Abstract
Background:
Autonomic dysfunctions including sudomotor abnormalities commonly occur in early Parkinson’s disease (PD), but little is known about potential sudomotor abnormalities in idiopathic REM Sleep Behavior Disorder (iRBD), a strong prodromal marker of PD.
Objective:
Our aim was to assess sudomotor dysfunction by galvanic skin response using SudoScan, as well as other autonomic markers in 49 iRBD, 40 PD (21 with RBD, 19 without), 20 atypical parkinsonisms, and 41 age-matched controls.
Methods:
All subjects underwent SudoScan of their hands and feet, a 30-second electrocardiogram with assessment of beat-to-beat variability, assessment of orthostatic blood pressure changes and autonomic symptom questionnaires.
Results:
The galvanic skin response in the hands of PD patients with RBD was significantly smaller than controls (hand mean difference = –7.877, 95% CI (–13.283, –2.470), p-value = 0.004) and PD patients without RBD (hand mean difference = –9.578, 95% CI (–17.215, –1.941), p-value = 0.014). iRBD and atypical parkinsonism did not have different SudoScan profiles than controls.
Conclusions:
Galvanic skin responses, as measured by SudoScan did not demonstrate significant sudomotor dysfunction in iRBD, but decreases were seen in the PD subtype associated with RBD.
INTRODUCTION
Parkinson’s disease (PD) is more than just a motor disorder, and autonomic symptoms like orthostatic hypotension, constipation, urinary dysfunction and sudomotor abnormalities are common. These autonomic changes are related in part to synuclein-mediated neurodegeneration in the peripheral nervous system [1, 2]. Autonomic dysfunction occurs in prodromal stages [3], and can predict development of clinical PD. Sudomotor function is currently assessed by thermoregulatory sweat tests, which are time-consuming and often confounded by factors such as room temperature and body temperature.
Recently, several simple measures have been proposed to assess autonomic dysfunction. One is a test of skin conductance, SudoScan [4]. It tests the function of sweat glands in the skin, which are innervated by thin, unmyelinated, predominantly cholinergic fibers. The SudoScan applies low-current DC stimulation of sweat glands to the palms and soles via stainless steel electrodes. Sweat production is assessed by changes in electrical conductance related to chloride ionic release in sweat. Electrochemical skin conductance (ESC) was found to correlate with sudomotor nerve fiber density [5] and feet Quantitative Sudomotor Axon Reflex Test (QSART) [6]. So far, studies have shown profound deficits in diabetic autonomic neuropathy [4]; however, whether such abnormalities can be detected in the subtler PD-associated neurodegeneration remains unclear.
In this study, we primarily aimed to assess galvanic skin response (i.e. SudoScan) in five groups, idiopathic REM Sleep Behavior Disorder (iRBD) (a very strong marker of prodromal PD), PD with RBD, PD without RBD, atypical parkinsonism, and age-matched controls. Secondary analyses were also performed to compare other autonomic-related measures namely heart rate variability [7–9], orthostatic drop in blood pressure and the Scales for Outcomes in Parkinson’s Disease-Autonomic questionnaire (SCOPA-AUT) scores between groups, and to correlate to the SudoScan measures.
METHODS
Subjects
150 Subjects (49 iRBD, 40 PD, 20 atypical parkinsonism and 41 age-matched controls) were recruited from June 2016 till May 2017 at McGill University Movement Disorder Clinic, and the Center for Advanced Research in Sleep Medicine, Hôpital du Sacré-Cœur de Montréal. iRBD subjects were polysomnography confirmed and were free of parkinsonism and dementia based on expert evaluation at the time of SudoScan and autonomic measures acquisition. PD subjects met criteria for probable PD, according to the new MDS Clinical Diagnostic criteria [10]. PD subjects were subdivided as PD with RBD and PD without RBD according to the RBD single question screen (RBD1Q) [11] followed by expert interview (by RP/AQ). Atypical parkinsonism cases were diagnosed clinically and included progressive supranuclear palsy (n = 12), vascular Parkinsonism (n = 2), Lewy body dementia (n = 1), multisystem atrophy (MSA) (n = 1), corticobasilar degeneration (n = 1), drug induced parkinsonism (n = 1), primary gait freezing (n = 1) and one patient with ferritinopathy. Exclusion criteria were age <40, diabetes mellitus, pre-existing diagnoses of autonomic neuropathy (i.e. from a cause other than PD), prescription of medications that directly alter peripheral autonomic function (beta adrenergic blockers and non-specific alpha-adrenergic blockers), and dementia of severity sufficient to preclude informed consent. The study was approved by the ethics board of McGill University Health Centre and of Hôpital du Sacré-Coeur, and all subjects gave written informed consent to participate.
