Abstract
Background:
Sporadic Parkinson’s disease (PD) patients have lower
Objective:
Determination of PD prevalence in FD and clinical, biochemical and vascular neuroimaging description of FD pedigrees with concomitant PD.
Methods:
Clinical screening for PD in 229 FD patients belonging to 31 families, harbouring
Results:
Prevalence of PD in FD was 1.3% (3/229) (3% in patients aged ≥50 years). Three FD patients, one female (73 years old) (P1) and two males (60 and 65 years old) (P2 and P3), three different pedigrees, presented akinetic-rigid PD, with weak response to levodopa (16% – 36%), and dopaminergic deficiency on 18F-DOPA PET. No pathogenic mutations were found in a PD gene panel. FD+/PD+ patients had worse clinical severity of FD (above upper 75% IQR in Mainz scores), and cortico-subcortical white matter/small vessel lesions. P3 patient was under enzyme therapy, started 1 year before PD diagnosis. P2-P3 patients had higher leucocyte
Conclusion:
We have shown a high prevalence of PD in a late-onset phenotype of FD, presenting high cerebrovascular burden and weak response to levodopa. Further studies will untangle how much of this PD phenotype is due to Gb3 deposition versus cerebrovascular lesions in the nigro-striatal network.
INTRODUCTION
Anderson-Fabry Disease (FD) is a rare X-linked lysosomal storage disorder caused by deficient activity of the enzyme
The potential molecular link was primarily driven by the description of small series of atypical Parkinsonism in Gaucher type I [4] and in “non-neuronopathic” GD [5, 6], and later on by the description of increased risk of developing PD in asymptomatic carriers of
Parnetti et al., when analysing the cerebrospinal fluid of PD patients (
Following two clinical reports of FD patients that later on developed parkinsonism [12, 13], Wise et al. [14] performed an online survey and family history questionnaire to determine the prevalence of PD in 90 FD patients. The authors included 63 females and 27 males and identified 2 PD patients (2.2%), carrying the p.Y134X mutation and the p.E59V variant in the
Herein we present the prevalence of PD in a large cohort of 229 FD patients, and analyse the pedigrees of three FD patients with concomitant PD, with respect to clinical, biochemical and vascular brain neuroimaging markers.
METHODS
This research was approved by the hospital Ethics Committee of the Hospital Senhora da Oliveira, Guimarães, Portugal (Ref. CAc 42/2018). The study was thoroughly explained to all patients, who provided written informed consent. We have included 229 adult patients (≥18 years) from 31 families carrying the
FD patients were submitted to a biannual follow-up, including a systematic multidisciplinary clinical assessment with characterization of the multiorgan involvement by FD [18]. Mainz severity score index (MSSI) was estimated in all patients. The neurological evaluation was performed by the same neurologist and was focused on central and peripheral nervous system complications associated with FD (cerebrovascular disease, cognitive and behaviour complications, depression, anxiety, acroparesthesias, neuropathic pain). Mainz score (neurological sub-score) [19] and general neuropsychological evaluation by Montreal Cognitive Assessment (MoCA) [20] were also evaluated. Brain imaging, by magnetic resonance imaging (MRI) or computed tomography in patients with pacemaker, was performed in all patients, and qualitative description of the vascular brain imaging findings (presence and location of white matter/ischemic/haemorrhagic lesions, global cortical atrophy scale) was specifically collected for the current study based on neuroradiology records. Comorbid cardiac and cerebrovascular risk factors (previous transient ischemic attack, previous ischemic/haemorrhagic stroke, cardiac disease, hypertension, dyslipidaemia, diabetes, smoking) were collected based on clinical records. Patients with FD were diagnosed with parkinsonism according to MDS-clinical criteria for PD [21], and grouped as FD+PD+, and performed a brain 18F-DOPA PET. Genetic testing targeting the major known PD genes (ANO3, ATP13A2, ATP1A3, ATP6AP2, C190RF12, CHCHD2, COASY, CSF1R, DCTN1, DNAJC6, EIF4G1, FBXO7, FTL, GBA, GCH1, GNAL, GRN,LRRK2, MAPT, NR4A2, OPA3, PANK2, PARK7, PDE10A, PDGFB, PDGFRB, PINK1, PLA2G6, POLG, PRKN, PRKRA, RAB39B, SGCE, SLC18A2,SLC20A2, SLC30A10, SLC39A14, SLC6A3, SNCA, SNCAIP, SPG11, SPR, SYNJ1, TAF1, TH, THAP1, TOR1A, TUBB4A, TWNK, UCHL1, VCP, VPS13A,VPS13C, VPS35, WDR45 and XPR1) were requested for PD patients. In order to ascertain family history of PD, each FD+PD+ participant was asked about participant’s personal and family history of PD, specifically if other family members without FD, had been diagnosed or treated for PD or showed common symptoms of PD (resting tremor, shuffling gait, stooped posture, decreased arm swing, rigidity). PD clinical markers [MDS-UPDRS I-IV score off/on stage), subtype (tremor, akinetic-rigid, mixed, PIGD), Hoehn-Yahr stage, levodopa equivalent daily dose (LED)] were analysed.
