Abstract
Background:
Periodic limb movements in sleep (PLMS) are repetitive movements usually of the legs strongly associated with Restless-legs syndrome (RLS), which appear more frequently in males, older age and other sleep disturbances, such as sleep-disordered breathing (SDB). Patients with Parkinson’s disease (PD) suffer from various sleep disturbances including REM sleep behavior disorder, RLS and PLMS. Although a dopaminergic pathophysiology of PLMS is discussed, no systematic data on PLMS side-to-side distribution in PD and its correlation with asymmetry of motor symptoms are available.
Objective:
This study aimed at elucidating PLMS asymmetry in correlation to that of motor symptoms in PD compared to SDB and RLS.
Methods:
Cross-sectional, retrospective analysis of two polysomnography (PSG) recordings per patient scoring PLMS separately for both legs.
Results:
Of 105 patients (44 PD, 44 age- and sex-matched SDB and 17 RLS patients) PLMS measures (number of PLM, PLM-Index, PLM-arousal index) showed significant side-to-side differences in all disease entities in both PSGs (P < 0.001; Wilcoxon rank test). PLM-Index asymmetry (PLM-I difference of >5/h between both sides) was observed less frequently in PD (34% of patients) compared to RLS (77% , P < 0.05) and SDB (59% , P < 0.05; χ2 test). In asymmetric PD patients, predominant side of PLMS was more stable than in SDB and RLS comparing the two PSGs, but we did not detect an agreement between PLMS predominant side with that of motor symptoms in PD patients.
Conclusions:
Only the minority of PD patients shows asymmetric PLMS distribution with relatively high night-to-night stability but no correlation with motor symptom asymmetry.
Keywords
INTRODUCTION
Parkinson’s disease (PD) is one of the most frequent neurodegenerative diseases defined by the key motor symptoms akinesia, rigor, tremor and postural instability [1], which— in most cases— appear asymmetrical at least at the beginning of the clinical course. Sleep disturbances, e.g., REM sleep behavior disorder, sleep disordered breathing, insomnia, circadian rhythm disorders and excessive daytime sleepiness, are frequent in PD, even in de novo patients [2, 3] and affect quality of life adversely [4]. Periodic limb movements in sleep (PLMS) belong to the sleep-related movement disorders [5] and are characterized by repetitive, stereotyped, involuntary movements usually of the lower limbs occurring during sleep [5–7] with high night-to-night variability [8, 9]. PLMS may also appear as an isolated phenomenon [7] without clinical symptoms even in healthy persons [10, 11]. However, the diagnosis of periodic limb movement disorder (PLMD) requires additional symptoms such as sleep disturbances or functional impairment, PLM frequency (PLM index, PLM-I) exceeding 15/h in adults and exclusion of other sleep, medical or neurological disorders made responsible for PLMS [5]. PLMS are closely associated with Restless-legs syndrome (RLS or Willis-Ekbom disease) affecting 40 up to 80% of RLS patients [12–14] constituting a possible susceptibility marker for RLS [15–17]. PLMS may also occur in other sleep disorders such as sleep-disordered breathing (SDB) [18] or REM-sleep behavior disorder [19]. Other factors that seem to influence the occurrence of PLMS in the general population are male sex [20, 21] and age [13, 22]. Whether PLMS and RLS prevalence is increased in PD is still debated controversially [4, 23–25], but RLS prevalence [26–29] and also PLM indices (PLM-I) seem to increase with PD duration [30]. PLMS are considered responsible for impairment of subjective, but not objective sleep quality and quality of life in PD and seem to be associated with disease severity [4]. Although isolated PLMS, PD and RLS were all shown to improve with dopaminergic medication strongly implying a dopaminergic pathology in PLMS generation [31–33], no clear pathophysiological association of these entities has been proven yet [34]. Indeed, the pathophysiology of PLMS is still not fully elucidated, but currently, impaired inhibitory influences on bilateral spinal pattern generators [35, 36] as part of a complex network of spinal cord, brainstem and supratentorial structures [6] modified by the autonomic nervous system [37] and genetic predisposition [20] are supposed to be responsible for PLMS generation [38].
