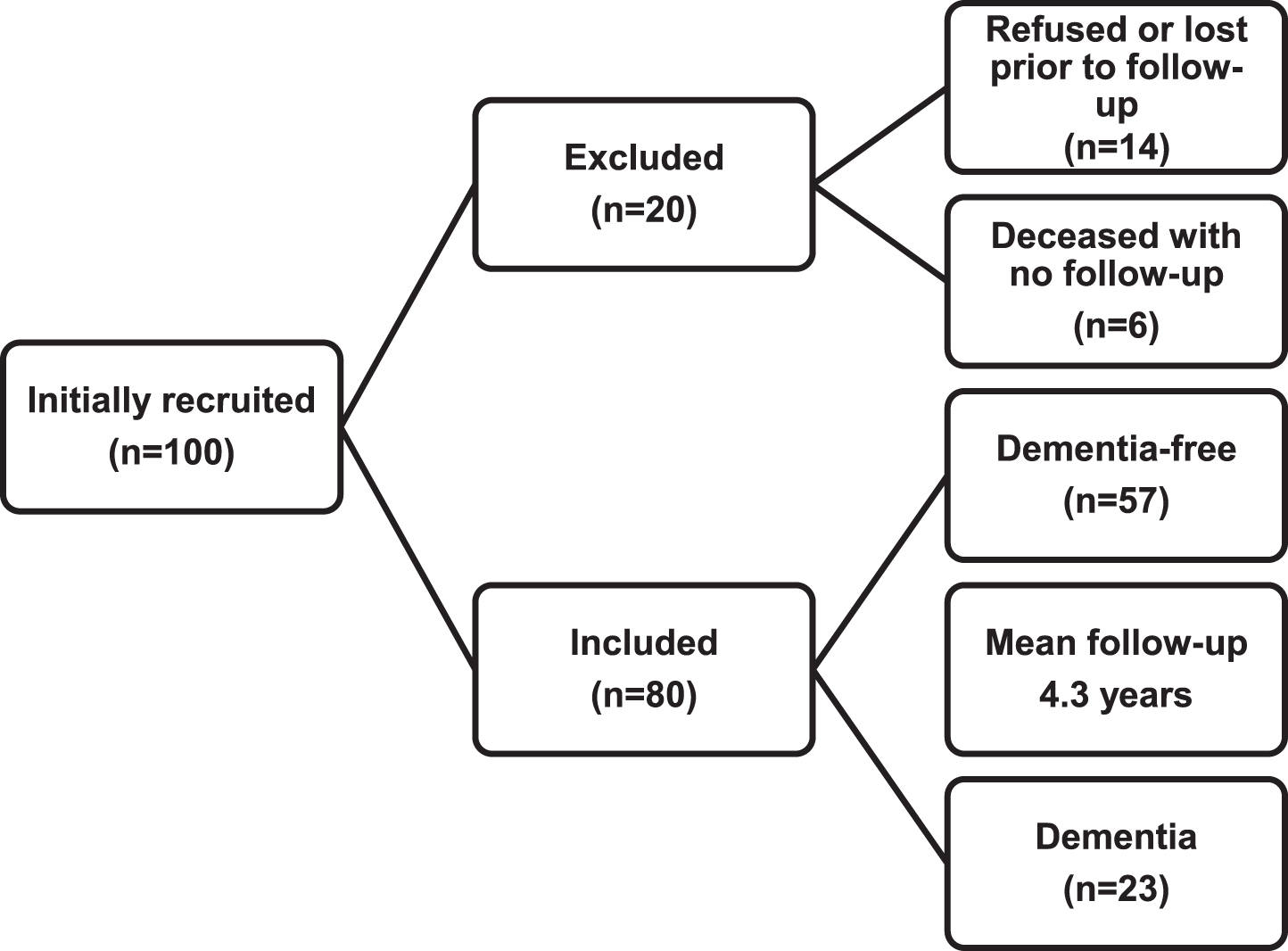
Patient flowchart.
