Abstract
Background:
Parkinson’s disease (PD) frequently causes progressive deterioration in speech, voice and cognitive aspects of communication. These affect wellbeing and quality of life and are associated with caregiver strain and burden. Therapeutic singing groups can ameliorate PD-related communication disorders and increase social interaction and wellbeing for caregivers and care recipients.
Objective:
To analyse the effects of ParkinSong group singing sessions on Parkinson’s communication and wellbeing outcomes for people with PD and caregivers over 12 months.
Methods:
A 4-armed controlled clinical trial compared ParkinSong with active non-singing control conditions over 12 months. Two dosage levels (weekly versus monthly) were available for each condition. ParkinSong comprised high-effort vocal, respiratory and speech exercises, group singing, and social interaction. PD-specific outcomes included vocal loudness, speech intelligibility, maximum phonation time, respiratory muscle strength, and voice related quality of life (QoL). Wellbeing outcomes were also measured for caregivers and care recipients.
Results:
We recruited 75 people with PD and 44 caregivers who attended weekly ParkinSong, monthly ParkinSong, weekly control or monthly control groups. We found significant improvements in the primary outcome of vocal loudness (p = 0.032), with weekly singers 5.13 dB louder (p = 0.044) and monthly singers 5.69 dB louder (p = 0.015) than monthly controls at 12 months. ParkinSong participants also showed greater improvements in voice-related QoL and anxiety. Caregivers who attended ParkinSong showed greater reductions in depression and stress scores.
Conclusions:
This 12-month controlled clinical trial of ParkinSong demonstrated improvements in speech loudness and voice-related QoL for participants with PD, and enhanced wellbeing for both caregivers and care recipients. No adverse effects were reported over 12 months and improvements were sustained.
Keywords
INTRODUCTION
One of the challenges for people living with Parkinson’s disease (PD) is knowing which therapies, activities and programs are supported by scientific evidence as well as benefiting health, wellbeing and social engagement. Each individual living with PD has a different array of motor and non-motor symptoms [1–3] and these vary over time [4, 5]. Personalised medicine is now advocated [6] given that symptoms change according to the daily Parkinson’s medication cycle, disease progression and co-morbidities. Alongside the well-recognised gait disorders, slowness, tremor, freezing and postural instability that characterize Parkinsonism [7, 8], speech, voice and communication are often affected [9] and neuropsychiatric non-motor symptoms such as depression, anxiety and apathy are prevalent [2]. Debilitating speech and voice sequelae are under-recognised yet surprisingly common, with up to 90% of people with PD and related conditions experiencing communication difficulties (such as reduced loudness, reduced pitch variation, reduced stress, imprecise consonants, inappropriate silences [10] and impaired sensory processing [11]) which negatively impact health-related quality of life [12, 13]. One of the most socially debilitating aspects of PD-related communication impairment is reduced speech loudness [14]. This, in combination with reduced self-perception of speech, reduced nonverbal communication and cognitive-linguistic impairments make communication difficult and often lead to avoidance of social interaction [14]. Loudness is also the main aspect of communication that is targeted in some PD-specific speech treatments, such as LSVT Loud [11], with reported secondary benefits from targeting loudness, such improved articulation and pitch variation. There is a need for accessible and evidence-based therapies to enable people living with PD to continue to speak clearly and loudly, and to communicate effectively throughout the time course of this debilitating disease, which can last for up to 25–30 years.
The range of evidence-based treatment options for speech and communication difficulties in people with PD is limited [15, 16]. Traditionally, speech pathology in PD has been provided as a one to one intervention over short and infrequent bursts, typically of around 4–6 weeks [16]. Despite an estimated 7–10 million people currently living with PD worldwide [17], the workforce of health professionals with PD-specific expertise cannot adequately meet this need. Speech therapy can be particularly challenging to access and costly to deliver. People with PD in remote and rural regions have even less access to allied health, nursing and comprehensive care services [18]. Alongside evidence-based speech pathology delivered in clinical settings, there is a need for community-based programs that are beneficial, enjoyable and accessible.
PD is also associated with caregiver strain and burden [19]. Caregivers often seek help with stress management, social support, information about disease management and coping with emotional distress [20]. The progressive nature of PD warrants a suite of treatment options that encourage and reinforce long-term maintenance of communication and social engagement as well as caregiver involvement to promote adherence and long term-compliance [19, 21]. In addition to the need to manage stress and burden for caregiver wellbeing, there is evidence to suggest that decreases in caregiver distress are associated with better care recipient wellbeing and functional outcomes [22–24].
Group singing is a promising medium through which to address both speech and non-motor symptoms in PD [25]. Singing shares neural networks and structural mechanisms used during speech [26]. It requires greater respiratory support, higher vocal effort, and can provide rhythmic cues to regulate tempo, and stress, through stimulation and organisation of motor output. Further, during singing the brain releases neurochemicals that increase feelings of pleasure and alertness and decrease anxiety and stress [27]. Targeted group therapy programs that utilise singing also afford social participation and active engagement in addition to creative musical expression. There is currently limited evidence on the effects of singing interventions in PD, but some promising preliminary studies have suggested potential to improve vocal loudness and other areas of speech and respiratory function [28–31]. However, methodological limitations include small samples, short intervention length, selection bias, and lack of control groups. Recently we implemented “ParkinSong”, an interdisciplinary program designed to improve communication and social connectedness for individuals with PD. Weekly group singing intervention session frequency has been previously reported to be useful and acceptable in PD [28, 33]. Initial findings from our 3-month trial showed improved vocal loudness, expiratory pressure and voice-related quality of life [25]. Whether a longer-term program has stronger or more lasting effects remains uncertain.
The aim of the current investigation was to evaluate the outcomes of a 12-month program of “ParkinSong” in people with PD of mild and moderate severity. We predicted that after a year of targeted group singing, the participants with PD would maintain improvements gained at 3 months in their vocal loudness and voice-related quality of life. We predicted that weekly group frequency would be more beneficial than monthly groups and also anticipated that caregivers would benefit from regular social engagement and support.
