Abstract
Background:
It is debatable whether transcranial sonography (TCS) could be a biomarker for monitoring disease progression. Various phenotypes of Parkinson’s disease (PD) may be a major reason contributing to the inconsistency.
Objective:
We classified PD patients into different subtypes and evaluated the correlation between SN echogenicity and disease progression.
Methods:
A total of 411 PD patients were included in this study. TCS evaluations of the substantia nigra (SN) were performed, and motor and non-motor symptoms were assessed by a series of rating scales in all PD patients.
Results:
Three hundred and thirteen patients had appropriate temporal acoustic bone windows, and they were divided into three subgroups according to disease onset age. SN hyperechogenicity (SN+) was found to be associated with age, gender, disease duration, H-Y stage and UPDRS-II scores in 220 middle-age onset patients. Regression analysis identified both disease duration and gender as independent predictors for SN+. When this distinct group was separated into male and female subgroups, the correlation between larger SN echogenicity (SNL) and disease duration was positive in males rather than females. When these middle-age onset male patients were classified as tremor dominant (TD) and non-TD subtypes, it turned out that correlation between disease duration and SNL only existed in male non-TD PD patients.
Conclusions:
Our study demonstrated correlation between the size of SN echogenicity and disease duration in Chinese patients with PD who were male non-TD subtypes with middle-age onset, suggesting the formation of SN echogenicity might be a dynamic process following disease progression in this distinct subtype.
INTRODUCTION
Parkinson’s disease (PD) is the second most common neurodegenerative disorder and its movement dysfunction mainly results from the progressive death of dopaminergic neurons in the substantia nigra (SN) [1]. Transcranial sonography (TCS) can display specific structural changes in the SN of PD patients, and hyperechogenicity in the anatomical area of the SN is believed to be a marker of PD [2–6]. More and more evidence has indicated that TCS could be a useful tool not only for diagnosis of PD, but also for differential diagnosis from other forms of Parkinsonism [7–10]. However, it is still unclear whether TCS could be a biomarker for monitoring disease progression or disease severity. Although some research has demonstrated that enlarged hyperechogenic size of the SN was associated with the functional impairment of the nigrostriatal system and disease severity [11–13], others have not shown the congruent results [3, 14–19]. One of the major reasons for this inconsistency could be the complexity of PD phenotypes or variety of subtypes. Different subtypes might imply different potential pathogenic mechanisms, leading to the different echogenic patterns. Here, our study classified PD patients into different subtypes with regard to onset age and motor features, and evaluated the correlation of the sonographic findings with the clinical features including motor and non-motor characteristics.
MATERIALS AND METHODS
Subjects
Four hundred and eleven Chinese patients with PD meeting the criteria of the UK Parkinson’s Disease Society Brain Bank [20] from the movement disorders clinic of our hospital were included in this study. None of the PD patients demonstrated atypical features such as pyramidal signs, gaze palsy, or cerebellar ataxia, or had a history of stroke, neuroleptic intake or other identifiable possible causes of secondary parkinsonism. This study was approved by the Ethics Committee of Ruijin Hospital affiliated to Shanghai Jiao Tong University School of Medicine. All participants have given written informed consent.
Three hundred and thirteen patients (76.2%) had appropriate temporal acoustic bone windows. Those PD patients were then divided into three groups with respect to the disease onset age, including young onset PD whose age of onset were less than 40 years, middle-age onset PD whose age of onset were between 40 and 65years, and late onset PD with age of onset equal to or above 65 years [21–23].
To further analyze motor subtypes, 220 middle-age onset male patients were further classified into two subgroups: tremor-dominant (TD) group (n = 55) and non-TD group (n = 165). The classification was based on previously reported methods [24]. Briefly, an average global tremor score was calculated as the mean of the following 9 items: right and left arm tremor as determined by history, resting tremor of face, lips, or chin, resting tremor of four limbs, and action or postural tremor in both arms as determined by the investigator’s examination. A mean score for the complex of postural instability and gait difficulty (PIGD) was calculated as the mean of the following 5 items: falling, freezing, a history of walking difficulty, and gait and postural instability by examination. The TD group was defined as patients with a ratio of mean tremor score/mean PIGD score≥1.5; the non-TD group included all patients with a ratio of <1.5.