Unilateral PLMS were already described in 1986 by Lugaresi and colleagues [39, 40]. In RLS, where in the majority of patients PLMS are detectable, asymmetry of clinical symptoms has been reported in either idiopathic [14, 41–43] as well as secondary forms [44–46]. However, most of the studies reporting asymmetric RLS symptoms did not include polysomnography (PSG) measurements of PLMS or did not quantify PLMS asymmetry [48, 49]. Only in a few case reports and small case series comprising PSG recordings unilateral or alternating predominance of PLMS in RLS patients [47] and in patients with either peripheral (e.g., spinal cord injury) [50] or cerebral lesions (e.g., stroke) were documented [51–55]. However, even in secondary forms the laterality of one-sided PLMS is not always concordant with its suspected causative cerebral lesion and also imaging data in RLS patients underline the variable involvement of cerebral structures either bilateral or contralateral to PLMS occurrence [51, 57]. In PD unilateral or predominantly one-sided RLS symptoms or leg motor restlessness (LMR)— not fulfilling the diagnostic criteria for restless legs syndrome— seem to occur considerably more prevalent [58–60] than in RLS patients [61], but also in PD the accordance of motor symptom asymmetry with the laterality of RLS/LMR is not consistent comparing different studies [58–61].
Despite these findings, no systematical analysis of PLMS asymmetry are available in general and not even in PD, where asymmetry of the pathology of the nigro-striatal dopaminergic system as well as the corresponding asymmetry of key motor symptoms is a characteristic hallmark [1]. Taken together with the fact that PLMS share some common features with PD such as dopamine sensitivity, this study was aimed at elucidating PLMS asymmetry in correlation to that of motor symptoms in PD patients in comparison to SDB and RLS patients.
PATIENTS AND METHODS
This retrospective, cross-sectional cohort study was conducted at the Technische Universität Dresden, Germany, Department of Neurology and Interdisciplinary Sleep Centre. It was approved by the local institutional review board (Ethics committee of the Technische Universität Dresden; EK7012017).
Eligible medical records of patients aged 45 to 90 years, who underwent a standardized, full-night attended, digital video-PSG (PSG1 [baseline, diagnostic PSG] Alice 5.0 software, Heinen & Loewenstein) according to American Academy of Sleep Medicine recommendations [62] at the Dresden University Sleep Centre and if available a further PSG (PSG2), e.g., with treatment, between 2012 and 2018 were screened for study inclusion. In detail PSG recordings included electroencephalography (EEG), electrooculogramm (EOG), electromyography (EMG) of the submentalis muscle and both tibialis anterior muscles, oronasal airflow, microphone, thoracic and abdominal respiratory effort, position sensor, oxygen saturation and electrocardiogram (ECG) according to AASM standard recommendations [62]. Subjects were excluded if EMG showed high levels of EMG noise or artefacts.
Time difference between the first and the second PSG recording varied widely between one day and a few months due to different indications for further PSGs, e.g., initiation of PAP treatment or control of therapy efficacy. PD patients had to be diagnosed according to UK Brain Bank criteria [1]. Disease controls with sleep-disordered breathing (SDB) and a Respiratory Distress Index (RDI)>15/h classifying SDB as at least moderate [63] and RLS patients fulfilling the diagnostic criteria of the International RLS Study Group were screened for eligibility [64]. Exclusion criteria were atypical parkinsonian features in PD patients, known RLS, RLS symptoms or treatment with dopaminergic agents in SDB patients, and documented RDI >15/h in RLS patients. In both control groups, patients with known history or clinical signs of other medical conditions, which might influence symmetry of PLMS (e.g., cerebral ischemia, spinal cord lesions), and relevant comorbid sleep disorders (e.g., narcolepsy) were excluded.
Demographic and clinical data including lateralization of PD symptoms, medication, Unified PD Rating Scale (UPDRS), Hoehn & Yahr staging and PSG sleep measures (e.g., total sleep time [TST]) and PLM indices (number of PLM, PLM index [PLM-I], PLM arousal index [PLM-AI]) combined for both sides were extracted of the medical records. Levodopa equivalent dose was calculated according to Tomlinson and co-workers [65]. Sleep disturbances, e.g., “urge to move legs or arms”, were conveyed from questionnaires or interviews within the medical records without the possibility to differentiate between LMR and RLS and were thus classified as LMR. Presence of PLMS symptoms, of which the patient or the bed partner was aware, was also extracted of the medical records based on questionnaires.