METHODS
This controlled clinical trial was designed to explore the effects of a 12-month program of “ParkinSong” singing group participation, compared to active PD control group participation, on vocal loudness, voice-related quality of life, well-being and relationship quality. Control participants with PD attended non-singing PD-specific groups such as painting, dancing, tai chi or peer support groups. Participants were not randomly allocated to groups in this small pragmatic exploratory trial. Caregivers of participants with PD from both intervention and control groups were also recruited and invited to attend the respective group activity program. To better understand dosage effects, outcomes from different group frequencies (weekly versus monthly sessions) were explored over the 12 months of group participation. To be eligible for the study participants needed to have diagnosis of PD using the MDS criteria [34] (or be a caregiver to a person with PD) and exclusion criteria included MMSE cores below 18 and severe language or hearing impairments. A further inclusion criterion was some degree of communication change in the person with PD, as reported by the person themselves, their caregiver or a health professional.
The trial was registered with the Australian and New Zealand Clinical Trials Registry (ACTRN12617000528358). Written informed consent was obtained from all participants and ethical approval was provided by Human Research Ethics Committees at The University of Melbourne (1544384) and Monash Health (15355A). English-speaking people with a diagnosis of PD according to the Movement Disorders Society criteria [34] and alternative parkinsonian syndromes were eligible to participate. Recruitment for the study was facilitated by Parkinson’s Victoria and participants enrolled from multiple areas across metropolitan Melbourne, Australia. Caregivers of consenting participants with PD were also recruited to assess the effect of group participation on caregiver wellbeing and relationship quality. Caregiver attendance was mandatory for participants with mild cognitive impairment (i.e., baseline MMSE 18–23). The MMSE was administered at the start of the baseline assessment. All additional therapies and PD medication use were recorded.
Measurements
Disease severity was measured at baseline and 12 months using the MDS Unified Parkinson’s Disease Rating Scale MDS-UPDRS [35]. Measures of speech, respiratory pressures, quality of life and wellbeing were obtained at baseline, after 3 months of group participation and after 12 months of group participation. We conducted all assessments at approximately the same time of day post-medication intake with participants in the ‘on’ state, at the peak effect of the PD medication cycle. We could not use blinded assessors due to budget limitations. Full details of the methodology have been previously reported [25]. The primary outcome was vocal loudness during conversational speech, elicited through a monologue speaking task [36]. Vocal loudness was selected as the primary outcome as it is the first and most pervasive component of speech to deteriorate in PD and also negatively effects intelligibility and communication participation [11]. Additional speech tasks also included a standardized loud sentence, maximum phonatory time of sustained vowel /a/, and reading of sentences randomly generated from the Speech Intelligibility Test [37]. All speech tasks were recorded using a Shure SM35-XL headset microphone and RPM626 XLR preamp (with a mouth-to-microphone distance of 5 cm), via Roland UA-55 Quad-Capture and calibrated with a sound level meter. De-identified recordings were analysed acoustically via PRAAT software [38] and perceptually by two expert raters who were blinded to both allocation and time. Intelligibility was determined via sentence transcription (Speech Intelligibility Test) and Direct Magnitude Estimation (DME) as per Vogel et al. [39]. Participants with PD also completed assessments of maximum inspiratory and expiratory pressure (using a portable MicroRPM respiratory pressure meter) and voice-related quality of life using the Voice Activity and Participation Profile (VAPP) [40]. We included maximum respiratory pressures as a secondary outcome as we hypothesized that these might be the mechanism by which to increase vocal loudness. The VAPP evaluates the perception of voice problem, activity limitation, and participation restriction using the World Health Organisation International Classification of Impairments, Disabilities and Handicaps concept. Other secondary outcomes were measured using the Quality of Carer Patient Relationship (QCPR) scale [41], the Depression, Anxiety and Stress Scale (DASS) [42] and the Euroqol-5 Dimensions (EQ-5D) [43]. Caregivers also completed the QCPR, DASS and EQ-5D. Weekly ParkinSong participants completed the Affect Grid [44], a single-item measure of valence and arousal, before and after every session.
Intervention
The 2-hour ParkinSong session protocol included 30 minutes of vocal warm-ups, exercises and activities designed to develop and extend respiratory strength and control, vocal loudness, articulation, pitch control, and communication confidence, and address sensory processing deficits [25]. The communication strategies were taught and practised to increase vocal loudness and to improve self-monitoring of communication and internal cueing. These were built on during 60 minutes of singing activities that used familiar songs and rounds, with a continued focus on respiratory support and high intensity vocal effort. Each session concluded with 30 minutes for socialisation and refreshments, where there were additional informal opportunities to practise communication strategies in a safe and supportive peer environment.
All group facilitators received training in the ParkinSong protocol with regular fidelity checking from the research team. Weekly sessions were facilitated by a music therapist, speech pathologist and allied health assistant. Monthly sessions were facilitated by community musicians and volunteers with training and support from music therapists and speech pathologists. Songs were not standardized but were selected according to participant preference and in accordance with a set of ParkinSong guidelines for song selection.
Control participants attended weekly PD dancing, painting, or tai chi classes or monthly peer support groups with a similar socialisation component. We chose active non-singing PD groups as our comparison in order to control for any potential effects of regular social interaction, peer support, and engagement in enjoyable group activity. We measured the effects of participation over 12 months, but many of these groups are ongoing.
Statistical analysis
Multiple imputation was conducted for missing data using data from 72 participants with PD (i.e., excluding the 3 participants who died). Imputation analyses were carried out using the MICE (Multivariate Imputations by Chained Equations) package in R software [45] using predictive mean matching with 20 imputed data sets. Missing outcomes at 3 and 12 months were separately imputed, but missing values at baseline were imputed at the participant level.