Baseline demographic, clinical, and sleep characteristics
Characteristics of patients at baseline are presented in Table 1 (demographic and clinical) and Table 2 (sleep). Patients who developed dementia (PDD) were equivalent to patients still dementia-free on the majority of variables, except for older age, higher FM-100 score (higher score means a poorer performance), reduced N2 sleep percentage (a trend was observed after FDR correction), and higher proportion of MCI, particularly the multiple domain subtype. Age correlated with the following variables and was added as a covariate in subsequent analyses: MMSE; Sleep efficiency; Mean O2 saturation; TMT part A; TMT part B; Verbal Fluency, semantic; Verbal Fluency, phonetic; SCWT III–II (sec); RAVLT (total, immediate and delayed recalls); ROCF (immediate and delayed recalls); and Block Design.
Baseline demographic and clinical characteristics of patients
aMann-Whitney, bANCOVA (age). BDI-II, Beck Depression Inventory Second Edition; BAI, Beck Anxiety Inventory; UPDRS-III, Unified Parkinson Disease Rating Scale, part III; UPSIT, University of Pennsylvania Smell Identification Test; FM-100, Farnsworth-Munsell 100 Hue Color Vision Test; MMSE, Mini-Mental State Examination; MCI, mild cognitive impairment.
Baseline sleep characteristics of patients
aMann-Whitney, bANCOVA (age), *False Discovery Rate adjusted p value. ESS, Epworth Sleepiness Scale; ISI, Insomnia Severity Index; REM, Rapid eye movement.
Baseline cognitive performance
Cognitive performance of patients at baseline is presented in Table 3 and Fig. 2. PDD patients performed worse than patients who remained dementia-free on the following tests (after FDR adjustment): SCWT part III–II, time; TMT part B, time; TMT part B – A, time; Verbal Fluency, semantic; RAVLT, sum of trials 1 to 5 and immediate recall; ROCF, copy, immediate and delayed recalls; Block Design subtest; and Bells test. Moreover, the proportion of PDD patients with significantly impaired performance (1.5 SD below normative data) was higher on the following tests (after FDR adjustment): SCWT part III–II, time (52% vs. 11%, p = 0.002); TMT part B, time (74% vs. 40%, p = 0.021); TMT part B – A, time (61% vs. 20%, p = 0.003); Verbal Fluency, semantic (59% vs. 16%, p = 0.002); ROCF, delayed recall (48% vs. 16%, p = 0.017); Block Design subtest (41% vs. 11%, p = 0.016); and Bells test (59% vs. 21%, p = 0.005).
