Abstract
Background:
Trouble concentrating during orthostasis is one symptom of orthostatic hypotension (OH), but it is often ignored in clinical practice because OH incorporates many nonspecific symptoms. Blunted cognition during orthostasis may affect various aspects of the clinical status of patients with Parkinson’s disease (PD), but its impact on drug-naïve, early PD patients is not clear.
Objective:
This study investigated the association between trouble concentrating during orthostasis and orthostatic blood pressure changes and the influence of trouble concentrating during orthostasis on other non-motor symptoms and health-related quality of life in de novo PD.
Methods:
A total of 124 patients with early PD were enrolled and received a structured clinical interview, physical examination, and validated questionnaires to evaluate motor and non-motor symptoms and quality of life. Disturbed concentration was evaluated with question 5 of the Orthostatic Hypotension Questionnaire system assessment (part I). Supine blood pressure and blood pressure change during tilt table testing were recorded, and any association was analyzed.
Results:
Among the study population, 76 (61.3%) patients had trouble concentrating. Patients with blunted concentration had a steeper decrease in blood pressure during orthostasis. The score for trouble concentrating was positively associated with non-motor symptoms and quality of life scores.
Conclusions:
Trouble concentrating when standing was a frequent finding in early, drug-naïve PD patients. It was positively associated with non-motor burden, mood and quality of life in patients with PD. OH was correlated with the incidence of trouble concentrating. Such findings complicate treatment but provide valuable information for managing early PD.
INTRODUCTION
Neurogenic orthostatic hypotension (OH) occurs in about 30–40% [1] of patients with Parkinson’s disease (PD), and its prevalence increases as the disease progresses [2]. However not all blood pressure (BP) fluctuations are symptomatic [1, 3]. Episodes of profound OH could be asymptomatic (33%) and atypical presentations (24%) hamper proper management [4]. In addition, many symptoms of OH are ignored by both physicians and patients.
Trouble concentrating in association with autonomic failure is a frequent complaint though it is often overlooked because it is nonspecific [5, 6]. Patients neglect it because they do not realize that it is related to their disease [7, 8]. Trouble concentrating is also interwoven with various other symptoms of orthostatic challenge, such as mood, dysexecutive function, and global cognitive dysfunction [9–11]. This concurrence prevents patients from recognizing any trouble concentrating. In addition, these symptoms might be related to disrupted blood pressure homeostasis but may not be identified without proper testing.
The hypothesis tested in this study was that trouble concentrating during orthostasis is a result of orthostatic dysregulation, and it affects various non-motor symptoms and quality of life in early PD. We asked if trouble concentrating during orthostasis was related to blood pressure changes in head-up tilt testing. In addition, we investigated the influence of trouble concentrating during orthostasis on other non-motor symptoms and health-related quality of life in patients with de novo PD.
METHODS
Patients
This study was approved by the Institutional Review Board of our institution, and all subjects provided written informed consent to participate. All experiments were performed in accordance with relevant guidelines and regulations.
One hundred twenty-four patients with newly diagnosed, early PD who visited the movement disorder clinic at a tertiary university hospital between April 2015 and October 2017 were enrolled. Patients were diagnosed with Parkinson’s disease according to the criteria of the UK PD Society Brain Bank [12]. The clinical diagnosis was substantiated by positron emission tomography using 18F-N-(3-fluoropropyl)-2beta-carbon ethoxy-3beta-(4-iodophenyl) nortropane imaging studies. All patients had presynaptic dopamine transporter uptake deficiency in the striatum.
Clinical information, including age, sex, disease duration, history of hypertension, diabetes mellitus, and smoking status, was obtained. All enrolled patients were PD naïve and did not take any dopaminergic agents and antidepressants such as serotonin reuptake inhibitors and tricyclic antidepressants. All hypertensive PD patients underwent the study protocol maintaining their antihypertensive medication. The motor symptoms of PD were measured using the Unified Parkinson’s Disease Rating Scale (UPDRS) Part III and modified Hoehn and Yahr (H&Y) stage scores. Non-motor symptoms, depression, and health related quality of life were assessed with the Non-Motor Symptoms Scale (NMSS) [13], Montgomery-Asberg Depression Rating Scale (MADRS) [14], and Parkinson’s Disease Quality of Life-39 (PDQ-39) [15], respectively.