Continuous outcomes were summarized as means (SD). The outcomes were modelled using the imputed data in R. For some outcomes, the assumptions about the residuals were of concern, so transformations were used. These were log transformations (DASS, MIP, SIT, with scale adjustments to avoid zero scores) and square root transformations (VAPP). Group allocation was considered as a single factor with four levels, corresponding to the different combinations of activity and frequency. Two models were considered. The most complex, a two-way interaction model, considered the interaction of time (a factor with two levels: 3 months and 12 months) and group; baseline scores were included as a covariate, and participant as a random effect. Pairwise comparisons were obtained from this analysis to estimate the mean differences between the groups at each time point. In cases where the interaction of time and group was small, a simpler, main effects model was fitted with time and group as the main effects, baseline scores as a covariate and participant as a random effect. For this model, the pairwise comparisons estimated the mean differences between the groups, averaged across time. Differences between groups in baseline demographics were tested using one-way analysis of variance for continuous measures, and Fisher’s exact test for categorical variables. We also examined whether any other baseline variables influenced the main effect of group for the primary outcome of vocal loudness by including the following potential explanatory variables in the model: disease severity (H & Y score), speech severity (UPDRS speech score), PD medications, deep brain stimulation, and caregiver attendance. This analysis was carried out at each timepoint separately.
RESULTS
A total of 75 participants with PD (46 male) and 44 caregivers (14 male) were recruited (Fig. 1). Baseline demographics and group comparison results are presented in Table 1. See Tamplin et al. [25] for a detailed summary of baseline demographics.
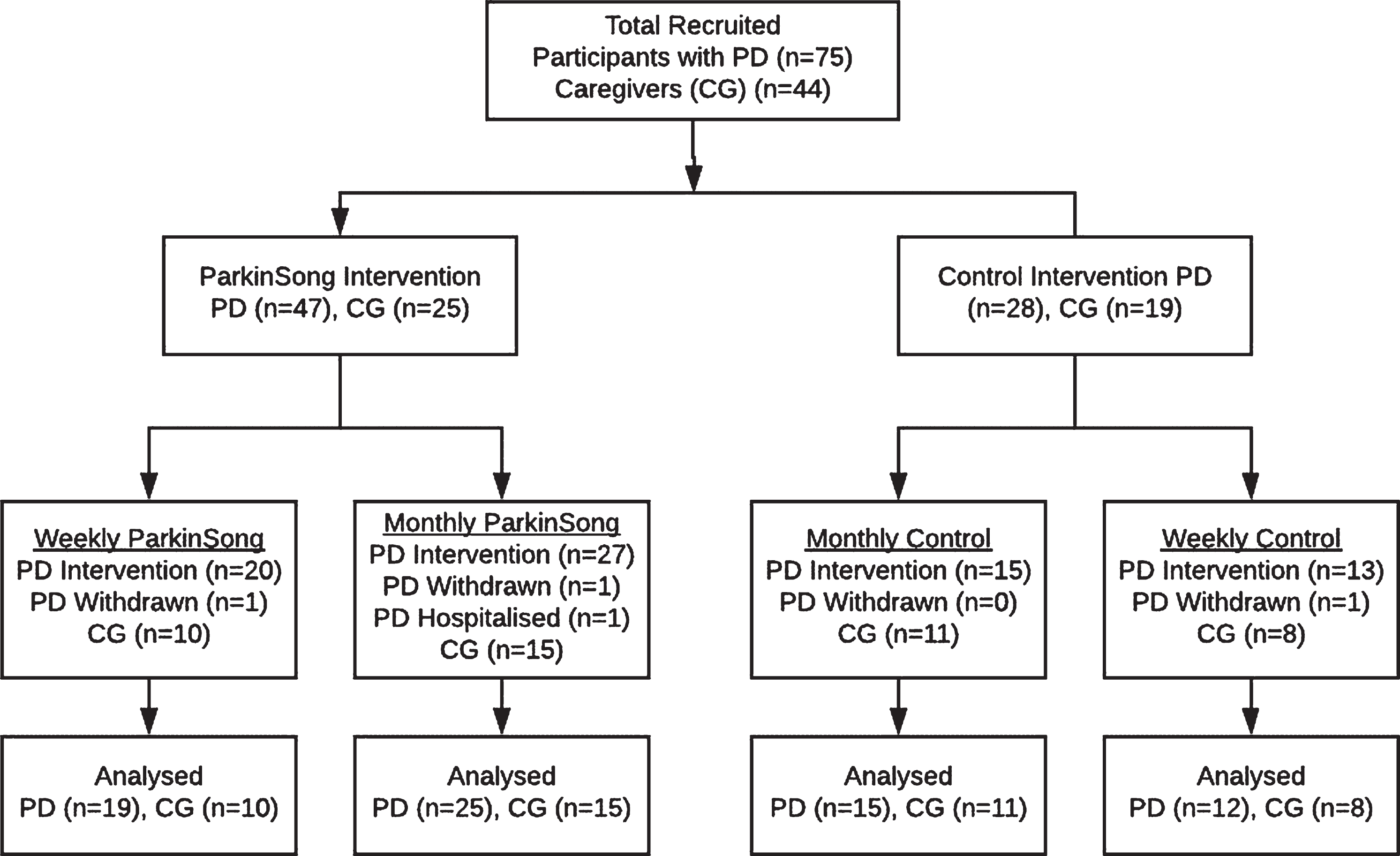
CONSORT Flowchart. PD, Parkinson’s disease; CG, caregiver.
Baseline demographics of participants with PD (includes ParkinSong singing groups and active control groups)
HY, Modified Hoehn & Yahr scale; MDS-UPDRS, Movement Disorders Society Unified Parkinson’s. Disease Rating Scale; MMSE, Mini-Mental State Exam. aTaken from question 3.1 of the Movement Disorders Society Unified Parkinson’s Disease Rating Scale.
