Abstract
Background:
In Parkinson’s disease (PD), postural instability-gait disorder (PIGD) has been associated with more rapid cognitive decline, dementia, and greater non-motor symptom (NMS) burden.
Objective:
To assess the importance of balance-gait disorder, relative to and in combination with other clinical measures, as a risk factor for cognitive impairment, dementia and NMS burden in PD.
Methods:
164 PD subjects were evaluated in a retrospective cross-sectional study using the MDS-UPDRS scale, MMSE and Clinical Dementia Rating Scale. Using univariate comparisons followed by multiple stepwise regression, we identified factors most closely associated with NMS burden and concurrent dementia. Nominal logistic regression and linear discriminant analysis was used to compute receiver operating characteristic curves and to measure sensitivity and specificity of predictors of dementia.
Results:
Dementia was more frequent in those with the PIGD phenotype (χ2 = 11.49, p = 0.003). The MDS-UPDRS balance-gait measure, excluding freezing of gait, was the single best predictor not only of concurrent cognitive impairment and dementia (F = 37.16, p < 0.001) but also of NMS burden, predicting 29% of the MDS-UPDRS total non-motor experiences of daily living score (F = 67.14, p < 0.0001). This measure, together with combined severity of hallucinations/psychosis, daytime sleepiness and urinary problems, a closely correlated symptom cluster (r = 0.63 p < 0.0001), accurately classified 84% of patients with dementia.
Conclusions:
In PD, balance-gait impairment is closely associated with dementia and NMS burden, particularly the linked symptoms of cognitive impairment, psychosis, daytime sleepiness and urinary dysfunction. This phenotype characterizes patients likely to require closer surveillance and more comprehensive care. Confirmation of these findings in prospective longitudinal studies might help refine predictive algorithms designed to identify PD patients more likely to progress from mild cognitive impairment to dementia.
Keywords
INTRODUCTION
Parkinson’s disease (PD) is characterized pathologically by progressive and widespread misfolding and subsequent aggregation of alpha-synuclein in specific nuclei of the central and peripheral nervous systems [1]. This pathology underlies not only the motor symptoms but a panoply of non-motor symptoms (NMS) of PD [2, 3]. Almost universally present, some NMS antedate the appearance of motor symptoms and many increase with disease duration and severity [4–6]. Ultimately, some NMS may become the most dominant clinical feature, critically impacting quality of life, functional status and prognosis [6–11]. Cognitive impairment and dementia in particular are associated with adverse outcomes, including gait impairment and falls [12–15], greater disability and caregiver burden [16], higher economic costs [17], and increased mortality [18, 19].
While some neuropsychological predictors for the progression from mild cognitive impairment (MCI) to dementia in PD have been recognized [20–23], reliable predictors of PD dementia, including simple clinical predictors, continue to be sought. Compared with tremor-predominant patients, those with the postural instability-gait disorder (PIGD) phenotype of PD [24] exhibit not only a more rapid rate of cognitive decline, but also a greater burden of NMS with earlier development of psychopathology, and a poorer quality of life [25–32]. Although the prognostic significance of PIGD in PD, both as an initial clinical presentation and as a later evolving phenotype [31] is recognized, the importance of PIGD symptom severity, relative to other clinical factors, as a risk factor for both cognitive impairment and dementia in PD has differed from study to study [3, 34] and the relationship of such dysfunction to the expression of individual NMS has only recently started to receive attention [28, 35]. Longitudinal studies evaluating risk factors for cognitive impairment in PD have predominantly used the Hoehn and Yahr stage or axial impairment, the latter variously assessed, as indicators of postural instability-gait disorder [3], while a more recent study focused instead on freezing and falls [36]. Some information is available regarding the relationship between postural instability in PD and freezing of gait (FOG) [37–39], but the unique contribution, if any, of FOG as an element of PIGD has not previously been explored when studying the prognostic significance of the PIGD phenotype.
The primary aims of this study were to explore, in a cross-sectional, retrospective study of PD patients: (1a) the relative importance of PIGD symptom severity, relative to other clinical factors, as a risk factor for concurrent cognitive impairment, dementia and overall NMS burden, and (1b) its accuracy, alone and in combination with other factors, as a predictor of concurrent dementia. Secondly, in pursuing the above analysis, we sought to: (2a) parse the PIGD phenotype, with a view to determining whether or not FOG contributes to the predictive value of balance-gait impairment with respect to risk for dementia, and (2b) compare measures of postural instability and gait disorder, using the MDS-UPDRS, Hoehn and Yahr stage and occurrence of falls, to determine the balance/gait measure most sensitive as a risk factor for concurrent dementia. Finally, we sought to determine the unique profile of NMS associated with balance-gait disorder in PD.