Statistical analysis
Cohort characteristics and demographic data, for the two groups (FD+PD+ and their FD family members, FD+PD–), were reported using medians and inter-quartile range (IQR) for continuous variables and frequencies for categorical variables. As FD is X-linked, we stratified statistical analyses by gender. In order to perform a comparison between the two groups, due to the limited number of FD+PD+ subjects, we outlined their position in respect to the 95% confidence intervals estimated for the largest group (FD+PD–).
RESULTS
In 229 adult patients with FD due to the p.F113L mutation (157 patients aged ≥35 years; 101 patients aged ≥50 years; 61 patients aged >60 years), we found three patients with PD (prevalence of PD in FD: 3/229, 1.3%; 3/157, 1.9%; 3/101, 3%; 3/61, 4.9%, according to age cut-off).
FD+PD+ patients, one female (73 years old) (P1) and two males (60 and 65 years old) (P2 and P3), presented akinetic-rigid PD with a weak response to levodopa (6% – 36%) (LED: 300–500 mg) (Table 1) and no dyskinesias. The female patient (P1) presented PD 8 and 12 years later in comparison to male patients (P2 and P3, respectively), clearly offsetting as a more advanced stage of PD (Table 1), albeit without postural instability (HY = 2), presenting gait impairment and flexed posture refractive to levodopa (off/on MDS-UPDRS subcores: 3.10. (1/1); 3.13. (2/2)). Patient P1, besides p.F113L mutation, also carried a genetic variant of uncertain significance, in homozygous state, in the SLC30A10 gene (c.284C>T; p.Thr95Ile). This variant has been described in individuals manifesting with a syndrome of Hepatic Cirrhosis, Dystonia, Polycythemia and Hypermanganesemia; and individuals with juvenile-onset Dystonia and adult-onset Parkinsonism [22]. Patient P2 carried a genetic variant of uncertain significance, in heterozygous state, in LRRK2 gene (c.5687C>G; p.Ala1896Gly) that, to our knowledge, has not been reported yet. Nevertheless, in both P1 and P2 cases, no family history of parkinsonism was found in the previous generation. P3 patient had a negative test in the PD gene panel. 18F-DOPA PET scan revealed nigro-striatal dopaminergic presynaptic deficit in P1, P2 and P3 (Fig. 2). All FD+PD+ patients presented brain WML and the female patient (P1) had suffered a clinical stroke (Table 2, Fig. 3).
Demographics and clinical characteristics of FD+PD+ patients (P1, P2, P3) and their FD+PD–relatives
Data is presented individually for each of the three patients with Parkinson’s disease (PD) and Fabry disease (FD) and collectively for their relatives with FD in the three pedigrees (median/ interquartile range (IQR)) for continuous variables and number of patients or Yes/No for categorical variables). 1-two females and one male without MRI; 2-data available for 16 females; 3-data available for 9 females; 4- Data available for 11 females;.5-data available for 6 females; CRF, cerebrovascular risk factors; LED, Levodopa equivalent dose; ERT, enzyme replacement therapy; LVH, left ventricular hypertrophy; HBP, high blood pressure; NA, non-available data; MoCA, Montreal Cognitive Assessment; OT, other meds; PDQ-39, Parkinson’s disease Questionary-39; TIA, transient ischemic attack; UPRDRS, Unified Parkinson’s Disease Rating Scale in “Off” stage (without levodopa medication) and “On” stage (on levodopa medication); WML, white matter lesions. Reference values: enzymatic activity of
Neurological and Neuroimaging findings in FD+PD+ patients and their FD+PD–relatives
Data is presented individually for each of the three patients (P1–P3) with Parkinson’s disease (PD) and Fabry disease (FD) and collectively (median/ interquartile range (IQR)) for continuous variables and number of patients Yes/No for categorical variables) for their relatives with FD in the three pedigrees stratified by gender; 1-two females without MRI; 2-one male without MRI; ACGS, Global cortical atrophy scale; CT, computerized tomography; MRI, magnetic resonance imaging; WML, white matter lesions.