The average age of participants with PD was 74 years (range 51 to 93 years). Most participants (n = 71) had idiopathic PD. There were two participants with Progressive Supranuclear Palsy and two with Multiple System Atrophy in the weekly ParkinSong group. Mean disease length was 9 years (range 1 to 26 years) and the median Hoehn and Yahr score was II. Analysis of baseline demographics indicated that disease severity, cognitive function, and severity of speech impairment did not vary significantly between groups. Caregivers were mostly spouses (n = 42) and 2 were adult children. Three participants with PD died between the 3 and 12-month time points. No adverse responses to the ParkinSong intervention were reported. The results of this study at the primary time-point of interest (3 months) have been previously published [25]. The focus of this paper is to examine outcome trajectories at 12 months for participants with PD and also caregivers.
Primary outcome: vocal loudness for participants with PD
Participants with PD who attended ParkinSong therapeutic singing groups (both weekly and monthly) increased their conversational speech loudness over 12 months. The statistically significant group by time interaction effect (p = 0.032) for mean sound pressure level on the monologue task (Table 2) also indicated that differences between groups was not the same at 3 and 12-month timepoints. At 3 months the weekly singers had increased their vocal loudness more than the other three groups. They were louder than weekly controls by 4.9 dB (95% Confidence Interval (CI) 0.03 to 9.8, p = 0.048), monthly singers by 5.8 dB (95% CI 1.6 to 9.9, p = 0.007), and monthly controls by 7.2 dB (95% CI 2.4 to 11.9, p = 0.003). At 3 months the other three groups either retained a similar speech volume to baseline or deteriorated (Fig. 2). Between 3 and 12 months mean vocal loudness decreased for weekly singers (but remained significantly higher than baseline). Between 3 and 12 months, vocal loudness for monthly singers continued to increase and almost matched weekly singers. Post hoc analysis indicated that this difference (3.7 db higher) between 3-month and 12-month mean sound pressure level for monthly singers was statistically significant (95% CI 0.9 to 6.5, p = 0.011). In addition, post hoc analysis showed that both weekly singers (5.13 dB, 95% CI 0.15 to 10.10, p = 0.044) and monthly singers (5.69 dB, 95% CI 1.13 to 10.25, p = 0.015) had significantly higher vocal loudness at 12 months than monthly control group participants. The control groups continued on the same trajectories from 3–12 months (weekly control group participants remained stable, and the monthly controls declined) as can be seen in Fig. 2.
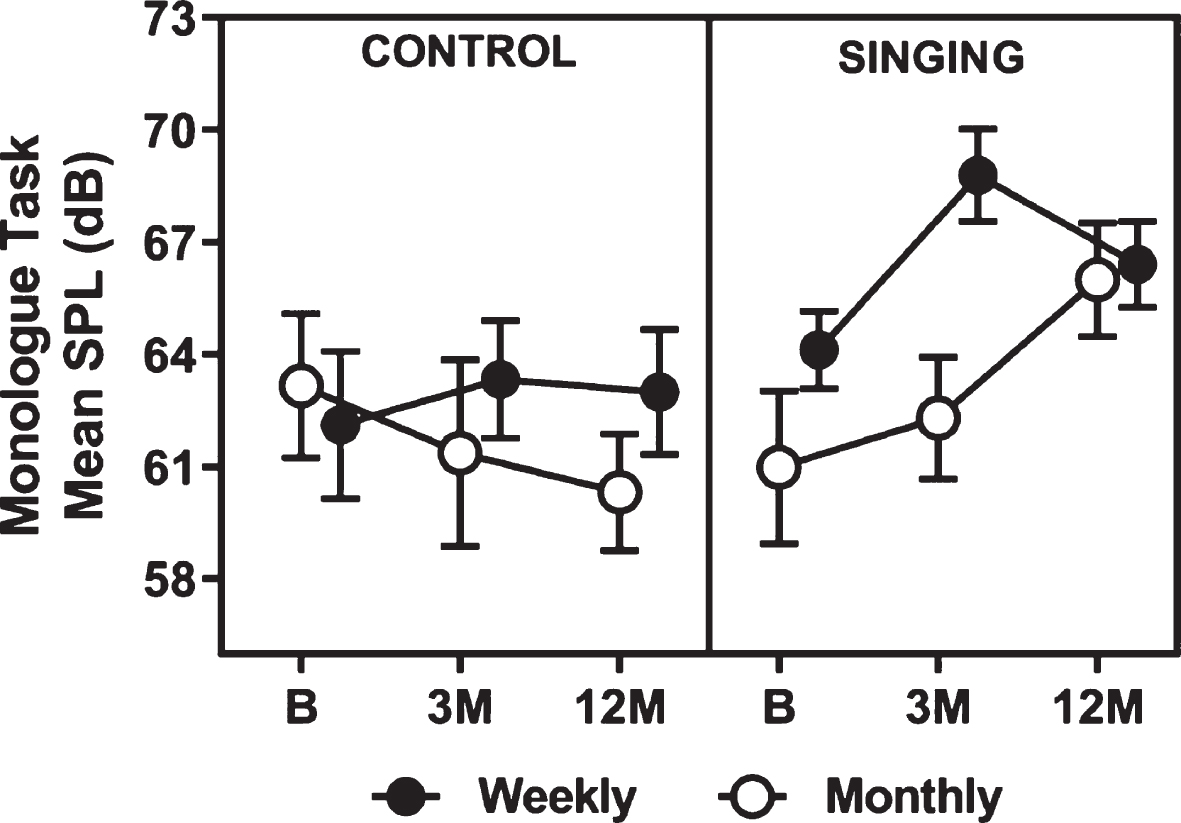
Mean raw data for Sound Pressure Level (dB) on the monologue task. SPL, sound pressure level; dB, decibels; B, baseline; 3 M, 3 months; 12 M, 12 months.
Raw mean scores for all voice, speech and respiratory measures for participants with PD in each group and timepoint
*p values are reported for ANCOVA analyses (including imputed data) using baseline scores as the co-variate, based on a main effect of group unless otherwise indicated. ∧statistically significant group difference. # analysis based on transformed data. dB, decibels; sd, standard deviation; SIT, Speech Intelligibility Test; Max, Maximum; MEP, Maximum Expiratory Pressure; MIP, Maximum Inspiratory Pressure; cm/H2O, centimeters of water; VAPP, Voice Activity and Participation Profile.