PATIENTS AND METHODS
164 patients with PD, as defined by the UK Brain Bank criteria [40], evaluated at the Buffalo VA Medical Center and at a second tertiary Movement Disorders Center in Buffalo, NY, were included in the study (Table 1). All patients were diagnosed and assessed by a movement disorders neurologist trained and certified in use of the MDS-UPDRS scale (DGL) [41]. For case inclusion, a minimum data set was required that included: (1) At least one complete MDS-UPDRS rating, performed in an “on” state; (2) information regarding frequency of falls; and (3) MMSE and Clinical Dementia Rating (CDR) [42] evaluation, obtained within 3 months of the MDS-UPDRS rating. Patients with other parkinsonian syndromes or comorbid disorders unrelated to PD which might affect gait or balance or otherwise independently influence MDS-UPDRs subscales were excluded. The latter included: major psychiatric illness considered unrelated to PD, such as schizophrenia or bipolar disorder, prior traumatic brain injury, history of stroke as determined by clinical or radiologic evaluation, prominent deep white matter microvascular disease, hydrocephalus, significant endocrine or metabolic disorder, clinically relevant visual, vestibular or cerebellar impairment, myelopathy, lower extremity radiculopathy or neuropathy, and orthopedic or rheumatological condition affecting gait. Patients with a prior history of DBS or other brain surgery were also excluded, as were those with a diagnosis of multifactorial dementia or dementia with Lewy bodies, according to consensus criteria [43], and those with dementia sufficient to affect validity for Part 1 MDS-UPDRS ratings.
Subjects
MDS-UPDRS, International Parkinson and Movement Disorder Society Unified Parkinson’s Disease Rating Scale; PIGD, Postural Instability Gait Disorder; EDL, Experiences of Daily Living.
A retrospective, cross-sectional study was performed in an effort to clarify clinical risk factors for overall non-motor symptom (NMS) burden and concurrent dementia in the PD cohort.
Age of onset of PD was defined as the age at the first motor symptom of PD. Duration of PD was defined as the period between the first motor symptom of PD and the MDS-UPDRS rating. The motor phenotype at the time of the assessment was defined by the tremor/PIGD (postural instability-gait disorder) ratio, as follows: (1) tremor-predominant (ratio≥1.15); (2) PIGD (ratio≤0.90); (3) indeterminate (ratio 0.90–1.15) [44].
Total score on part 1 of the MDS-UPDRS scale, the non-motor experiences of daily living (nM-EDL), was used to assess non-motor symptom burden. This score has been demonstrated to have a strong relationship with a composite score of validated scales for the non-motor aspects of PD [45]. Part 1A is administered by the rater, with all pertinent information from the patient and caregivers, and focuses on six complex behaviors: cognitive impairment, hallucinations and psychosis, depressed mood, anxious mood, apathy, and features of dopamine dysregulation syndrome. Part 1B is a component of the self-administered Patient Questionnaire that covers seven NMS: sleep problems, daytime sleepiness, pain and other sensations, urinary problems, constipation problems, light headedness on standing, and fatigue. Subjects are asked to assess their average or usual function over the past week, without concern regarding separation of Parkinson’s disease from other conditions. The patient was generally the primary data source but when appropriate, for example for some patients with dementia, the ratings were provided either by the caregiver or by the patient and caregiver in equal proportion.
Dementia was defined by the DSM-5 criteria for major neurocognitive disorder [46], and was diagnosed following review of medical records, interview of patient and knowledgeable informant (when present), and cognitive assessments by a neurologist, supplemented in 72% of cases by data from a clinical neuropsychological examination. Level 1 criteria for the diagnosis of Parkinson’s disease dementia (PDD) as proposed by the Movement Disorder Society Task Force [47] were required. This includes PD development prior to the onset of dementia, MMSE score below 26, cognitive deficits severe enough to impact daily living (caregiver interview or Pill Questionnaire), and impairment in more than one cognitive domain. The latter included impairment in at least two of the following tests: serial seven subtraction; clock drawing or lexical fluency; MMSE pentagons; and 3-word recall. Also required was exclusion of other primary explanations for cognitive impairment (e.g. delirium, stroke, major depression, metabolic or endocrinologic abnormalities, head trauma, or adverse effects of medication) or other PD-associated comorbid conditions (e.g. severe depression) that, in the opinion of the clinician, might significantly influence cognitive testing [47, 48].