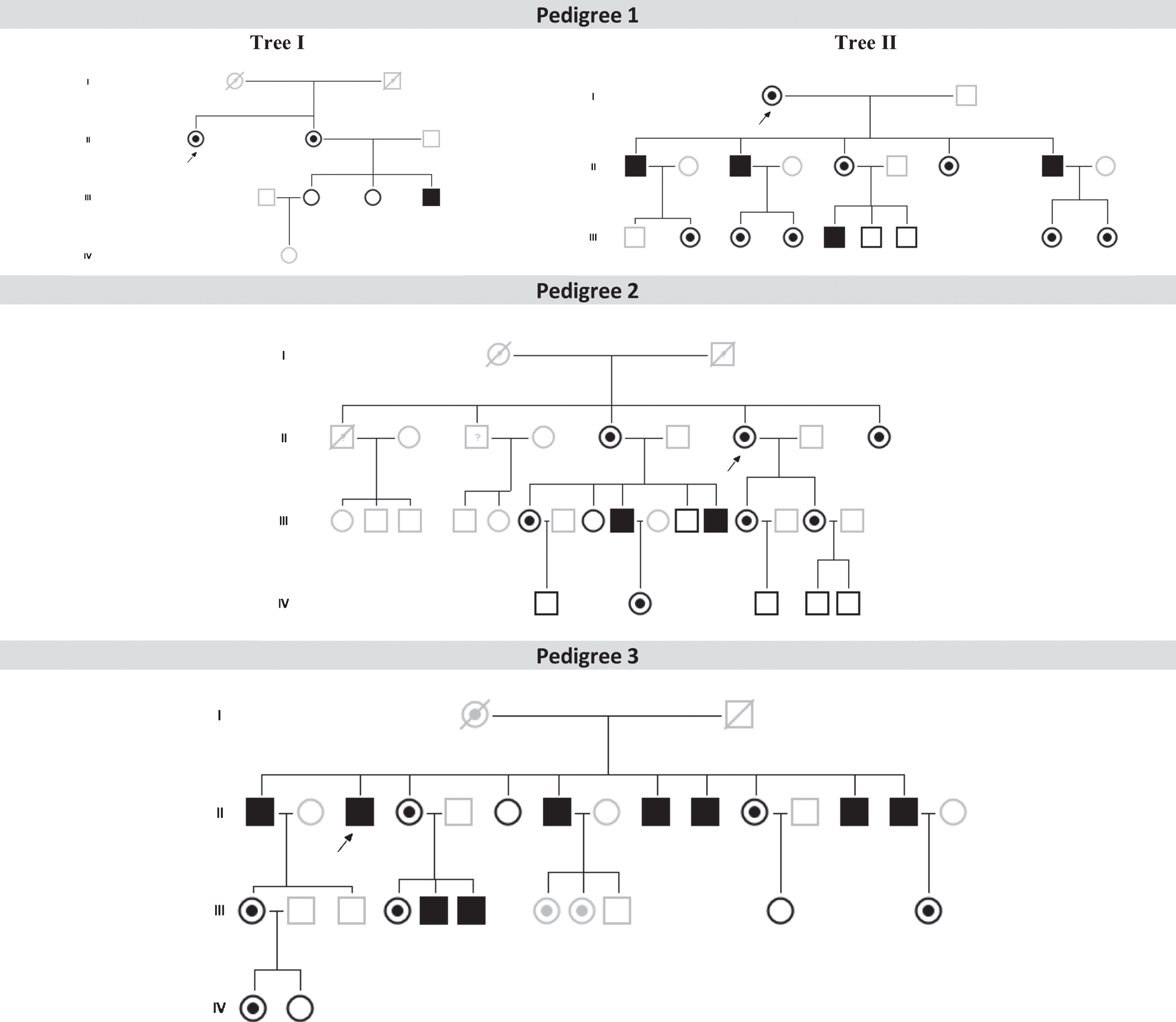
Family pedigrees from the three patients with Parkinson’s disease patients and Fabry disease (FD+PD+) (p. F113L mutation) and their relatives with or without FD. Index FD patients are marked with a black arrow (in Pedigree 1, individuals II-1 from Tree I and I-1 from Tree II are the same index patient). Patients with FD and PD: P1 (Pedigree 1, tree I, II-2); Patient 2 (Pedigree 2, III-9); Patient 3 (Pedigree 3, II-1). Dark symbols represent FD patients. White symbols represent relatives without FD. Grey symbols represent untested individuals.