For the primary outcome of change in conversational vocal loudness over 12 months, additional analyses were carried out that adjusted for five potential explanatory variables (disease severity, speech severity, PD medications, deep brain stimulation, and caregiver attendance). In these analyses the effects of baseline vocal loudness and group were accounted for in the presence of these additional variables. None of these variables significantly affected vocal loudness results.
For the standardized loud sentence task there was a statistically significant group by time interaction effect (p = 0.013). The weekly singers were significantly louder than the other three groups at 3 months (8.5 dB louder than weekly controls, 95% CI 1.3 to 15.6, p = 0.021; 15.7 dB louder than monthly controls, 95% CI 8.3 to 23.1, p < 0.001; and 8.7 dB louder than monthly singers 95% CI 2.4 to 14.2, p = 0.008). Loudness deteriorated between 3 and 12 months for weekly singers and there were no significant differences between groups at 12 months.
Secondary outcomes: speech and respiratory measures for participants with PD
There were no statistically significant differences between groups at 12 months on maximum expiratory pressure (p = 0.15), maximum inspiratory pressure (p = 0.31) or maximum phonation time (p = 0.74). Although not statistically significant in this analysis, the weekly singers showed clinically relevant improvements in maximum expiratory pressure scores at 3 months and higher mean scores were maintained at 12 months.
A significant group main effect (p = 0.001) was found for perceptual ratings of speech intelligibility using the Direct Magnitude Estimation data. This was primarily due to the large decrease in intelligibility observed in the monthly controls, which was significantly different to the other groups (who maintained or improved on high baseline intelligibility scores over the 12 months). There were no statistically significant results for the Speech Intelligibility Test data (p = 0.11).
Voice-related quality of life improved significantly (i.e., scores decreased) for people with PD who attended weekly ParkinSong sessions for 12 months (Fig. 3). This was demonstrated by a significant group main effect (p = 0.039) on the Voice Activity and Participation Profile (VAPP) total score. Total VAPP scores decreased at 3 months for weekly singers, but increased again between 3 and 12 months, while all other group scores worsened over 12 months. Pairwise comparisons revealed a significant difference between weekly singers and monthly controls raw scores of 437 (using square root transformation 8.68, 95% CI 2.87 to 14.4, p = 0.004).
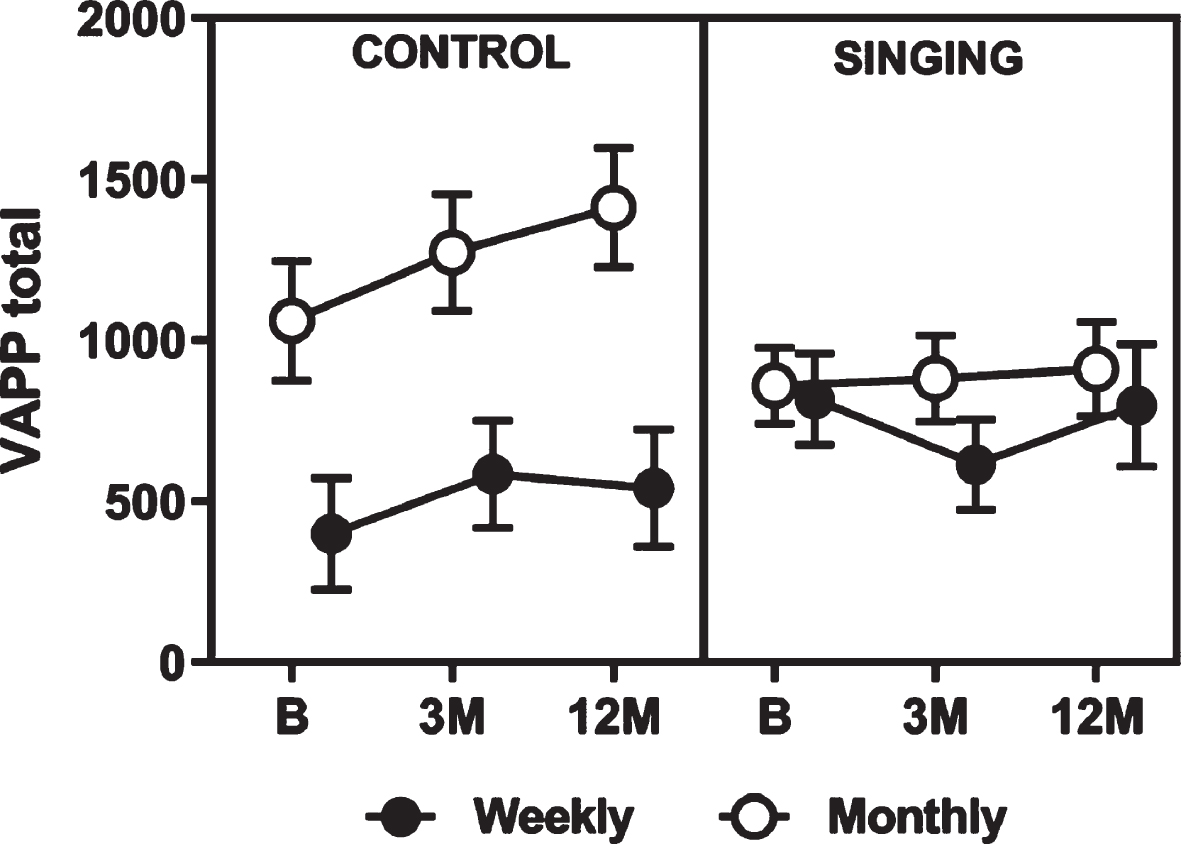
Mean raw data for Voice Activity and Participation Profile (VAPP) total scores. VAPP, Voice Activity and Participation Profile; B, baseline; 3 M, 3 months; 12 M, 12 months.