For diagnosis of mild cognitive impairment (MCI), we used a modification of the Movement Disorder Society (MDS) task force guidelines for PD-MCI [48] which was consistent with the DMS-V criteria for mild neurocognitive disorder [46, 49] and recognized established findings of discriminant validity of the MMSE as a screening and diagnostic instrument for MCI in PD [50] as well as the lack of meaningful differences in performance between the MMSE and the MoCA as a screening or diagnostic tool for PD-MCI [51, 52]. Thus, all subjects met the MDS task force exclusion and inclusion criteria for PD-MCI, including gradual decline in cognitive ability reported by either the patient or informant, or observed by the clinician; cognitive deficits on either formal neuropsychological testing or a scale of global cognitive abilities (see modification below); and cognitive deficits not sufficient to interfere significantly with functional independence. Nineteen (29%) subjects met the task force specific Level II guidelines for PD-MCI using a comprehensive neuropsychological assessment. The remainder underwent an abbreviated assessment, consistent with Level I guidelines but using the MMSE as the global scale, and applying the previously defined optimal screening cut-off score of 29/30 for the MMSE for PD-MCI [50, 52]. For all subjects, the diagnosis of MCI was supported by the Clinical Dementia Rating Scale (CDR score = 0.5) [42].
Clinical predictors of total nM-EDL score and presence of dementia were sought, including patient sex, age, age at PD symptom onset, disease duration, LD equivalent dose [53], the modified Hoehn and Yahr stage, and the following motor subscores from the MDS-UPDRS scale: bradykinesia (sum of scores for items 3.4a –3.8b and 3.14); tremor (sum of scores for items 2.9 and 3.15a –3.18); rigidity (sum of scores for items 3.3a –3.3e); PIGD score (sum of scores for items 2.12, 2.13, 3.10, 3.11 and 3.12) [44]; PIGD minus freezing, or “Bal/Gait” score (the sum of scores for items 2.12, 3.10 and 3.12, i.e. a clinical rating concerning problems with walking and balance over the past week and observation of gait and postural stability, excluding freezing of gait from the historical and observational ratings); the presence of motor complications, total motor complications score (items 4.1–4.6, including both dyskinesias and motor fluctuations), and total motor fluctuations score (items 4.3–4.6).
Ethical approval for the study was obtained from the local Institutional Review Board, with waiver of informed patient consent considering the retrospective nature of the study.
Statistical analysis
In the initial assessment of predictors of total nM-EDL score, correlational analysis was used for continuous, normally distributed predictor variables, analysis of variance or covariance (ANCOVA) for ordinal predictors and Student’s t tests for comparison of means of dichotomous predictors. Normality of distribution was assessed using the Shapiro-Wilk W test. We examined the scatterplot of residuals to ensure they fulfilled all linear regression assumptions. Multiple stepwise regression was used to identify the relative importance of PIGD severity relative to other independent clinical predictors of total nM-EDL score. All variables with p < 0.25 in the univariate models were included in a forward stepwise procedure with p-removal = 0.10 [54]. For highly correlated or collinear variables, that with the lower p value was entered as the independent variable. We performed a sensitivity analysis using a backward selection procedure to verify the consistency of the selection of predictors. Statistical significance was considered achieved at p < 0.05. The profile of individual NMS significantly associated with balance-gait disorder was then defined, with a Bonferroni correction for multiple comparisons.
For subjects with normal cognition, MCI and dementia, comparisons of demographic and clinical variables were made using ANOVA or ANCOVA for continuous, normally distributed data, with 2-sample t tests for comparison of subjects with and without dementia, Kruskall-Wallis or Mann-Whitney U tests for non-parametric data, and the chi-square test for categorical variables. Variables associated with dementia at P value < 0.10 on bivariate analysis were then included in multivariate analyses using nominal logistic regression models. For the regression analyses, a single variable was entered at a time for variables demonstrating multicollinearity (i.e. correlation >0.6). Variables with p-values for effect likelihood ratio tests <0.05 in the logistic regressions were retained. A stepwise approach was used, comparing the receiver operating characteristic (ROC) curve for the best predictor with that including the next best indicator. For each regression, nonsignificance of the Hosmer and Lemeshow Test was confirmed, to ensure goodness-of-fit of the model. In an effort to maximize clinical relevance, both optimal and reduced-factor models were constructed on the basis of the most significant and clinically meaningful predictive factors. The aim of this analysis was to understand which clinical factors other than PIGD symptom severity might be useful in identifying individuals more likely to be suffering from dementia. Three different combinations of variables were considered in the nominal logistic regressions on dementia: (1) age and disease duration only; (2) severity of balance-gait disorder, with and without other motor and demographic variables; (2) all clinical variables, including NMS severity. Odds ratios (ORs) and 90% confidence intervals (CIs) for each variable in the models were assessed. Linear discriminant analysis was used to compute the ROC curves and to measure sensitivity and specificity of the baseline predictors, as well as other measures of their diagnostic accuracy. The optimal cutoff values for each predictor of concurrent dementia were defined by Youden’s index [55]. JMP 7 software (SAS Institute Inc., Cary, NC) was used for the statistical analyses.