Patients were excluded from the study if they had any of the following: (1) any signs or symptoms of atypical PD and secondary parkinsonism, (2) history of diabetic neuropathy or other etiology indicating autonomic neuropathy, (3) history of cardiac abnormalities that affect blood pressure, (4) history of peripheral arterial disease (PAD) and (5) medications such as tricyclic antidepressants that influence autonomic functions.
Each patient was assessed for occurrence of orthostatic intolerance and its severity with a validated clinimetric questionnaire for OH [16]. OHQ Part I, question 5, which states “Have you ever experience trouble concentrating during orthostasis? Please tick the number on the scale that best rates how severe trouble concentrating from low blood pressure has been on the average over the past week. If you do not experience the symptom, circle zero” was used to assess trouble concentrating during orthostasis. Patients were defined to have “trouble concentrating” if they graded the severity more than one.
Clinical characteristics
PD, Parkinson’s disease; H&Y, modified Hoehn and Yahr stage; UPDRS, Unified Parkinson’s Disease Rating Scale; SBP, systolic blood pressure; DBP, diastolic blood pressure; OHQ, Orthostatic hypotension questionnaire. Analyses were performed by independent t test for continuous variables and χ2 test for categorical variables. Non-normally distributed continuous variables were analyzed by Mann-Whitney U test.
Head up tilt test
Patients were instructed not to drink alcohol or coffee the day before the tilt test. Smokers were restricted from smoking for at least one hour before the examination. All patients were at a full resting state before the exam began. Continuous electrocardiograph leads and non-invasive BP monitoring equipment were attached to the patients (YM6000, Mediana Tech, Redmond, WA, USA). A supine position was maintained for 20 minutes before tilting to 60 degrees (ENRAF NONIUS, Rotterdam, The Netherlands). While in the supine position, BP and heart rate were measured every 5 minutes for 20 minutes. At 60 degrees, BP and heart rate were recorded at 0, 3, 5, 10, 15, 20 minutes. The first supine BP (at 0 minutes) was abandoned and the measurements at 5, 10, 15, 20 minutes were used for analysis. The arithmetic mean BP was calculated for systolic and diastolic pressure. The supine mean baseline and lowest tilt BP were recorded. For statistical analysis, the lowest systolic and diastolic pressure within 5 minutes was selected. The degree of BP fall for systolic (ΔSBP) and diastolic pressure (ΔDBP) was calculated by subtracting the standing BP from the supine mean BP. Patients were classified as having OH if orthostatic ΔSBP≥20 mmHg, ΔDBP≥10 mmHg or both [17].
Statistical analysis
All statistical analyses were performed with the Statistical Package for the Social Sciences, version 24.0 for the Mac (SPSS, Inc.; Chicago, IL, USA). Demographic characteristics were evaluated by descriptive statistics. Difference in questions 1 and 5 of OHQ Part I was assessed by the McNemar test. The relationships between trouble concentrating during orthostasis (OHQ part I-5) and other symptoms on OHQ were calculated by Spearman’s correlation test and the internal consistency among OHQ Part I questions were assessed by Cronbach alpha. Between-group characteristics were compared by independent t-test, the Mann-Whitney U test, and the Chi-square test for continuous and categorical variables. The Jonckheere-Terpstra test was performed to assess any dose-dependent association between increasing concentration problems and orthostatic BP drop. Any relationship between trouble concentrating and other validated clinimetric scales was also evaluated with a partial correlation analysis. A p-value <0.05 was considered significant.