Midbrain transcranial sonography
The sonographic examinations were performed by one experienced sonographer who was blind to the clinical data of all patients. The procedure was done as previously described [25]. In brief, TCS was performed through the preauricular acoustic bone windows using a 2.5 MHz sonographic device (MyLab90, ESAOTE, Italy) with a depth of 16 cm and a dynamic range of 45 dB. The SN was scanned through both temporal bone windows in the axial plane. Initially, the butterfly shaped hypoechogenic midbrain, which was surrounded by the hyperechogenic basal cistern was identified. Then, the one with the hyperechogenic signal at the anatomical region of SN in its maximum extent was stored. Both sides of SN echogenic areas from stored images were then manually encircled and measured. Reproducibility of the SN sonographic measurement had been previously validated by two independent investigators [25]. Those with both of SN echogenic sizes less than 18 mm2 were classified as normal (SN–), the others with at least one SN echogenic size≥18 mm2 were classified as hyperechogenic (SN+) as previously described [25]. The larger value of bilateral SN hyperechogenicity was defined as SNL.
Motor and non-motor symptoms assessment
The disease disability of the PD patients was rated by the unified Parkinson’s disease rating scale (UPDRS) and Hoehn & Yahr Stage (H-Y stage) while patients were on treatment. A battery of scales were used to evaluate non-motor characteristics as previously described [25], including the Non-Motor Symptoms Questionnaire (NMS-Quest), the Non-Motor Symptoms Scale (NMSS), the 16-item odor identification test from extended version of sniffin’ sticks (SS-16; Burghart Messtechnik, Wedel, Germany), the rapid eye movement (REM) sleep behavior disorder screening questionnaire (RBDSQ), the RBD questionnaire Hong Kong version (RBDQ-HK), the scale for outcomes in PD for autonomic symptoms (SCOPA-AUT), the Mini-Mental State Examination (MMSE) and the 17-item Hamilton Rating Scale for Depression (HAMD-17).
Statistical analysis
All analyses were performed by using SPSS 16.0. Continuous variables were given as means (±standard deviation, SD). Categorical variables were summarized by counts of patients and percentages. All variables were tested for normality by Kolmogorov-Smirnov. Two-sample T test, ANOVA or Chi-square test was applied for the comparisons between the SN+ and SN– groups. Tukey method was used as p ost hoc test. To account for multiple testing, resulting nominal p values were adjusted according to the Bonferroni procedure. Since 18 T-tests were applied in the comparison of subgroups, the adjusted p values were equal to 18 times nominal p value. Adjusted p values below 0.05 were considered significant. Then a step-wise logistic regression model was used to determine the most significant variables which were independently correlated with SN+. Threshold for entry and exit of a variable was p = 0.05. All variables including age, gender, disease duration, H-Y stage and UPDRS-II score were included in the logistic regression model. Correlation analyses between the variables and SNL were performed by Spearman correlation coefficients. The significant level was set at α= 0.05.
RESULTS
TCS examination could be performed in 313 of the 411 PD patients. Ninety-eight subjects (23.8%) had no appropriate temporal acoustic bone windows and were therefore excluded from further analysis, which was in line with our previous study [25].
Association of variables with SN+ in patients with different age of onset
The detailed clinical data of the 311 patients were shown in Table 1. Comparison between patients with SN+ and SN– was performed under each subgroup. It turned out that in the middle-age onset group, SN+ was significantly associated with older age (59.9±7.6 vs. 58.0±6.1, p = 0.036), male gender (p < 0.001), longer disease duration (5.9±4.6 vs. 4.2±3.4, p = 0.002), higher H-Y stage (2.0±0.7 vs. 1.8±0.6, p = 0.031) and higher UPDRS part II (UPDRS-II) scores (12.1±6.7 vs. 10.3±5.6, p = 0.030). However, in the other two groups with different onset ages, no significant difference in clinical features between SN+ and SN– patients was found. Finally, after Bonferroni correction, only gender (p < 0.001) and disease duration (p = 0.036) were related to SN+ echogenicity in the middle-age onset group (Table 1).
Clinical data of 311 PD patients grouped according to disease age of onset
SN, substantia nigra; UPDRS, Unified Parkinson’s Disease Rating Scale; MMSE, Mini-Mental State Examination; SCOPA-AUT, scale for outcomes in PD for autonomic symptoms; SS-16, 16-item odor identification test from extended version of Sniffin’ Sticks; HAMD-17, 17-item Hamilton Depression Rating Scale; NMS-Quest, Non-Motor Symptoms Questionnaire; NMSS, Non-Motor Symptoms Scale; RBDSQ, rapid eye movement sleep behavior disorder screening questionnaire; RBD-HK, rapid eye movement sleep behavior disorder questionnaire Hong Kong version. Two-sample T test, ANOVA or Chi-square test was applied for the comparisons between the SN+ and SN– groups. Tukey method was used as Post hoc test. *p values below 0.05 were considered significant. To account for multiple testing, resulting nominal p values were adjusted according to the Bonferroni procedure. Adjusted #p values below 0.05 were considered significant.