Secondary outcomes: wellbeing measures for participants with PD
There was a statistically significant group main effect (p = 0.037) for anxiety (measured using DASS data). Anxiety scores were lower for weekly singers and weekly controls at 3 months in comparison to both monthly groups and stayed low at 12 months whereas anxiety scores increased for monthly controls (Fig. 4). Mean difference in raw anxiety scores for weekly singers was 2.5 points (log transformed mean difference = 0.45, p = 0.010) and 2.32 points for weekly controls (log transformed mean difference = 0.44, p = 0.021). All scores were within the normal range for anxiety (0–7) for all groups at all timepoints.
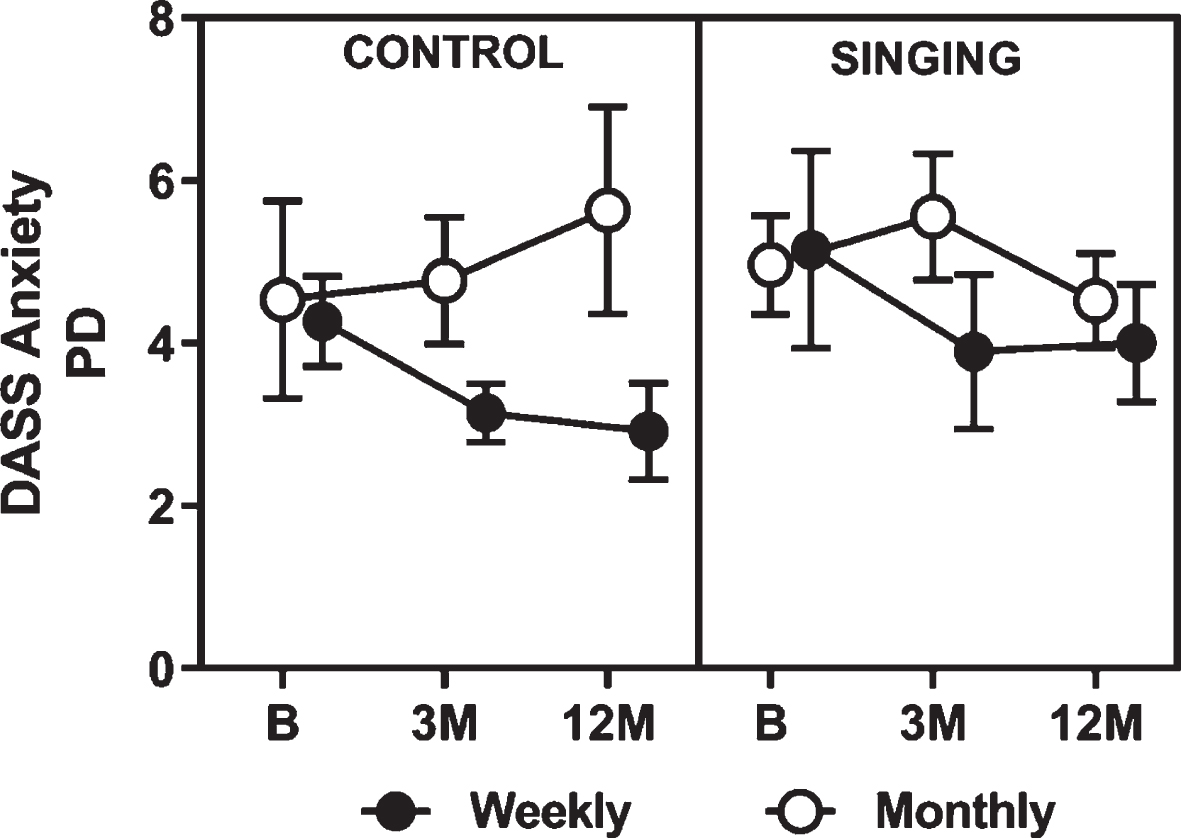
Mean raw PD participant data for anxiety scale of the Depression, Anxiety & Stress Scale. DASS, Depression, Anxiety and Stress Scale; PD, Parkinson’s disease; B, baseline; 3 M, 3 months; 12 M, 12 months.
No statistically significant differences were found between groups in ratings of depression or stress (measured using DASS data). Trends in raw mean scores showed decreases in both depression and stress for weekly singers and increases for monthly controls, which although not statistically significant, may still be clinically relevant. In comparison, raw mean depression and stress scores for weekly controls and monthly singers remained relatively stable over 12 months (Table 3). No statistically significant differences were found between groups in ratings of overall health (EQ-5D, p = 0.51) or relationship quality (QCPR, p = 0.89).
Raw mean scores for wellbeing measures for each group and time point
*p values are reported for ANCOVA analyses (including imputed data) using baseline scores as the co-variate, based on a main effect of group unless otherwise indicated. ∧statistically significant group difference. # analysis based on transformed data. PD, Parkinson’s disease, CG, caregiver; QCPR, Quality of Carer Patient Relationship scale; EQ-5D-OH, European Quality of life scale – 5 Dimensions - Overall Health; DASS, Depression, Anxiety and Stress Scale, DASS-D, Depression scale on DASS; DASS-A, Anxiety scale on DASS; DASS_S, Stress scale on DASS.
The Affect Grid measure was only implemented with the weekly ParkinSong participants (both PD and caregivers). On the 9-point scale there was an improvement in affect from pre to post session over the intervention period. Results showed a mean improvement in both dimensions of affect: increase in valence (positive emotion) of 0.88 points (95% CI 0.78 to 0.99, p < 0.001), and arousal (energy level) of 0.87 (95% CI 0.75 to 0.99, p < 0.001) from pre to post session over the 12-month intervention period. Caregiver improvements in affect from pre to post session were even greater, with a mean increase in valence of 1.7 (95% CI 0.12 to 1.47, p < 0.001) and arousal of 1.9 points (95% CI 0.13 to 1.59, p < 0.001).