Baseline cognitive performance
aMann-Whitney, bANCOVA (age), * False Discovery Rate adjusted p value. TMT, Trail Making Test; RAVLT, Rey Auditory-Verbal Learning Test; ROCF, Rey-Osterrieth Complex Figure test; MMSE, Mini-Mental State Examination; BNT, Boston Naming Test; Stroop components: II = Naming, III = Interference, IV = Flexibility.
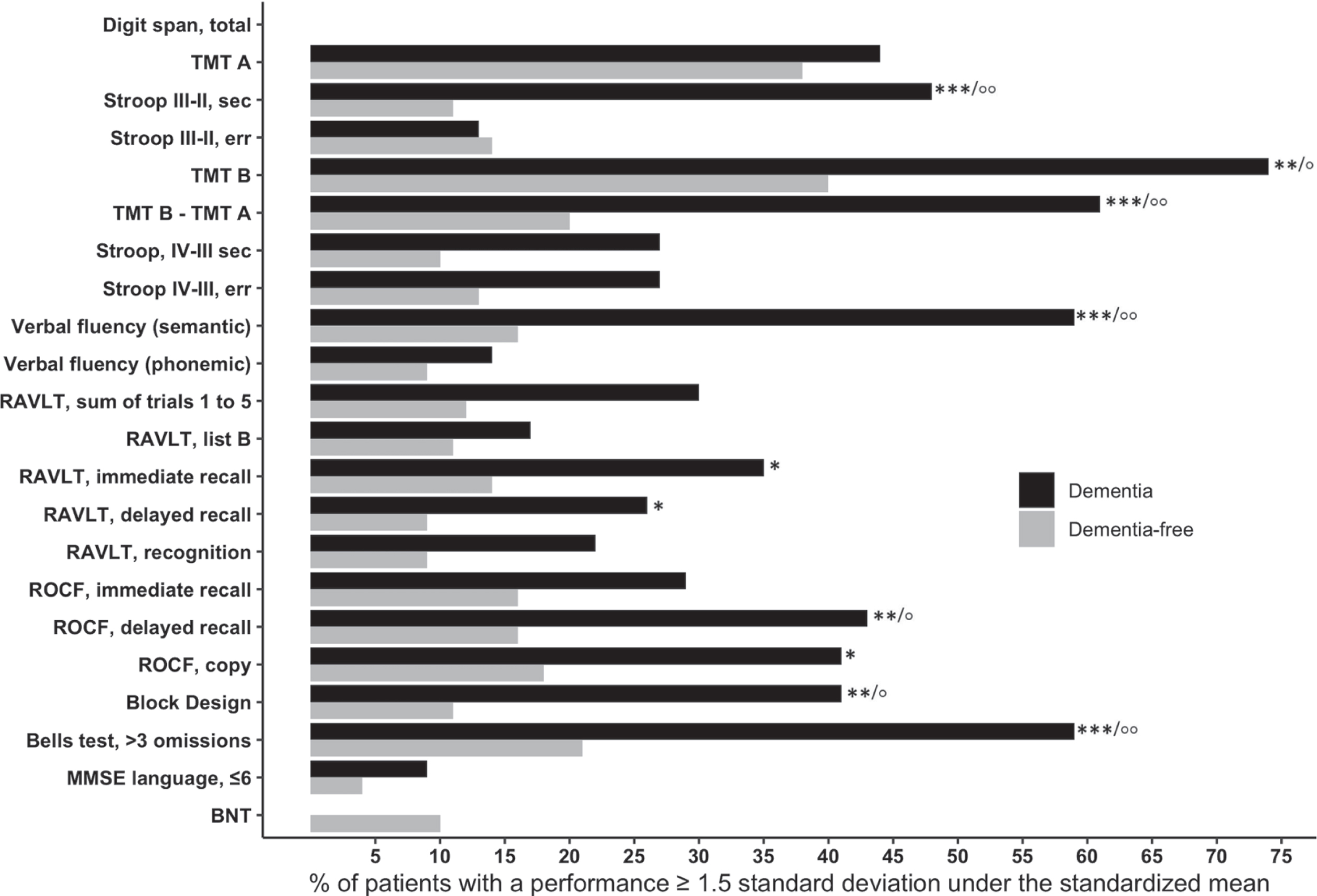
Percentage of participants with clinically impaired performance on cognitive tests at baseline. TMT, Trail Making Test; RAVLT, Rey Auditory-Verbal Learning Test; ROCF, Rey-Osterrieth Complex Figure Test; MMSE, Mini-Mental State Examination; BNT, Boston Naming Test; Stroop components: II = Naming, III = Interference, IV = Flexibility. ***p < 0.001, **p < 0.01, *p < 0.05/°°p < 0.001 false discovery rate adjusted value, °p < 0.05 false discovery rate adjusted value.
Dementia outcomes according to baseline cognitive performance
Kaplan-Meier survival curves with significant difference are shown in Fig. 3. Baseline abnormal performance (1.5 SD below normative data) on the following cognitive tests was a strong predictor of dementia after controlling for age, sex, education, disease duration, severity of motor symptoms, and levodopa dose: Block Design subtest (HR = 14.16, p < 0.001, 95% CI: 3.96–50.7); SCWT III–II, time (HR = 5.95 p = 0.006, 95% CI: 1.69–21.0); RAVLT, delayed recall (HR = 5.13, p = 0.006, 95% CI: 1.59–16.62); RAVLT, sum of trials 1 to 5 (HR = 4.98, p = 0.004, 95% CI: 1.65–14.84); RAVLT, immediate recall (HR = 1.16, p = 0.027, 95% CI: 1.16–11.68); Verbal Fluency, semantic (HR = 3.04, p = 0.045, 95% CI: 1.03–9.0). Furthermore, patients with multiple domain MCI were associated with increased risk of developing dementia compared to patients with normal cognition (HR = 10.64, p < 0.001, 95% CI: 3.13–36.2). In the combined regression model, impaired performance on the Block Design subtest was the only independent cognitive predictor of dementia in PD (HR = 8.0, p = 0.015, 95% CI: 1.51–42.4).