Further step-wise logistic regression analysis was used to identify the relationship between variables (including age, gender, disease duration, H-Y stage and UPDRS-II score) and SN+. Only gender and disease duration were included in the final model. It demonstrated that there was significant association between disease duration and SN+ (odds ratio = 1.095, 95% CI = 1.022–1.183, p < 0.001), as well as significant relationship between male gender and SN+ (odds ratio = 3.322, 95% CI = 1.863–5.925, p < 0.001). In order to eliminate PD patients with possible misdiagnosis, we also checked the correlation of those variables in a subgroup with disease duration equal to or longer than 5 years. It turned out that the association still existed between disease duration and SN+ (odds ratio = 1.122, 95% CI = 1.007–1.345, p < 0.001), as well as between male gender and SN+ (odds ratio = 6.912, 95% CI = 2.568–18.605, p < 0.001).
Figure 1A demonstrated that there was significant correlation between disease duration and SNL (R = 0.191, p = 0.005) in the middle-age onset PD group, but not in the other two groups (R = –0.123, p = 0.450, or R = 0.104, p = 0.464, respectively). The correlation even became slightly stronger (R = 0.302, p = 0.003) when only patients with disease duration equal to or longer than 5 years were included in this middle-age onset group (Fig. 1B).
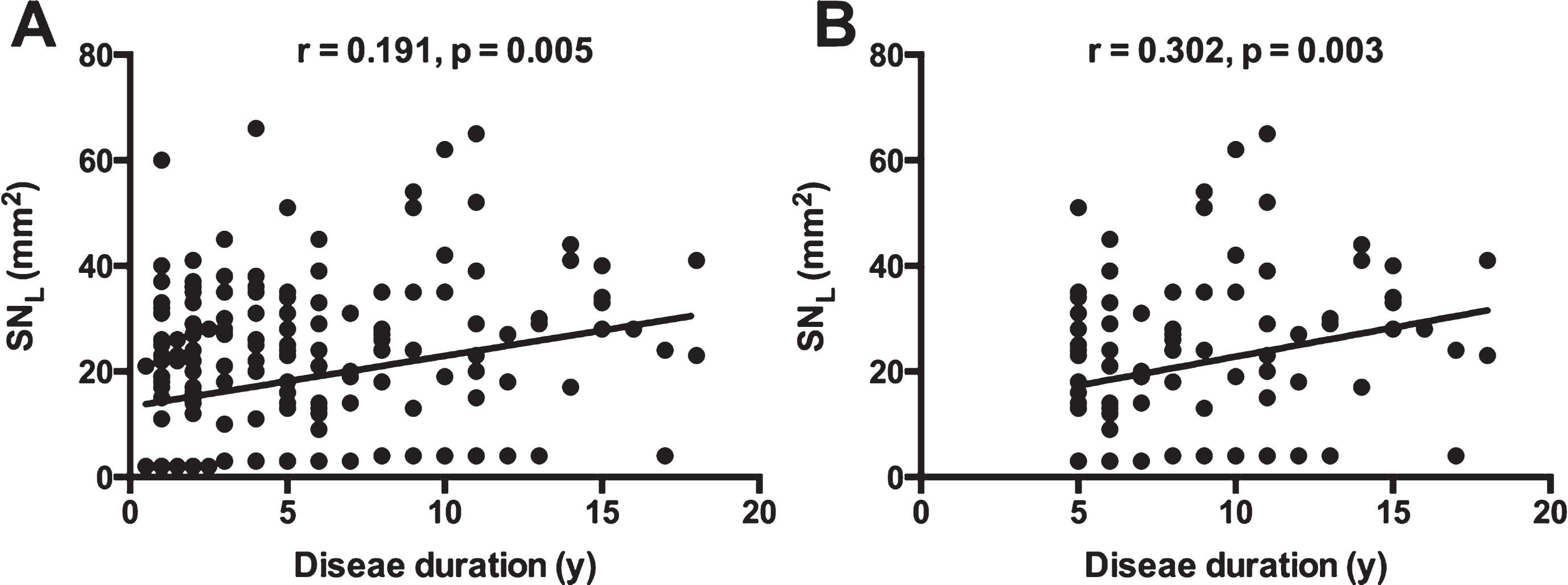
Correlation between SNL and disease duration in the middle-age onset PD patients. A demonstrated significant correlation between SNL and disease duration in the whole middle-age onset PD, and B showed stronger correlation when only patients with disease duration equal to or longer than 5 years were included. (SNL, larger value of bilateral substantia nigra hyperechogenicity).