Scoring of LM was performed manually by a trained board-certified Sleep Medicine specialist (WH; TS). LM were scored according to AASM standard criteria [62], if EMG activity increased ≥8μV above baseline level lasting 0.5 s up to 10 s and decreased for at least 0.5 s ≤2μV above baseline level. PLM were scored if LM groups of at least four with an inter movement interval of 5 s to 90 s appeared. PLMS-Index (PLM-I) is defined as the number of PLM/hour sleep (PLMS/h). LMs were excluded as respiratory related, if LMs occurred overlapping with an interval of±0.5 s at the start and the end of the respiratory events (apnea/hypopnea). PLM were scored for both legs combined according to AASM standard scoring criteria and afterwards manually rescored for both legs separately. PLM occurring bilaterally were scored separately on either side regardless of co-occurrence on the other side. Number of PLM and PLM indices were calculated for both sides separately and side-to-side differences were normalized to the respective values of the more affected side. If not mentioned otherwise, PLM were classified as being asymmetric, if PLM-I difference was >5/h between the more and the less affected side.
Results are presented as mean±SDs, median (IQR) or as numbers (%) as appropriate. Demographic characteristics were compared by Kruskal-Wallis- or Pearson’s χ2, side differences of PLMS were compared using Wilcoxon, Kruskal-Wallis, Pearson’s χ2 or Fisher’s exact test as appropriate. Percentage agreement and Cohen’s κ were used to determine the agreement of PLM-I asymmetry between PSG1 and PSG2 and the agreement of PLM-I asymmetry with PD motor asymmetry and handedness. Pearson’s correlation test and multivariate linear regression modelling was used for correlations of PLMS measures and candidate covariates potentially influencing PLMS severity such as levodopa equivalent dose [65], age, gender, PD symptom duration and disease severity (UPDRS III motor score). κ or Pearson’s correlation coefficient |r|<0.3 was considered a weak, κ/|r| = 0.3–0.59 a moderate, κ/|r|≥0.6 a strong agreement/correlation. All reported P values are two-sided. Due to the explorative character of the study, α adjusting of P values was carried out, when comparing the three patient groups using the Bonferroni method, but not for multiple testing. Statistical analyses were performed using SPSS version 23.0 or higher (SPSS, Chicago, IL, USA).
RESULTS
A total cohort of 105 patients comprising of 44 patients with PD (mean age 68.9±8 years; 43% female; mean UPDRS total score 32.1±10.8), 44 sex- and age-matched patients with SDB (69.6±7 years, P = 0.631 vs. PD (post-hoc Mann-Whitney test); 43% female: P = 0.266 (χ2 test) vs. PD/RLS) and 17 patients with RLS (63±10 years, P = 0.027 vs. PD; 65% female) met eligibility criteria and were included into the analysis. Due to multiple exclusion criteria, especially comorbid SDB and spinal cord lesions, RLS patients were not age and sex-matched (demographic details in Table 1). The majority of PD patients (86%) and 53% of RLS patients received dopaminergic treatment (P = 0.014; Fisher’s exact test). LED differed significantly between PD and RLS (P < 0.001, Table 1). Between the first and second PSG, positive airway pressure treatment was initiated in 11 of PD patients (27% of patients with second PSG, n = 41) and nearly all SDB patients with second PSG (n = 37/38). Dopaminergic medication was adapted in some of the PD and RLS patients (Supplementary Table 1).
Baseline demographic and clinical characteristics of study cohortsa
Data are mean±SD, median (IQR) or number (%) as appropriate. P values are from §Kruskal-Wallis test, †Mann-Whitney-U-test, ‡Chi-square test or ‡‡Fisher’s exact test. **represents P < 0.01 when compared to SDB; # represents P < 0.05; ###P < 0.001 when compared to PD (all from post-hoc Mann-Whitney test with Bonferroni adjustment). ADL, Activities of daily living; IQR, Interquartile range; PAP, Positive airway pressure therapy; PD, Parkinson’s disease; RLS, Restless-Legs-Syndrome; SDB, Sleep-disordered breathing; UPDRS, Unified Parkinson’s disease rating scale (part I: evaluation of mentation, behavior and mood; part II: activities of daily life; part III: motor function; part IV: complications). aLevodopa equivalent dose was calculated according to Tomlinson and co-workers [66].