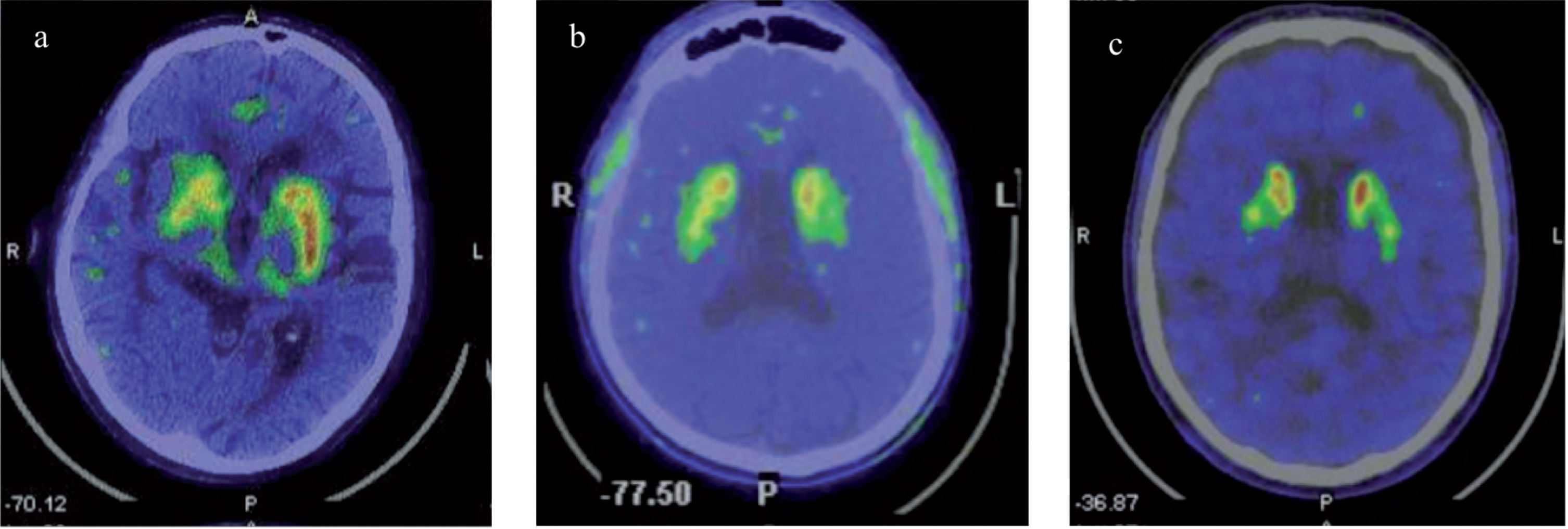
18F-DOPA PET scan performed in patients with Fabry Disease and Parkinson’s Disease revealed: a) reduced right striatal dopaminergic presynaptic deficit in patient P1; b) reduced left striatum dopaminergic presynaptic deficit in patient P2; c) and reduced right nigro-striatal dopaminergic presynaptic deficit in patient P3.