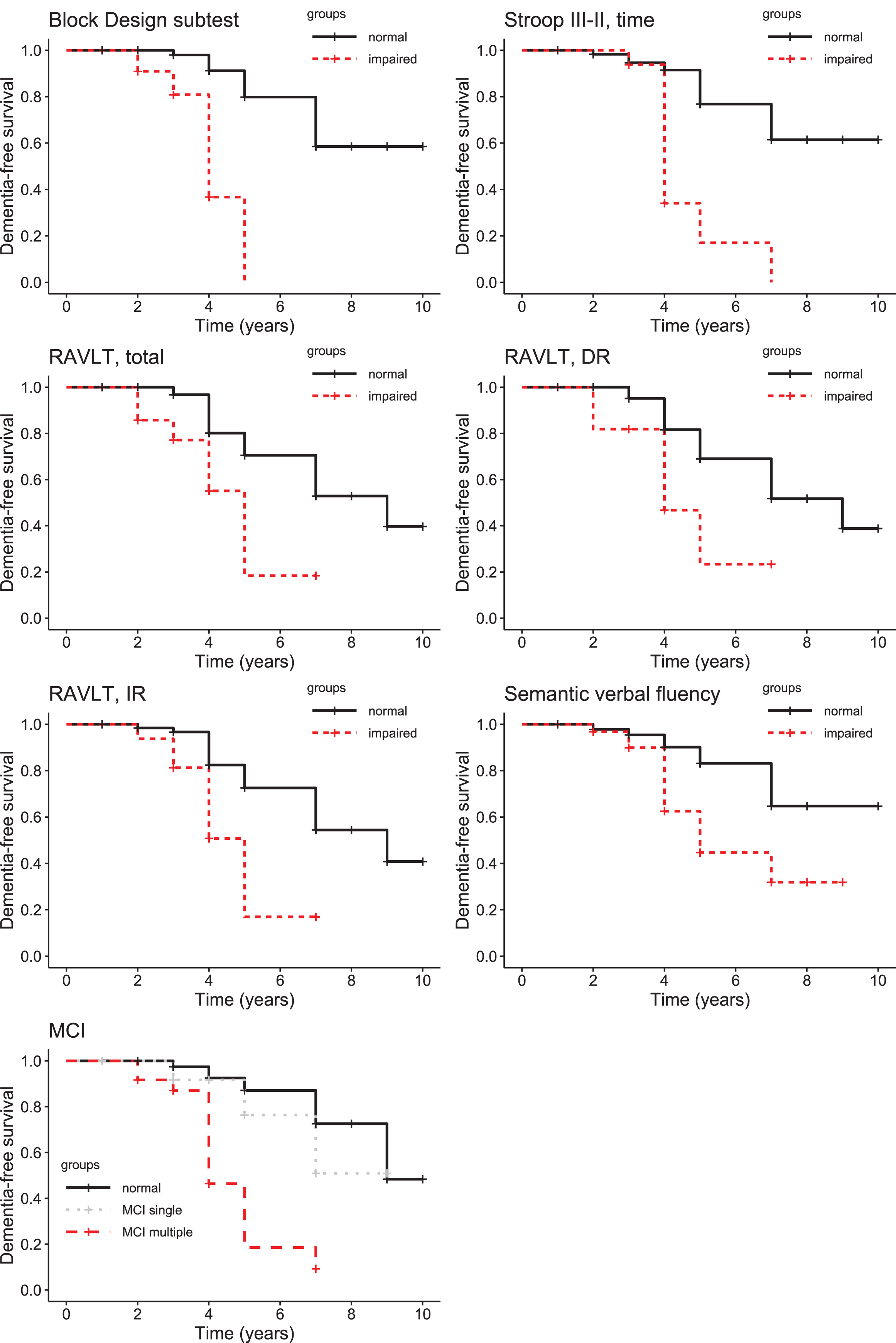
Cognitive decline rates in relation to baseline cognitive performance on cognitive variables. RAVLT, Rey Auditory-Verbal Learning Test; IR, Immediate recall; DR, Delayed recall Stroop components: II = Naming, III = Interference; MCI, mild cognitive impairment.