Clinical assessment and autonomic function measures
All subjects were evaluated for the four parts of the modified Movement Disorder Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS), Hoehn and Yahr stage, SCOPA-AUT Multisystem Atrophy (MSA) rating scale and RBD1Q. Neuropathy screening was done with the 5-item peripheral neuropathy screening interview. Orthostatic blood pressure(BP) was measured in the supine position, and after 1 and 3 minutes of standing.
The primary outcome measure, sudomotor function from palms and feet, was assessed as electrochemical skin conductance (ESC) using the SudoScan (Impeto Medical ®, France). Normal values approximate 70–80 MicroSiemens (μS) in the hands and 80–85μS in the feet [4]. Lower results indicate abnormal sudomotor function. A 30-second electrocardiography (ECG) trace at rest was obtained for each subject using HeartCheck™ ECG device. ECGs were manually screened for abnormality and quality by MD (AQ). RR intervals were measured manually for each tracing and heart beat to beat variability was assessed by calculating the square root of the mean squared differences of successive normal to normal RR intervals (RMSSD) using the following formula [7]:
Statistical analysis
Prior to main analysis, outliers of SudoScan and heart rate variability measures were excluded using the Tukey’s fences method. For measures with non-normal distribution, we applied the non-parametric Kruskal Wallis test for the between-group comparisons and Spearman Rho for bivariate correlations. Equivalent parametric tests namely one-way ANOVA and Pearson correlation were used for normally-distributed variables. For post-hoc testing, we performed multivariate linear regression analysis. In each model, the autonomic measure of interest (i.e. SudoScan of the hand and feet) was set as the dependent outcome, and the study group was defined as the independent categorical variable (with the controls as the reference condition), while adjusting for age, sex and disease duration.
RESULTS
As shown in Table 1, most demographic variables were similar. The atypical parkinsonism group was older, had higher motor score (UPDRS-III) and lower MOCA compared with other groups, with similar disease duration as the PD group. SCOPA-AUT score and orthostatic drop in systolic blood pressure were significantly higher in PD with RBD compared to PD without RBD and to other groups.
Baseline characteristics
Median and interquartile range (IQR) are reported except for Gender (%). *p < 0.05; **p < 0.01. aMissing values (n = 9, Control 2, RBD 2, PD with RBD 2, PD without RBD 0, parkinsonism 3). 5 subjects moved during SudoScan recording, one subject was on beta-blocker and 3 values were excluded as outliers. bMissing values (n = 30, Control 4, RBD 8, PD with RBD 6, PD without RBD 3, parkinsonism 9). 16 tracing with movement artifacts, 8 subjects with arrhythmias, one subject was on beta-blocker and one subject was post cardiac transplant. 4 values were excluded as outliers. RMSSD, square root of the mean squared differences of successive normal to normal RR intervals.
On the primary outcome, adjusting for age and sex, SudoScan of the hand and feet was significantly lower in PD with RBD compared to controls, (hand mean difference = –7.877, 95% CI (–13.283, –2.470), p-value = 0.004, feet mean difference = –6.354, 95% CI (–9.76, –2.948), p-value <0.001), and PD patients without RBD (hand mean difference = –9.578, 95% CI (–17.215, –1.941), p-value = 0.014, feet mean difference = –6.390, 95% CI (–11.428, –1.351), p-value <0.013). In general, although there was overlap between groups, we observed a decreasing trend of SudoScan results as we moved from control to RBD with and without PD (Fig. 1A, B). However, patients with idiopathic RBD did not have significant differences from controls. Furthermore, there was no significant difference in hand and feet SudoScan of PD without RBD and atypical parkinsonism compared to controls.