No differences in sleep quality parameters (e.g., sleep efficiency) were detected comparing patient groups (Table 2, Supplementary Table 1). Total PLMS measures (standard examinations with both sides combined) were significantly lower in PD (median (IQR) PLM-I of 5.2/h (0.9–29.8); mean±SD 17.7/h±24.7) compared to SDB (median (IQR) PLM-I of 39.0/h (18.5–62.7); mean±SD: 44.3±38.6) and RLS (median (IQR) 36.5/h (20.8–47.0); mean±SD 35.2±20.3; both P < 0.00144; post-hoc Mann-Whitney test). Initiation or adaption of standard-of-care treatment for SDB (PAP treatment) and RLS (dopaminergic compounds) resulted in improvement of night-time respiratory parameters in SDB and the PAP-subcohort of PD patients (median (IQR) RDI at PSG1 of 37.9/h (18.5–44.9) and at PSG2 of 4.7/h (2.6–11.7), P < 0.001; median (IQR) AHI at PSG1 of 31.9/h (13.0–35.3) and at PSG2 of 4.4/h (1.2–11.0), P < 0.001; Wilcoxon rank test for both) and PLM-I and PLM-AI in RLS in PSG2 compared to PSG1 (Supplementary Table 1).
Baseline polysomnographic sleep characteristics of study cohortsa
Data are mean±SD, median (IQR) or number (%) as appropriate. P values are from §Kruskal-Wallis test. *** represents P < 0.001 when compared to SDB; #represents P < 0.05; ##P < 0.01; ###P < 0.001 when compared to PD (all from post-hoc Mann-Whitney test with Bonferroni adjustment). AHI, Apnea-Hypopnea index; IQR, Interquartile range; LM, leg movements; PD, Parkinson’s disease; PLM, Periodic limb movements; RDI, respiratory distress index (sleep-associated events including respiratory effort related arousals); SDB, Sleep-disordered breathing; TIB, Time in bed; TST, Total sleep time. aThis table reports baseline PSG data (from PSG1 recordings). For comparative data of the two PSG recordings, refer to Supplementary Table 1.
When analyzing PLMS separately for both sides in PSG1, numbers of PLM, PLM-I and PLM-AI differed significantly between less affected and more affected side regardless of disease entity (Wilcoxon test, P≤0.002) with median side-to-side differences ranging from 30 to 62% (Table 3). No differences of side-to-side distribution patterns of PLMS measures between the patient groups were observed. Similar results were obtained in PSG2 (Table 3).
Side differences of PLM in sleep
§P values from Wilcoxon rank tests comparing more affected and less affected side. Similar results were obtained for PSG2. ‡Side-to-side differences normalized to more affected side. P values from Wilcoxon rank tests comparing PSG1 and PSG2 data. $Kruskal-Wallis tests comparing disease groups revealed P values of 0.125 for PLM, 0.091 for PLM index and 0.276 for PLM-arousal index for PSG1 data, and P values of 0.404 for PLM, 0.385 for PLM index and 0.616 for PLM-arousal index for PSG2 data. IQR, Interquartile range; PD, Parkinson’s disease; PLM, Periodic limb movements; PSG, polysomnography; RLS, Restless-Legs-Syndrome; SDB, Sleep-disordered breathing.
Asymmetric PLMS distribution (ΔPLM index >5/h between sides) was observed more frequently in RLS (77%) and SDB (59%) compared to PD (34%; P < 0.05; post-hoc Fisher exact tests) in PSG1 (Fig. 1, Table 4). In PSG2, asymmetry of PLMS distribution occurred more frequently in SDB compared to RLS and PD but these differences did not reach statistical significance (Fig. 1, Table 4). No significant differences regarding right versus left predominance of asymmetric PLM-I were detected neither for PSG1 (PD: P = 0.092; SDB: P = 0.208; RLS: P = 0.250) nor for PSG2 (PD: P = 1.000; SDB: P = 0.725; RLS: P = 1.000; Fisher’s exact test for all comparisons) in any of the patient groups. In contrast to the relatively high asymmetry rates in both PSGs, we found no relevant agreement between PSG1 and PSG2 with respect to PLM-I symmetry/asymmetry or its dominant side in RLS (Cohen’s κ: 0.084 and –0.429, respectively). In SDB, we detected no agreement of PLM-I symmetry/asymmetry between the two PSGs (Cohen’s κ: 0.009), but a moderate agreement rate between the PLM-I dominant side in asymmetric patients (Cohen’s κ: 0.417). However, in PD patients, either agreement of PLM-I symmetry/asymmetry between the two PSGs (Cohen’s κ: 0.629) as well as agreement of PLM dominant side in asymmetric patients were moderate (Cohen’s κ: 0.476; Fig. 1, Table 4). Similar results were also obtained with a threshold of PLM-I difference >15/h (Table 4).