Baseline predictive value of cognitive tests for detecting dementia
We performed ROC curves analyses on the cognitive tests administered at baseline and identified by the previous analyses as significantly affected in patients who were found to have developed dementia at follow-up. Their psychometric properties are presented in Table 4. The 23 patients who developed dementia were paired with 46 controls (normal value), equivalent for age (71.96±5.09 vs. 70.98±5.92, p = 0.501), education (13.57±3.81 vs. 13.78±3.03, p = 0.798), and sex (78% vs. 63% men, p = 0.201). The following cognitive tests had an AUC > 0.90 (mean and SD for controls): TMT part B, time (82.02±26.60); and Verbal Fluency, semantic (37.70±6.93). Other cognitive tests also had an AUC > 0.85 (mean and SD for controls): TMT part B – A, time (44.70±28.58); ROCF, delayed recall (18.39±6.62); ROCF, immediate recall (17.30±5.48); RAVLT, sum of trials 1 to 5 (47.33±8.64); RAVLT, immediate recall (9.91±2.58); Block Design subtest, raw score (34.28±10.35); SCWT part III–II, time (41.57±39.70); and ROCF, copy (31.01±2.96). The psychometric properties of the cognitive tests administered at baseline for detecting PD patients who developed dementia versus patients still dementia-free are presented in Supplementary Table 3.
Psychometric properties of the cognitive tests (AUC > 0.85) for detecting patients who developed dementia compared to healthy controls
aMaximum accuracy value calculated by the Youden Index (y = sensitivity + specificity – 1); bOptimal screening value, or the lowest value with >80% sensitivity and NPV; b*Optimal screening value, or the highest value with >80% sensitivity and NPVs; cOptimal diagnostic value, or the highest value with > 80% specificity and PPV; c*Optimal diagnostic value, or the lowest value with >80% specificity and PPV. AUC, Area under the curve; PPV, Positive predictive value; NPV, Negative predictive value; TMT, Trail Making Test; RAVLT, Rey Auditory-Verbal Learning Test; ROCF, Rey-Osterrieth Complex Figure test.
DISCUSSION
The aim of this prospective cohort study was to use complementary statistical methods to identify the optimal cognitive predictors of dementia in PD patients. First, we found that several cognitive tests differentiated PD patients who had developed dementia at follow-up from patients who remained dementia-free. Second, the survival analyses revealed that baseline impaired performance on several cognitive tests was associated with a significantly greater risk of developing dementia; in the combined regression model, impaired performance on the Block Design subtest was the only independent cognitive predictor of dementia in PD. Third, the ROC curve analyses revealed that the most valid tests (AUC > 0.90) for identifying PD patients at risk of developing dementia from normal values are the TMT part B (time) and Verbal Fluency (semantic). The Verbal Fluency (semantic) test was also very good (AUC > 0.84) at differentiating patients who developed dementia from dementia-free patients. These three cognitive tests require a broad range of cognitive resources (i.e., executive and motor functions, visuospatial abilities, semantic), which are based on large brain networks, including frontal (executive), temporal (semantic) and parietal (visuospatial) structures [33]. This may explain their higher sensitivity for detecting cognitive decline in PD. Moreover, they are non-invasive, inexpensive, and available in a clinical and research setting, and could be considered for use in identifying PD patients at risk of dementia in intervention trials.