RESULTS
A total of 218 charts were reviewed and 164 were analyzed. Of the 54 subjects excluded, 46 were based on the following exclusion criteria: prior cortical infarction (n = 3); subcortical microvascular disease and/or lacunar infarction considered contributory to impairments in balance, gait or cognition (n = 9); prior traumatic brain injury (n = 2); hydrocephalus (n = 2); schizo-affective disorder (n = 1); bipolar disorder or severe major depression (n = 4); metabolic encephalopathy (n = 2); vitamin B12 deficiency (n = 2); prior alcoholism (n = 1); unrelated central nervous system, spinal cord, peripheral root/nerve, orthopedic or rheumatological condition considered contributory to balance or gait dysfunction (n = 12); prior DBS surgery (n = 2); revised diagnosis of multiple systems atrophy (n = 1); diagnosis of Lewy body or multifactorial dementia (n = 4); and concurrent aripiprazole therapy (n = 2). An additional eight patients were excluded based on missing elements of the clinical ratings or demographic data. For the 54 subjects excluded, demographic data, including patient age and disease duration, total motor MDS-UPDRS score, and the MDS-UPDRS Pt 1 (total nM-EDL) score did not differ significantly from that of the patients included in the analysis (data not shown).
Demographics, including relationship between cognitive status and gender
Demographic data of study subjects are shown in Table 1. The age of males and females was comparable, both for the group as a whole (72.0±8.9 versus 72.2±11.8 respectively) and for the subset with dementia (78.0±5.7 versus 79.5±10.0 respectively). Males comprised 60.6% (40/66) of the subjects with normal cognition, 53% (17/32) of those with dementia, and 83% (55/66) of those with MCI (χ2 = 12.67, df = 2, p = 0.0018).
Relationship of cognitive status with age and disease duration
Grouped by cognitive status, patients differed significantly in age and disease duration (p < 0.0001) (Table 1). Dementia was present in 29.8% (28/94) subjects≥age 70 years, compared with 5.7% (4/70) of subjects younger than age 70 (χ2 = 16.72, p < 0.0001). Compared with non-demented subjects, those with dementia were more likely to have a disease duration≥10 years (71.0% versus 34.1%, χ2 = 14.04, p = 0.0002). Age of PD onset was not significantly associated with dementia (Table 1).
Association of cognitive status with measures of balance and gait, including falls
Dementia was more frequent in subjects with the PIGD subtype (27/100, 27%) than in those with the tremor-predominant (3/52, 5.8%) or indeterminate subtypes (2/12, 16.7%) (χ2 = 11.49, df = 2, p = 0.003). In the month prior to their clinic evaluation, 66% (21/32) patients with dementia suffered at least one fall, compared to 26% (34/132) subjects without dementia (p < 0.0001). Cognitive subgroups also differed with respect to the Modified Hoehn and Yahr stage (χ2 = 64.30, df = 10, p < 0.0001) and the MDS-UPDRS PIGD rating. Thus, for subjects with normal cognition, MCI and dementia, ratings on the MDS-UPDRS PIGD measure were 3.8±3.5, 6.2±4.5 and 10.7±4.4 respectively (F = 30.43, p < 0.0001). Comparable subgroup ratings for the Bal/Gait (PIGD minus freezing score) measure were 3.1±2.7, 4.8±3.1 and 8.5±2.7 respectively (F = 37.16, p < 0.0001) (Fig. 1). Comparing the five measures of balance-gait impairment assessed, the MDS-UPDRS-Bal/Gait Score had the closest association with dementia, at its optimal cut-off score (≥6) (Table 2). In the nominal logistic regression, the effect of the Bal/Gait score on cognitive status remained significant when adjusting for age and PD duration (effect likelihood χ2 = 15.02, p = 0.0005; χ2 for age = 8.19, p = 0.017; χ2 for PD duration = 6.28, p = 0.04). Paralleling the findings for the three cognitive subgroups, the Bal/Gait score increased incrementally for each of the four subgroups defined by the MDS-UPDRS nM-EDL Cognitive Impairment score, ranging from a mean Bal/Gait score of 3.1 (±2.6) for subjects with cognition rated normal to a score of 7.6 (±3.5) for those judged to have at least moderate cognitive impairment (F = 17.60, p < 0.0001) (Fig. 1).
The Association of Dementia with Measures of Balance-Gait Dysfunction
OR, odds ratio; RR, relative risk; CI, confidence interval. MDS-UPDRS Bal/Gait Sc = MDS-UPDRS PIGD score – freezing score. Optimal cutoff scores were used for the MDS-UPDRS measures.