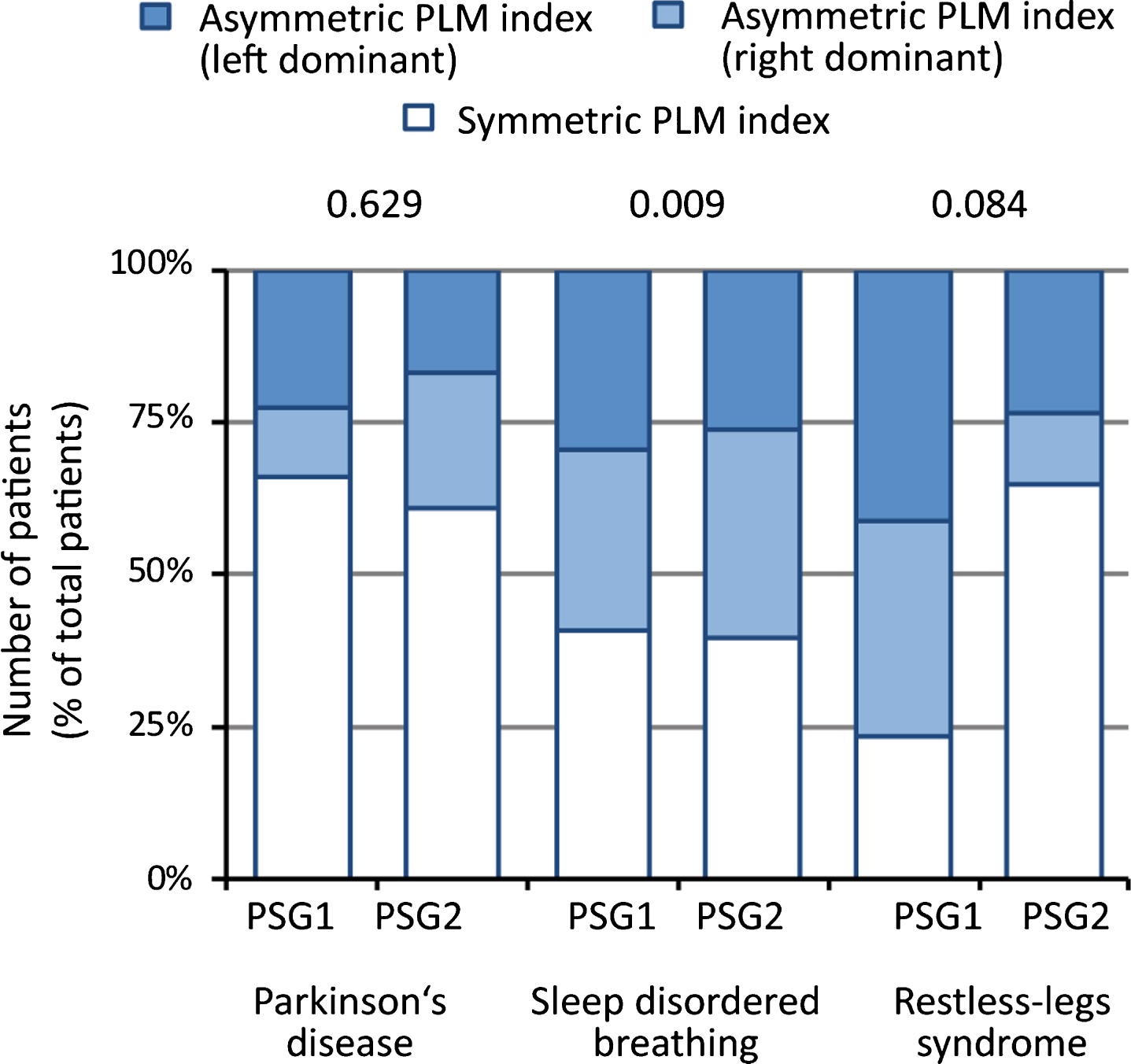
Side-to-side symmetry of Periodic limb movements (PLM) index in Parkinson’s disease (PD), sleep-disordered breathing (SDB) and Restless-legs-Syndrome (RLS). PLM indexes are considered asymmetric with differences between both sides of >5/h. Displayed are data from two polysomnographies (PSG). Values in the diagram are Cohen’s κ values displaying the agreement rate of PLM index symmetry (asymmetric / symmetric) between PSG1 and PSG2 (see Table 4 for details).