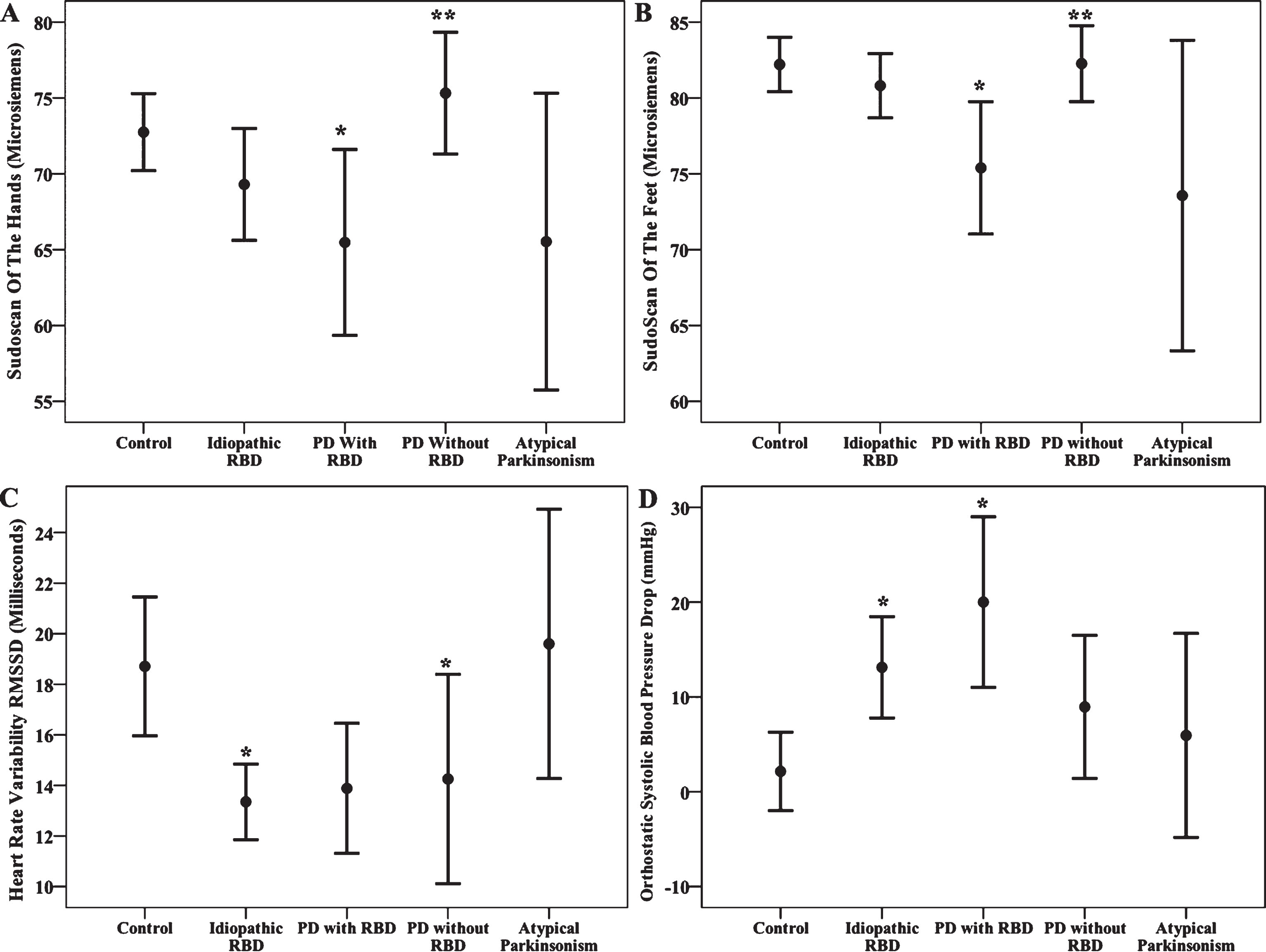
95% confidence interval error bars for Hand SudoScan (A), Feet SudoScan (B), Heart Rate Variability (C) and Orthostatic Systolic Blood Pressure Drop (D) across all groups. *significant difference compared with Control; **significant difference compared with PD with RBD.
For measures of heart rate variability, the median of RMSSD was less in the PD without RBD group compared to atypical parkinsonism and controls. RMSSD was also significantly lower in the iRBD group than controls (Fig. 1C). Compared to controls, the highest decline in systolic blood pressure after 3 minutes of standing occurred in PD with RBD (mean difference = 16.769, 95% CI (8.199,25.34), p-value <0.001, Fig. 1D) followed by the iRBD group (mean difference = 10.279, 95% CI (3.323, 17.236), p-value = 0.004).
Hand SudoScan of PD with RBD correlates with total SCOPA-AUT (Spearman Rho = –0.412, P = 0.04) and urinary scores (Spearman Rho = –488, P = 0.034). Systolic blood pressure drop was not correlated with SudoScan across all groups.
DISCUSSION
We demonstrated that galvanic skin measures in PD with RBD were significantly lower than both controls and PD patients without RBD, suggesting a difference in peripheral alpha-synuclein burden between PD with RBD and PD without RBD [1, 2]. By contrast, there were no significant difference in SudoScan profile between PD without RBD and atypical parkinsonism when compared to controls. Similar to previous studies, we found that heart rate variability, orthostatic blood pressure, and autonomic symptoms are abnormal in idiopathic RBD, and often correlate with RBD status in PD [3, 12].