Gender difference in middle-age onset PD patients
Considering that gender was an independent risk factor for SN+ in the middle-age onset PD patients, correlation analyses between disease duration and SNL were performed in male and female patients separately. Age was used as a covariate. Figure 2A showed that significant correlation was only found in males (R = 0.243, p = 0.005), not in females (R = 0.057, p = 0.605).
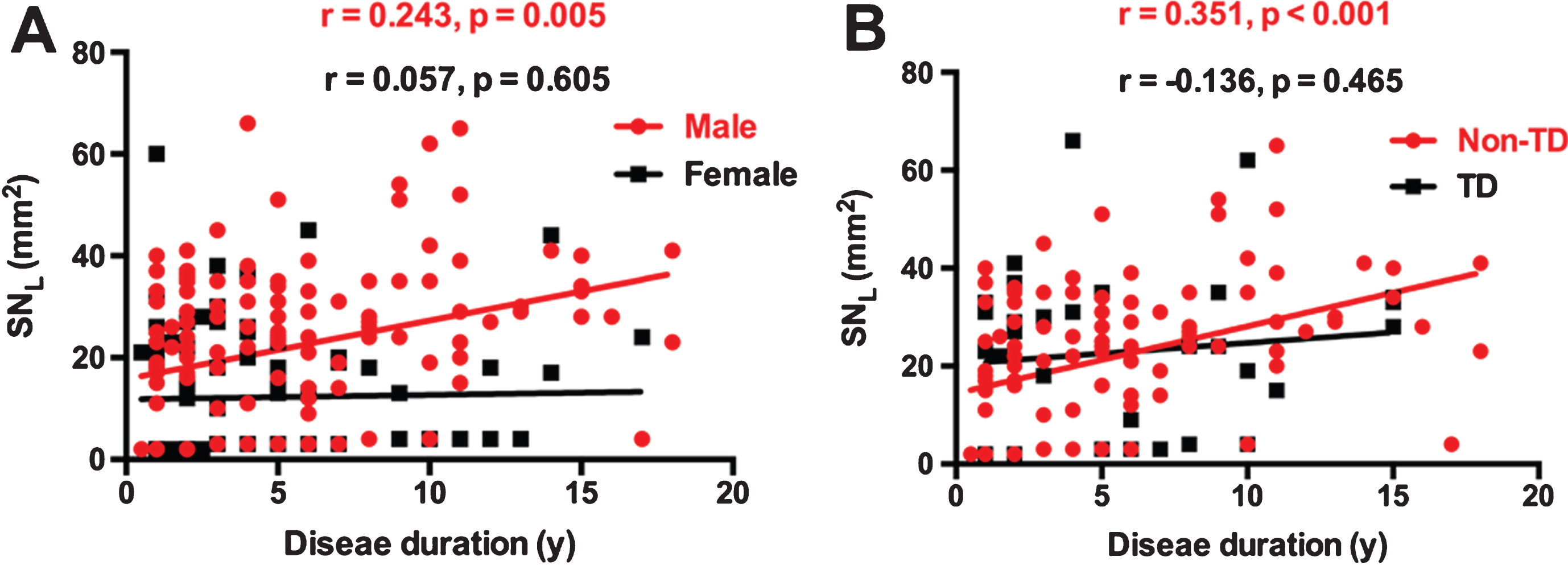
Gender difference and motor subtypes difference in the correlation between SNL and disease duration. A showed that significant correlation was only found in males, but not in females. B showed significant correlation was found in non-TD group rather than in TD group. (SNL, larger value of bilateral substantia nigra hyperechogenicity; TD, tremor dominant; non-TD, non-tremor dominant).
Motor subtypes difference in middle-age onset male PD patients
To explore whether motor subtypes had effects on the correlation of disease duration and SNL, we further divided the middle-age onset male patients into TD (n = 55) and non-TD (n = 165) subgroups. In the non-TD males, the association was positive between disease duration and SN+ (odds ratio = 1.224, 95% CI = 1.093–1.439, p < 0.001), while no association was found in TD male patients (odds ratio = 0.959, 95% CI = 0.865–1.215, p = 0.404). The results of Spearman correlation analysis also showed significant correlation in non-TD group (R = 0.351, p < 0.001) as opposed to the TD group (R = –0.136, p = 0.465) (Fig. 2B).