Asymmetry of PLM index
Data are presented as numbers of patients (%). §P values from Pearson’s χ2 test or Fisher exact test as appropriate comparing all three disease groups. *represents P < 0.05 when compared to values from SDB, $ represents P < 0.05 when compared to values from RLS by post-hocχ2 test or Fisher exact test as appropriate with Bonferroni adjustment. Comparing SDB and RLS revealed no statistically significant differences. Note that between PSG1 and PSG2 positive-airway pressure therapy was initiated in 11 (25%) PD and in nearly all SDB patients. In PD patients, positive airway pressure treatment did not lead to effects on PLM index asymmetry (P = 0.069 when comparing patients with and without treatment using Fisher exact test). PD, Parkinson’s disease; PLM, Periodic limb movements; RLS, Restless-Legs-Syndrome; PSG, polysomnography; SDB, Sleep disordered breathing.
Comparison of predominant side of PLMS and that of motor symptoms in PD in both PSGs revealed no relevant agreement (PSG1: agreement rate: 8/15 (53%), Cohen’s κ: 0.000; PSG2: agreement rate: 9/16 (56%), Cohen’s κ: 0.125). Results were similar for PLM-I difference of >15/h (data not shown). This is in agreement with numerical data on PLM, PLM-I and PLM-AI when comparing PD dominant with non-dominant side (Table 5). Moreover, PLM-I dominant side and handedness in PD patients were not associated (data not shown).
PLMS values with respect to PD predominant side
§P values from Wilcoxon test comparing ipsilateral and contralateral side. PD, Parkinson’s disease; PLM, Periodic limb movements; PSG, polysomnography.
When analyzing the subcohort of patients with high PLM indices (PLM-Index>5/h), we confirmed the results of the overall cohort in all disease entities (Supplementary Tables 2–4) by showing significant side-to-side differences of PLMS distribution. Comparison of predominant side of asymmetric PLMS with that of motor symptoms in this PD subcohort also revealed no agreement (PSG1: Agreement rate of 7/14 (50%), Cohen’s κ: 0.043; PSG2: Agreement rate of 6/12 (50%), Cohen’s κ: 0.000). The only difference we detected, when analyzing patients with higher PLM-index >5/h was a significant reduction of PLM measures in the RLS cohort in PSG2 compared to PSG1 (Supplementary Table 2), indicating a better treatment response to the initiation of dopaminergic treatment.
When selecting subgroups of PD patients, e.g., patients in early disease stages (H&Y stage 1, n = 10), patients with PLMS symptoms (n = 10) or patients with LMR (n = 13), results were similar to the overall PD cohort showing side-to-side differences of PLMS measures in PSG 1. Accordingly, analyses of PLMS values with respect to PD predominant side were similar to results in the overall cohort (data not shown). Also in subgroups of PD patients without PLMS symptoms or LMR, findings were similar to the results of the overall PD cohort (data not shown).
The adaption of dopaminergic therapy with an increase of LED in PD and RLS patients did not lead to changes in PLMS asymmetry parameters (Tables 3 and 4). Consistently, we did not observe differences between subcohorts of PD and RLS with and those without changes in dopaminergic therapy (data not shown). Indeed, we could not detect any relevant correlations between PLMS asymmetry measures and relevant covariates such as LED [65], age, sex and UPDRSIII, but only a moderate association with PD symptom duration not surviving multivariate regression analysis (Supplementary Table 5). In contrast, in RLS there was a clear association of PLM and PLM-I, but not of their side-to-side differences, with LED (Supplementary Table 6). Initiation of PAP treatment did not lead to significant differences in PLM and PLM-I asymmetry in the PD group (P = 0.069 comparing PAP-treated with PAP-untreated PD patients; Fisher exact test). In SDB, we observed a mild increase of PLM and PLM-I asymmetry measures between PSG1 and PSG2 with initiation of PAP, particularly the extent of the side-to-side-difference between the more and the less affected side (Table 3), but no change of the number of patients with PLM-I asymmetry (Fig. 1, Tables 3 and 4).
DISCUSSION
We report a retrospective cross-sectional cohort study using two polysomnographic recordings to assess the (a)symmetry of PLMS in PD patients in relation to the asymmetry of motor symptoms and handedness compared to patients suffering from SDB and RLS as disease controls. This is, to our knowledge, the first systematic quantitative analysis on PLMS side-to-side differences in patients without neurological conditions predisposing for PLMS asymmetry such as stroke. We detected side-to-side asymmetry of PLMS (number of PLM and PLM-I) in all disease conditions with a higher proportion of patients with asymmetric PLMS in SDB and RLS compared to PD. We could neither show a relevant association of the predominant side of PLMS with that of PD motor symptoms nor with handedness in PD. By comparing different subgroups of PD patients, e.g., low H&Y stages and patients with and without PLMS symptoms or LMR, we confirmed our results implying a reasonable robustness of our findings.