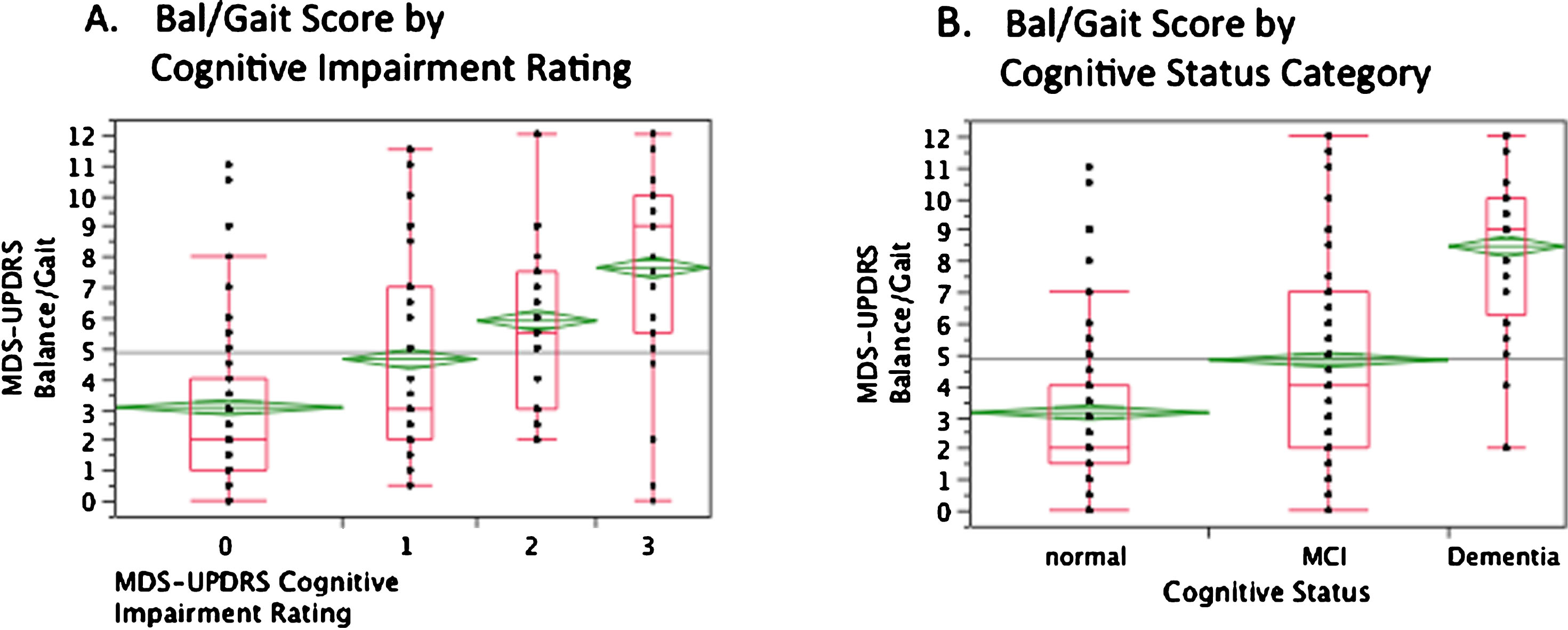
Relationship between the MDS-UPDRS Bal/Gait Score and (A) The MDS-UPDR nM-EDL Cognitive Impairment Rating and (B) Categorical Cognitive Status. A) Quantiles and means with upper and lower 95% confidence intervals for the MDS-UPDRS Bal/Gait score for subjects divided by degree of cognitive impairment, as assessed by the MDS-UPDRS nM-EDL scale. Categories 0–3 indicate subjects with no cognitive impairment (n = 63), slight cognitive impairment (n = 37), mild cognitive impairment (n = 33), and moderate cognitive impairment (n = 30), respectively. The category 3 figure also includes data from one patient with severe cognitive impairment. B) Quantiles and means with upper and lower 95% confidence intervals for the MDS-UPDRS Bal/Gait score for subjects with normal cognition (n = 66), MCI (n = 66), and dementia (n = 32).
The MDS-UPDRS total motor exam (Part III) score was also higher in demented subjects (Table 1) and these patients were receiving somewhat higher average LD equivalent doses compared to non-demented subjects (844.4±373.9 mg versus 702±492.5 mg, p = 0.038).
Relationship of dementia to non-motor symptoms
Relative to non-demented subjects, those with dementia had higher total scores on the MDS-UPDRS nM-EDL rating scale (12.1±4.3 versus 6.9±4.1, p < 0.0001) a difference which persisted after subtraction of the cognitive subscore on this scale (9.2±4.2 versus 6.1±3.7, p = 0.0002). Demented subjects were more likely to endorse the following non-motor experiences of daily living: hallucinations and psychosis (34% versus 8%, χ2 = 12.39, p = 0.0004); daytime sleepiness (81% versus 49%, χ2 = 11.53, p = 0.0007); urinary problems (78% versus 46%, χ2 = 13.63, p = 0.0002) and constipation problems (81% versus 55%, χ2 = 7.86, p = 0.005). The sum of the ratings for the three most distinguishing of these NMS, hallucinations/psychosis, daytime sleepiness and urinary problems, was then considered as a defining non-motor symptom cluster. This composite score, nM-EDL-3, was significantly higher in demented relative to non-demented subjects (3.7±2.0 versus 1.7±1.7, p < 0.0001).