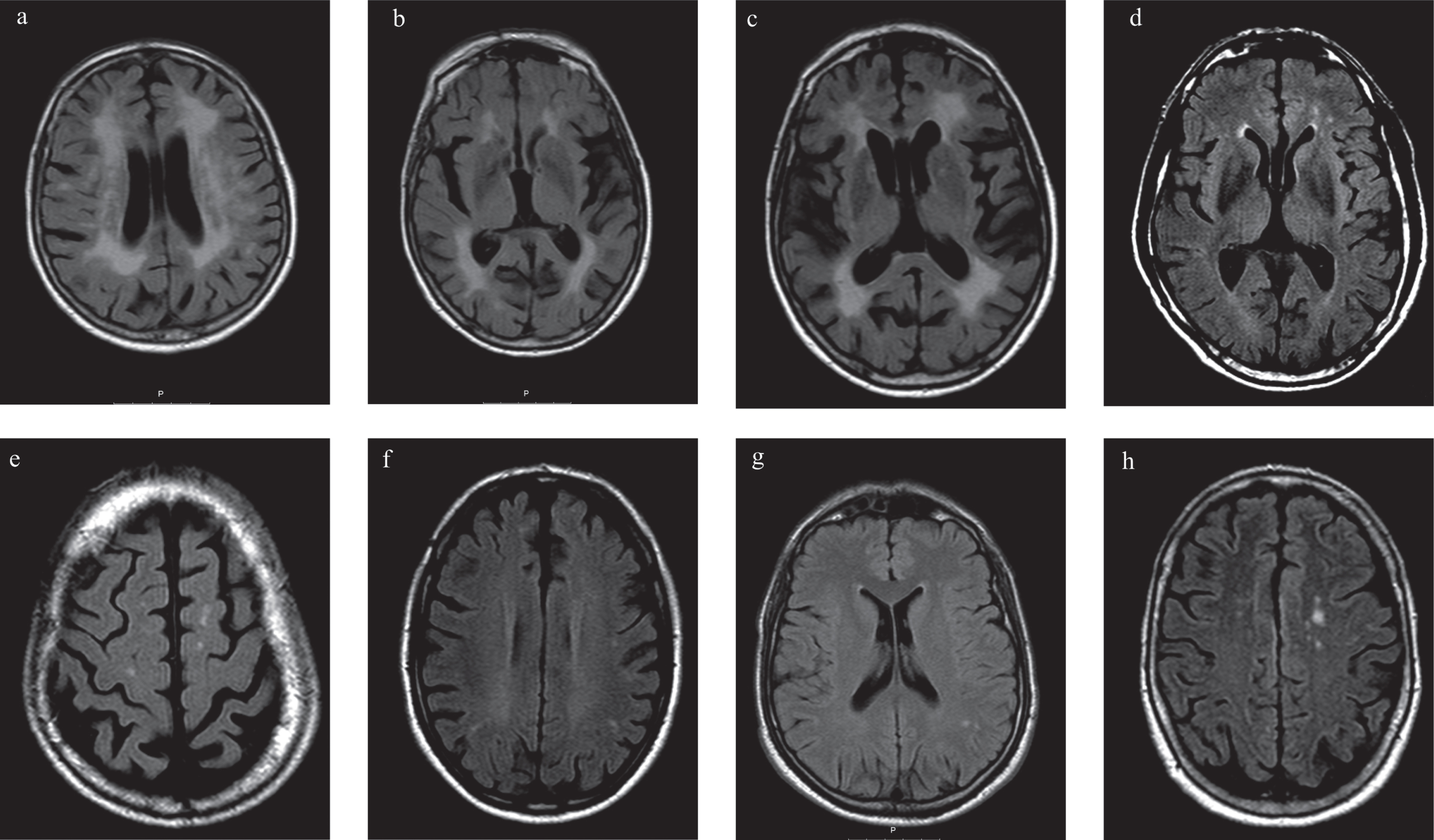
Brain magnetic resonance imaging (MRI) with
FD+PD+ patients belonged to three different pedigrees, with each pedigree having 8 (P1), 9 (P2) and 13 (P3) family members with confirmed FD, totaling 30 FD patients (Fig. 1). From this cohort of 30 FD patients, 24 FD patients were older than 35 years old (5 (P1), 9 (P2) and 10 (P3)) and were included in our final analysis (16 females/8 males). Overall, the FD+PD+ patients had worse clinical severity of FD than their FD+PD- family members (Tables 1 and 2), clearly offsetting the upper IQR of the MSSI total score (all); and neurological (P1, P2); cardiovascular (all); and renal subscores (P1, P2) (Table 2).