Secondary outcomes: caregiver wellbeing
Improvements in depression, anxiety and stress were evident from the DASS data for caregivers who attended weekly ParkinSong groups (Fig. 5). For depression, there was a significant interaction effect of group and time (p = 0.05), measured using transformed DASS data. Pairwise comparisons showed a significant difference of 3.6 points (log transformed mean difference = 0.63, 95% CI 0.06 to 1.21, p = 0.031) at 12 months between caregivers in the weekly singing groups (whose depression scores decreased) and monthly control groups (whose depression scores increased). All scores were within the normal range for depression (0–9) for all groups at all timepoints.
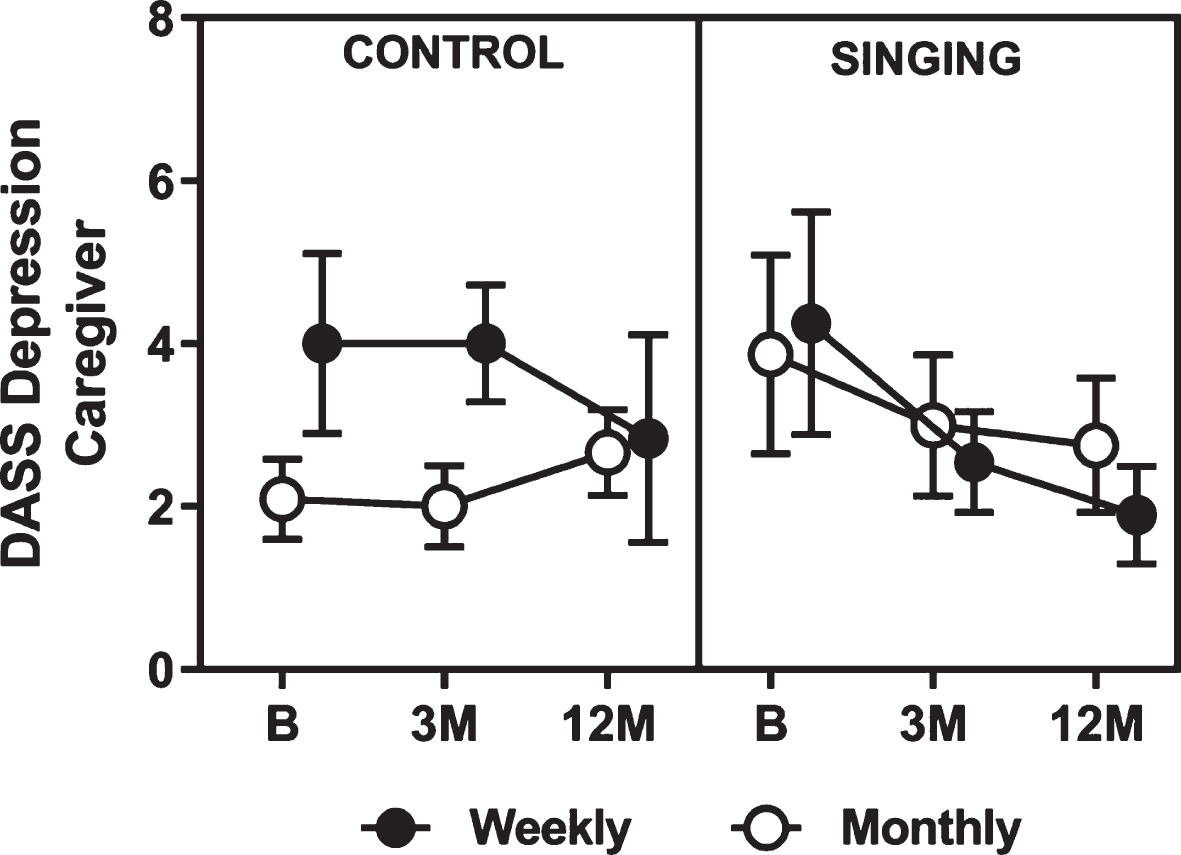
Mean raw caregiver data for the depression subscale of the Depression Anxiety and Stress Scale. DASS, Depression, Anxiety and Stress Scale; B, baseline; 3 M, 3 months; 12 M, 12 months.
There were no statistically significant differences between groups for caregiver anxiety, but clinically relevant trends towards a group main effect (p = 0.067) for caregiver stress (Table 3). Caregivers in the weekly singing groups had significantly lower stress scores than that of weekly controls by 3.6 points (log transformed mean difference = 0.53, p = 0.044) and of monthly controls by 3.5 points (log transformed mean difference = 0.57, p = 0.022) at 12 months. All scores were within the normal range for stress (0–14) for all groups at all time points.
There were no statistically significant differences between groups for caregiver quality of life (EQ-5D, p = 0.70) or perceived relationship quality as measured by the QCPR. However, the QCPR results were approaching significance (p = 0.09) and could be meaningful in a more highly powered study. As shown in Table 3, caregiver QCPR total scores increased by a mean of 15 points for the weekly caregiver singers (5 points for monthly singers, 4 points for weekly controls) and decreased by around 2.5 points for the monthly caregiver controls.
DISCUSSION
The 12-month ParkinSong therapeutic singing program was accepted by participants with no adverse effects and improved vocal loudness and voice-related quality of life in people living with PD. Participants with PD who attended either weekly or monthly ParkinSong groups showed increases in vocal loudness (by 5.13 dB and 5.69 dB, respectively) in comparison to monthly control participants whose vocal loudness declined as predicted with disease progression. These differences in decibel levels between treatment and control groups over 12 months are clinically relevant and greater than between-group differences achieved in LSVT-Loud therapy (2.4 dB) * over 7 months [11]. A greater improvement in vocal loudness was achieved earlier by weekly ParkinSong attendees, which is likely due to the higher dosage of therapy received by the weekly group. The ParkinSong participants who attended monthly sessions for 12 months eventually achieved a similar increase in vocal loudness as the weekly group, although the improvement trajectory was slower. The decay in loudness in the weekly singing group at 12 months may be due to a treatment ceiling effect, natural disease progression or a combination of both. These findings suggest that intensive singing and practice of voice and communication strategies might afford both an increase in functional reserve and/or a neuro-protective effect against a decline in vocal loudness in PD in line with results from a small study by Yinger et al. [30]. These findings are in contrast to other previous studies that found no significant improvements in vocal loudness following a singing intervention [29, 47]. Further, the improvements in voice-related quality of life seen in weekly ParkinSong participants show the functional benefit of increased vocal loudness and regular practise on communication confidence and participation. This is one of the first studies to look at the effects of a targeted singing intervention in PD over such a long timeframe (12 months), which is important given the lengthy trajectory of this degenerative disease. By increasing the sample size and intervention length and including active control groups, we have attempted to overcome some of the methodological limitations reported in previous studies.