Predictors of dementia and classification accuracy
The first nominal logistic regression on dementia included age and disease duration only. Patient age was a better unique predictor of dementia (χ2 = 20.29, p < 0.0001) than disease duration (χ2 = 12.63, p = 0.0004). Combining these two predictors resulted in a model which was significant (χ2 = 34.8, p < 0.0001), increasing the ROC area under the curve (AUC) to 0.826 compared with 0.741 for age alone. The unit ORs for age and PD duration in this model were 1.13 (1.07–1.21) (χ2 = 22.43, p < 0.0001) and 1.15 (1.07–1.25) (χ2 = 14.91, p = 0.0001) respectively. Using the optimal cut-off scores for age (≥70) and PD duration (≥6 years), 60.5% of 81 subjects having both or neither of these predictors were correctly classified, with 100% sensitivity but only 45% specificity (See Table 3).
Clinical Predictors of Concurrent Dementia
Accuracy, % of subjects correctly classified; PPV, positive predictive value; NPV, negative predictive value; +ve LR, positive likelihood ratio; – ve LR, negative likelihood ratio; RR, relative risk; CI, confidence interval; Bal/Gait Sc, MDS-UPDRS Balance-Gait score (PIGD minus freezing score); nM-EDL-3 Sc, summed score for the items hallucinations/psychosis, daytime sleepiness and urinary problems on Part 1 (Non-Motor Aspects of Experiences of Daily Living) of the MDS-UPDRS scale.
The second logistic regression on dementia included PIGD symptom severity and was significant when including as a single variable the Bal/Gait score: χ2 = 44.56, p < 0.0001, AUC = 0.851. The unit OR for the Bal/Gait variable was 1.53 (1.33–1.80). Using the optimal cutoff score for Bal/Gait (≥6), 77% of the 164 subjects were correctly classified (Table 3). Adding sequentially the demographic variables age and PD duration to this model failed to significantly increase the ROC AUC.
The final logistic regression model for dementia assessed first the unique predictive value of the composite score for the three non-motor symptoms most closely associated with dementia, the nM-EDL-3 score. This model was significant (χ2 = 27.26, p < 0.0001; AUC = 0.808). The unit OR for the nM-EDL-3 score was 1.73 (1.39 –2.22). Using the optimal cut-off score for nM-EDL-3 (≥3), 77% of the study subjects were correctly classified. Addition of the Bal/Gait score to this model increased the ROC AUC to 0.870. Using the optimal cutoff scores for both variables, 84% of 131 subjects were accurately classified (Table 3). Addition of age and disease duration did not improve the predictive accuracy of this model.
Considering the predictive models for dementia, we investigated the relationship between the Bal/Gait and nM-EDL-3 scores. These clinical variables were highly correlated (r = 0.63, p < 0.0001).
Predictors of non-motor symptom burden (Total MDS-UPDRS nM-EDL Score)
Of the 13 NMS evaluated, the median number of NMS endorsed was 5 (range: 0–12), with a total nM-EDL score of 7.9±4.6 (range: 0–24). There was no association of total nM-EDL score with age or sex, but there was a significant association with PD duration (r = 0.40, p < 0.0001) with a trend for nM-EDL score to be higher in those with an earlier age of PD onset (r = –0.16, p = 0.049). The total nM-EDL score was also associated with the MDS-UPDRS total motor score (p < 0.0001), bradykinesia score (p < 0.0001) and rigidity score (p = 0.006) but not with the tremor score (p = 0.40). In addition, the total nM-EDL score was associated with presence of motor complications (p = 0.0003) and total LD Equiv. dose (p < 0.0001).
A strong relationship of total nM-EDL score with measures of gait and balance impairment was demonstrated by association with the Modified Hoehn and Yahr stage (F = 10.52, p < 0.0001), PIGD score (r = 0.54, p < 0.0001), Bal/Gait score (r = 0.54, p < 0.0001), and motor subtype. Thus, the total nM-EDL mean scores for the tremor-predominant, indeterminant and PIGD subgroups were 5.5±3.3, 8.7±5.6 and 9.1±4.5 respectively (F = 12.92, p < 0.0001). In a regression analysis, the Bal/Gait score alone predicted 29% of the variance of the total nM-EDL score (F = 67.14, p < 0.0001). This effect remained significant when controlling for PD duration (F = 39.21, p < 0.0001). In a stepwise regression analysis on total MDS-UPDRS nM-EDL score, four primary predictors accounted for 46.6% of the total nM-EDL score variance: Bal/Gait score (29.3% : F ratio 21.1, p < 0.0001), total LD equivalent dose (11.8% : F ratio 14.61, p = 0.0002), bradykinesia score (3.4% : F ratio 11.68, p = 0.0008)), and the MDS-UPDRS motor fluctuations subscore (2.1% : F ratio 6.43, p = 0.012).