Since we, in contrast to most previous studies, analyzed two PSGs in almost all patients, we were able to investigate night-to-night variability of PLMS asymmetry and largely exclude coincidental asymmetry. We found relatively stable night-to-night symmetry/asymmetry distributions of PLMS in PD patients including reasonable stability of the predominant side of PLMS in asymmetric patients. This was in contrast to findings in RLS and SDB with no relevant agreement of the symmetry/asymmetry distribution of PLMS between the two PSG recordings showing a high night-to-night variability of PLMS. In line with our results, high night-to-night variability of dominant PLMS side and asymmetric PLMS in RLS patients have been reported previously [47]. Our results are also in agreement with electrophysiological findings showing asymmetric muscle recruitment for the generation of PLMS [66] as well as PSG recorded high night-to-night variability of PLMS in various sleep disorders [8, 9].
In contrast to our findings of relatively stable PLM asymmetry in PD, we were not able to detect an association of asymmetry of PLMS with that of motor symptoms. As our study is the first one to systematically analyze PLMS asymmetry in PD by PSG, there are no other studies arguing for or against our findings. However, PSG results of asymmetric PLMS in our study are in line with clinical descriptions of asymmetric leg motor restlessness symptoms in PD without accordance to the PD dominant side [58, 59]. On the contrary, two cross-sectional studies revealed consistency of LMR with PD dominant side [60, 61]. However, our results do not support the hypothesis that the pathophysiology of PLMS in PD is mediated through its dopaminergic pathology in the nigro-striatal system as implicated by functional imaging [67].
Unilateral or asymmetric PLMS were already described in patients with cerebral or spinal cord lesions [50–53], but these patients were excluded in our groups by detailed clinical/neurological investigation and medical history data. Although lesion effects in our control groups cannot be ruled out completely due to the retrospective analysis approach, the likelihood for a relevant influence of other medical conditions mediating PLMS asymmetry is low. In line with our results, asymmetric PLM in RLS patients have been reported in a small case series [47], but no repetitive PSGs with detailed analysis of side-to-side-variances were performed with their major importance to evaluate asymmetry over several nights. In a case-control study unilateral or predominantly one-sided LMR were reported more frequently by PD patients (60–94%) than by iRLS patients (17–22%) [46, 61], but these studies did not include objective measures of PLMS. In contrast to these findings, in our cohort asymmetry of PLMS as measured objectively by PSG was detectable in RLS /SDB to a greater extent than in PD. However, discrepancies between objective and subjective sleep quality in patients are well known and do not speak against our findings.
Since we report a retrospective study, we were not able to fully control for treatment regimes in our patient cohorts. We thus performed ancillary statistical analyses using demographic and clinical factors as confounders. Treatment of SDB with PAP led to improved night-time respiratory parameters in the SDB and the PD group, but did not alter PLM or PLM asymmetry measures in the PD group. In SDB, we detected a mild increase of PLM and PLM-I asymmetry, most likely as an expression of the high night-to-night variability. Notwithstanding, influences of PAP therapy on the extent of LM are already documented. However, these effects are complex leading to suppression of respiratory related LM, but also unmasking of previously hidden genuine PLM in severe OSA by the PAP treatment [68]. Though we excluded respiratory related LM according to AASM scoring rules the definition of respiratory-related LM is currently under debate [69, 70]. Therefore, partial inclusion of respiratory related LMs might lead to a bias in the results as respiratory related PLMS seem to completely disappear after initiation of ventilation therapy, but might not be influenced by dopaminergic therapy [71]. However, the reasons for our findings remain enigmatic yet and further investigations are needed to clarify the influence of PAP on SDB-associated PLMS asymmetry.
Increasing the dose of dopaminergic medication between the two PSGs resulted in reduced PLM and PLM-I in RLS, but not in PD patients. This is consistent with RLS literature showing a clear dopaminergic effect on PLMS in RLS [31, 73], whereas in PD results are more complex. Högl and co-workers reported a positive effect of the dopamine agonist cabergoline on PLMS in rather early PD [33], while in the cross-sectional study by Puligheddu and colleagues dopaminergic therapy led to complete suppression of PLMS in early but not in more advanced PD [23]. However, PLMS distribution over night was altered by dopaminergic therapy, but in line with our results, no change of PLM or PLM-I was detectable [23]. This is also congruent to a four-year longitudinal study showing that RLS in PD only partially responded to dopaminergic treatment [29]. These non-responsive PLMS might be a different not “genuine” PLMS sub-type with other time structure features than those seen in RLS [23, 74]. These findings indicate that PLMS responsiveness to therapy might be attributed to diverging pathophysiology and the possible involvement of other neurotransmitters in RLS generation in PD [74]. However, changes of dopaminergic therapy did not alter PLMS symmetry patterns in PD and SDB.