Concerning
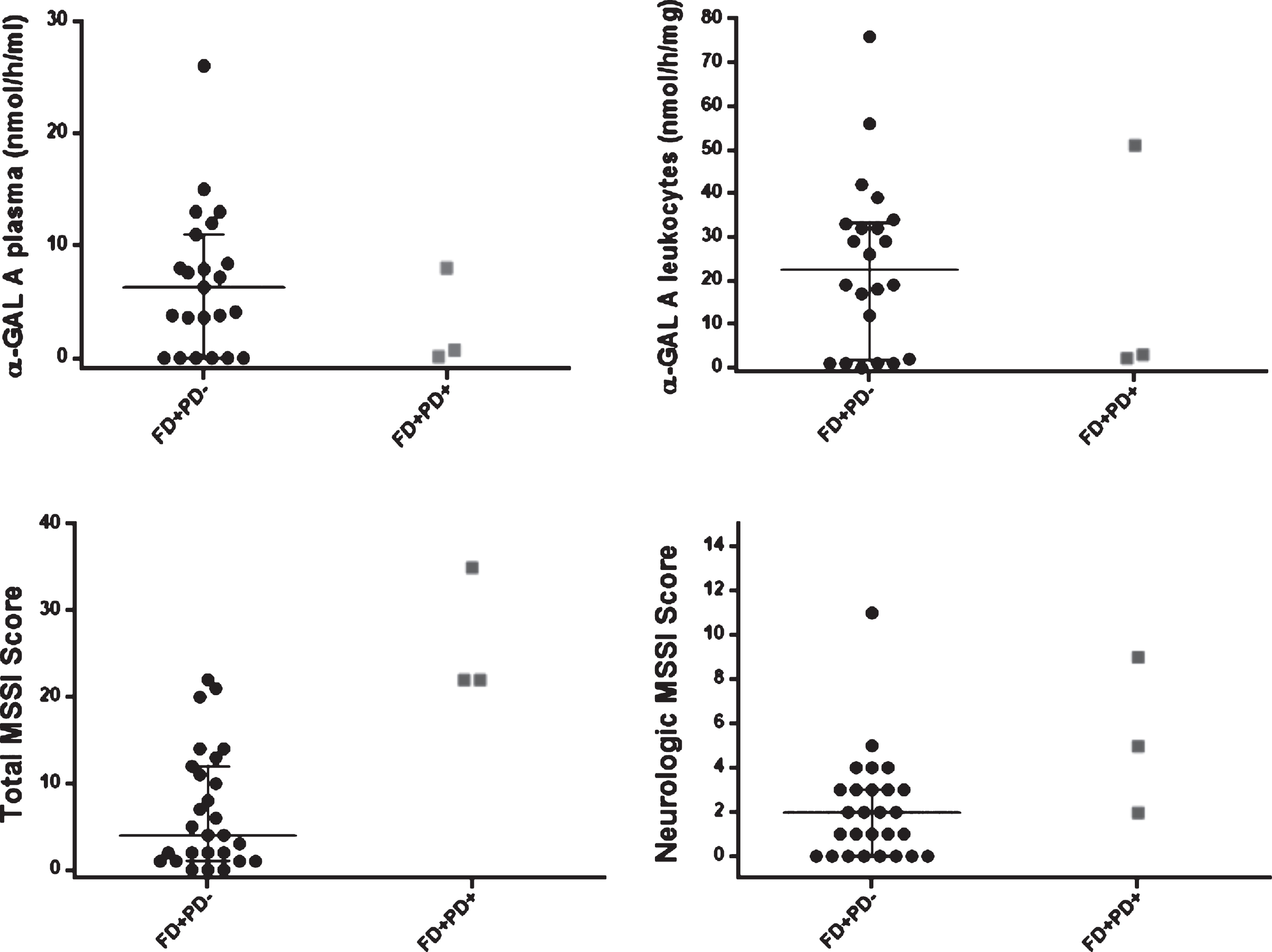
Scatter dot plot, with median and IQ intervals (25–75%) for
Concerning FD biomarkers, P3 male patient had lower value of Plasma Lyso-Gb3 (P3:5.76 versus. Males FD+PD–: 9.2 (median)(ng/mL)), which is most likely explained by the fact of being under enzyme replacement therapy (ERT), agalsidase beta (70 mg iv, every other week) for 24 months, since the age of 63 years old, 1 year previous to PD diagnosis.