Non-motor symptoms associated with balance-gait disorder
The Bal/Gait score was associated with presence of cognitive impairment (p < 0.0001), hallucinations/psychosis (p < 0.0001), daytime sleepiness (p < 0.0001), urinary problems (p = 0.0002) and constipation (p < 0.0001), the NMS shown to differentiate demented from non-demented subjects. Associations of the Bal/Gait score with all other NMS were not significant with the Bonferroni correction (p > 0.004).
DISCUSSION
The primary aim of this study was to ascertain the relative importance of PIGD, and especially the severity of balance-gait disorder, as a risk factor for concurrent dementia and NMS burden in PD. Relative to patients with the tremor-predominant or indeterminant subtypes, we confirmed that dementia was more frequent among PD subjects with the PIGD subtype, being associated strongly with the severity of the balance-gait disorder. Our data also showed that balance and gait disturbances predicted concurrent non-motor symptoms other than cognitive impairment, especially hallucinations, daytime sleepiness, and urinary problems.
Dementia is the most important of the non-motor symptoms of PD, developing in up to 83% of 20-year survivors [10]. We found a strong relationship of dementia to age, as noted by others [56–59], the mean age of demented subjects being 79 years, with prediction of dementia status improved when also considering disease duration. Nevertheless, the combination of these risk factors, at their optimal cut-off values, while providing a high sensitivity (100%), had a specificity of only 45% and positive predictive value of only 0.42 for concurrent dementia status. Thus, while acknowledging the general “inevitability of dementia at 20 years” in PD [10], other factors clearly influence the risk of dementia at specific time points and are particularly relevant when attempting to define the course of patients with MCI, who progress to dementia at variable rates [20–23, 60].
Studies have shown that PIGD as a motor subtype predicts not only a more rapid rate of motor progression in PD but also early cognitive decline, with an increased risk of dementia [24, 63]. Transition from the tremor-predominant (TD) to the PIGD subtype in PD is also associated with accelerated cognitive decline and a highly increased risk for subsequent dementia [31]. In our study, using regression models which excluded collinear variables of lesser statistical significance, the MDS-UPDRS-derived Bal/Gait measure correctly classified 77% of demented subjects at its optimal cut-off value, being selected over the motor phenotype, Modified Hoehn and Yahr stage, falls, and the MDS-UPDRS PIGD measure, which includes assessment of freezing of gait (FOG), as the strongest predictor of concurrent dementia. The latter finding is consistent with other evidence that FOG is an independent motor symptom in PD, caused by a paroxysmal pathology that is different from that responsible for other motor symptoms and resulting from a unique neural network dysfunction [64, 65].
Supporting the findings of Polletti et al. [62] of an increased frequency of MCI in PD subjects with more prominent axial involvement, we found balance-gait dysfunction to be a strong risk factor not only for dementia but also for any cognitive impairment in PD. Thus, the Bal/Gait score had a linear relationship with subjective cognitive impairment, as measured by the MDS-UPDRS nM-EDL cognitive subscore, and clearly differentiated, in an incremental manner, subjects with normal cognition, MCI, and dementia (Fig. 1).
The reasons underlying the association between development of balance-gait disorder and both cognitive decline and dementia in PD remains incompletely understood. Neuropathological and pharmacological findings support a major role for the pedunculopontine nucleus (PPN) in the etiology of postural instability and gait difficulties in PD and other disorders [66–70]. Unilateral PPN stimulation has reduced falls and freezing of gait in some patients [71, 72]. The caudal PPN provides cholinergic input to the associative and intralaminar nuclei of the thalamus and projects to arousal-and attention-promoting areas of the hypothalamus and brainstem, including the forebrain nucleus basalis [73]. Ascending PPN neurons are involved in thalamocortical activity and animal studies support a role for these projections in attentional and learning processes [74, 75]. In addition, PPN stimulation in PD may produce significant improvements in executive functions, working memory, delayed recall, and verbal fluency and language, coinciding with widespread bilateral activation of prefrontal regions, as shown by FDG-PET imaging [76–78]. A parallel loss of cholinergic PPN neurons and other central cholinergic neurons linked with cognitive functioning could underlie the association between balance-gait disorder and cognitive dysfunction in PD, a hypothesis that is strengthened by the observation that the acetylcholinesterase inhibitor donepezil may reduce falls in PD [79]. However, there is currently no evidence that the PPN is directly involved in the processes that ultimately lead to PD dementia [31, 80]. In contrast, there is substantial evidence that PD patients with the PIGD phenotype have more widespread neuropathology, including more cortical Lewy bodies and amyloid beta plaques, compared with tremor dominant or younger onset patients [81]. A specific role for amyloid pathology in the development of balance-gait dysfunction in PD is suggested by the study of Alves et al. [82], which found that PD patients with the PIGD phenotype had significant alterations in several Abeta species.