RESULTS
Clinical characteristics of enrolled patients are summarized in Table 1. The mean age was 70.1±8.9 years old, and 66 (53.2%) were male. Their median disease duration was 12.0 months (interquartile range, 18.0 months). The mean UPDRS part III score was 16.2±9.3 and median modified Hoehn and Yahr stage was 1.5 (interquartile range, 1.0). Thirty-four (27.4%) patients had OH defined as orthostatic ΔSBP≥20 mmHg, ΔDBP≥10 mmHg, or both.
The median value of trouble concentrating score (OHQ part I-5) was 2.0 (interquartile range, 5.0). Seventy-six (61.3%) patients with PD complained of trouble concentrating during orthostasis. Orthostatic dizziness, assessed by OHQ Part I-1, was reported in 50 (40.3%) patients. Concentration problems were significantly more frequent than dizziness (McNemar test, p < 0.001). Trouble concentrating during orthostasis (OHQ part I-5) was correlated with other symptoms on OHQ (Spearman’s rho = 0.628, p < 0.001) and a high agreement between OHQ Part I questions was also found (Cronbach alpha = 0.785).
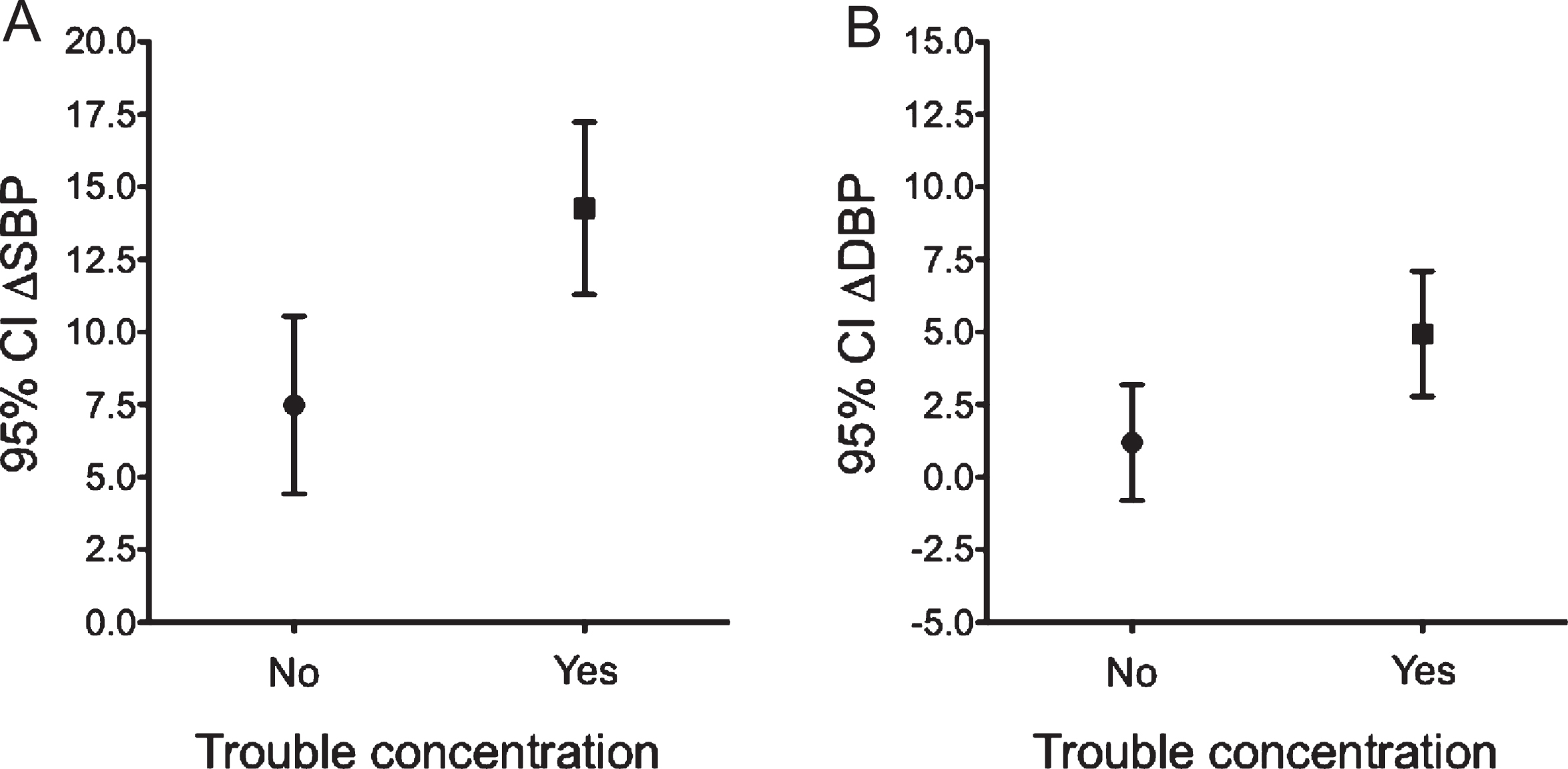
Mean and 95% confidence interval for orthostatic blood pressure changes in Parkinson’s disease with/without trouble concentrating. (A) ΔSBP, (B) ΔDBP.