There are a number of possible mechanisms relevant to the production of speech that could potentially be activated through musical experiences and singing. Firstly, engagement in musical behaviour might bypass the basal ganglia neural circuits that are compromised in PD [48, 49]. Motor speech output involves sequences of sub-movements. The facilitating effect of music on motion during external cueing [50, 51] and during mental singing [52], might occur during singing. This is further supported by a recent study, which found that expressive and receptive prosodic aspects of music were preserved in people with PD, affording a facilitatory effect on vocal behaviour [53]. The role of abnormal central sensory processing, internal cueing and self-monitoring in speech disorders in PD is widely recognised [54–56]. Therefore, contemporary methods of speech and language therapy for PD incorporate interventions targeting sensory perception [11]. ParkinSong included a combination of intensive high-effort vocal exercise and targeted structured singing activities (rather than casual sing-alongs). This inter-disciplinary “package” of speech-based vocal exercises, vocal warm-ups and singing interventions aimed to activate multiple neural networks [57] subserving sensory perception, self-monitoring of speech and voice, together with motor elements of the speech mechanism. We believe that the inclusion of caregiver participation in the ParkinSong intervention was also key to its success. In addition to promoting adherence and long term-compliance we were able incorporate caregivers in treatment provision and carryover by providing education around PD-related communication issues and teaching strategies to overcome these.
Singing has also been described as a form of respiratory muscle training due to the higher levels of respiratory support and control that are required in singing in comparison to speaking [58, 59]. By placing increasing demands on the respiratory support system via vocal and singing exercises we can stimulate increases in vocal loudness. We found significant improvements in maximum expiratory pressure at 3 months for weekly ParkinSong attendees [25], in line with a previous pre-post PD singing intervention study [29]. The use of multiple imputation in the current analysis increased the variability in scores, which likely reduced the potential to detect an effect in the current analysis.
The improvements in mood and energy levels during ParkinSong sessions demonstrated by the Affect Grid results showed that group singing is enjoyable and motivating. Singing can be facilitated in a social context, further supporting its application in therapy to address goals for wellbeing and social connection. Singing interventions have previously been reported to improve symptoms of depression [60–62], anxiety [62–64] and stress indicators [62, 66] in other populations, but to our knowledge have not yet been examined in prior singing-based PD research. In the current study we found decreases in anxiety in both weekly PD singers and weekly PD control participants, which may suggest a positive effect of regular social interaction and peer support. Given that the weekly control participants attended dancing and tai chi groups, this finding may also support previous research suggesting that these activities are also beneficial for improving wellbeing in PD [67, 68]. Stress scores decreased significantly at 3 months for weekly singers and remained low at 12 months. This supports previous research showing reductions in cortisol following group singing experiences [65].
The reduction in caregiver ratings of depression, anxiety and stress after the ParkinSong program shows that benefits can extend beyond those living with basal ganglia dysfunction to the significant other people in their lives. Given that caregiver strain is common in PD [21], this finding is notable, particularly as decreases in caregiver distress have been associated with better care recipient wellbeing and functional outcomes in previous research [22–24]. Relationship quality as perceived by caregivers was not significantly different between groups over the 12-month intervention period of this study. It is possible that additional interventions to support relationships are warranted.
There were several limitations of this clinical trial. The participants were not randomly allocated to conditions, hence there was potential for selection bias to influence the results. The lack of assessor blinding creates a further risk of bias. The difference in facilitators between monthly and weekly groups is also a limitation. In addition, people with PD and caregivers reported that the Quality of Carer Patient Relationship scale was difficult to complete due to the use of double negatives in some questions. Further proposed limitations around the measurement of intelligibility, and lack of control for medication changes and additional therapy (although comparable across groups) were reported previously [25].
Conclusions
Twelve months of participation in a PD-specific therapeutic singing program had positive and sustained effects on vocal loudness and voice-related quality of life. ParkinSong participants who attended monthly sessions reached comparable gains in vocal loudness over 12 months, although the rate of improvement was much slower than for weekly attendees. The ParkinSong intervention has also shown potential to address various aspects of wellbeing such as depression, anxiety and stress, to different degrees in people with PD and their caregivers.
CONFLICT OF INTEREST
Dr. Tamplin receives salaried support from the National Health and Medical Research Council, Australia (Fellowship ID 1106603) and also receives institutional support from The University of Melbourne.
Prof. Morris is funded by NHMRC grants (APP1134495, APP1104237, APP1114210) and Parkinson’s Victoria. She has a joint appointment with Health scope and La Trobe University.
Prof. Vogel is chief science officer of Redenlab who provided assistance with the acoustic analysis. He receives salaried support from the National Health and Medical Research Council, Australia (Fellowship ID1152223) and also receives institutional support from The University of Melbourne.
Mrs. Marigliani, Prof. Baker, and Dr. Noffs report no disclosures.
Footnotes
as calculated using monologue change from baseline scores of LSVT LOUD participants minus change scores from untreated participants at 7 months post therapy.
ACKNOWLEDGMENTS
This research was funded by The University of Melbourne, La Trobe University, and Parkinson’s Victoria (Australia). J Tamplin and A Vogel were supported by National Health and Medical Research Council Fellowships (#1106603 and #1135683 respectively). We would like to acknowledge and thank Monash Health and the Hugh Williamson Foundation for their support of this research. Special thanks also to the people living with Parkinson’s who gave so generously of their time to participate in this research.