Our study is the first to assess the SudoScan device as a marker of galvanic skin response in iRBD. A previous study has used SudoScan in PD including 48 subjects (19 PD, 7 E46-synuclein mutation carriers, 3 Lewy body dementia and 19 controls) [13]. The SudoScan profile, for the hand in particular, was lower in E46-SCNA mutation carriers and Lewy body dementia. Like this study, we also found abnormalities more in the hands than the feet. This is in accord with proposed craniocaudal spread of cutaneous alpha-synuclein deposition [1]. Whereas we did see abnormalities when combining averages between groups, it is important to note that there was considerable overlap between groups. Moreover, there were some outlier values, with readings below the level of biological plausibility. Therefore, it is unlikely that SudoScan could be used alone as a diagnostic test for autonomic dysfunction in PD. Furthermore, the absence of significant differences between idiopathic RBD patients and controls, despite clear changes on other autonomic parameters, suggests low sensitivity to prodromal stages of PD/DLB.
In the current study, cardiac beat-to-beat variability was significantly lower in iRBD compared to controls, consistent with previous studies [8, 9]. However, in contrast to previous studies, we did not see clear reduction of variability in PD patients with RBD compared to those without RBD. This could be a reflection of low statistical power due to the smaller sample size compared to iRBD group. We used a handheld ECG recorder which resulted in some tracing with movement artifact, contributing to the lower sample size. Note also that we used a single summary measure on a 30-second trace, rather than a detailed analysis of variability patterns that can emerge over longer periods (i.e. very low frequency and low frequency variability). These low frequency variability patterns are driven more by sympathetic denervation and are not assessed on short traces.
We found no correlation between SudoScan measure and systolic blood pressure drop across all group. A potential explanation is that SudoScan measures cholinergic sudomotor function as opposed to orthostatic BP response which is mostly adrenergic [14]. Hand SudoScan in PD with RBD appears to be the main variable correlated with autonomic scores like total SCOPA-AUT and urinary score of MSA scale. Overall, the lack of correlation between SudoScan and the other autonomic measures is not clearly understood. One explanation is simply that the SudoScan is insufficiently precise, leading to lack of statistical power. Another explanation comes from recalling that sweat production is dependent both on integrity of sweat glands and upon sudomotor innervation [15]. Perhaps the lack of correlation between ESC and other autonomic measures is due to the presence of underlying small fiber neuropathy in the skin of PD patients [1] leading to sweat gland atrophy (a process that does not correlate with autonomic dysfunction). This is supported by existing correlation between skin nerve fiber density and ESC [5].
Some limitations should be pointed out. One is that stratifying the PD into those with and without RBD reduced the sample size of the PD subgroup. However, we felt this important, since RBD is a strong marker of a diffuse malignant phenotype of PD [16], and has been linked to autonomic dysfunction [3, 12]. Another limitation is that RBD was not confirmed by polysomnography in PD group, but was assessed by questionnaire and expert interview. Although at our center, expert interview has high specificity [17], residual misclassification would likely wash out differences between groups. Furthermore we had one case of MSA only, it would be of particular interest to assess SudoScan in more subjects with this form of atypical parkinsonism. Finally, it should be emphasized that this study used one measure of galvanic skin response. Although SudoScan results found differences between groups, these were modest; alternate studies of sweating responses may be better biomarkers of autonomic dysfunction in synucleinopathy.
CONFLICT OF INTEREST
The authors have no conflict of interest to report.
FULL FINANCIAL DISCLOSURE FOR THE PREVIOUS 36 MONTHS
A. AL-Qassabi reports the following disclosure: Scholarship from Sultan Qaboos University, Muscat, Oman.
A. Pelletier reports the following disclosures: Employment with Research Institute of the McGill University Health Centre (Montreal, QC, Canada).
S.M. Fereshtehnejad reports the following disclosures: Employment with McGill University (Montreal, QC, Canada); Grants from Canada First Research Excellence Fund for the Healthy Brains for Healthy Lives (HBHL) initiative Postdoctoral Fellowship, Richard and Edith Strauss Postdoctoral Fellowship (McGill University), Preston Robb Fellowship (Montreal Neurological Institute).
R.B. Postuma reports the following disclosures: Employment with Department of Neurology, Montreal General Hospital (Montreal, QC, Canada); Grants from Fonds de la Recherche en Sante Quebec, the Canadian Institute of Health Research, the Parkinson Society of Canada, the Weston-Garfield Foundation, the Michael J. Fox Foundation and the Webster Foundation; Honoraria from Novartis Canada, Teva Neurosciences; Consultancies for Biotie, Roche.
Footnotes
ACKNOWLEDGMENTS
We thank Michael J Fox Foundation for funding this work. Also we thank Chun Yao PhD student for help in heart rate variability formula and Marie Corbeil for secretarial support.