Previous neuropsychological studies investigating the predictors of dementia in PD show a lack of consensus (see Supplementary Table 1) [6–16]. Cognitive tests identified as good predictors of dementia are heterogeneous, and assess different cognitive domains: attention [11, 16], executive functions [6, 9–15], episodic memory [8, 15], visuospatial functions [12–14], and language [16]. As a result, it is difficult to draw a conclusion from the available literature. In the present study, impaired performance on the Block Design subtest was strongly and independently associated with the subsequent development of dementia in PD. To our knowledge, only one previous study used that test [10], and found no association with the development of dementia in PD. However, in that study, the mean performance on this test was significantly lower for patients who developed dementia compared to patients who remained dementia-free. Moreover, the authors used the median score of their sample to dichotomise the performance, while we used standardised normative data to determine whether a patient was impaired or not on this test. This suggests that a clinically impaired performance on the Block Design subtest, based on normative data, is a better predictor of dementia in PD than a score below the median. We found two other cognitive tests, the TMT part B (time) and Verbal Fluency (semantic), with excellent psychometric characteristics for detecting PD patients at high risk of dementia. A previous study using the TMT part B found a positive association with the risk of developing dementia in PD [8]. For the Verbal Fluency (semantic) test, seven studies looked at its validity to predict dementia in PD, with three reporting a positive association and four a negative one. This inconsistency might be due to heterogeneity in their protocols and methodological aspects. Furthermore, the results of previous studies are difficult to generalize to a clinical or research setting since the optimal cut-offs at which a patient is more likely to convert to dementia are not provided. Moreover, none of these previous studies used complementary statistical methods to determine the optimal cognitive predictors of dementia in PD. The present study addresses several of these limitations. First, we used the criteria and cognitive tests proposed by the MDS task force to diagnose dementia in PD, which allow generalization and validation [1, 2]. This approach also enabled us to assess a broader range of cognitive functions and diagnose MCI. Furthermore, we used complementary statistical methods to determine the optimal cognitive markers of dementia in PD: baseline group comparisons, survival analyses and ROC curve analyses. The ROC curves calculated with a group of healthy controls allowed us to identify the psychometric properties of several cognitive tests for assessing prodromal dementia in PD. We also presented three different cut-offs for different neuropsychological markers, which provide options for different trade-offs between sensitivity, specificity, PPV and NPV that could be used for research or by clinicians to better target patients at high risk of developingdementia.
The present study also identified multiple domain MCI as a predictor of dementia in PD. Previous studies consistently associated MCI in PD with a greater risk of developing dementia [8, 38–43]. To our knowledge, only one other study has investigated the risk of dementia in PD in relation to MCI subtypes, according to the criteria recommended by the MDS task force [41]. The previous study and this one found that the MCI multiple domain subtype is associated with an increased risk of developing dementia, which is not the case for the single domain subtype of MCI. However, the presentation as well as the progression of MCI is heterogeneous in PD, and not all patients with concomitant PD and MCI will progress to dementia in a linear trajectory [44]. Given this heterogeneity, and the great burden associated with conducting an exhaustive neuropsychological assessment to cover all cognitive domains and MCI criteria in PD, it is important to consider the individual contribution of specific cognitive tests to the prediction of dementia in this population.
The present study also found that the percentage of sleep stage N2 was reduced in patients who developed dementia compared to patients still dementia-free. This confirms, in a larger sample of patients, our previous results showing reduced sleep spindle (density and amplitude) and sigma power activity (12–15 Hz), two components of sleep stage N2, in the posterior brain regions in PD patients who developed dementia compared to patients still dementia-free [19, 20]. Several other biomarkers of cognitive decline have been identified in PD, such as neuroimaging, waking EEG features, genetic and cerebrospinal fluid-based markers [5, 45]. Most of these techniques are invasive or expensive, which limits their use and accessibility in research and clinical settings. Also, despite the advances in biomarker research for cognitive decline in PD, none has been found to have adequate accuracy for use in a clinical context [46]. Various other demographic or clinical risk factors of dementia have been identified in PD, such as age at the time of PD diagnosis, male sex, lower education level, visual hallucinations, REM sleep behavior disorder (RBD), abnormal color vision, autonomic dysfunction, and gait abnormalities [36, 47]. In this regard, the Montreal Parkinson Risk of Dementia Scale (MoPARDS), an office-based screening tool for the risk of dementia in PD, has been developed and validated recently [48]. This scale is composed of eight clinical items, defined as: 1) age older than 70 years, 2) male sex, 3) falls and/or freezing, 4) bilateral onset of motor symptoms, 5) history of RBD, 6) orthostatic hypotension, 7) MCI, and 8) visual hallucinations. Cognitive tests that have excellent accuracy in targeting patients who are more likely to develop dementia, such as the Block Design subtest, TMT part B (time), and Verbal Fluency (semantic), could be included among future screening tools.