Notwithstanding other cardiovascular risk factors (e.g., diabetes, hypertension) that were also frequent (Table 2), brain WML (100% of FD+PD+ patients, 38% of FD+PD–females and 60% of FD+PD–males) and small vessels ischaemic lesions (100% of FD+PD+ patients, 15% of FD+PD- females and 20% of FD+PD- males) were widely present in FD patients, with higher prevalence in FD+PD+ patients.
DISCUSSION
To the best of our knowledge, this is the first study surveying PD in a large cohort of FD patients with the p.F113L mutation. We found a higher prevalence of PD in FD patients (1.3% in the total cohort of adult FD patients and 3.0% in the subgroup of FD patients aged ≥50 years), when compared to the 0.24% adjusted prevalence of PD in community-dwelling Portuguese population, aged ≥50 years [23]. With respect to previous reports, we have found at least two common similarities, which can be highlighted as characteristic of FD with PD: higher burden of cardiac and cerebrovascular lesioning.
Orimo et al. [12] reported a 68-year-old male with FD (Gb3 deposits in cardiac and kidney autopsy, but without genetic investigation) presenting an atypical form of parkinsonism, beginning at the age of 63 years old, with axial signs (gait and postural instability), mild symmetrical rigidity, and pyramidal signs (mild right hemiparesis, generalized hyperreflexia and positive Babinski sign on both sides). Brain MRI showed multiple T2 hyperintensities in the basal ganglia and deep white matter regions, suggesting the possibility of vascular and/or neuronal dysfunction in FD [24]. Buechner et al. [13] reported a 57-year old woman with FD due to the p.R301P
In our cohort, even though the two FD+/PD+ male patients (P2-P3) suffered a mild parkinsonism and LED dose can be further increased, the female patient (P1) presented, in less than 18 months, a moderate-severe parkinsonism with very low response to levodopa. This refractoriness to levodopa can be explained by concomitant cerebrovascular lesions affecting directly nigro-striatal neural structures (P1 patient with basal ganglia and brainstem infarcts), and/or indirectly subcortical fibers (P2-P3 patients).
As we previously reported, the p.F113L
It is still open to discussion how lower
Study strengths and limitations
Our study approach has substantial strengths in comparison to previous literature, as it accounts for a prospective screening for PD in patients with an already established diagnosis of FD, complemented with biochemical and neuroimaging analysis.
Our description of FD patients with PD and respective pedigrees has several limitations. Screening for the major known PD genes in these three patients led to the identification of a variant in the
Future directions
The prodromal phase of PD begins decades before its clinical onset (when more than 60% of nigrostriatal loss is already present), namely some non-motor symptoms (TREND and PRIPS studies) [34]. Lohle et al. [35] found that, while FD patients (60 females and 50 males with
Conclusion
In summary, this study showed a higher prevalence of PD in FD patients than in the general population (1.3% in the total cohort of adult FD patients and 3.0% in the subgroup of FD patients aged ≥50 years), further highlighting the importance of the autophagy-lysosomal pathway in PD. All FD+PD+ patients presented an akinetic-rigid PD with weak response to levodopa as well as worse clinical severity of FD, brain WML and high cerebrovascular burden. Further studies will be needed to understand how much of this PD phenotype is due to deposition of Gb3 versus cerebrovascular lesions in the nigro-striatal network. Although the discussion of Vascular Parkinsonism and overlapping syndromes of Vascular Parkinsonism with PD is controversial, it is plausible, at this moment, to foresee that cerebrovascular burden will be a very important co-variable determining the final phenotype of PD in FD patients. Moreover, although ERT has not been shown to cross the blood-brain barrier [36], prospective follow-up will demonstrate if its cardiovascular and renal benefits have meaningful impact in PD history. Finally, future studies will be needed to corroborate if, upon PD neurodegeneration, patients activate a compensatory mechanism of increased
CONFLICT OF INTEREST
The authors have no conflict of interest to report
Footnotes
ACKNOWLEDGMENTS
The authors are grateful for the clinical support provided by all colleagues working in the Reference Centre on Lysosomal Storage Disorders, and in the Neurology and Cardiology Departments, Hospital da Senhora da Oliveira, EPE, Guimarães.