In addition to the severity of the balance-gait disorder, we found that several non-motor symptoms were also strongly associated with PD dementia, especially hallucinations and psychosis, daytime sleepiness and urinary problems. The combined score on this cognition-related NMS cluster (nM-EDL-3) was highly correlated with the severity of the balance-gait disorder and, when considered alone, predicted concurrent dementia with the same accuracy as balance-gait dysfunction. Visual illusions and hallucinations are core symptoms of the dementing process [83, 84] and strongly predict cognitive decline and PD-dementia (PDD) [85–90]. Visual perceptual dysfunction and visual hallucinations in cognitively impaired PD patients have been linked with grey matter atrophy in posterior visual cortices and white matter microstructural alterations in bilateral posterior cingulate bundles, through which cholinergic projections from the nucleus basalis of Meynert travel to visual cortices [91]. Greater daytime sleepiness is associated with both increasing cognitive impairment and psychosis in PD [3, 92–95], and has been linked with cholinergic dysfunction [3, 96], and the presence of cortical Lewy bodies [97].
Our findings of increased frequency of urinary problems and constipation in PD subjects with dementia supports other data suggesting that some autonomic features may emerge more prominently later in the course of PD, with other non-motor symptoms [10, 98], although autonomic dysfunction, including orthostatic hypotension, may also present at an earlier stage, especially in individuals with the PIGD subtype [30, 99]. In one study, early urinary dysfunction characterized a “nonmotor dominant” subgroup associated with more prominent axial symptoms and a more malignant course [100]. In the Sydney multicenter study, orthostatic hypotension was found in 35% and urinary dysfunction in 41% of PD patients after 15 years follow-up [101], prevalence of these conditions increasing to 48% and 71%, respectively, after 20 years of follow-up [10]. It was not specified in this study whether autonomic symptoms were more frequent in association with dementia, which developed in 48% of subjects at 15 years and 83% of subjects at 20 years, but autonomic dysfunction was most frequent in those with higher Hoehn and Yahr stages [10, 101]. Together with our own findings, this suggests that some autonomic symptoms do progress in parallel with cognitive and balance-gait impairment in PD, and may have a pathophysiologic relationship.
In our study, the Bal/Gait score was the strongest unique predictor of total NMS burden, accounting for 29% of the total nM-EDL score variance and 63% of its explained variance, and showing a specific association with the three NMS linked most closely with dementia: hallucinations and psychosis, day-time sleepiness, and urinary problems. This corroborates the findings of Ba et al., who found significant associations between higher NMS burden and both the PIGD phenotype and the axial subscore of the UPDRS-III [28].
In summary, our study supports and extends previous literature regarding the primary importance of balance-gait disorder, irrespective of freezing of gait, as a risk factor for cognitive dysfunction and dementia in PD. Balance-gait impairment also emerged as the major risk factor for NMS burden, reflecting the close association of this measure with a cluster of NMS linked to cognitive impairment.
There are some limitations of this study. As a retrospective study, it is possible that unintentional selection bias may have affected the profile of the study cohort. Against this is the observation that patients screened but not included in the study did not differ significantly in their demographic data or clinical ratings from the study subjects. Furthermore, the study cohort was similar to that of PD cohorts evaluated in previous prognostic studies, and approximates the clinical profile of PD patients in the general population. A strength of our study was the close screening of subjects to exclude conditions other than PD which might affect balance, gait or cognitive functioning. As typically employed in evaluation of parkinsonism, our assessment of gait and balance was based on a recommended clinical measure, rather than posturography [31, 44]. Although it is likely that the great majority of our patients classified as MCI, who met current DSM-5 criteria for mild neurocognitive disorder, would also meet current consensus guidelines for PD-MCI [48], use of the MoCA or other approved cognitive scales [102] in future studies would allow greater generalizability of our findings.
The results of this study draws attention to a simple clinical measure that defines a subgroup of PD patients likely to require close clinical surveillance and more comprehensive care. From a different perspective, the findings should be considered in the context of related prospective studies, and with regard to the sensitivity of differing measures of gait-balance disorder as predictors of PD dementia. If supported by further prospective longitudinal studies, our observations suggest that the MDS-UPDRS Bal/Gait score, a measure strongly correlated with both increasing degrees of cognitive impairment and concurrent dementia, and more robust in our study as a predictor of such status than either the Hoehn and Yahr stage, freezing or falls, could find a place in algorithms designed to predict the likelihood of progression to dementia in PD.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
The authors thank the patients and family members who participated in this study. The study was unfunded.