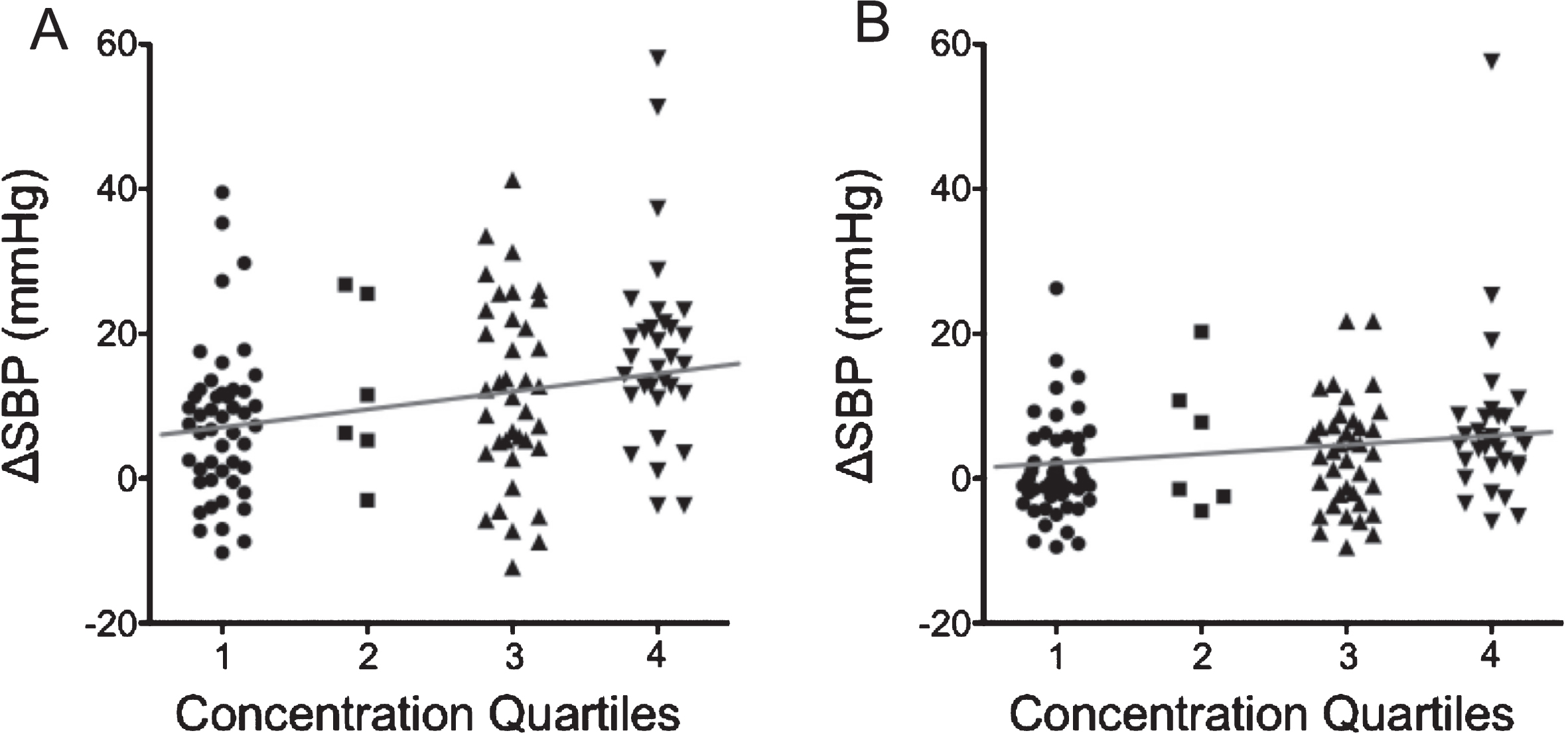
Dose-dependent association between trouble concentrating score and orthostatic blood pressure change. Trouble concentrating score was divided into quartiles, and the magnitude of trouble concentrating was compared to blood pressure drop by using the Jonckheere-Terpstra test. (A) ΔSBP, p < 0.001; (B) ΔDBP, p = 0.006.
Baseline demographics were compared for patients without and with trouble concentrating. Age, sex, body mass index, disease duration, history of hypertension, diabetes mellitus, and smoking status did not differ between groups (Table 1). All hypertensive patients were on anti-hypertensive medications, and these medications did not influence on the OHQ part I score (no hypertension vs. hypertension; 13.5±9.6 vs. 15.4±11.5, p = 0.298). Those with trouble concentrating had significantly higher H&Y stage and UPDRS part III scores (no trouble concentrating group vs. trouble concentrating group: H&Y stage score, 1.5 vs 2.0, p = 0.024; UPDRS part III score, 13.6±9.0 vs. 17.9±9.2, p = 0.011, respectively). They also had higher mean supine systolic BP (no trouble concentrating group vs. trouble concentrating group: 120.3±15.6 mmHg vs. 126.6±15.2 mmHg, p = 0.028). Symptomatic patients had a steeper BP drop when standing (no trouble concentrating group vs. trouble concentrating group: median orthostatic ΔSBP = 7.4 mmHg vs. 13.4 mmHg, p = 0.001, median orthostatic ΔDBP = –0.4 mmHg vs. 4.8 mmHg, p = 0.010; Fig. 1), and they were more often diagnosed with OH (no trouble concentrating group vs. trouble concentrating group: 14.6% vs. 35.5%, p = 0.011).
Orthostatic BP change was strongly associated with trouble concentrating magnitude. When trouble concentrating score was divided into quartiles, the magnitude of trouble concentrating was positively related to BP drop (Fig. 2).