DISCUSSION
Our study shows a significant correlation between SNL and disease duration in male non-TD subtypes of PD patients, whose disease onset age was between 40 and 65 years.
Several previous studies also demonstrated that SN+ in PD patients was positively correlated with longer disease duration [3, 26]. However, other studies suggested that SN hyperechogenicity was a stable marker during disease progression [13–15, 27]. The controversy could be explained by misdiagnosis of atypical PD with shorter disease duration as idiopathic PD [26]. Indeed, most of the patients in our cohort were relatively at early H-Y stages, which might lead to misdiagnosis of PD in some patients. However, this could not explain the whole scenario, since the positive correlation existed not only in all middle-age onset patients, but also in those patients whose disease duration were equal to or above 5 years, which could almost exclude the possibility of misdiagnosis. In accordance to previous studies including ours [3, 25], UPDRS III scores was not related to SN+. Indeed, it was debatable whether the extent of SN echogenecity correlated with the degeneration of the nigrostriatal dopaminergic system and disease severity [11, 28]. The origin of SN hyperechogenecity was not fully understood, but was proposed to be related to nonchelated forms of iron, protein bound iron and microglia activation [3, 30]. We could speculate that with the course of disease, those increased iron, iron binding protein structure and gliosis might produce a detectable hyperechogenic signal in the SN, but not be sufficient to destroy the nigrostriatal function. One interesting finding in our study was that the non-TD rather than TD male patients had a significant correlation between SNL and disease duration. Walter et al. [15] also demonstrated that the akinetic-rigid or mixed PD group showed larger hyperechogenicity than the TD group. Different neuropathological processes affecting the SN between akinetic-rigid PD and TD PD [31, 32] might be the explanation for different SN echogenic patterns.
Another distinct factor found in our study was gender difference. In middle-age onset patients, male gender was not only related to SN+, but also as an independent risk factor for it, which was in line with our previous study [25]. The comparable sonographic penetration rate between female (75.7%) and male (72.9%) in this distinct cohort further helped to exclude the bias due to different penetration rate. It has been shown that gender could influence the prevalence and onset age of PD, as well as clinical phenotypes [33]. However, to the best of our knowledge, this was the first report addressing gender influence on the relationship between SN echogenicity and disease duration. Hormone-related pathologic mechanisms might contribute to the different SN echogenic patterns between genders.
The great strength of our study was the large number of patients enrolled and analyzed. However, there was a big concern about the low sensitivity of TCS in this cohort. Only 52.1% of PD patients had positive hyperechogenic signals in the SN, which was much lower than most of earlier studies [13–17, 35]. The inaccurate diagnosis of PD could be one reason, since patients in the early stage of the disease were easily to be misdiagnosed [36]. Indeed, in our cohort, the mean H-Y value between groups was between 1.7 and 2, therefore misdiagnosis of PD might be the case in some patients. However, race-ethnic difference could not be absolutely excluded. Both our group and another Chinese research group have previously shown that the sensitivity of TCS was only about 60% even when using single photon emission computed tomography as a clinical gold standard [19, 37]. Spectrum bias also could be another main cause. In our cohort, 12.9% of PD patients were under 40, which was higher than expected. This might be because that most of our enrolled patients were from one expertise’s clinic, and also only the patients who had good compliance were selected. Another limitation of our study was different sizes of three groups. Although there was no positive correlation found in the other two subgroups, we could not exclude the possibility that the lack of significance in the other groups might be simply due to the relatively smaller size.
In summary, our present study demonstrated that the size of SNL was correlated with disease duration in a distinct cohort whose disease onset age was between 40 and 65 years and who were male non-TD patients. This might give us a hint that SN echogenicity could be a dynamic marker following the progression of disease in this specific PD subtype. However, our study was a cross-sectional study, so that we did not see the natural progression of the disease and its relation with SN echogenicity. More convincing conclusion could be drawn by future long term longitudinal prospective follow up studies which would take different PD subtypes into account at the first place.
CONFLICT OF INTEREST
All authors have no conflict of interest to report.
Footnotes
ACKNOWLEDGMENTS
This study was supported by a National Key Basic Research Program of China (No.2011CB504104), Natural Science Fund of China (No. 81430022, 81371407) and Natural Science Foundation of Science and Technology of Shanghai (No. 15ZR1426700). We also thank all the subjects who participated in the study.