Our study has several limitations: First, the retrospective character of the analysis with its known restrictions including limited control of confounding factors such as different/changing therapies. Our detailed statistical analyses however largely exclude major effects of demographic or therapeutic parameters on PLMS asymmetry patterns. Second, the RLS control group was not sex- and age matched and relatively small compared to the other disease entities with a female predominance. This was due to the high proportion of RLS patients with comorbidities as exclusion criteria in our Sleep Centre largely serving as an institution for differential diagnosis questions. This might lead to a bias in our conclusions as male patients seem to exhibit higher PLMS numbers. However, we did not detect an influence of sex on PLMS and PLMS symmetry parameters and therefore adding more male patients is not likely to change our findings. Additionally, we could not show differences in asymmetry parameters between cohorts of patients with low and high PLM-I underlining the robustness and reliability of our data. Only in RLS patients we detected that patients with higher PLM-I experienced better treatment responses to dopaminergic therapy, which was revealed by excluding one patient with lower PLM index (<5/h), thus underlining a more favorable effect of dopaminergic treatment in patients with higher PLM measures.
Our analysis is furthermore limited by the relatively small sample size of the overall cohort, which also has an impact on the statistical reliability of analysis of subcohorts of patients, e.g., with and without LMR. However, analyses of various subcohorts of PD patients showed consistent results with the overall cohort, particularly regarding PLM asymmetry and the lack of agreement between PLM predominant and PD motor symptoms dominant side. Although these findings indicate a reasonable validity of our results, future studies are warranted to re-confirm our findings in larger cohorts including de novo PD patients and patients in the early disease stages.
PLM indices in our PD cohort of patients mostly on dopaminergic treatment are lower compared to other studies [4, 23]. However, in line with our findings two other studies showed similar PLM numbers in PD patients on dopaminergic treatment or rather after the initiation of dopaminergic therapy and also confirmed our results of lower PLM numbers in PD compared to iRLS [28, 33]. Since we did not detect relevant differences between the PD and the RLS cohort or patients with high vs. low PLMS numbers with respect to PLMS asymmetry measures as our major readout, these aspects should not relevantly limit our analyses or interpretations. In addition, no agreed cut-off values to determine asymmetry of PLMS exist yet, and we thus set these cut-offs as >5/h and >15/h between the more and the less affected side without the possibility to validate their clinical significance.
Together, only the minority of PD patients shows an asymmetric PLMS distribution pattern with relatively high night-to-night stability but no correlation with motor symptom asymmetry. This is in contrast to SDB and RLS with higher frequencies of PLMS asymmetry but lower night-to-night consistency of asymmetry patterns. These findings underline that motor symptoms of PD and PLMS, at least in PD, do not share the same pathophysiological background. Although further studies with shorter periods of PLMS analysis intervals are needed to elucidate the side-to-side changes of PLMS asymmetry, our study is one step further on the way to understand non-motor symptoms in PD and to further elucidate the relation of PLMS and PD.
CONFLICT OF INTEREST
Disclosures related to the manuscript
The authors declare that they have no financial or non-financial disclosures related to the manuscript.
Full disclosures (last 12 months)
Wiebke Hermann, Theresa Flemming, Moritz Brandt and Simona Langner report no disclosures.
Heinz Reichmann was acting on Advisory Boards, received grants and honoraria and received research grants from Abbott Abbvie, Bayer Health Care, Bial, Boehringer/Ingelheim, Brittania, Cephalon, Desitin, GSK, Lundbeck, Medtronic, Merck-Serono, Novartis, Orion, Pfizer, TEVA, TEVA & Lundbeck GmbH, UCB Pharma, Valeant and Zambon outside the submitted work.
Alexander Storch has received funding from the Deutsche Forschungsgemeinschaft (German Re-search Association) and the Helmholtz-Association. He has received honoraria for presentations/advisory boards/consultations from AbbVie, Bayer, Bial, Desitin, Grünenthal, Teva, and UCB. He has received royalties from Kohlhammer Verlag and Elsevier Press. He serves as an editorial board member of Stem Cells, Stem Cells International, Open Biotechnology Journals, and The Journal of Biological Chemistry.