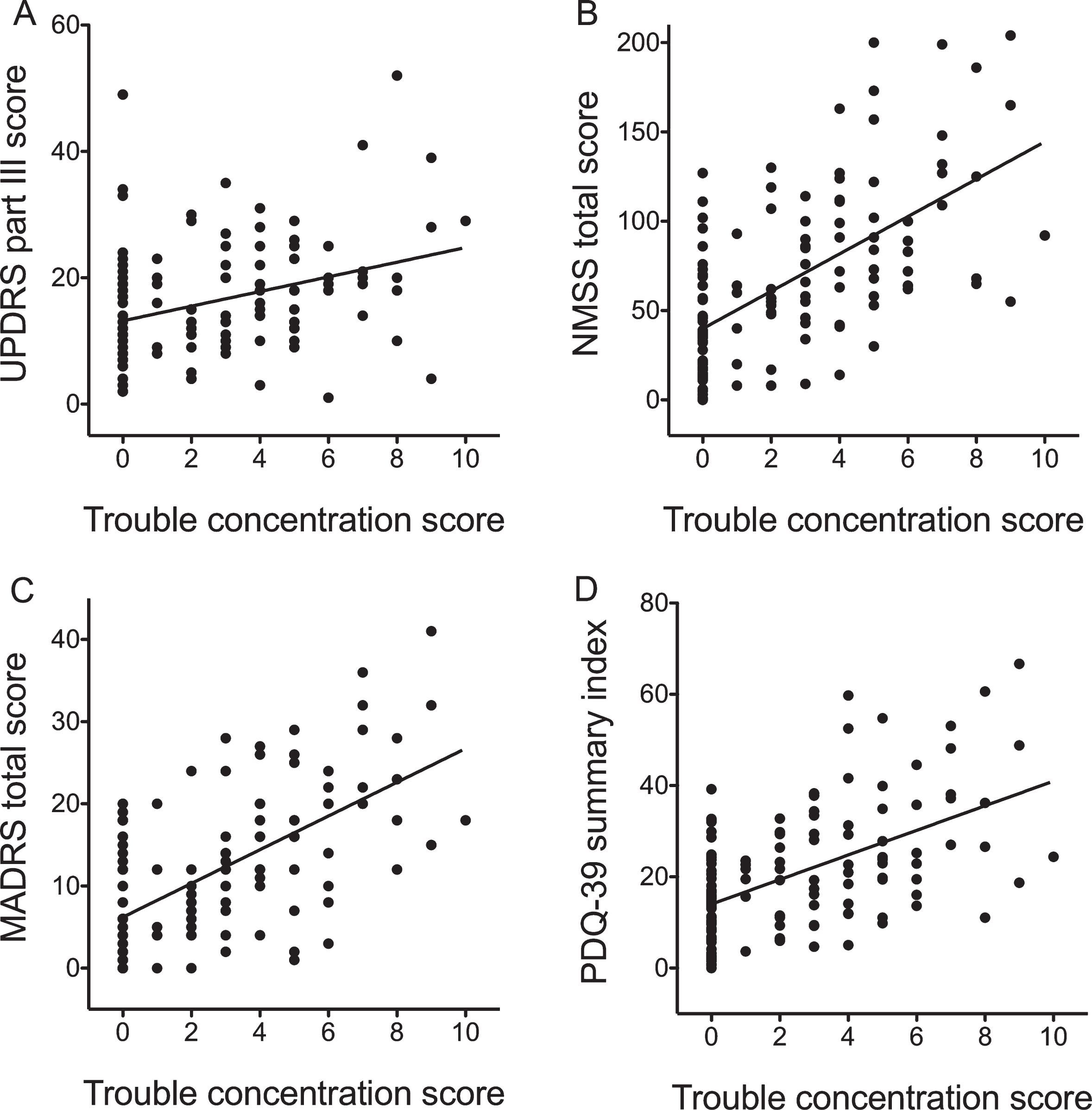
Correlation analysis between trouble concentrating score and motor and non-motor symptoms, mood, and health-related quality of life. Trouble concentrating was positively associated with the Unified Parkinson’s Disease Rating Scale (UPDRS) Part III score (A), Non-Motor Symptoms Scale (NMSS) (B), Montgomery-Asberg Depression Rating Scale (MADRS) (C), and Parkinson’s Disease Quality of Life-39 (PDQ-39) summary index (D).
Correlation between Trouble concentration (OHQ Part I–5) and NMSS, MADRS and PDQ-39
NMSS, Non-motor Symptoms Scale; MADRS; Montgomery-Asberg depression rating scale, PDQ-39, Parkinson’s Disease Quality of Life-39; Analysis was performed by partial correlation test adjusted for age and disease duration.
The relationships between trouble concentrating and motor and non-motor symptoms, mood, and health-related quality of life were assessed. Trouble concentrating was positively associated with UPDRS part III, NMSS, MADRS, and PDQ-39 summary index (Fig. 3 and Table 2; r = 0.293, p = 0.001; r = 0.592, p < 0.001; r = 0.598, p < 0.001; r = 0.504, p < 0.001, respectively) after controlling for age and disease duration.