The mechanisms underlying cognitive decline in PD are still poorly understood. Post-mortem studies suggest that the primary pathological substrate for dementia in PD is cortical Lewy body pathology, but Alzheimer’s disease pathology, vascular changes, and other pathologies also can play a role [5]. Furthermore, the anatomical distribution of these substrates varies between cases and does not always correlate with the clinical symptoms observed [49]. In addition to the heterogeneous cellular pathology underlying cognitive decline in PD, synaptic and mitochondrial dysfunction, neuroinflammation, and neurotransmitter activity are known to be involved in PD associated with dementia [5]. Therefore, the factors contributing to dementia in PD are complex and difficult to generalize to all patients. The multiple pathological substrates of cognitive decline in PD suggest that combined markers or predictive algorithms should be used in future research, since a single marker is unlikely to detect these complex and heterogeneous mechanisms. In this regard, the three cognitive tests identified in our study have excellent accuracy and could be included in these future studies.
Some limitations of this study should be noted. First, the sample was relatively small and the patients were recruited from two different university-hospital based movement disorder clinics. Further multicenter independent studies would be needed to validate the proposed tools in a broader PD population. Our results may also not be generalizable to either newly diagnosed patients or those with very advanced-stage PD. Future validation studies should thus include a broader disease duration range. Moreover, we were unable to follow up 20% of our study sample. The patients who were not included in follow-up were statistically equivalent to those who were reevaluated for age, education, severity of the disease and MMSE score at baseline, but attrition still has the potential to introduce bias in longitudinal studies. Second, not all PD patients underwent a formal neuropsychological assessment at the follow-up visit. However, among those patients who were unable to participate in an in-person evaluation, many had severe disability/dementia and some were institutionalized. Moreover, the number of tests within each cognitive domain was unequal, with executive and memory functions heavily represented. However, we were limited by the duration of the neuropsychological assessment. We cannot exclude the possibility that other cognitive tests measuring attention, visuospatial, or language abilities, not used in the present study, might be sensitive for detecting dementia in PD. Another limitation of this study is that the exact time of conversion to dementia in our PD patients is not always known. The follow-up duration was defined as the time between the baseline assessment and the diagnosis. Therefore, it is possible that some patients developed dementia between assessments. Finally, the group of patients who had developed dementia at follow-up were significantly older than the group that remained dementia-free. However, we used age as a covariate in our analyses, as well as aged-matched controls and age adjusted z scores to control for this effect.
Therapeutic interventions are available to improve motor symptoms in PD, yet no treatments or interventions are effective in slowing or preventing cognitive decline. A better understanding of the prodromal stages of dementia in PD is important to ensure adequate care for patients and the future development of targeted intervention.
FINANCIAL DISCLOSURES/CONFLICT OF INTEREST
Jessie De Roy received PhD scholarships from the Fonds de Recherche du Québec – Santé (FRQ-S) and Parkinson Canada.
Ronald B. Postuma reports grants and personal fees from the FRQ-S, the Canadian Institutes of Health Research (CIHR), Parkinson Canada, The W. Garfield Weston Foundation, The Michael J. Fox Foundation, The Webster Foundation, and personal fees from Roche/Prothena, Teva Neurosciences, Novartis Canada, Biogen, Boehringer Ingelheim, Theranexus, Takeda, Jazz Pharmaceuticals, Abbvie, Jannsen, and GE HealthCare, outside the submitted work.
Marina Brillon-Corbeil, Jacques Montplaisir, Daphné Génier Marchand, and Frédérique Escudier report no disclosures.
Michel Panisset has received research support from Abbvie and serves on advisory boards for Allergan and Merz.
Sylvain Chouinard has received research support from Abbvie.
Jean-François Gagnon received grants from The W. Garfield Weston Foundation, the CIHR, and the FRQ-S. He holds a Canada Research Chair in Cognitive Decline in Pathological Aging.
The authors have no conflicts of interest to report.
Footnotes
ACKNOWLEDGMENTS
Karen Grislis for linguistic revision of the manuscript.
This work was supported by the Canadian Institutes of Health Research, the Fonds de Recherche du Québec – Santé, and the Réseau Québécois de Recherche sur le Vieillissement. Dr. Gagnon holds a Canada Research Chair in Cognitive Decline in Pathological Aging.