DISCUSSION
This cross-sectional study found that trouble concentrating upon standing was common in early PD and was even more prevalent than orthostatic-specific symptoms. Blunted concentration was related to the magnitude of the blood pressure drop. Concentration was clearly associated with motor and non-motor burdens, mood, and overall quality of life.
In this study, the prevalence of OH was lower than in previous research [1, 3]. The difference could be attributed to the methodology of determining the tilt test results. Rather than selecting the highest supine BP, the values were averaged and compared with the lowest upright BP. The mean BP will be lower than the highest BP, thus, decreasing the difference between supine and upright pressure and leading to more negative results. This method might underestimate the overall OH prevalence, but OH is largely asymptomatic [3], and our approach could reduce the gap between the operational and actual symptomatic incidences. Despite such a trade-off, we believe our calculation can detect symptomatic OH that corresponds to the OHQ.
One peculiar finding in our research was that trouble concentrating (OHQ Part I-5) was significantly more common than specific symptoms such as dizziness and lightheadedness (OHQ Part I-1). The actual prevalence of acute impaired concentration during orthostasis has seldom been studied. One of the largest studies reported the incidence of symptomatic OH to be 18% [18], but the definition of “symptomatic OH” did not include trouble concentrating. Trouble concentrating is nonspecific and could be attributed to other causes, despite being evaluated by the validated OHQ. In this study, a trend analysis was performed and we found a positive association between the severity of trouble concentrating and blood pressure drop. Trouble concentrating was caused by orthostatic challenge and is thought to be a result of cerebral hypoperfusion.
Difficulty concentrating during orthostatic challenge has been studied previously [6, 19]; however, its associations with other aspects of PD such as motor and non-motor symptoms, depression, and quality of life were seldom explored. This study reveals that not only the UPDRS part III, NMSS, and MADRS scores, and PDQ-39 summary index but also the subscales are highly associated with trouble concentrating during orthostasis, and they are positively related to each other. This result implies that meticulous blood pressure management can influence on many clinical manifestations of PD. No consensus has been reached on how to treat blood pressure variability in PD patients; however, non-pharmacological managements such as elevated head-up position, isometric exercise before standing, avoidance of alcohol and a pharmacological treatment (domperidone, fludrocortisone, midodrine or droxidopa in severe cases) could be beneficial for blood pressure optimization [20].
Our study has some limitations. First, the number of subjects in each subgroup was relatively small for the purpose of drawing firm conclusions, although this study used drug-naïve patients with de novo PD. A long-term follow-up study is needed to characterize the prognosis of the PD with OH group. Second, the magnitude of trouble concentrating was relatively mild in these subjects of the study. However, the median value was increased to 4.0 for those who complained of trouble concentrating during orthostasis. Third, we did not completely exclude other causes of trouble concentrating such as ophthalmologic, otologic, dysexecutive function by PD itself or other medical problems. Fourth, we have not completely ruled out the effects of various medications and their interactions on the autonomic manifestations. In particular, the use of antihypertensive medication can contribute to trouble concentrating and hypotension during orthostasis, although this study did not show such a result. Fifth, we did not check a real-time report of cognitive blunting during head up tilt test to see if there is a real-time correlation. Future experiments to monitor each orthostatic symptom undergoing tilt-table testing would substantiate our hypothesis. Finally, delayed OH [16, 21] was not included. Further extensive research including delayed OH would have stronger clinical implications.
In summary, trouble concentrating is a common symptom of OH, and it is widely linked with other features of PD. Optimizing blood pressure may assist in alleviating motor and non-motor symptoms, and improving health-related quality of life in PD.
CONFLICT OF INTEREST
The authors have no conflicts of interest or financial support to report.
Footnotes
ACKNOWLEDGMENTS
This research was supported by Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Science, ICT and Future Planning (NRF-2017R1D1A1B06028086).
